Abstract
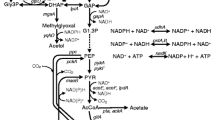
Aminolevulinic acid (ALA), a type of natural non-protein amino acid, is a key precursor for the biosynthesis of heme, and it has been broadly applied in medicine, agriculture. Several strategies have been applied to enhance ALA synthesis in bacteria. In the present study, we employed synthetic antisense RNAs (asRNAs) of hemB (encodes ALA dehydratase) to weaken metabolic flux of ALA to porphobilinogen (PBG), and investigated their effect on ALA accumulation. For this purpose, we designed and constructed vectors pET28a-hemA-asRNA and pRSFDuet-hemA-asRNA to simultaneously express 5-ALA synthase (ALAS, encoded by hemA) and PTasRNAs (2 inverted repeat DNA sequences sandwiched with the antisense sequence of hemB), selecting the region ranging from − 57 nt upstream to + 139 nt downstream of the start codon of hemB as a target. The qRT-PCR analysis showed that the mRNA levels of hemB were decreased above 50% of the control levels, suggesting that the anti-hemB asRNA was functioning appropriately. ALA accumulation in the hemB weakened strains were 17.6% higher than that obtained using the control strains while accumulating less PBG. These results indicated that asRNAs can be used as a tool for regulating ALA accumulation in E. coli.





Similar content being viewed by others
References
Choi HP, Hong JW, Rhee KH, Sung HC (2004) Cloning, expression, and characterization of 5-aminolevulinic acid synthase from Rhodopseudomonas palustris KUGB306. FEMS Microbiol Lett 236:175–181
Ding W, Weng H, Du G, Chen J, Kang ZJ (2017) 5-Aminolevulinic acid production from inexpensive glucose by engineering the C4 pathway in Escherichia coli. Ind Microbiol Biotechnol 44(8):1127–1135
Hamza I, Dailey HA (2012) One ring to rule them all: trafficking of heme and heme synthesis intermediates in the metazoans. Biochim Biophys Acta 1823:1617–1632
Jaffe EK, Ali S, Mitchell LW, Taylor KM, Volin M, Markham GD (1995) Characterization of the role of the stimulatory magnesium of E. coli porphobilinogen synthase. Biochem 34:244–251
Kang Z, Wang Y, Wang Q, Qi Q (2011) Metabolic engineering to improve 5-aminolevulinic acid production. Bioeng Bugs 2:1–4
Kang Z, Zhang J, Zhou J, Qi Q, Du G, Chen J (2012) Recent advances in microbial production of delta-aminolevulinic acid and vitamin B12. Biotechnol Adv 30:1533–1542
Kang Z, Ding W, Gong X, Liu Q, Du G, Chen J (2017) Recent advances in production of 5-aminolevulinic acid using biological strategies. World J Microbiol Biotechnol 33(11):200
Lee DH, Jun WJ, Kim KM, Shin DH, Cho HY, Hong BS (2003) Inhibition of 5-aminolevulinic acid dehydratase in recombinant Escherichia coli using D-glucose. Enzyme Microb Technol 32:27–34
Li F, Wang Y, Gong K, Wang Q, Liang Q, Qi Q (2014) Constitutive expression of RyhB regulates the heme biosynthesis pathway and increases the 5-aminolevulinic acid accumulation in Escherichia coli. FEMS Microbiol Lett 350:209–215
Liu S, Zhang G, Li X, Zhang J (2014) Microbial production and applications of 5-aminolevulinic acid. Appl Microbiol Biotechnol 98:7349–7357
Mauzerall D, Granick S (1956) The occurrence and determination of delta-amino-levulinic acid and porphobilinogen in urine. J Biol Chem 219:446
Mills-Davies N, Butler D, Norton E et al (2017) Structural studies of substrate and product complexes of 5-aminolaevulinic acid dehydratase from humans, Escherichia coli and the hyperthermophile Pyrobaculum calidifontis. Acta Crystallogr D 73:9–21
Mobius K, Arias-Cartin R, Breckau D, Hannig AL, Riedmann K, Biedendieck R (2010) Heme biosynthesis is coupled to electron transport chains for energy generation. Proc Natl Acad Sci USA 107:10436–10441
Na D, Yoo SM, Chung H, Park H, Park JH, Lee SY (2013) Metabolic engineering of Escherichia coli using synthetic small regulatory RNAs. Nat Biotechnol 31:170–174
Nakashima N, Tamura T, Good L (2006) Paired termini stabilize antisense RNAs and enhance conditional gene silencing in Escherichia coli. Nucleic Acids Res 34:e138
Noh MH, Lim HG, Park S, Seo SW, Jung GY (2017) Precise flux redistribution to glyoxylate cycle for 5-aminolevulinic acid production in Escherichia coli. Metab Eng 43:1–8
Peng Q, Warloe T, Berg K, Moan J, Kongshaug M, Giercksky KE (1997) 5-Aminolevulinic acid-based photodynamic therapy. Clin Res Fut Challenges Cancer 79:2282–2308
Sasaki K, Ikeda S, Nishizawa Y, Hayashi M (1987) Production of 5-aminolevulinic acid by photosynthetic bacteria. J Ferment Technol 65:511–515
Sasaki K, Watanabe M, Tanaka T, Tanaka T (2002) Biosynthesis, biotechnological production and applications of 5-aminolevulinic acid. Appl Microbiol Biotechnol 58:23–29
Schon A, Krupp G, Gough S, Berry-Lowe S, Kannangara CG, Soll D (1986) The RNA required in the first step of chlorophyll biosynthesis is a chloroplast glutamate tRNA. Nature 322:281–284
Sorensen MA, Kurland CG, Pedersen S (1989) Codon usage determines translation rate in Escherichia coli. J Mol Biol 207:365–377
Su T, Guo Q, Zheng Y, Liang Q, Wang Q, Qi Q (2019) Fine-tuning of hemB Using CRISPRi for Increasing 5-aminolevulinic acid production in Escherichia coli. Front Microbiol 31(10):1731
Sun T, Li S, Song X, Pei G, Diao J, Cui J, Shi M, Chen L, Zhang W (2018) Re-direction of carbon flux to key precursor malonyl-CoA via artificial small RNAs in photosynthetic Synechocystis sp. PCC 6803. Biotechnol Biofuels 5(11):26
Warnick GR, Burnham BF (1971) Regulation of prophyrin biosynthesis. Purification and characterization of 5-aminolevulinic acid synthase. J Biol Chem 246:6880–6885
Wu J, Yu O, Du G, Zhou J, Chen J (2014) Fine-tuning of the fatty acid pathway by synthetic antisense RNA for enhanced (2S)-naringenin production from l-tyrosine in Escherichia coli. Appl Environ Microbiol 80:7283–7292
Yang Y, Lin Y, Li L, Linhardt RJ, Yan Y (2015) Regulating malonyl-CoA metabolism via synthetic antisense RNAs for enhanced biosynthesis of natural products. Metab Eng 29:217–226
Yang P, Liu W, Cheng X, Wang J, Wang Q, Qi Q (2016) A new strategy for production of 5-aminolevulinic acid in recombinant Corynebacterium glutamicum with high yield. Appl Environ Microbiol 82(9):2709–2717
Yu X, Jin H, Liu W, Wang Q, Qi Q (2015) Engineering Corynebacterium glutamicum to produce 5-aminolevulinic acid from glucose. Microb Cell Fact 14:183
Zhang B, Ye BC (2018) Pathway engineering in Corynebacterium glutamicumS9114 for 5-aminolevulinic acid production. 3 Biotech 8(5):247
Zhang L, Chen J, Chen N, Sun J, Zheng P, Ma Y (2013) Cloning of two 5-aminolevulinic acid synthase isozymes HemA and HemO from Rhodopseudomonas palustris with favorable characteristics for 5-aminolevulinic acid production. Biotechnol Lett 35:763–768
Zhang JL, Kang Z, Chen J, Du GC (2015) Optimization of the heme biosynthesis pathway for the production of 5-aminolevulinic acid in Escherichia coli. Sci Rep 5:8584
Zhang J, Weng H, Ding W, Kang Z (2017) N-terminal engineering of glutamyl-tRNA reductase with positive charge arginine to increase 5-aminolevulinic acid biosynthesis. Bioengineered 8:424–427
Zhang J, Weng H, Zhou Z, Du G, Kang Z (2019) Engineering of multiple modular pathways for high-yield production of 5-aminolevulinic acid in Escherichia coli. Bioresour Technol 274:353–360
Acknowledgements
This study was funded by the Sichuan provincial Science & Technology Department (2018JY0106, 2018JY0310).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors has any financial or personal relationships that could inappropriately influence or bias the content of the research paper. The authors declare that they have no conflict of interest in the publication.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ge, F., Wen, D., Ren, Y. et al. Downregulating of hemB via synthetic antisense RNAs for improving 5-aminolevulinic acid production in Escherichia coli. 3 Biotech 11, 230 (2021). https://doi.org/10.1007/s13205-021-02733-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02733-8