Abstract
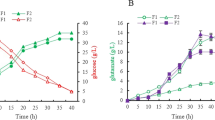
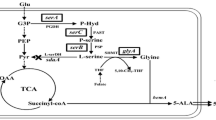
5-Aminolevulinic acid (ALA) is a non-protein amino acid with a significant potential for cancer treatment and plant stress resistance. Microbial fermentation has gradually replaced the traditional chemical-based method for ALA production, thus increasing the need for high-ALA-producing strains. In this study, we engineered the glutamate producing strain, Corynebacterium glutamicum S9114, for ALA production. To efficiently convert l-glutamate to ALA, hemA and hemL from Salmonella typhimurium and Escherichia coli were tandemly overexpressed. In addition, ncgl1221 encoding a glutamate transporter was deleted to block glutamate secretion and thus improve ALA production. Furthermore, the intrinsic ribosome-binding site (RBS) of hemB was replaced by a relatively weak RBS to reduce the conversion of ALA to porphyrin. Transcriptional and fermentation data confirmed that inactivation of lysE and putP reduced the conversion of glutamate to arginine and proline, which also contribute to ALA production. The final SA14 strain produced 895 mg/L concentration of ALA after 72 h incubation in a shake flask. This amount was 58-fold higher than that obtained by the parent strain C. glutamicum S9114. The results demonstrate the potential of C. glutamicum S9114 for efficient ALA production and provide new targets for the development of ALA-producing strains.





Similar content being viewed by others
Abbreviations
- RBS:
-
Ribosome-binding sites
- ALA:
-
5-Aminolevulinic acid
- ALAD:
-
5-Aminolevulinic acid dehydratase
References
Akram NA, Ashraf M (2013) Regulation in plant stress tolerance by a potential plant growth regulator, 5-aminolevulinic acid. J Plant Growth Regul 32:663–679
Becker J, Wittmann C (2016) Industrial Biotechnology: Microorganisms. In: Industrial Microorganisms: Corynebacterium glutamicum, chap 6. Wiley‐VCH Verlag GmbH & Co. KGaA, pp 183–220
Chen M, Chen X, Wan F, Zhang B, Chen J, Xiong Y (2015) Effect of Tween 40 and DtsR1 on l-arginine overproduction in Corynebacterium crenatum. Microb Cell Fact 14:119
Chung S-C, Park J-S, Yun J, Park JH (2017) Improvement of succinate production by release of end-product inhibition in Corynebacterium glutamicum. Metab Eng 40:157–164
Dong X, Zhao Y, Hu J, Li Y, Wang X (2016) Attenuating l-lysine production by deletion of ddh and lysE and their effect on l-threonine and l-isoleucine production in Corynebacterium glutamicum. Enzyme Microb Technol 93–94:70–78. https://doi.org/10.1016/j.enzmictec.2016.07.013
Feng L, Zhang Y, Fu J, Mao Y, Chen T, Zhao X, Wang Z (2015) Metabolic engineering of Corynebacterium glutamicum for efficient production of 5-aminolevulinic acid. Biotechnol Bioeng 113:1284–1293
Fu W, Lin J, Cen P (2007) 5-Aminolevulinate production with recombinant Escherichia coli using a rare codon optimizer host strain. Appl Microbiol Biotechnol 75:777–782
Heider SA, Peters-Wendisch P, Wendisch VF (2012) Carotenoid biosynthesis and overproduction in Corynebacterium glutamicum. BMC Microbiol 12:198. https://doi.org/10.1186/1471-2180-12-198
Huang J, Wu Y, Wu W, Zhang Y, Liu D, Chen Z (2017) Cofactor recycling for co-production of 1, 3-propanediol and glutamate by metabolically engineered Corynebacterium glutamicum. Sci Rep 7:42246
Ishikawa T, Kajimoto Y, Inoue Y, Ikegami Y, Kuroiwa T (2015) Chapter seven-critical role of ABCG2 in ALA-photodynamic diagnosis and therapy of human brain tumor. Adv Cancer Res 125:197–216
Jakoby M, Ngouoto-Nkili C-E, Burkovski A (1999) Construction and application of new Corynebacterium glutamicum vectors. Biotechnol Tech 13:437–441
Jo S, Yoon J, Lee S-M, Um Y, Han SO, Woo HM (2017) Modular pathway engineering of Corynebacterium glutamicum to improve xylose utilization and succinate production. J Biotechnol 258:69–78
Kang Z, Wang Y, Gu P, Wang Q, Qi Q (2011) Engineering Escherichia coli for efficient production of 5-aminolevulinic acid from glucose. Metab Eng 13:492–498
Kang M-K, Eom J-H, Kim Y, Um Y, Woo HM (2014) Biosynthesis of pinene from glucose using metabolically-engineered Corynebacterium glutamicum. Biotechnol Lett 36:2069–2077
Kennedy JC, Pottier RH, Pross DC (1990) Photodynamic therapy with endogenous protoporphyrin IX: basic principles and present clinical experience. J Photochem Photobiol B 6:143–148
Lubitz D, Jorge JM, Pérez-García F, Taniguchi H, Wendisch VF (2016) Roles of export genes cgmA and lysE for the production of l-arginine and l-citrulline by Corynebacterium glutamicum. Appl Microbiol Biotechnol 100:8465–8474
Lv Y, Wu Z, Han S, Lin Y, Zheng S (2011) Genome sequence of Corynebacterium glutamicum S9114, a strain for industrial production of glutamate. J Bacteriol 193:6096–6097. https://doi.org/10.1128/JB.06074-11
Mei J, Xu N, Ye C, Liu L, Wu J (2016) Reconstruction and analysis of a genome-scale metabolic network of Corynebacterium glutamicum S9114. Gene 575:615–622
Mimitsuka T, Sawai H, Hatsu M, Yamada K (2007) Metabolic engineering of Corynebacterium glutamicum for cadaverine fermentation. Biosci Biotechnol Biochem 71:2130–2135. https://doi.org/10.1271/bbb.60699
Nguyen AQ, Schneider J, Reddy GK, Wendisch VF (2015) Fermentative production of the diamine putrescine: system metabolic engineering of Corynebacterium glutamicum. Metabolites 5:211–231
Nowroozi FF, Baidoo EE, Ermakov S, Redding-Johanson AM, Batth TS, Petzold CJ, Keasling JD (2014) Metabolic pathway optimization using ribosome binding site variants and combinatorial gene assembly. Appl Microbiol Biotechnol 98:1567–1581
Park SH, Kim HU, Kim TY, Park JS, Kim S-S, Lee SY (2014) Metabolic engineering of Corynebacterium glutamicum for l-arginine production. Nat Commun 5:4618
Peter H, Bader A, Burkovski A, Lambert C, Krämer R (1997) Isolation of the putP gene of Corynebacterium glutamicum and characterization of a low-affinity uptake system for compatible solutes. Arch Microbiol 168:143–151
Ramzi AB, Hyeon JE, Kim SW, Park C, Han SO (2015) 5-Aminolevulinic acid production in engineered Corynebacterium glutamicum via C5 biosynthesis pathway. Enzyme Microb Technol 81:1–7. https://doi.org/10.1016/j.enzmictec.2015.07.004
Sasaki K, Watanabe M, Tanaka T (2002) Biosynthesis, biotechnological production and applications of 5-aminolevulinic acid. Appl Microbiol Biotechnol 58:23–29
Sassa S (1982) Delta-aminolevulinic acid dehydratase assay. Enzyme 28:133–145
Schafer A, Tauch A, Jager W, Kalinowski J, Thierbach G, Puhler A (1994) Small mobilizable multi-purpose cloning vectors derived from the Escherichia coli plasmids pK18 and pK19: selection of defined deletions in the chromosome of Corynebacterium glutamicum. Gene 145:69–73
Siebert D, Wendisch VF (2015) Metabolic pathway engineering for production of 1, 2-propanediol and 1-propanol by Corynebacterium glutamicum. Biotechnol Biofuels 8:91
Tetard M-C et al (2016) Interstitial 5-ALA photodynamic therapy and glioblastoma: preclinical model development and preliminary results. Photodiagn Photodyn Ther 13:218–224
Tian T, Salis HM (2015) A predictive biophysical model of translational coupling to coordinate and control protein expression in bacterial operons. Nucleic Acids Res 43:7137–7151
Wang J et al (2015) Fine-tuning of ecaA and pepc gene expression increases succinic acid production in Escherichia coli. Appl Microbiol Biotechnol 99:8575–8586
Wieschalka S, Blombach B, Bott M, Eikmanns BJ (2013) Bio-based production of organic acids with Corynebacterium glutamicum. Microb Biotechnol 6:87–102
Xu J, Xia X, Zhang J, Guo Y, Qian H, Zhang W (2014) A method for gene amplification and simultaneous deletion in Corynebacterium glutamicum genome without any genetic markers. Plasmid 72:9–17
Yang P, Liu W, Cheng X, Wang J, Wang Q, Qi Q (2016) A new strategy for production of 5-aminolevulinic acid in recombinant Corynebacterium glutamicum with high yield. Appl Environ Microbiol 82:2709–2717
Yao LL, Ye BC (2015) Reciprocal regulation of GlnR and PhoP in response to nitrogen and phosphate limitations in Saccharopolyspora erythraea. Appl Environ Microbiol 82:409–420. https://doi.org/10.1128/AEM.02960-15
Yu X, Jin H, Liu W, Wang Q, Qi Q (2015) Engineering Corynebacterium glutamicum to produce 5-aminolevulinic acid from glucose. Microb Cell Fact 14:183
Yu X, Jin H, Cheng X, Wang Q, Qi Q (2016) Transcriptomic analysis for elucidating the physiological effects of 5-aminolevulinic acid accumulation on Corynebacterium glutamicum. Microbiol Res 192:292–299
Zhang C, Shi Z, Gao P, Duan Z, Mao Z (2005) On-line prediction of products concentrations in glutamate fermentation using metabolic network model and linear programming. Biochem Eng J 25:99–108
ZHANG B, Fang W, QIU YL, CHEN XL, Li T, CHEN JC, XIONG YH (2015a) Increased l-arginine production by site-directed mutagenesis of N-acetyl-l-glutamate kinase and proB gene deletion in Corynebacterium crenatum. Biomed Environ Sci 28:864–874
Zhang B, Zhou N, Liu Y-M, Liu C, Lou C-B, Jiang C-Y, Liu S-J (2015b) Ribosome binding site libraries and pathway modules for shikimic acid synthesis with Corynebacterium glutamicum. Microb Cell Fact 14:71
Zhang J, Kang Z, Chen J, Du G (2015c) Optimization of the heme biosynthesis pathway for the production of 5-aminolevulinic acid in Escherichia coli. Sci Rep 5:8584. https://doi.org/10.1038/srep08584
Zhang B, Ren L-Q, Yu M, Zhou Y, Ye B-C (2017a) Enhanced l-ornithine production by systematic manipulation of l-ornithine metabolism in engineered Corynebacterium glutamicum S9114. Biores Technol. https://doi.org/10.1016/j.biortech.2017.11.017
Zhang B, Yu M, Zhou Y, Li Y, Ye B (2017b) Systematic pathway engineering of Corynebacterium glutamicum S9114 for l-ornithine production. Microb Cell Fact 16:158
Acknowledgements
We thank Dr. Andreas Burkovski and Dr. Xue-Lan Chen for providing pXMJ19 and pk18mobsacB. Moreover, we thank Dr. Zhong-Gui Mao and Dr. Li-Ming Liu for providing C. glutamicum S9114 strains. This study was supported by China NSF 21575089 and 21335003 and the National Key Technologies R&D Programs (2014AA021502).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declared that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, B., Ye, BC. Pathway engineering in Corynebacterium glutamicum S9114 for 5-aminolevulinic acid production. 3 Biotech 8, 247 (2018). https://doi.org/10.1007/s13205-018-1267-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-018-1267-2