Abstract
The efficient utilization of all of the key lignocellulosic biomass residues is important for the development of an integrated biorefineries uassing this as its feedstock. In this study, a process involving a simultaneous pre-treatement process with anaerobic digestion (AD) were implemented for processing oil palm empty fruit bunches (OPEFB) in order to produced both vanillin and methane. Before and after treatment with the dry rot fungus (Serpula lacrymans) the biomass was analyzed for total reducing sugar (TRS), total soluble phenols (TSP), pH, and weighed. Change in the physical characteristic and morphology of untreated and OPEFB following fungal culture was observed using scanning electron microscopy (SEM). The biomethane potential (BMP) of pretreated OPEFB significantly increased (62.07%) compared to untreated sample and in addition. The amount of vanillin extracted reached 3.48ug mL−1. This study therefore confirms that multiple products can effectively be isolated using a fungal preatreatment of lignocellulosic OPEFB followed by anerobic fermentation of biomass residual thus maximizing the potential returns and reducing environmentally impacts compared to other pretreatments.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Introduction
Lignocellulosic biomass is an abundant and renewable resource mostly composed of polysaccharides and lignin [1, 2]. Indonesia is one of the largest palm oil producers countries in the world [3, 4]. It produced 37.97 million tons of crude palm oil (CPO) in 2017 which increased to 42.87 million tons in 2019 [5]. The fruit bunches consists of 21.8% oil, 6% kernel, 14.3% mesocarp, 7% shell, and 22.5% oil palm empty fruit bunches (OPEFB) [6, 7]. Accumulation of OPEFB can have a negative impact on the environment if this is not suitably managed [5, 8, 9]. OPEFB could be utilized as a source of renewable energy [8] and value added chemicals production. Its benefits include the fact that it is produced throughout the year and as it is considered a waste has a low cost and carbon footprint [10] OPEFB lignocellulose is comprised of cellulose (24–65%), hemicellulose (21–34%) and lignin (14–31%) [9, 11]. Due to the complexity and recalcitrance of its lignocellulose, to fully utilize the biomass, pretreatment is necessary to make the constitutive polymers more accessible [12, 13] through changing both the physical and chemical structure. Biological pretreatment utilising enzymes [14] is considered to be safer, less energy consuming and more environmentally friendly than other forms of pretreatment [14,15,16,17]. The use of lignin degrading microbes also shows promise as the microorganisms can convert the lignocellulosic polymers into simple compounds through enzymatic activity in both aerobic and anaerobic conditions [18]. A fungal pretreatment of 5 weeks at 30 °C was shown to be capable of simultaneously degrading the recalcitrant interlinked structure of lignin, cellulose, and hemicellulose [19]. The brown rot fungus Serpula lacrymans has been identified as one of fungi most capable of modifying lignin [20,21,22]. It is thought to act through a combination of enzymes and the Fenton reaction to produce hydroxyl radicals capable of depolymerizing lignin [23, 24]. The activity of such microorganisms can release various economically important chemicals compounds including sugars, organic acids, and phenolic derivatives [25]. The products that can be generated from lignin include aromatic aldehydes (vanillin, p-hydroxybenzaldehyde, dan syringaldehyde) and hydroxycinnamic or phenolic acids (vanillic acid, ferulic acid, and p-coumaric acid [26, 27]. Vanillin can be produced through bioconversion using microbes from ferulic acid, eugenol, isoeugenol [28, 29], guaiacol [30], and lignin [26, 30, 31]. The use of lignin for vanillin production is more environmentally friendly compare to guaiacol [30]. However, due to the complexity structure of lignin, the separation of vanillin from other lignin degradation products is still to be effectively resolved [32] although organic solvents such as ethyl acetate can separate bioactive compound such as vanillin [33, 34].
The biorefinery concept envisages the production of value added products and energy from biomass feedstock [35]. The concept being the complete utilization of all lignocellulosic components and other plant components to produce various biobased products and to convert the low value remaining elements to energy if no other more valuable use can be found [36]. The efficient use of all such lignocellulose biomass is important if a sustainable biorefinery model is to be developed [37]. Combining different processes can increase the efficacy of the entire process through reducing energy consumption and the environmental impacts [38, 39]. In this study, fungal treatment of lignocellulosic OPEFB was used to release lignin components and the remaining residues were tested for their potential for biogas production via anaerobic digestion (AD), which is recognised as one of the most sustainable means to remove organic matter from the waste streams [40] through converting residual biomass into renewable energy [41]. Various studies have reported that biomass pretreatments can enhance AD performance. Previously it has been shown that the pretreatment of OPEFB with Trichoderma reesei resulted in increased methane yields and a 60–75% higher biodegradation efficiency without any additional energy input or undesired by-product inhibition [42].
The objective of this study was to investigate the production of vanillin as an example of a value-added chemicals produced from degradation of lignocellulosic OPEFB and to demonstrate that the residue following its extraction the waste biomass can be utilized as a substrate for biogas production using as measured using Biochemical Methane Potential (BMP) test. Therefore, it demonstrates the implementation of an integrated biorefinery system could be implemented through the sequential bioconversion of OPEFB.
Materials and Methods
Feedstock, Fungal Strain, and Inoculum
The OPEFB was obtained from PT Sawit Madani, Blitar, Indonesia. OPEFB was dried in the sun for 48 h and chopped (0.5–1.0 cm), then put 10 g in honey jars and 10 mL of distilled water was added. The samples were then double sterilized at 121 °C for 20 min each time to ensure no other microbes were present. The pure culture of S. lacrymans was supplied by Laboratory Bioindustry, Department Agroindustry, Universitas Brawijaya and maintained in malt extract agar (MEA) at 22 ± 2 °C. Agar plugs with mycelia were added to barley grain to produce inoculum (grain spawn). Anerobic digestate for use as this inoculum was obtained from anaerobic digestion plant at Balai Besar Penelitian Peternakan (BBPP), Batu City, Indonesia. The inoculum was filtered through a 60-mesh sifter to separate dirt or large particles. The filtrate then was degassed for 48 h at 37 °C before being used for BMP test.
Pretreatment and Aqueous Extraction
The sterilized of OPEFB jars were inoculated with 0.2 g S. lacrymans grain spawn of and incubated at 22 ± 2 °C for 42 days. The aqueous extraction was conducted to determine the amount of total reducing sugar and total soluble phenols. The incubated samples were analyzed for sugar and phenols released at every 7 days. For aqueous extraction, 100 mL of distilled water was heated to 80 °C, then added to each inoculated sample and shaken at 150 rpm for 1 h. The jars were subsequently emptied separately into fine muslin netting and the extract sample was filtered again using Whatman filter paper No.1. The filtrate was used to measure total reducing sugars (TRS) and total soluble phenols (TSP), while the residue was subsequently used for vanillin analysis and as the substrate for BMP testing.
Physical and Chemical Characterization
Physical and chemical characterization was carried out on non-treated and fungal treated samples of OPEFB. The physical analysis measured water content, ash, total solid (TS) and volatile solid (VS) based on the NREL (National Renewable Energy Laboratory) method [43,44,45]. The chemical analysis included lignin and cellulose analysis, proximate analysis, elemental analysis (CHNS) and calorific value (CV).
Lignin and cellulose content were measured every 7 days during the 42 days of cultivation. This was carried out according to Anderson and Ingram [46], using the Tropical Soil, Biology and Fertility (TSBF) Program method. Elemental analysis was calculated to estimate the methane production potential according to the the Buswell equation [47].
Total Soluble Phenol (TSP), Total Reducing Sugar (TRS), pH, and Weight Loss Analysis
Total soluble phenols (TSP), total reducing sugars (TRS), pH and weight loss were also measured every 7 days over the 42 days period. Both TSP and TRS was carried out using UV–Vis spectrophotometer (Thermo Scientific type Genesys 10 UV). TSP measurement was conducted according to Singleton and Rossi [48] and used Folin-Ciocalteu reagent, while the TRS method refers to Miller [49] and uses 3,5-Dinitrosalicylic acid (DNS). pH was measured using a digital pH meter, calibrated using pH standard solution at 4 and 7 [50] and weight loss calculated using the oven drying method [51]. Each sample was analysed in triplicate.
Scanning Electron Microscope (SEM) Analysis
The morphology of non-treated and pretreated OPEFB samples were compared using scanning electron microscope (a NanoSEM-FEI Nova 200, type Inspect-S50, Hillsboro, Oregon, USA) analysis using a range of magnifications. The OPEFB samples were fixed to a carbon tip and placed on a sputter coater (mini sputter coater EMITECH type SC7620, Laughton, East Sussex, UK) for the vacuum coating process to prepare the samples for SEM.
Vanillin Identification
Vanillin extraction was carried out using 100 mL ethyl acetate. The solid cake from aqueous extraction was weighed and placed into a conical flask and heated in a shaking water-bath at 30 °C; 150 rpm for 2 h. The residue was then filtered and centrifuged at 13,000 g at 4 °C for 10 min. The filtrate was evaporated using a rotary evaporator (RV 10 control, IKA, Goteborg, Sweden) at 40 °C, 100 rpm. LC–MS/MS (Thermo Scientific Triple Quadrupole MS TSQ Quantum Access Max) analysis was carried out according to Deepa and Dhepe [52] and Tešević et al. [53] using a Hypersil Gold C18 column (2.1 mm × 50 mm × 1.9 m) to determine the vanillin content quantitatively by comparison to a standard. The column heater was set at 30 °C with 0.1% formic acid in water and 0.1% formic acid in acetonitrile (ACN) as the mobile phases. The mobile phase flow rate was maintained at 0.5 mL/min and the injection volume was 1μL. The retention time used was 0–5.5 min. The column was controlled at 30ºC, and the autosampler compartment was set to 16ºC.
Biochemical Methane Potentials (BMP) Test
The estimated methane produced during the experiment was measured using the BMP test [54] to study the methane formation potential in OPEFB residue after extraction for vanillin analysis. BMP test was carried out over 28 days at 37 °C in a water bath. 140 mL bottles with a working volume of 40 mL were used. Blank control samples (inoculum only), positive control samples (α-cellulose and inoculum), sample A (OPEFB non-treated and inoculum), and sample B (pre-treated OPEFB and inoculum) were used for the assay. All the samples were carried out in triplicate. Samples A and B in the form of a mixture of residual and inoculum samples were added to each reactor with a 6:1 ratio of inoculum to substrate (I/S ratio) based on the VS. Gas pressure was measured daily using a Digitron 2026P (Electron Technology, UK). The methane potential is the amount of methane produced on a daily basis. It was calculated using the following equation:
where, BMP is the normalization methane volume (m3 CH4/kg VS); VS is the mean value of accumulated methane volume from reactor (with substrate and inoculum); VB is mean value of methane volume from the reactor with inoculum only (blank sample); mIS is the mass of VS of inoculum added in the sample; mB is the mass of VS of inoculum added in the blank sample; and mVS,sS is the mass of substrate added in the reactor.
The cumulative biogas volume, cumulative methane volume, and specific methane potential were subsequently calculated. Cumulative biogas volume was obtained from the accumulated results of pressure measurements per day to obtain the average gas pressure. Calculation of cumulative biogas volume based on the equation as follows [55]:
where P is bottle headspace pressure (kPa); Vol is bottle headspace volume (mL); Vm is molar volume of ideal gas (22.414 Lmol−1); R is ideal gas constant (8.314m3 Pa K−1 mol−1); and T is incubation temperature (K).
The methane concentration used to calculate specific methane potential (SMP) was the value obtained from calculation using the following Buswell equation [47] which based on the elemental analysis:
where C, H, O, N, S is the fraction weight or percentage of the element weight based on VS; and c, h, o, n, s are the moles of each element. The theoretical methane was used as an assumption of methane percentage in biogas to determine the daily methane volume where the assumption that biogas only consists of methane (CH4) and carbon dioxide (CO2). Biogas and methane potentials or called specific biogas potential (SBP) and specific methane potential (SMP), indicated the volume of biogas and methane per gram of organic matter. The SBP and SMP values can be calculated using Eq. (1) based on Strömberg et al. [56].
Stastistical Analysis
The result data was analyzed by one-way analysis of variance (ANOVA) using SPSS version 16.0 software. The significance test and level of treatment were tested using a Duncan Multiple Range Test (DMRT). Mean and standard deviation (error bars) were calculated using Microsoft Excel software. The tests were carried out in three replicates with a confidence level of 95%. The Cronbach's alpha reliability test was performed using SPSS version 16.0 to evaluate the reproducibility of the BMP experiment in this study. The acceptable values of Cronbach’s alpha range from 0.70 to 0.95 [57].
Results and Discussions
Characteristics of OPEFB Before and After Pretreatment
The characteristic of OPEFB before and after pretreatment was determined using proximate analysis in which water content, ash content, total solid (TS), volatile solid (VS) and other parameters were measured (Table 1). The water content of OPEFB slightly increased after pretreatment. According to Aghdam et al. [58], a high water content in the substrate can increase methane production as the high water content of substrate can support the movement of microbes, dissolve nutrients for the assimilation of microorganism, dilute inhibitory compounds and limit oxygen transport. The ash content of OPEFB before pretreatment and in the residue were 0.25% and 0.26% respectively. A higher ash content of biomass indicates a low level of substrate digestibility because biomass contains non-volatile parts.
TS and VS values decreased in pretreated OPEFB, this is presumably due to the degradation in the OPEFB following fungal culture as it has initiated breakdown of the complex structure and therefore more organic components are released [59,60,61]. The value of fat, protein and carbohydrates are indicators of the presence of precursors of inhibitory compounds, such as long chain fatty acids and ammonia [62]. The amount of carbohydrate in both non-treated and the pretreated samples were 77.21% and 79.93% respectively. While the number of proteins were 4.35% and 6.89% and fat contents were 9.71% and 3.52%. According to Ohemeng-Ntiamoah and Datta [63], samples which contain high amount of fats (> 60%) produced more biogas than samples with higher protein and carbohydrate. However, samples with high amount of carbohydrate (> 60%) have a faster biogas production rate. Samples which contain high amount of fat and protein in the substrate can inhibit the production of biogas.
The calorific value test aims to determine the amount of energy produced per unit mass on the OPEFB as a substrate. In this study the value of CV in both samples were 11.88 and 9.71 MJ kg-1TS. Theoretically the calorific value was obtained from a calculation using the Boie formula (Table 1). The theoretically value of CV obtained is in accordance with a previous study by Solarte-Toro et al. [64], where the CV of its lignocellulosic biomass ranged between 17-20 MJ/kg. According to Ilminnafik et al. [65], a high calorific value will result in the high production of methane gas (CH4), while a low calorific value will lead to higher carbon dioxide (CO2) production.
C/N ratio is an indicator of the potential to produce methane gas by microorganisms. The C/N ratio of non-treated samples of OPEFB was higher than pretreated OPEFB (Table 1). The optimum C/N ratio for AD process is between 20–30 [66]. If C/N ratio is above 30, methanogenic bacteria will consume more nitrogen to meet their need to produce protein, therefore it will react with carbon in substrate [67]. However, if carbon content is too high, then the degradation process will take longer [68], and this will result in lower biogas production. Therefore, in most biogas production facilities which use lignocellulosic biomass, livestock manure is mixed with the biomass to produce allowing the ideal C/N ratio.
Evaluation of Lignocellulose Degradation From OPEFB
The breakdown of OPEFB lignocellulose by the fungus S. lacrymans is indicated by a change in specific parameters, including increases in total soluble phenol (TSP), total reducing sugars (TRS), pH, and weight loss over 42 days (Fig. 1). The increased availability of organic compounds resulting from lignocellulose degradation will also stimulate fungal growth which results in the absorption of some of the lignocellulose breakdown products by the fungus [69,70,71]. The quantification of phenols released from the substrate during cultivation is therefore one of the strongest indicators of breakdown by the fungus as these are not reabsorbed. The value of TSP was observed to increase during the period of cultivation, the highest levels of phenolic compound release being seen after 35 days (0.195 mg g−1) with these levels subsequently falling. This decreases is most likely due to the participation of these phenolic compounds as reducing agent in Fenton reaction [72], which helps to depolymerize lignin structure. The brown rot fungi are known to produce oxalic acid during the fungal growth which then dissolved iron and initiates the Fenton reaction [73, 74]. S. lacrymans has been shown to be capable of depolymerized lignin through Fenton reaction [22, 75, 76].
It has previously been reported that the change to the lignocellulose structure can be caused by the Basidiomycota and that this dominates the earlier stages of wood decomposition [77]. In this study, the average detectable total reducing sugar fluctuated during the period of fungal growth, this ranged from 14.289 mg g−1 to 27.938 mg g−1. The highest TRS was detected at 7 days incubation (27.938 mg g−1), and susbsequently dropped during the later incubation period of 7–21 days. The increase in TRS during early incubation is likely related to fungal enzyme activity, as it coincides with the breakdown of cellulose and hemicellulose [78, 79]. However the fungus also needs carbon sources to grow and the consumption of these resources will affect the detectable sugar as the fungus grows [80, 81] and therefore the sugar values fluctuate. Previous studies using either chemical or physicals pretreatments resulted in reducing sugars yields ranging from 388.26–728.45 mg g−1 [82] however in a previous study, using S. lacrymans grown on corn leaves it peaked at 207.37 mg g−1. The low TRS released from OPEFB treated by S. lacrymans was probably due to a relatively small amount of lignin breakdown, which consequently restricted access by the enzymes required to degrade the cellulose.pH is also be used as an indicator for lignocellulose breakdown by fungus. In this study, the of pH value decreased during the first two weeks incubation, dropping from 7.65 to 6.65 (Fig. 1). The decrease of pH is likely due to the production of organic acid released during fungal growth over the first two weeks of incubation [83]. After two weeks the pH then increased possibly as a consequence of an increase of ammonium alkali produced [84].
The measurement of biomass sample before and after the fungal treatment allowed the change of weight loss to be determined [85]. Weight loss is a useful parameter to determine the effectiveness of any pretreatment method [86] as weight loss of lignocellulosic biomass can also indicate the extent of lignin-carbohydrate degradation [16]. In this study, the rate of weight loss increased during S. lacrymans cultivation (Fig. 1). The weight loss detected following S. lacrymans cultivation ranged up to 12.35% (Fig. 1), the longer the cultivation period, the greater the percentage of weight loss obtained. This indicates that a greater breakdown of the lignocellulose structure is achieved using fungus and corresponds with Zhang et al. [87].
Pretreatment can enhance the digestibility of lignocellulosic substrates by reducing cellulose crystallinity, thus increasing the porosity of substrate and its ability to release value added compounds [88]. The amount of cellulose and lignin obtained from OPEFB inoculated with S. lacrymans for 42 days culture is shown in Fig. 2. The cellulose percentage following S. lacrymans cultivation dropped from 35.58% to 28.08% and corelates with the change in total reducing sugar (Fig. 1). Previous study have shown some fungi are able to degrade lignin effectively, but prefer to consume cellulose [16].
Lignin content is one of the key factors that controls the gas produced during AD of lignocellulosic biomass. It appears to be important to breakdown lignin in order to aid the release of sugars from both the cellulose and hemicellulose component of the cell walls. A decreasing trend in the amount of lignin detectable was observed during S. lacrymans cultivation (Fig. 2), as a result of the modification of the lignocellulose and its lignin by the fungus [17]. The lignin content in OPEFB is normally high which acts as a shield for cellulose and hemicellulose, and therefore OPEFB cannot be easily to be converted to biogas.
Microscopic Analysis (Scanning Electronic Microscopy)
Scanning electronic microscopy (SEM) of OPEFB before and after pretreatment indicates structural changes in OPEFB biomass (Fig. 3). Biological pretreatment of OPEFB using S. lacrymans caused significant changes in the surface structure of OPEFB. The non-treated OPEFB structure shows that the surface is normally rigid, dense, and well structured (Fig. 3a). This is supported by Razali et al. [6] which stated that the initial OPEFB before pretreatment had a hard, coarse texture and pores were filled with silica. The surface structure of OPEFB after pretreatment shows damage, the surface is cracked, and the structure is stretched when it is compared to samples without pretreatment (Fig. 3b).
The changes in the OPEFB indicates that the biological pretreatment with this fungus is effective and contributes to modification of the lignocellulosic structure. Kamcharoen et al. [16], showed that during Trametes versicolor cultivation, mycelium covered and penetrated into OPEFB surface which led to greater porosity and roughness than OPEFB without pretreatment and indicated that cellulose digestibility of the pretreated sample was higher than untreated. Singh et al. [89], showed pores and cracks caused by the weakening of cell wall during delignification which led to increase dissolution efficiency of cellulose and increased yield of reducing sugars.
The Identification of Vanillin Using LC–MS/MS
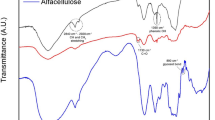
Vanillin is a key component of natural vanillin extract, and is used as a flavoring agent in food, beverages, and medicines [90]. In this study, the release of vanillin as a derivative of lignin breakdown from OPEFB cultured with S. lacrymans was monitored using LC–MS/MS. The total ion chromatogram (TIC) can be seen in Fig. 4. The identification of vanillin was based on its molecular weight (152.149 g mol−1) and comparison to industrial standards. The main peak of vanillin was identified along with several other minor lignin derivative products were detcted. This is in accordance with the study conducted by Shen et al. [91] that also used a molecular weight of vanillin 153 g mol−1. Tešević et al. [53], carried out a full scan mass spectrum of aldehydes for vanillin with the mass spectrometer set to monitor the transition of the precursor to product ion m/z 153/93. The qualitative analysis on this study showed that vanillin was detected in OPEFB extracted samples with retention time of 2.27 min out of a total retention of 5.5 min (Fig. 4a, b). This indicates that during pretreatment process using S. lacrymans, the metabolite products were formed due to biotransformation of phenolic components into vanillin.
In this study, vanillin production was assayed quantitatively using a standard curve based on vanillin solution as a comparison. The amount of vanillin released was calculated based on a peak area of vanillin standard. Based on the LC–MS/MS result, vanillin yield obtained was 3.48ug mL−1. This result is much lower compared to Harshvardhan et al. [31], which used lignin degrader bacteria to degrade 100 g bamboo. The natural consortium of bacteria can directly convert biomass to vanillin, which is comparatively equivalent to ferulic acid. However, the results of this study are not much different when it is compared to Taira et al.[92], that used Aspergillus luchuensis to rice koji fermentation. The vanillin obtained from Taira et al.[92] was maximally about 3 g mL−1 but decreased to around 2 g mL−1 after 15 days of cultivation. This evidence revealed that vanillin yield produced from bioconversion process depends on microorganism type. According to Messaoudi et al. [93], vanillin is formed in small quantities, that known as precursors in lignin biosynthesis and degradation by some rot fungi.
BMP Measurements
Theoretically, the biogas component can be determined by analyzing the C, H, O, and N content of untreated and pretreated OPEFB samples. The theoretical methane potential of non-treated and pretreated samples of OPEFB sample was calculated based on the Buswell equation [47]. The theoretical methane potential value in non-treated and pretreated OPEFB samples was 50.81% and 51.66%, respectively. This study was assumed that the methane content in all treatments obtained the same methane percentage as the pretreated OPEFB (51.66%).
Specific biogas and methane potential (SBP and SMP) are shown in Fig. 5a and b. The highest SBP and SMP were obtained from α-cellulose sample as a positive control which showed an increase in the cumulative biogas and methane volume as well as the specific biogas and methane potential during fungal cultivation for 28 days (Fig. 5b). SBP and SMP from non-treated samples were lower than pretreated OPEFB samples. This result indicated that pretreatment causes an increase in the value of SBP and SMP which is thought to be due to an increase in VS value of the substrate used. The presence of pretreatment can increase the depolymerization of lignin and accelerate the anaerobic digestion process for biogas production [59]. The accessibility of cellulose and hemicellulose by fungus is due to the loss of some lignin structure, which enables microorganisms to convert the substrate to produce methane [94].
The average of SBP in non-treated and pretreated OPEFB was 0.058m3 kg−1 VS and 0.091m3 kg−1 VS. While the average of SMP in non-treated and pretreated OPEFB was 0.029 m3 kg−1 VS and 0.047 m3 kg−1 VS. Thus, the increase in SBP and SMP of non-treated to pretreated OPEFB were 56.89% and 62.07%, respectively. This study showed that both SBP and SMP average of non-treated OPEFB was lower than pretreated OPEFB sample (Fig. 6a and b), which is presumably because parts of cellulose and hemicellulose structures in non-pretreated samples are still bound with lignin. This is highly recalcitrant and therefore limits methane production. Lignin has a complex structure that can limit biomass conversion to biogas by the AD system. Furthermore, microbial activity at all stages of the AD process depends on substrate type, particle size, surface area, and type of AD system applied [95]. The reduction of substrate particle size can increase the lignocellulosic degradation, while the larger particle size can reduce methane production and make it difficult for microbes to degrade the organic compounds.
The reliability test showed that all tested samples have Cronbach's alpha values higher than 0.7. Cronbach's alpha for SBP and SMP were 0.866, and 0.867, respectively. This showed that the reproducibility and consistency of BMP experiment were acceptable and reliable, as stated by Adadana and Savasci [96].
Mass Balance
The overall mass balance in the sequential bioconversion process of lignocellulosic OPEFB for the generation of vanillin and biomethane shows the total lignocellulosic content, vanillin yield, and methane potential produced (Fig. 7). The biological pretreatment of OPEFB using S. lacrymans removed 25.48% of initial lignin following 42 days of cultivation, 19.4% of total solids content, and the vanillin yield obtained was 3.48 µg g−1. While for the BMP test, the results after 28 days showed a biogas production of 15.92 mL, and also biomethane and SMP were 8.22 mL and 0.077 m3 kg−1 VS respectively. Where the results of biogas volume, biomethane volume and SMP for non-treated OPEFB were 12.79 mL, 6.50 mL, and 0.059 m3 kg−1 VS, respectively.
Lignin removal was lower than study by Angelo et al. [97], where during OPEFB pretreatment using white rot fungus Pleurotus ostreatus for 60 days, the highest lignin removal was 30.72%, obtained on 30 days pretreatment. BMP test results, compared to non-treated OPEFB, showed an increase in biogas volume, methane volume, and SMP were 24.47%, 26.46% and 30.51%, respectively. This result is lower than research by Suksong et al. [42] where after pretreatment using Trichoderma reesei and Pleurotus ostreatus, methane yields increased by 44–52% compared to raw OPEFB.
Conclusion
S. lacrymans was shown to be capable of modifying OPEFB to increase the release of biomethane from residual biomass following vanillin extraction in agreement with previous studies utilising dry rot fungi. The degree and selectivity of delignification following the fungal treatment of OPEFB was improved. This was reflected in all parameters used to measure lignocellulose breakdown such as the value of TSP, TRS, weight loss, cellulose, and lignin. The depolymerization of lignin from OPEFB resulted in the production of phenolic compounds as revealed by the detection of vanillin. Following subsequent extraction, vanillin yields of 3.48ug mL−1 was obtained. This results clearly show that fungal treatment with S. lacrymans also enhanced its specific biomethane potential reflecting an improvement in the residues potential for conversion into biogas. The solvent extraction of vanillin carried out did not significantly impair the subsequent methane production. The advantages of using fungal pretreatment followed by solvent extraction of high value prodcuts before susbsequent use of the residues is clearly indicated. The results suggest that further development of the approach could lead to improved treatment of OPEFB reducing the environmental consequences of its accumulation.
Data Availability
Data can be made available upon reasonable request.
References
Vu, H.P., Nguyen, L.N., Vu, M.T., Johir, M.A.H., McLaughlan, R., Nghiem, L.D.: A comprehensive review on the framework to valorise lignocellulosic biomass as biorefinery feedstocks. Sci. Total Environ. 743, 140630 (2020). https://doi.org/10.1016/j.scitotenv.2020.140630
Kaparaju, P., Serrano, M., Thomsen, A.B., Kongjan, P., Angelidaki, I.: Bioethanol, biohydrogen and biogas production from wheat straw in a biorefinery concept. Bioresour. Technol. 100, 2562–2568 (2009). https://doi.org/10.1016/j.biortech.2008.11.011
Ministry of Agriculture: Outlook Kelapa Sawit Komoditas Pertanian Subsektor Perkebunan. Pusat Data dan Sistem Informasi Pertanian Sekretariat Jenderal - Kementerian Pertanian, Jakarta (2016)
Isroi, M.I., Millati, R., Syamsiah, S., Cahyanto, M.N., Niklasson, C., Taherzadeh, M.J.: Structural changes of oil palm empty fruit bunch (OPEFB) after fungal and phosphoric acid pretreatment. Molecules 17, 14995–15012 (2012). https://doi.org/10.3390/molecules171214995
Ministry of Agriculture: Produksi Kelapa Sawit Menurut Provinsi di Indonesia, 2015 - 2019. , Jakarta (2019)
Razali, W.A.W., Baharuddin, A.S., TarmezeeTalib, A., Sulaiman, A., Naim, M.N., Hassan, M.A., Shirai, Y.: Degradation of oil palm empty fruit bunches (OPEFB) fibre during composting process using in-vessel composter. BioResources 7, 4786–4805 (2012). https://doi.org/10.15376/biores.7.4.4786-4805
Omar, R., Idris, A., Yunus, R., Khalid, K., Isma, M.I.A.: Characterization of empty fruit bunch for microwave-assisted pyrolysis. Fuel 90, 1536–1544 (2011). https://doi.org/10.1016/j.fuel.2011.01.023
Santi, L.P., Kalbuadi, D.N., Goenadi, D.H.: Empty fruit bunches as a potential source for biosilica fertilizer for oil palm. In: Prosiding Seminar Nasional Biologi Tropika 2018. hal. 1–12. , Yogyakarta (2018)
Chang, S.H.: An overview of empty fruit bunch from oil palm as feedstock for bio-oil production. Biomass Bioenerg. 62, 174–181 (2014). https://doi.org/10.1016/j.biombioe.2014.01.002
Zulkarnain, A., Bahrin, E.K., Ramli, N., Phang, L.Y., Abd-Aziz, S.: Alkaline hydrolysate of oil palm empty fruit bunch as potential substrate for biovanillin production via two-step bioconversion. Waste and Biomass Valorization. 9, 13–23 (2018). https://doi.org/10.1007/s12649-016-9745-4
Suksong, W., Mamimin, C., Prasertsan, P., Kongjan, P., O-Thong, S.: Effect of inoculum types and microbial community on thermophilic and mesophilic solid-state anaerobic digestion of empty fruit bunches for biogas production. Ind. Crops Prod. 133, 193–202 (2019). https://doi.org/10.1016/j.indcrop.2019.03.005
Diaz, A.B., Blandino, A., Caro, I.: Value added products from fermentation of sugars derived from agro-food residues. Trends Food Sci. Technol. 71, 52–64 (2018). https://doi.org/10.1016/j.tifs.2017.10.016
Jiang, T.T., Liang, Y., Zhou, X., Shi, Z.W., Xin, Z.J.: Optimization of a pretreatment and hydrolysis process for the efficient recovery of recycled sugars and unknown compounds from agricultural sweet sorghum bagasse stem pith solid waste. PeerJ 6, e6186 (2019). https://doi.org/10.7717/peerj.6186
Salvachúa, D., Prieto, A., López-Abelairas, M., Lu-Chau, T., Martínez, Á.T., Martínez, M.J.: Fungal pretreatment: An alternative in second-generation ethanol from wheat straw. Bioresour. Technol. 102, 7500–7506 (2011). https://doi.org/10.1016/j.biortech.2011.05.027
Dai, Y., Si, M., Chen, Y., Zhang, N., Zhou, M., Liao, Q., Shi, D., Liu, Y.: Combination of biological pretreatment with NaOH/Urea pretreatment at cold temperature to enhance enzymatic hydrolysis of rice straw. Bioresour. Technol. 198, 725–731 (2015). https://doi.org/10.1016/j.biortech.2015.09.091
Kamcharoen, A., Champreda, V., Eurwilaichitr, L., Boonsawang, P.: Screening and optimization of parameters affecting fungal pretreatment of oil palm Empty Fruit Bunch (EFB) by experimental design. Int. J. Energy Environ. Eng. 5, 303–312 (2014). https://doi.org/10.1007/s40095-014-0136-y
Arbaain, E.N.N., Bahrin, E.K., Ibrahim, M.F., Ando, Y., Abd-Aziz, S.: Biological pretreatment of oil palm empty fruit bunch by Schizophyllum commune ENN1 without washing and nutrient addition. Processes. 7, 1–8 (2019). https://doi.org/10.3390/pr7070402
Tahir, A.A., Barnoh, N.F.M., Yusof, N., Said, N.N.M., Utsumi, M., Yen, A.M., Hashim, H., Noor, M.J.M.M., Akhir, F.N.M.D., Mohamad, S.E., Sugiura, N., Othman, N., Zakaria, Z., Hara, H.: Microbial diversity in decaying Oil Palm Empty Fruit Bunches (OPEFB) and isolation of lignin-degrading bacteria from a tropical environment. Microbes Environ. 34, 161–168 (2019). https://doi.org/10.1264/jsme2.ME18117
Millati, R., Wikandari, R., Ariyanto, T., Putri, R.U., Taherzadeh, M.J.: Pretreatment technologies for anaerobic digestion of lignocelluloses and toxic feedstocks. Bioresour. Technol. 304, 122998 (2020). https://doi.org/10.1016/j.biortech.2020.122998
Korripally, P., Timokhin, V.I., Houtman, C.J., Mozuch, M.D., Hammel, K.E.: Evidence from Serpula lacrymans that 2,5-dimethoxyhydroquinone is a lignocellulolytic agent of divergent brown rot basidiomycetes. Appl. Environ. Microbiol. 79, 2377–2383 (2013). https://doi.org/10.1128/AEM.03880-12
Schilling, J.S.: Effects of calcium-based materials and iron impurities on wood degradation by the brown rot fungus Serpula lacrymans. Holzforschung 64, 93–99 (2010). https://doi.org/10.1515/HF.2010.009
Kauserud, H., Knudsen, H., Högberg, N., Skrede, I.: Evolutionary origin, worldwide dispersal, and population genetics of the dry rot fungus Serpula lacrymans. Fungal Biol. Rev. 26, 84–93 (2012). https://doi.org/10.1016/j.fbr.2012.08.001
Tamaru, Y., Yoshida, M., Eltis, L.D., Goodell, B.: Multiple iron reduction by methoxylated phenolic lignin structures and the generation of reactive oxygen species by lignocellulose surfaces. Int. J. Biol. Macromol. 128, 340–346 (2019). https://doi.org/10.1016/j.ijbiomac.2019.01.149
Xie, S., Qin, X., Cheng, Y., Laskar, D., Qiao, W., Sun, S., Reyes, L.H., Wang, X., Dai, S.Y., Sattler, S.E., Kao, K., Yang, B., Zhang, X., Yuan, J.S.: Simultaneous conversion of all cell wall components by an oleaginous fungus without chemi-physical pretreatment. Green Chem. 17, 1657–1667 (2015). https://doi.org/10.1039/c4gc01529k
Jönsson, L.J., Martín, C.: Pretreatment of lignocellulose: Formation of inhibitory by-products and strategies for minimizing their effects. Bioresour. Technol. 199, 103–112 (2016). https://doi.org/10.1016/j.biortech.2015.10.009
Gomes, E.D., Rodrigues, A.E.: Lignin biorefinery: Separation of vanillin, vanillic acid and acetovanillone by adsorption. Sep. Purif. Technol. 216, 92–101 (2019). https://doi.org/10.1016/j.seppur.2019.01.071
Tang, P.L., Hassan, O., Maskat, M.Y., Badri, K.: Production of monomeric aromatic compounds from oil palm empty fruit bunch fiber lignin by chemical and enzymatic methods. Biomed Res. Int. (2015). https://doi.org/10.1155/2015/891539
Kundu, A.: Vanillin biosynthetic pathways in plants. Planta 245, 1069–1078 (2017). https://doi.org/10.1007/s00425-017-2684-x
Shakeri, A., Rad, S.M., Ghasemian, A.: Oxidative production of vanillin from industrial lignin using oxygen and nitrobenzene: a comparative study. Int. J. Farming Allied Sci. 2, 1165–1171 (2013)
Wang, Y., Sun, S., Li, F., Cao, X., Sun, R.: Production of vanillin from lignin: the relationship between β-O-4 linkages and vanillin yield. Ind. Crops Prod. 116, 116–121 (2018). https://doi.org/10.1016/j.indcrop.2018.02.043
Harshvardhan, K., Suri, M., Goswami, A., Goswami, T.: Biological approach for the production of vanillin from lignocellulosic biomass (Bambusa tulda). J. Clean. Prod. 149, 485–490 (2017). https://doi.org/10.1016/j.jclepro.2017.02.125
Xu, C., Arancon, R.A.D., Labidi, J., Luque, R.: Lignin depolymerisation strategies: towards valuable chemicals and fuels. Chem. Soc. Rev. 43, 7485–7500 (2014). https://doi.org/10.1039/c4cs00235k
Di Gioia, D., Luziatelli, F., Negroni, A., Ficca, A.G., Fava, F., Ruzzi, M.: Metabolic engineering of Pseudomonas fluorescens for the production of vanillin from ferulic acid. J. Biotechnol. 156, 309–316 (2011). https://doi.org/10.1016/j.jbiotec.2011.08.014
Shakeel, F., Haq, N., Siddiqui, N.A.: Solubility and thermodynamic function of vanillin in ten different environmentally benign solvents. Food Chem. 180, 244–248 (2015). https://doi.org/10.1016/j.foodchem.2015.01.102
Bittencourt, G.A., da Barreto, E.S., Brandão, R.L., Baêta, B.E.L., Gurgel, L.V.A.: Fractionation of sugarcane bagasse using hydrothermal and advanced oxidative pretreatments for bioethanol and biogas production in lignocellulose biorefineries. Bioresour. Technol. 292, 121963 (2019). https://doi.org/10.1016/j.biortech.2019.121963
Dong, Y., Yu, H.: Research progress on biorefinery of lignocellulosic biomass. IOP Conf. Ser. Earth Environ. Sci. 295, 1–6 (2019). https://doi.org/10.1088/1755-1315/295/2/012078
Bilatto, S., Marconcini, J.M., Mattoso, L.H.C., Farinas, C.S.: Lignocellulose nanocrystals from sugarcane straw. Ind. Crops Prod. 157, 112938 (2020). https://doi.org/10.1016/j.indcrop.2020.112938
Zang, G., Shah, A., Wan, C.: Techno-economic analysis of an integrated biorefinery strategy based on one-pot biomass fractionation and furfural production. J. Clean. Prod. 260, 120837 (2020). https://doi.org/10.1016/j.jclepro.2020.120837
Banu, J.R., Preethi, S.K., Tyagi, V.K., Gunasekaran, M., Karthikeyan, O.P., Kumar, G.: Lignocellulosic biomass based biorefinery: a successful platform towards circular bioeconomy. Fuel 302, 121086 (2021). https://doi.org/10.1016/j.fuel.2021.121086
Rabelo, S.C., Carrere, H., Filho, R.M., Costa, A.C.: Production of bioethanol, methane and heat from sugarcane bagasse in a biorefinery concept. Bioresour. Technol. 102, 7887–7895 (2011). https://doi.org/10.1016/j.biortech.2011.05.081
Kainthola, J., Kalamdhad, A.S., Goud, V.V.: A review on enhanced biogas production from anaerobic digestion of lignocellulosic biomass by different enhancement techniques. Process Biochem. 84, 81–90 (2019). https://doi.org/10.1016/j.procbio.2019.05.023
Suksong, W., Wongfaed, N., Sangsri, B., Kongjan, P., Prasertsan, P., Marie, S., Insam, H., O-thong, S.: Enhanced solid-state biomethanisation of oil palm empty fruit bunches following fungal pretreatment. Ind. Crop. Prod. 145, 112099 (2020). https://doi.org/10.1016/j.indcrop.2020.112099
Sluiter, A., Hames, B., Hyman, D., Payne, C., Ruiz, R., Scarlata, C., Sluiter, J., Templeton, D., Nrel, J.W.: Laboratory Analytical Procedure (LAP): Determination of Total Solids in Biomass and Total Dissolved Solids in Liquid Process Samples. Colorado (2008)
Amano, O.A.K., Offei, F.D., Adom, E., Appiah-Danquah, E.: Determination of Biogas Content in Biomass after Anaerobic digestion: Laboratory Analytical Procedure (LAP). Kumasi, Ghana (2017)
Umor, N.A., Abdullah, S., Mohamad, A., Ismail, S.B., Ismail, S.I., Misran, A.: Energy potential of oil palm Empty Fruit Bunch (EFB) fiber from subsequent cultivation of volvariella volvacea (Bull). Singer. Sustainability. 13, 13008 (2021). https://doi.org/10.3390/su132313008
Anderson, J.M., Ingram, J.S.I.: Tropical Soil Biology and Fertility: A Handbook of Methods. CAB International, Wallingford (1993)
Buswell, A.M., Mueller, H.F.: Mechanism of Methane Fermentation. Ind. Eng. Chem. 44, 550–552 (1952). https://doi.org/10.1021/ie50507a033
Singleton, V.L., Rossi, J.A.: Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am. J. Enol. Vitic. 16, 144–158 (1965)
Miller, G.L.: Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal. Chem. 31, 426–428 (1959). https://doi.org/10.1021/ac60147a030
López, M.J., Elorrieta, M.A., Vargas-García, M.C., Suárez-Estrella, F., Moreno, J.: The effect of aeration on the biotransformation of lignocellulosic wastes by white-rot fungi. Bioresour. Technol. 81, 123–129 (2002). https://doi.org/10.1016/S0960-8524(01)00112-2
Pitt, J., Hocking, A.: Fungi and Food Spoilage. Springer, New York, USA (2009)
Deepa, A.K., Dhepe, P.L.: Solid acid catalyzed depolymerization of lignin into value added aromatic monomers. RSC Adv. 4, 12625–12629 (2014). https://doi.org/10.1039/c3ra47818a
Tešević, V., Aljančić, I., Vajs, V., Živković, M., Nikićević, N., Urošević, I., Vujisić, L.: Development and validation of an LC- MS/MS method with a multiple reactions monitoring mode for the quantification of vanillin and syringaldehyde in plum brandies. J. Serbian Chem. Soc. 79, 1537–1543 (2014). https://doi.org/10.2298/JSC140225079T
Holliger, C., Alves, M., Andrade, D., Angelidaki, I., Astals, S., Baier, U., Bougrier, C., Buffière, P., Carballa, M., De Wilde, V., Ebertseder, F., Fernández, B., Ficara, E., Fotidis, I., Frigon, J.C., De Laclos, H.F., Ghasimi, D.S.M., Hack, G., Hartel, M., Heerenklage, J., Horvath, I.S., Jenicek, P., Koch, K., Krautwald, J., Lizasoain, J., Liu, J., Mosberger, L., Nistor, M., Oechsner, H., Oliveira, J.V., Paterson, M., Pauss, A., Pommier, S., Porqueddu, I., Raposo, F., Ribeiro, T., Pfund, F.R., Strömberg, S., Torrijos, M., Van Eekert, M., Van Lier, J., Wedwitschka, H., Wierinck, I.: Towards a standardization of biomethane potential tests. Water Sci. Technol. 74, 2515–2522 (2016). https://doi.org/10.2166/wst.2016.336
Suhartini, S., Heaven, S., Zhang, Y., Banks, C.J.: Antifoam, dilution and trace element addition as foaming control strategies in mesophilic anaerobic digestion of sugar beet pulp. Int. Biodeterior. Biodegrad. 145, 1–13 (2019). https://doi.org/10.1016/j.ibiod.2019.104812
Strömberg, S., Nistor, M., Liu, J.: Towards eliminating systematic errors caused by the experimental conditions in Biochemical Methane Potential (BMP) tests. Waste Manag. 34, 1939–1948 (2014). https://doi.org/10.1016/j.wasman.2014.07.018
Tavakol, M., Dennick, R.: Making sense of Cronbach’s alpha. Int. J. Med. Educ. 2, 53–55 (2011). https://doi.org/10.5116/ijme.4dfb.8dfd
Aghdam, E.F., Scheutz, C., Kjeldsen, P.: Assessment of methane production from shredder waste in landfills: The influence of temperature, moisture and metals. Waste Manag. 63, 226–237 (2017). https://doi.org/10.1016/j.wasman.2016.11.023
Patowary, D., Baruah, D.C.: Effect of combined chemical and thermal pretreatments on biogas production from lignocellulosic biomasses. Ind. Crops Prod. 124, 735–746 (2018). https://doi.org/10.1016/j.indcrop.2018.08.055
Yavini, T.D., Chia, A.I., John, A.: Evaluation of the effect of total solids concentration on biogas yields of agricultural wastes. Int. J. Environ. Sci. 3, 70–75 (2014)
Momayez, F., Karimi, K., Horváth, I.S.: Sustainable and efficient sugar production from wheat straw by pretreatment with biogas digestate. RSC Adv. 9, 27692–27701 (2019). https://doi.org/10.1039/c9ra05285b
Kim, M.J., Kim, S.H.: Evaluation of the degradation of carbohydrate-based material during anaerobic digestion for high-efficiency biogas production. J. Biosyst. Eng. 43, 110–118 (2018). https://doi.org/10.5307/JBE.2018.43.2.110
Ohemeng-Ntiamoah, J., Datta, T.: Evaluating analytical methods for the characterization of lipids, proteins and carbohydrates in organic substrates for anaerobic co-digestion. Bioresour. Technol. 247, 697–704 (2018). https://doi.org/10.1016/j.biortech.2017.09.154
Solarte-Toro, J.C., Chacón-Pérez, Y., Cardona-Alzate, C.A.: Evaluation of biogas and syngas as energy vectors for heat and power generation using lignocellulosic biomass as raw material. Electron. J. Biotechnol. 33, 52–62 (2018). https://doi.org/10.1016/j.ejbt.2018.03.005
Ilminnafik, N., Setiyawan, D.L., Sutjahjono, H., Rofiq, A., Hadi, A.S.: Flame Characteristics of Biogas from Coffee Waste Materials. J. Phys. Conf. Ser. 1175, 012273 (2019). https://doi.org/10.1088/1742-6596/1175/1/012273
Romagnoli, F., Dorella, M., Gruduls, A., Collotta, M., Tomasoni, G.: Anaerobic co-digestion of Baltic seaweeds with wheat straw and straw pellets: synergetic effects on biomethane yield and kinetic biodegradability constant. Energy Procedia. 158, 854–860 (2019). https://doi.org/10.1016/j.egypro.2019.01.221
Chandra, R., Takeuchi, H., Hasegawa, T.: Methane production from lignocellulosic agricultural crop wastes: a review in context to second generation of biofuel production. Renew. Sustain. Energy Rev. 16, 1462–1476 (2012). https://doi.org/10.1016/j.rser.2011.11.035
Náthia-Neves, G., Berni, M., Dragone, G., Mussatto, S.I., Forster-Carneiro, T.: Anaerobic digestion process: technological aspects and recent developments. Int. J. Environ. Sci. Technol. 15, 2033–2046 (2018). https://doi.org/10.1007/s13762-018-1682-2
Zeng, G., Yu, M., Chen, Y., Huang, D., Zhang, J., Huang, H., Jiang, R., Yu, Z.: Effects of inoculation with Phanerochaete chrysosporium at various time points on enzyme activities during agricultural waste composting. Bioresour. Technol. 101, 222–227 (2010). https://doi.org/10.1016/j.biortech.2009.08.013
Arantes, V., Jellison, J., Goodell, B.: Peculiarities of brown-rot fungi and biochemical Fenton reaction with regard to their potential as a model for bioprocessing biomass. Appl. Microbiol. Biotechnol. 94, 323–338 (2012). https://doi.org/10.1007/s00253-012-3954-y
Mustabi, J., Wedawati, A.A.K.: Improving quality and digestibility of cocoa pod with white rot fungi. IOP Conf. Ser. Earth Environ. Sci. 157, 1–7 (2018). https://doi.org/10.1088/1755-1315/157/1/012002
Arantes, V., Qian, Y., Milagres, A.M.F., Jellison, J., Goodell, B.: Effect of pH and oxalic acid on the reduction of Fe3+ by a biomimetic chelator and on Fe3+ desorption/adsorption onto wood: Implications for brown-rot decay. Int. Biodeterior. Biodegrad. 63, 478–483 (2009). https://doi.org/10.1016/j.ibiod.2009.01.004
Aguiar, A., Gavioli, D., Ferraz, A.: Extracellular activities and wood component losses during Pinus taeda biodegradation by the brown-rot fungus Gloeophyllum trabeum. Int. Biodeterior. Biodegrad. 82, 187–191 (2013). https://doi.org/10.1016/j.ibiod.2013.03.013
Shah, F., Mali, T., Lundell, T.K.: Polyporales brown rot species fomitopsis pinicola: enzyme activity profiles, oxalic acid production, and Fe3+-reducing metabolite secretion. Appl. Environ. Microbiol. 84, e02662-e2717 (2018)
Zhu, Y., Mahaney, J., Jellison, J., Cao, J., Gressler, J., Hoffmeister, D., Goodell, B.: Fungal variegatic acid and extracellular polysaccharides promote the site-specific generation of reactive oxygen species. J. Ind. Microbiol. Biotechnol. 44, 329–338 (2017). https://doi.org/10.1007/s10295-016-1889-5
Nurika, I., Suhartini, S., Barker, G.C.: Biotransformation of tropical lignocellulosic feedstock using the brown rot fungus Serpula lacrymans. Waste Biomass Valor. 11, 2689–2700 (2020). https://doi.org/10.1007/s12649-019-00581-5
Janusz, G., Pawlik, A., Sulej, J., Świderska-Burek, U., Jarosz-Wilkolazka, A., Paszczyński, A.: Lignin degradation: Microorganisms, enzymes involved, genomes analysis and evolution. FEMS Microbiol. Rev. 41, 941–962 (2017). https://doi.org/10.1093/femsre/fux049
Gupta, R., Mehta, G., Khasa, Y.P., Kuhad, R.C.: Fungal delignification of lignocellulosic biomass improves the saccharification of cellulosics. Biodegradation 22, 797–804 (2011). https://doi.org/10.1007/s10532-010-9404-6
Cianchetta, S., Di Maggio, B., Burzi, P.L., Galletti, S.: Evaluation of selected white-rot fungal isolates for improving the sugar yield from wheat straw. Appl. Biochem. Biotechnol. 173, 609–623 (2014). https://doi.org/10.1007/s12010-014-0869-3
Hamad, H.O., Alma, M.H., Ismael, H.M., Göçeri, A.: The effect of some sugars on the growth of Aspergillus niger. Kahramanmaraş Sütçü İmam Üniversitesi Doğa Bilim. Derg. 17, 7–11 (2014). https://doi.org/10.18016/ksujns.28479
Lalak, J., Kasprzycka, A., Martyniak, D., Tys, J.: Effect of biological pretreatment of Agropyron elongatum “BAMAR” on biogas production by anaerobic digestion. Bioresour. Technol. 200, 194–200 (2016). https://doi.org/10.1016/j.biortech.2015.10.022
Cai, J., Wang, Y., Liu, J., Zhang, X., Li, F.: Pretreatment enhanced structural disruption, enzymatic hydrolysis, fermentative hydrogen production from rice straw. Int. J. Hydrogen Energy. 47, 11778–11786 (2022). https://doi.org/10.1016/j.ijhydene.2022.01.214
Liaud, N., Giniés, C., Navarro, D., Fabre, N., Crapart, S., Gimbert, I.H., Levasseur, A., Raouche, S., Sigoillot, J.-C.: Exploring fungal biodiversity: organic acid production by 66 strains of filamentous fungi. Fungal Biol. Biotechnol. 1, 1–10 (2014). https://doi.org/10.1186/s40694-014-0001-z
Hau, L.J., Shamsuddin, R., May, A.K.A., Saenong, A., Lazim, A.M., Narasimha, M., Low, A.: Mixed composting of Palm Oil Empty Fruit Bunch (EFB) and Palm Oil Mill Effluent (POME) with various organics: an analysis on final macronutrient content and physical properties. Waste Biomass Valor (2020). https://doi.org/10.1007/s12649-020-00993-8
Costa, S., Rugiero, I., Uria, C.L., Pedrini, P., Tamburini, E.: Lignin degradation efficiency of chemical pre-treatments on banana rachis destined to bioethanol production. Biomolecules 8, 141 (2018). https://doi.org/10.3390/biom8040141
Zabihi, S., Alinia, R., Esmaeilzadeh, F., Kalajahi, J.F.: Pretreatment of wheat straw using steam, steam/acetic acid and steam/ethanol and its enzymatic hydrolysis for sugar production. Biosyst. Eng. 105, 288–297 (2010). https://doi.org/10.1016/j.biosystemseng.2009.11.007
Zhang, S., Jiang, M., Zhou, Z., Zhao, M., Li, Y.: Selective removal of lignin in steam-exploded rice straw by Phanerochaete chrysosporium. Int. Biodeterior. Biodegradation. 75, 89–95 (2012). https://doi.org/10.1016/j.ibiod.2012.09.003
Sun, Y., Cheng, J.: Hydrolysis of lignocellulosic materials for ethanol production: a review. Bioresour. Technol. 83, 1–11 (2002)
Singh, J.K., Chaurasia, B., Dubey, A., Noguera, A.M.F., Gupta, A., Kothari, R., Upadhyaya, C.P., Kumar, A., Hashem, A., Alqarawi, A.A., Allah, E.F.A.: Biological characterization and instrumental analytical comparison of two biorefining pretreatments for water hyacinth (Eicchornia crassipes) biomass hydrolysis. Sustainability. 13, 245 (2021). https://doi.org/10.3390/su13010245
Jha, H., Arora, R.: Microbial-Assisted Systems for Lignin-Based Product Generation. In: Verma, P. (ed.) Industrial Microbiology and Biotechnology. hal. 555–587. Springer, Singapore (2022)
Shen, Y., Han, C., Liu, B., Lin, Z., Zhou, X., Wang, C., Zhu, Z.: Determination of vanillin, ethyl vanillin, and coumarin in infant formula by liquid chromatography-quadrupole linear ion trap mass spectrometry. J. Dairy Sci. 97, 679–686 (2014). https://doi.org/10.3168/jds.2013-7308
Taira, J., Toyoshima, R., Ameku, N., Iguchi, A., Tamaki, Y.: Vanillin production by biotransformation of phenolic compounds in fungus, Aspergillus luchuensis. AMB Express 8, 40 (2018). https://doi.org/10.1186/s13568-018-0569-4
Messaoudi, Y., Smichi, N., Bouachir, F., Gargouri, M.: Fractionation and biotransformation of lignocelluloses-based wastes for bioethanol, xylose and vanillin production. Waste Biomass Valor. 10, 357–367 (2019). https://doi.org/10.1007/s12649-017-0062-3
Wadchasit, P., Siripattana, C., Nuithitikul, K.: The effect of pretreatment methods for improved biogas production from oil-palm empty fruit bunches (EFB): Experimental and model. IOP Conf. Ser. Earth Environ. Sci. 463, 012126 (2020). https://doi.org/10.1088/1755-1315/463/1/012126
Saha, B., Sathyan, A., Mazumder, P., Choudhury, S.P., Kalamdhad, A.S., Khwairakpam, M., Mishra, U.: Biochemical methane potential (BMP) test for Ageratum conyzoides to optimize ideal food to microorganism (F/M) ratio. J. Environ. Chem. Eng. 6, 5135–5140 (2018). https://doi.org/10.1016/j.jece.2018.07.036
Adadan, E., Savasci, F.: An analysis of 16–17-year-old students’ understanding of solution chemistry concepts using a two-tier diagnostic instrument. Int. J. Sci. Educ. 34, 513–544 (2012). https://doi.org/10.1080/09500693.2011.636084
Angelo, N.R., Rudiyansyah, R., Gusrizal, G.: Microbial Degradation of Lignocellulose in Empty Fruit Bunch at Various Incubation Time. J. Trop. Biodivers. Biotechnol. 7, 1–5 (2022). https://doi.org/10.22146/jtbb.69743
Acknowledgements
The authors would like to thank University of Warwick, United Kingdom for supporting the open access publishing charge.
Funding
This work was financially supported by “Hibah Penelitian Kerjasama International (HAPKI)” Universitas Brawijaya, Indonesia. (Grant No.610.1/UN10.C20/2023).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nurika, I., Azizah, N., Suhartini, S. et al. Integrated Biorefinery Approach: The Generation of Bioproducts Vanillin and Biomethane Through a Sequential Bioconversion of Lignocellulose From Oil Palm Empty Fruit Bunch (OPEFB). Waste Biomass Valor 15, 903–915 (2024). https://doi.org/10.1007/s12649-023-02205-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-023-02205-5