Abstract
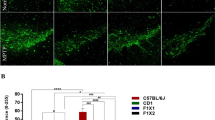
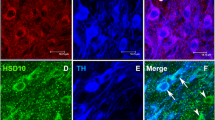
Parkinson disease (PD) prevalence varies by ethnicity. In an earlier study, we replicated the reduced vulnerability to PD in an admixed population, using 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-susceptible C57BL/6 J, MPTP-resistant CD-1 and their F1 crossbreds. In the present study, we investigated if the differences have a developmental origin. Substantia nigra was evaluated at postnatal days 2 (P2), P6, P10, P14, P18, and P22. C57BL/6 J mice had smaller nigra and fewer dopaminergic neurons than the CD-1 and crossbreds at P2, which persisted through development. A significant increase in numbers and nigral volume was observed across strains until P14. A drastic decline thereafter was specific to C57BL/6 J. CD-1 and crossbreds retained their numbers from P14 to stabilize with supernumerary neurons at adulthood. The neuronal size increased gradually to attain adult morphology at P10 in the resistant strains, vis-à-vis at P22 in C57BL/6 J. Accordingly, in comparison to C57BL/6 J, the nigra of CD-1 and reciprocal crossbreds possessed cytomorphological features of resilience, since birth. The considerably lesser dopaminergic neuronal loss in the CD-1 and crossbreds was seen at P2 and P14 and thereafter was complemented by attenuated developmental cell death. The differences in programmed cell death were confirmed by reduced TUNEL labelling, AIF, and caspase-3 expression. GDNF expression aligned with the cell death pattern at P2 and P14 in both nigra and striatum. Earlier maturity of nigra and its neurons appears to be better features that reflect as MPTP resistance at adulthood. Thus, variable MPTP vulnerability in mice and also differential susceptibility to PD in humans may arise early during nigral development.





Similar content being viewed by others
References
Alladi PA, Mahadevan A, Shankar SK, Raju TR, Muthane U (2010a) Expression of GDNF receptors GFRalpha1 and RET is preserved in substantia nigra pars compacta of aging Asian Indians. J Chem Neuroanat 40(1):43–52. https://doi.org/10.1016/j.jchemneu.2010.03.007
Alladi PA, Mahadevan A, Vijayalakshmi K, Muthane U, Shankar SK, Raju TR (2010b) Ageing enhances alpha-synuclein, ubiquitin and endoplasmic reticular stress protein expression in the nigral neurons of Asian Indians. Neurochem Int 57(5):530–539. https://doi.org/10.1016/j.neuint.2010.06.018
Alladi PA, Mahadevan A, Yasha TC, Raju TR, Shankar SK, Muthane U (2009) Absence of age-related changes in nigral dopaminergic neurons of Asian Indians: relevance to lower incidence of Parkinson’s disease. Neuroscience 159(1):236–245. https://doi.org/10.1016/j.neuroscience.2008.11.051
Andersen JK, Frim DM, Isacson O, Beal MF, Breakefield XO (1994) Elevation of neuronal MAO-B activity in a transgenic mouse model does not increase sensitivity to the neurotoxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). Brain Res 656(1):108–114. https://doi.org/10.1016/0006-8993(94)91371-4
Antonopoulos J, Dori I, Dinopoulos A, Chiotelli M, Parnavelas JG (2002) Postnatal development of the dopaminergic system of the striatum in the rat. Neuroscience 110(2):245–256
Arya R, White K (2015) Cell death in development: signaling pathways and core mechanisms. Semin Cell Dev Biol 39:12–19. https://doi.org/10.1016/j.semcdb.2015.02.001
Baquet ZC, Williams D, Brody J, Smeyne RJ (2009) A comparison of model-based (2D) and design-based (3D) stereological methods for estimating cell number in the substantia nigra pars compacta (SNpc) of the C57BL/6J mouse. Neuroscience 161(4):1082–1090. https://doi.org/10.1016/j.neuroscience.2009.04.031
Barroso-Chinea P, Cruz-Muros I, Aymerich MS, Rodríguez-Díaz M, Afonso-Oramas D, Lanciego JL, González-Hernández T (2005) Striatal expression of GDNF and differential vulnerability of midbrain dopaminergic cells. Eur J Neurosci 21(7):1815–1827. https://doi.org/10.1111/j.1460-9568.2005.04024.x
Bhaduri B, Abhilash PL, Alladi PA (2018) Baseline striatal and nigral interneuronal protein levels in two distinct mice strains differ in accordance with their MPTP susceptibility. J Chem Neuroanat 91:46–54. https://doi.org/10.1016/j.jchemneu.2018.04.005
Bjorklund A, Dunnett SB (2007) Dopamine neuron systems in the brain: an update. Trends Neurosci 30(5):194–202. https://doi.org/10.1016/j.tins.2007.03.006
Blum M, Weickert CS (1995) GDNF mRNA expression in normal postnatal development, aging, and in weaver mutant mice. Neurobiol Aging 16(6):925–929. https://doi.org/10.1016/0197-4580(95)02011-x
Burke RE (1998) Programmed cell death and Parkinson’s disease. Mov Disord 13(Suppl 1):17–23
Burke RE (2003) Postnatal developmental programmed cell death in dopamine neurons. Ann N Y Acad Sci 991:69–79
Buss RR, Sun W, Oppenheim RW (2006) Adaptive roles of programmed cell death during nervous system development. Annu Rev Neurosci 29:1–35. https://doi.org/10.1146/annurev.neuro.29.051605.112800
Cabello CR, Thune JJ, Pakkenberg H, Pakkenberg B (2002) Ageing of substantia nigra in humans: cell loss may be compensated by hypertrophy. Neuropathol Appl Neurobiol 28(4):283–291
Castagna C, Merighi A, Lossi L (2016) Cell death and neurodegeneration in the postnatal development of cerebellar vermis in normal and Reeler mice. Ann Anat 207:76–90. https://doi.org/10.1016/j.aanat.2016.01.010
Cheng S, Tereshchenko J, Zimmer V, Vachey G, Pythoud C, Rey M, Liefhebber J, Raina A, Streit F, Mazur A, Bähr M, Konstantinova P, Déglon N, Kügler S (2018) Therapeutic efficacy of regulable GDNF expression for Huntington’s and Parkinson’s disease by a high-induction, background-free “GeneSwitch” vector. Exp Neurol 309:79–90. https://doi.org/10.1016/j.expneurol.2018.07.017
Chocyk A, Przyborowska A, Dudys D, Majcher I, Mackowiak M, Wedzony K (2011) The impact of maternal separation on the number of tyrosine hydroxylase-expressing midbrain neurons during different stages of ontogenesis. Neuroscience 182:43–61. https://doi.org/10.1016/j.neuroscience.2011.03.008
Chu Y, Kompoliti K, Cochran EJ, Mufson EJ, Kordower JH (2002) Age-related decreases in Nurr1 immunoreactivity in the human substantia nigra. J Comp Neurol 450(3):203–214. https://doi.org/10.1002/cne.10261
Das SK, Misra AK, Ray BK, Hazra A, Ghosal MK, Chaudhuri A, Roy T, Banerjee TK, Raut DK (2010) Epidemiology of Parkinson disease in the city of Kolkata, India: a community-based study. Neurology 75(15):1362–1369. https://doi.org/10.1212/WNL.0b013e3181f735a7
Fahn S (1989) The endogenous toxin hypothesis of the etiology of Parkinson’s disease and a pilot trial of high-dosage antioxidants in an attempt to slow the progression of the illness. Ann N Y Acad Sci 570:186–196
Fatemi SH, Folsom TD (2009) The neurodevelopmental hypothesis of schizophrenia, revisited. Schizophr Bull 35(3):528–548. https://doi.org/10.1093/schbul/sbn187
Fearnley JM, Lees AJ (1991) Ageing and Parkinson’s disease: substantia nigra regional selectivity. Brain 114(Pt 5):2283–2301
Fu Y, Paxinos G, Watson C, Halliday GM (2016) The substantia nigra and ventral tegmental dopaminergic neurons from development to degeneration. J Chem Neuroanat 76(Pt B):98–107. https://doi.org/10.1016/j.jchemneu.2016.02.001
Fu Y, Yuan Y, Halliday G, Rusznák Z, Watson C, Paxinos G (2012) A cytoarchitectonic and chemoarchitectonic analysis of the dopamine cell groups in the substantia nigra, ventral tegmental area, and retrorubral field in the mouse. Brain Struct Funct 217(2):591–612. https://doi.org/10.1007/s00429-011-0349-2
Fujimiya M, Hosoda S, Kitahama K, Kimura H, Maeda T (1986a) Early development of serotonin neuron in the rat brain as studied by immunohistochemistry combined with tryptophan administration. Brain Dev 8(4):335–342
Fujimiya M, Kimura H, Maeda T (1986b) Postnatal development of serotonin nerve fibers in the somatosensory cortex of mice studied by immunohistochemistry. J Comp Neurol 246(2):191–201. https://doi.org/10.1002/cne.902460205
Harris GJ, Codori AM, Lewis RF, Schmidt E, Bedi A, Brandt J (1999) Reduced basal ganglia blood flow and volume in pre-symptomatic, gene-tested persons at-risk for Huntington’s disease. Brain 122(Pt 9):1667–1678
Jackson-Lewis V, Vila M, Djaldetti R, Guegan C, Liberatore G, Liu J, O’Malley KL, Burke RE, Przedborski S (2000) Developmental cell death in dopaminergic neurons of the substantia nigra of mice. J Comp Neurol 424(3):476–488
Jiang P, Gan M, Yen SH, McLean PJ, Dickson DW (2017) Histones facilitate α-synuclein aggregation during neuronal apoptosis. Acta Neuropathol 133(4):547–558. https://doi.org/10.1007/s00401-016-1660-z
Jyothi HJ, Vidyadhara DJ, Mahadevan A, Philip M, Parmar SK, Manohari SG, Shankar SK, Raju TR, Alladi PA (2015) Aging causes morphological alterations in astrocytes and microglia in human substantia nigra pars compacta. Neurobiol Aging 36(12):3321–3333. https://doi.org/10.1016/j.neurobiolaging.2015.08.024
Katunar MR, Saez T, Brusco A, Antonelli MC (2010) Ontogenetic expression of dopamine-related transcription factors and tyrosine hydroxylase in prenatally stressed rats. Neurotox Res 18(1):69–81. https://doi.org/10.1007/s12640-009-9132-z
Kawano H, Ohyama K, Kawamura K, Nagatsu I (1995) Migration of dopaminergic neurons in the embryonic mesencephalon of mice. Brain Res Dev Brain Res 86(1–2):101–113
Kilbourn M, Frey K (1996) Striatal concentrations of vesicular monoamine transporters are identical in MPTP-sensitive (C57BL/6) and -insensitive (CD-1) mouse strains. Eur J Pharmacol 307(2):227–232. https://doi.org/10.1016/0014-2999(96)00252-x
Kordower JH, Olanow CW, Dodiya HB, Chu Y, Beach TG, Adler CH, Halliday GM, Bartus RT (2013) Disease duration and the integrity of the nigrostriatal system in Parkinson’s disease. Brain 136(Pt 8):2419–2431. https://doi.org/10.1093/brain/awt192
Korunes KL, Goldberg A (2021) Human genetic admixture. PLoS Genet 17(3):e1009374. https://doi.org/10.1371/journal.pgen.1009374
Kreczmanski P, Heinsen H, Mantua V, Woltersdorf F, Masson T, Ulfig N, Schmidt-Kastner R, Korr H, Steinbusch HW, Hof PR, Schmitz C (2007) Volume, neuron density and total neuron number in five subcortical regions in schizophrenia. Brain 130(Pt 3):678–692. https://doi.org/10.1093/brain/awl386
Kuan CY, Roth KA, Flavell RA, Rakic P (2000) Mechanisms of programmed cell death in the developing brain. Trends Neurosci 23(7):291–297. https://doi.org/10.1016/s0166-2236(00)01581-2
Kupsch A, Sautter J, Götz ME, Breithaupt W, Schwarz J, Youdim MB, Riederer P, Gerlach M, Oertel WH (2001) Monoamine oxidase-inhibition and MPTP-induced neurotoxicity in the non-human primate: comparison of rasagiline (TVP 1012) with selegiline. J Neural Transm (vienna) 108(8–9):985–1009. https://doi.org/10.1007/s007020170018
Lee JK, Mathews K, Schlaggar B, Perlmutter J, Paulsen JS, Epping E, Burmeister L, Nopoulos P (2012) Measures of growth in children at risk for Huntington disease. Neurology 79(7):668–674. https://doi.org/10.1212/WNL.0b013e3182648b65
Lie DC, Dziewczapolski G, Willhoite AR, Kaspar BK, Shults CW, Gage FH (2002) The adult substantia nigra contains progenitor cells with neurogenic potential. J Neurosci 22(15):6639–6649. https://doi.org/10.1523/JNEUROSCI.22-15-06639.2002
Lieb K, Andersen C, Lazarov N, Zienecker R, Urban I, Reisert I, Pilgrim C (1996) Pre- and postnatal development of dopaminergic neuron numbers in the male and female mouse midbrain. Brain Res Dev Brain Res 94(1):37–43
Ma SY, Rinne JO, Collan Y, Roytta M, Rinne UK (1996) A quantitative morphometrical study of neuron degeneration in the substantia nigra in Parkinson’s disease. J Neurol Sci 140(1–2):40–45
Mabandla MV, Russell VA (2010) Voluntary exercise reduces the neurotoxic effects of 6-hydroxydopamine in maternally separated rats. Behav Brain Res 211(1):16–22. https://doi.org/10.1016/j.bbr.2010.02.045
Marras C, Beck JC, Bower JH, Roberts E, Ritz B, Ross GW, Abbott RD, Savica R, Van Den Eeden SK, Willis AW, Tanner CM (2018) Prevalence of Parkinson’s disease across North America. NPJ Parkinsons Dis 4:21. https://doi.org/10.1038/s41531-018-0058-0
Meredith GE, Rademacher DJ (2011) MPTP mouse models of Parkinson’s disease: an update. J Parkinsons Dis 1(1):19–33. https://doi.org/10.3233/jpd-2011-11023
Morrow BA, Roth RH, Redmond DE Jr, Sladek JR Jr, Elsworth JD (2007) Apoptotic natural cell death in developing primate dopamine midbrain neurons occurs during a restricted period in the second trimester of gestation. Exp Neurol 204(2):802–807. https://doi.org/10.1016/j.expneurol.2007.01.009
Naskar A, Mahadevan A, Philip M, Alladi PA (2019) Aging mildly affects dendritic arborisation and synaptic protein expression in human substantia nigra pars compacta. J Chem Neuroanat 97:57–65. https://doi.org/10.1016/j.jchemneu.2019.02.001
Oo TF, Burke RE (1997) The time course of developmental cell death in phenotypically defined dopaminergic neurons of the substantia nigra. Brain Res Dev Brain Res 98(2):191–196
Oo TF, Kholodilov N, Burke RE (2003) Regulation of natural cell death in dopaminergic neurons of the substantia nigra by striatal glial cell line-derived neurotrophic factor in vivo. J Neurosci 23(12):5141–5148. https://doi.org/10.1523/jneurosci.23-12-05141.2003
Pagida MA, Konstantinidou AE, Chrysanthou-Piterou MA, Patsouris ES, Panayotacopoulou MT (2020) Apoptotic markers in the midbrain of the human neonate after perinatal hypoxic/ischemic injury. J Neuropathol Exp Neurol 79(1):86–101. https://doi.org/10.1093/jnen/nlz114
Panzer A, Viljoen M (2005) Supportive neurodevelopmental evidence for ADHD as a developmental disorder. Med Hypotheses 64(4):755–758. https://doi.org/10.1016/j.mehy.2003.12.060
Park M, Kitahama K, Geffard M, Maeda T (2000) Postnatal development of the dopaminergic neurons in the rat mesencephalon. Brain Dev 22(Suppl 1):S38-44
Perneczky R, Wagenpfeil S, Lunetta KL, Cupples LA, Green RC, Decarli C, Farrer LA, Kurz A (2010) Head circumference, atrophy, and cognition: implications for brain reserve in Alzheimer disease. Neurology 75(2):137–142. https://doi.org/10.1212/WNL.0b013e3181e7ca97
Pienaar IS, Kellaway LA, Russell VA, Smith AD, Stein DJ, Zigmond MJ, Daniels WM (2008) Maternal separation exaggerates the toxic effects of 6-hydroxydopamine in rats: implications for neurodegenerative disorders. Stress 11(6):448–456. https://doi.org/10.1080/10253890801890721
Ragothaman M, Murgod UA, Gururaj G, Kumaraswamy SD, Muthane U (2003) Lower risk of Parkinson’s disease in an admixed population of European and Indian origins. Mov Disord 18(8):912–914. https://doi.org/10.1002/mds.10449
Reiss AL, Faruque F, Naidu S, Abrams M, Beaty T, Bryan RN, Moser H (1993) Neuroanatomy of Rett syndrome: a volumetric imaging study. Ann Neurol 34(2):227–234. https://doi.org/10.1002/ana.410340220
Rodrigues AJ, Leao P, Carvalho M, Almeida OF, Sousa N (2011) Potential programming of dopaminergic circuits by early life stress. Psychopharmacology 214(1):107–120. https://doi.org/10.1007/s00213-010-2085-3
Rosas HD, Goodman J, Chen YI, Jenkins BG, Kennedy DN, Makris N, Patti M, Seidman LJ, Beal MF, Koroshetz WJ (2001) Striatal volume loss in HD as measured by MRI and the influence of CAG repeat. Neurology 57(6):1025–1028
Sailaja K, Gopinath G (1994) Developing substantia nigra in human: a qualitative study. Dev Neurosci 16(1–2):44–52
Sailaja K, Gopinath G (1996) Ultrastructure of developing substantia nigra in humans. Int J Dev Neurosci 14(6):761–770
Saito H, Matsumoto M, Togashi H, Yoshioka M (1996) Functional interaction between serotonin and other neuronal systems: focus on in vivo microdialysis studies. Jpn J Pharmacol 70(3):203–205
Sanges D, Comitato A, Tammaro R, Marigo V (2006) Apoptosis in retinal degeneration involves cross-talk between apoptosis-inducing factor (AIF) and caspase-12 and is blocked by calpain inhibitors. Proc Natl Acad Sci U S A 103(46):17366–17371. https://doi.org/10.1073/pnas.0606276103
Schildknecht S, Di Monte DA, Pape R, Tieu K, Leist M (2017) Tipping points and endogenous determinants of nigrostriatal degeneration by MPTP. Trends Pharmacol Sci 38(6):541–555. https://doi.org/10.1016/j.tips.2017.03.010
Seldin MF, Pasaniuc B, Price AL (2011) New approaches to disease mapping in admixed populations. Nat Rev Genet 12(8):523–528. https://doi.org/10.1038/nrg3002
Seshadri A, Alladi PA (2019) Divergent expression patterns of Drp1 and HSD10 in the nigro-striatum of two mice strains based on their MPTP susceptibility. Neurotox Res 36(1):27–38. https://doi.org/10.1007/s12640-019-00036-8
Shan X, Chi L, Bishop M, Luo C, Lien L, Zhang Z, Liu R (2006) Enhanced de novo neurogenesis and dopaminergic neurogenesis in the substantia nigra of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinson’s disease-like mice. Stem Cells 24(5):1280–1287. https://doi.org/10.1634/stemcells.2005-0487
Sn S, Pandurangi J, Murumalla R, Dj V, Garimella L, Acharya A, Rai S, Paul A, Yarreiphang H, Pillai MS, Giridharan M, Clement JP, Alladi PA, Saiyed T, Manjithaya R (2019) Small molecule modulator of aggrephagy regulates neuroinflammation to curb pathogenesis of neurodegeneration. EBioMedicine. https://doi.org/10.1016/j.ebiom.2019.10.036
Strickland D, Bertoni JM (2004) Parkinson’s prevalence estimated by a state registry. Mov Disord 19(3):318–323. https://doi.org/10.1002/mds.10619
Sullivan RM, Brake WG (2003) What the rodent prefrontal cortex can teach us about attention-deficit/hyperactivity disorder: the critical role of early developmental events on prefrontal function. Behav Brain Res 146(1–2):43–55
Suresh SN, Chavalmane AK, Vidyadhara DJ, Yarreiphang H, Rai S, Paul A, Clement JP, Alladi PA, Manjithaya R (2017) A novel autophagy modulator 6-Bio ameliorates SNCA/alpha-synuclein toxicity. Autophagy 13(7):1221–1234. https://doi.org/10.1080/15548627.2017.1302045
Suresh SN, Chavalmane AK, Pillai M, Ammanathan V, Vidyadhara DJ, Yarreiphang H, Rai S, Paul A, Clement JP, Alladi PA, Manjithaya R (2018) Modulation of autophagy by a small molecule inverse agonist of erralpha is neuroprotective. Front Mol Neurosci 11:109. https://doi.org/10.3389/fnmol.2018.00109
Tomac A, Widenfalk J, Lin LF, Kohno T, Ebendal T, Hoffer BJ, Olson L (1995) Retrograde axonal transport of glial cell line-derived neurotrophic factor in the adult nigrostriatal system suggests a trophic role in the adult. Proc Natl Acad Sci U S A 92(18):8274–8278. https://doi.org/10.1073/pnas.92.18.8274
Vidyadhara DJ, Lee JE, Chandra SS (2019a) Role of the endolysosomal system in Parkinson’s disease. J Neurochem 150(5):487–506. https://doi.org/10.1111/jnc.14820
Vidyadhara DJ, Sasidharan A, Kutty BM, Raju TR, Alladi PA (2019b) Admixing MPTP-resistant and MPTP-vulnerable mice enhances striatal field potentials and calbindin-D28K expression to avert motor behaviour deficits. Behav Brain Res 360:216–227. https://doi.org/10.1016/j.bbr.2018.12.015
Vidyadhara DJ, Yarreiphang H, Abhilash PL, Raju TR, Alladi PA (2016) Differential expression of calbindin in nigral dopaminergic neurons in two mice strains with differential susceptibility to 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine. J Chem Neuroanat. https://doi.org/10.1016/j.jchemneu.2016.01.001
Vidyadhara DJ, Yarreiphang H, Raju TR, Alladi PA (2017) Admixing of MPTP-resistant and susceptible mice strains augments nigrostriatal neuronal correlates to resist MPTP-induced neurodegeneration. Mol Neurobiol 54(8):6148–6162. https://doi.org/10.1007/s12035-016-0158-y
Voelbel GT, Bates ME, Buckman JF, Pandina G, Hendren RL (2006) Caudate nucleus volume and cognitive performance: are they related in childhood psychopathology? Biol Psychiatry 60(9):942–950. https://doi.org/10.1016/j.biopsych.2006.03.071
von Campenhausen S, Bornschein B, Wick R, Botzel K, Sampaio C, Poewe W, Oertel W, Siebert U, Berger K, Dodel R (2005) Prevalence and incidence of Parkinson’s disease in Europe. Eur Neuropsychopharmacol 15(4):473–490. https://doi.org/10.1016/j.euroneuro.2005.04.007
Voorn P, Kalsbeek A, Jorritsma-Byham B, Groenewegen HJ (1988) The pre- and postnatal development of the dopaminergic cell groups in the ventral mesencephalon and the dopaminergic innervation of the striatum of the rat. Neuroscience 25(3):857–887
Wang X, Michaelis EK (2010) Selective neuronal vulnerability to oxidative stress in the brain. Front Aging Neurosci 2:12. https://doi.org/10.3389/fnagi.2010.00012
Weber K, Rockstroh B, Borgelt J, Awiszus B, Popov T, Hoffmann K, Schonauer K, Watzl H, Propster K (2008) Stress load during childhood affects psychopathology in psychiatric patients. BMC Psychiatry 8:63. https://doi.org/10.1186/1471-244x-8-63
Whone A, Luz M, Boca M, Woolley M, Mooney L, Dharia S, Broadfoot J, Cronin D, Schroers C, Barua NU, Longpre L, Barclay CL, Boiko C, Johnson GA, Fibiger HC, Harrison R, Lewis O, Pritchard G, Howell M, Irving C, Johnson D, Kinch S, Marshall C, Lawrence AD, Blinder S, Sossi V, Stoessl AJ, Skinner P, Mohr E, Gill SS (2019) Randomized trial of intermittent intraputamenal glial cell line-derived neurotrophic factor in Parkinson’s disease. Brain 142(3):512–525. https://doi.org/10.1093/brain/awz023
Yamaguchi Y, Miura M (2015) Programmed cell death in neurodevelopment. Dev Cell 32(4):478–490. https://doi.org/10.1016/j.devcel.2015.01.019
Yetnikoff L, Lavezzi HN, Reichard RA, Zahm DS (2014) An update on the connections of the ventral mesencephalic dopaminergic complex. Neuroscience 282:23–48. https://doi.org/10.1016/j.neuroscience.2014.04.010
Zhao M, Momma S, Delfani K, Carlen M, Cassidy RM, Johansson CB, Brismar H, Shupliakov O, Frisen J, Janson AM (2003) Evidence for neurogenesis in the adult mammalian substantia nigra. Proc Natl Acad Sci U S A 100(13):7925–7930. https://doi.org/10.1073/pnas.1131955100
Acknowledgements
We are grateful to authors are grateful to Dr. G.H. Mohan, Co-Head, Animal Care and Resource Center/Head Veterinarian for providing breeding pairs of CD-1 mice to establish the colonies. VDJ received a Movement Disorder Society (MDS) International Congress travel grant to present a part of the study at MDS Congress 2017, Canada. HY received Dept. of Biotechnology, GOI travel award to present a part at MDS Congress, 2019, at Nice, France.
Funding
The study was funded by Science and Engineering Research Board, DST, Govt. of India to PAA (No. SR/SO/HS-0121/2012). VDJ was a NIMHANS fellow and HY was a University Grants Commission (UGC) fellow.
Author information
Authors and Affiliations
Contributions
PAA conceptualised the project and obtained funds. VDJ, HY, and PAA performed the experiments and analysed the data. VDJ, HY, PAA, and TRR wrote the manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Animal experiments were performed in accordance with the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), New Delhi, India, that are based on NIH, USA guidelines. No human participants were present in the study.
Consent for Publication
All the authors have read the manuscript and agreed in its current format for the publication.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vidyadhara, D.J., Yarreiphang, H., Raju, T.R. et al. Differences in Neuronal Numbers, Morphology, and Developmental Apoptosis in Mice Nigra Provide Experimental Evidence of Ontogenic Origin of Vulnerability to Parkinson’s Disease. Neurotox Res 39, 1892–1907 (2021). https://doi.org/10.1007/s12640-021-00439-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-021-00439-6