Abstract
Introduction
The extent to which work productivity, emotional well-being, social interactions, and family life are impacted in patients who self-identify as having chronic inflammatory demyelinating polyneuropathy (CIDP) is not well characterized.
Methods
Data from an online survey of 595 individuals with self-reported CIDP, recruited by the Guillain–Barré syndrome (GBS)/CIDP Foundation, were used to assess disease and treatment burden. A total of 37% of patients were classified as “likely”, 34% as “somewhat likely”, and 28% as “unlikely” CIDP.
Results
Of ten symptoms that patients with CIDP may experience, each symptom was experienced by 77–94% of “likely”, 79–96% of “somewhat likely”, and 66–91% of “unlikely” patients. In “likely” CIDP patients 44% stopped working because of their symptoms and 24% moved to a new home. The most common treatments were intravenous immunoglobulin (IVIg) infusion and corticosteroids. IVIg was associated with venous access issues and work/school absenteeism.
Conclusions
CIDP diagnostic confirmation was not performed in any of the survey respondents. Our results do not add any knowledge on the diagnosis or treatment of CIDP. Our findings do provide insight into the symptoms that patients that think they have CIDP or have been told they have CIDP experience, explores how patients that are labeled as having CIDP view treatment expectations, and highlights how these symptoms affect home and work life. We hope that the findings are constructively used to get patients the services they need to improve quality of life, maintain employment, and ensure a safe home environment regardless of diagnostic accuracy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Patients who self-identify as having CIDP frequently report disturbance in balance, pain, and fatigue. These symptoms cannot be used to differentiate patients that are likely or unlikely to have CIDP. |
Changes in work, home, and social environments are frequently reported by patients that self identify as having CIDP. |
Because many patients are misdiagnosed with CIDP a greater emphasis on supportive management strategies and access to social services which focus on symptom management is needed. Unlike immunotherapy which is best reserved for well-defined CIDP, supportive interventions may be appropriate regardless of the accuracy of the diagnosis. |
Digital Features
This article is published with digital features to facilitate understanding of the article. You can access the digital features on the article’s associated Figshare page. To view digital features for this article go to https://doi.org/10.6084/m9.figshare.13079354.
Introduction
Chronic inflammatory demyelinating polyneuropathy (CIDP) is a rare disorder of peripheral nerves in which affected patients may experience numbness, paraesthesia, and weakness [1, 2]. Although approximately one-third of individuals have a monophasic course or are able to achieve a durable period of drug-free remission, more commonly the disease evolves over time in a slowly progressive or relapsing–remitting fashion [3, 4]. Regardless of the disease activity state, CIDP may result in substantial disability. In one study of 267 patients, mean Rankin disability score at diagnosis was 2.9, indicating a moderate level of disability where individuals could walk unassisted but required some help with day-to-day activities [5]. In another study of 94 patients, peak Rankin disability scores of level 4 or 5 were recorded in 54% of patients [6]. Collectively, these studies show that CIDP begins to erode independence early and that over half of patients are unable to walk without assistance at some point over the disease course.
The extent to which work productivity, emotional well-being, social interactions, and family life are impacted in patients who self-identify with CIDP is not well described [7]. Herein we sought to better understand the extent to which patients who carry a diagnosis of CIDP are limited in domestic and work activities. Although we have tried to put some boundaries on the accuracy of the CIDP diagnosis, it is not within the scope of this study to confirm or refute the diagnosis. This important caveat precludes the ability of a patient-reported survey to identify specific CIDP-related determinants of disability, but it does not limit the importance of understanding how symptoms that get labeled (rightly or wrongly) as CIDP lead to changes in a patient’s home, work, and social life. As these changes, and the symptoms that precede them, drive treatment and management strategies, we hope to shed insight onto factors that sometimes erroneously drive immunotherapy utilization. We aim also to use these survey results to highlight the importance of supportive management strategies and social service resource allocation for patients who are diagnosed and misdiagnosed with CIDP.
Methods
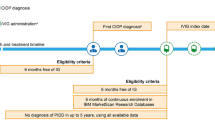
An online global Guillain–Barré syndrome (GBS)/CIDP Foundation survey from 595 adult patients was used to assess symptom and treatment burden in patients who self-reported CIDP. The survey was provided to USA-based members of the GBS/CIDP database between November 13, 2017 and December 12, 2017 and provided to non-USA-based members between January 5, 2018 and March 26, 2018. The survey consisted of 56 questions that gathered information about demographics, symptoms, work and social activities, and treatment history. Embedded within the survey were standardized patient-reported outcome measures including the Inflammatory Rasch-built Overall Disability Scale (I-RODS) questions and the Patient-Reported Outcomes Measurement Information System Physical Function (PROMIS PF) Short Form questions. All respondents provided informed consent as part of the survey completion, and ethics approvals were exempt for this type of survey analysis as The New England Independent Review Board (NEIRB) reviewed and deemed the study as exempt from NEIRB review.
Patients were stratified into “likely”, “somewhat likely”, and “unlikely” CIDP categories on the basis of patient-reported symptoms such as weakness, symptom progression, and nerve conduction study completion (Table 1). While we wanted to make some attempt to exclude patients without CIDP, we erred on the side of inclusivity as our focus was on symptom burden without necessarily attributing the symptoms to a confirmed specific disease state.
Responses were analyzed to determine the number and rank severity of symptoms experienced from a pre-defined list of symptoms. The impact of symptoms on respondents’ professional life, emotional well-being, living circumstances, mobility, and overall quality of life (QoL) were determined. This analysis included number of days missed from work/school as a result of symptoms versus as a result of intravenous immunoglobulin (IVIg) infusions during the past month. The reported treatment burden across common treatments was compared, including discontinuations and reasons for discontinuation. Descriptive statistics, including counts and percentages, were used for categorical (nominal or dichotomous) variables; mean, median, and standard deviations were used for continuous variables. Significance was assessed using z tests and Bonferroni correction was undertaken and samples were adjusted for pairwise comparisons.
Results
Respondent Demographics
Of the 595 respondents, 475 were USA-based and 120 were based outside the USA (Australia, Canada, France, Ireland, Italy, New Zealand, Spain, South Africa, and the UK). A total of 37% (n = 222) of patients were classified as “likely”, 34% (n = 204) “somewhat likely”, and 28% (n = 169) of patients were classified as “unlikely” CIDP (Table 2). Gender, employment status at diagnosis, treatment at the time of survey completion, and distribution of age at diagnosis was similar among the three groups, with the exception of a higher frequency of patients at older age at diagnosis (age 70–79 years) in the “unlikely” group (Table 3). In addition to IVIg and corticosteroids, respondents also indicated use of subcutaneous immunoglobulin infusion, plasma exchange, immunosuppressive agents, combination therapy of IVIg plus corticosteroids, and “other” treatments as current treatments.
Symptom Burden
Respondents indicated whether they had ever experienced any symptoms from a list of ten symptoms. Loss of balance/coordination, leg weakness, fatigue, and paresthesia were the most commonly reported symptoms (Fig. 1a). “Likely” and “somewhat likely” participants reported an increased frequency of any given symptom (Fig. 1a) and more symptoms overall (Fig. 1b) than the “unlikely” group. Significantly more patients in the “likely” group experienced weakness in the hips (p < 0.01), weakness in the feet (p < 0.01), tingling (p = 0.04), and pain (p = 0.01) compared with “unlikely” patients; significantly more “somewhat likely” patients experienced weakness in the shoulders (p = 0.01), weakness in the hips (p < 0.01), heaviness in the legs (p = 0.03), weakness in the feet (p < 0.01), pain (p < 0.01), or loss of balance (p < 0.01) compared with “unlikely” patients.
In all three groups the symptom that was most bothersome to patients (Fig. 2a) and the symptom that patients most wanted relief from (Table 2b) was loss of balance/coordination. Although for all groups leg weakness was the second most bothersome symptom at the start of treatment, at the time of the survey, pain management was more important than leg weakness. Fatigue also grew in importance from the time treatment started (5th most important) to the time of the survey (4th most important). There were no statistically significant differences in the frequency of any given symptom being bothersome between the three groups.
Participants were generally not optimistic that their symptoms would improve, and frequently reported concerns about disease progression, dependency on others, and disease-associated financial hardships (Fig. 3). Although the concerns were generally expressed more often in the “likely” and “somewhat likely” groups, only the financial aspects showed significant differences (Concerned that cost of treatments will lead to financial struggles in other aspects of my life: “likely” and “somewhat likely” significantly higher [p = 0.03 and p < 0.01, respectively] than “unlikely”; I won’t be able to afford my CIDP treatment: “somewhat likely” significantly higher than “unlikely”; p = 0.03). More than half of “likely” and “somewhat likely” respondents either stopped working or changed the way the worked (i.e., reduced hours, changed employer, changed occupation) because of their symptoms (Fig. 4a). “Unlikely” respondents changed their work environment less often, with significantly fewer “unlikely” respondents having to go on disability/start claiming disabled pay than “likely” and “somewhat likely” respondents and significantly more not having to change employment status and significantly fewer “unlikely” respondents stopping work. Respondents also made frequent modifications to their living environment to meet their disability needs, most often through home renovations or the addition of durable medical equipment (Fig. 4b). Over half of respondents in each group either made changes to their residence or moved residencies altogether.
Survey responses of “always” or “often”* on feelings about their future. *Respondent options: Never, Rarely, Sometimes, Often, Always. N=593 for questions ‘I won’t be able to afford my CIDP treatment’ and ‘Concerned that costs of treatment will lead to other financial struggles’, all other questions N=595
Treatment Burden
A total of 94% “likely”, 91% “somewhat likely”, and 88% “unlikely respondents had received IVIg at some point during their disease course (Fig. 5a). In each group about a third of respondents had discontinued IVIg by the time of the survey. The most common reason cited for IVIg discontinuation was because it was not felt to be beneficial; however, some patients discontinued as no more symptoms were experienced following IVIg. About 30% of respondents in each group that received IVIg missed school or work because of the infusions (Fig. 6). Among IVIg patients in each group, 48% of “likely”, 51% of “somewhat likely”, and 54% of “unlikely” CIDP patients did not miss work or school because they “were not in work or school”. Most (80% “likely”, 73% “somewhat likely”, 82% “unlikely”) IVIg users received infusions via a temporary IV line inserted for each infusion. Twelve percent of respondents reported that on at least one occasion the infusion was unable to be completed because of inadequate peripheral access.
Corticosteroids were prescribed at some point during the disease course in 63% “likely”, 63% “somewhat likely”, and 49% “unlikely” respondents (Fig. 5b). In each group about 2/3rd of respondents had discontinued corticosteroids by the time of the survey. Lack of benefit and poor tolerability were frequently cited as reasons for discontinuation in all groups.
Discussion
It is important to clarify that the results of this survey represent patients who self-identify as having CIDP and not patients with a confirmed diagnosis. Confirmatory review of clinical records, electrophysiological studies, and other CIDP supporting and exclusionary data were not within the scope of this study. On the basis of prior studies that explored the frequency of CIDP misdiagnosis, we can project that between 50% and 90% of respondents to our survey were misdiagnosed as having CIDP [8,9,10]. Even though efforts were made to identify a population of survey respondents who were more likely to have CIDP, we were still unable to confirm or exclude any particular diagnosis. As such, our results should not be interpreted as the characteristics that define CIDP. Instead, our findings provide insight into the concerns of patients who self-identify as having CIDP, whether the diagnosis is accurate or not.
We show here that the symptoms that are most troubling to patients and the degree to which those symptoms impact work and home life are similar in the “likely”, somewhat likely”, and “unlikely” CIDP cohorts. This observation highlights the importance of utilizing diagnostic guidelines during routine clinical care as a means of proportionally integrating the clinical features with characteristic electrophysiologic and laboratory abnormalities [11]. Considering that no symptom is by itself diagnostic of CIDP, and pain and fatigue (reported frequently by survey respondents) are especially difficult to interpret, routine use of guidelines may be an invaluable resource to improve CIDP diagnostic accuracy.
We show here that while patients who self-identify as having CIDP have many concerns, disturbance in balance is frequently the most bothersome and the symptom that patients most commonly want to improve with treatment. Considering that gait performance has been shown to correlate with quality of life in patients with CIDP [12] and functional status in patients without CIDP [13], this observation is both important and expected. Pain, weakness in the hips/legs, and fatigue are other symptoms that patients are frequently looking to improve. If the diagnosis is correct, then initiation of immunotherapy may lead to improvement (although not necessarily resolution) in symptoms in 80–90% of patients [5]. When the diagnosis is wrong, the (mis)diagnosis of CIDP may erroneously drive the treatment plan, disregarding potentially more effective supportive management strategies as well as diverting from the true causation of the symptoms. Our findings show that there are many opportunities to intervene with supportive management, and it is important that these are not overlooked. Just as would be the case for polyneuropathies of other causes, we encourage consideration of physical therapy for balance and gait, ability-appropriate exercise for fatigue, pain management, and durable medical equipment evaluations for all patients thought to have CIDP independent of immunotherapy.
The impact of chronic diseases on patients can extend beyond the physical symptoms. Chronic illness may lead to deterioration in emotional well-being and, in combination with the physical limitations, result in substantial changes in work activities, social interactions, and family life [9]. Again, our findings do not provide any meaningful data on the frequency with which CIDP leads to changes in work life or living situations. However, the shear frequency with which patients who self-identify as having CIDP change their work environment, stop working, modify their homes, or change living environments is striking. In addition to supportive care for the physical limitations of the illness, our findings also speak to the importance of allocating resources to patients with chronic illness such that they can access the necessary social services needed to mitigate the impact of changes to work and home environments.
This study has several limitations, the most important of which is the absence of diagnostic confirmation in any of the survey respondents. While this limitation precludes commentary on the disease burden in patients with CIDP, our findings are still informative on what is important to individuals who self-identify as having CIDP and how they are affected at work and at home. By highlighting that the burden is substantial in this group of patients, but that none of the burden is specific to CIDP, we hope that our findings reinforce the importance of utilization of supportive management strategies and social support networks. Our study is further limited by factors inherent to patient-reported surveys, including recall bias and the inability to verify the accuracy and reliability of self-reported data. We attempted to minimize such limitations by using approaches from similar surveys successfully conducted for other disease states [10, 14]. Because our study attempted to gather information from a worldwide population of patients, we cannot exclude the possibility that cultural bias was introduced. Although the majority of respondents were USA-based, when viewed as isolated cohorts the difference between USA and non-USA were generally minimal.
Conclusions
This study adds no new information on the diagnosis or treatment of CIDP. Our data does not reflect symptoms or disability in patients with CIDP as we were not able to confirm the diagnosis in any of the survey respondents. Rather, our findings may be helpful to better understand how patients that think they have CIDP or have been told they have CIDP view their expectations of treatment and how their symptoms impact home and work life. By highlighting the distinction between symptom and diagnosis, we hope to optimize supportive management approaches and minimize immunotherapy overutilization in cases where it may not be needed. We show herein that patients who self-identify as having CIDP are frequently worried about their condition. The prospect of becoming dependent on others is daunting. Financial concerns directly or indirectly related to treatment are common as well. There is generally a lack of optimism that symptoms will improve. Even without knowing the actual diagnosis of patients who completed the survey, this information may be helpful to efficiently direct resources to patients who are struggling to maintain their normal level of functionality. We hope that the findings are constructively used to get patients the services they need to improve quality of life, maintain employment, and ensure a safe home environment. While immunotherapy is an important part of treatment for patients with CIDP, it is only but one part of the treatment paradigm and only applicable to patients correctly diagnosed. Conversely, supportive management strategies including access to social support networks play an instrumental role when maximizing individual functionality and well-being and are appropriate regardless of diagnostic accuracy.
Change history
13 January 2021
A Correction to this paper has been published: https://doi.org/10.1007/s12325-020-01586-6
References
Dalakas MC. Advances in the diagnosis, pathogenesis and treatment of CIDP. Nat Rev Neurol. 2011;7(9):507–17.
Mathey EK, Park SB, Hughes RA, et al. Chronic inflammatory demyelinating polyradiculoneuropathy: from pathology to phenotype. J Neurol Neurosurg Psychiatry. 2015;86(9):973–85.
Chio A, Cocito D, Bottacchi E, et al. Idiopathic chronic inflammatory demyelinating polyneuropathy: an epidemiological study in Italy. J Neurol Neurosurg Psychiatry. 2007;78(12):1349–53.
Gorson KC, van Schaik IN, Merkies IS, et al. Chronic inflammatory demyelinating polyneuropathy disease activity status: recommendations for clinical research standards and use in clinical practice. J Peripher Nerv Syst. 2010;15(4):326–33.
Cocito D, Paolasso I, Antonini G, et al. A nationwide retrospective analysis on the effect of immune therapies in patients with chronic inflammatory demyelinating polyradiculoneuropathy. Eur J Neurol. 2010;17(2):289–94.
Lunn MP, Manji H, Choudhary PP, Hughes RA, Thomas PK. Chronic inflammatory demyelinating polyradiculoneuropathy: a prevalence study in south east England. J Neurol Neurosurg Psychiatry. 1999;66(5):677–80.
Megari K. Quality of life in chronic disease patients. Health Psychol Res. 2013;1(3):e27.
Levine TD, Katz JS, Barohn R, et al. Review process for IVIg treatment: lessons learned from INSIGHTS neuropathy study. Neurol Clin Pract. 2018;8(5):429–36.
Merkies IS, Kieseier BC. Fatigue, pain, anxiety and depression in Guillain-Barre Syndrome and chronic inflammatory demyelinating polyradiculoneuropathy. Eur Neurol. 2016;75(3–4):199–206.
Gibofsky A, Galloway J, Kekow J, et al. Comparison of patient and physician perspectives in the management of rheumatoid arthritis: results from global physician- and patient-based surveys. Health Qual Life Outcomes. 2018;16(1):211.
Van den Bergh PY, Hadden RD, Bouche P, et al. European Federation of Neurological Societies/Peripheral Nerve Society guideline on management of chronic inflammatory demyelinating polyradiculoneuropathy: report of a joint task force of the European Federation of Neurological Societies and the Peripheral Nerve Society - first revision. Eur J Neurol. 2010;17(3):356–63.
Ryltoft AK. Al-Zuhairy A, Sindrup SH, Andersen H, Markvardsen LK. Quality of life in chronic inflammatory demyelinating polyneuropathy patients treated with subcutaneous immunoglobulin. Acta Neurol Scand. 2020. https://doi.org/10.1111/ane.13322.
Venkataraman K, Tai BC, Khoo EYH, et al. Short-term strength and balance training does not improve quality of life but improves functional status in individuals with diabetic peripheral neuropathy: a randomised controlled trial. Diabetologia. 2019;62(12):2200–10.
Sunkureddi P, Gibson D, Doogan S, Heid J, Benosman S, Park Y. Using self-reported patient experiences to understand patient burden: learnings from digital patient communities in ankylosing spondylitis. Adv Ther. 2018;35(3):424–37.
Acknowledgements
We thank Bryter for their development and implementation of the survey.
Funding
This study and journal’s rapid service and open access fees were funded by CSL Behring.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Authorship Contributions
All authors contributed to the study design, data interpretation, review and approval of the manuscript.
Medical Writing and Editorial Assistance
Editorial assistance was provided by Joanna Musgrove of Meridian HealthComms, funded by CSL Behring.
Disclosures
Jeffrey A. Allen is an employee of the University of Minnesota; has received grants from CSL Behring, consulting fees/honorarium from Akcea, Alexion, Argenx, Momenta, CSL Behring and Biotest; received support for travel to meetings for the study from CSL Behring and payment for lectures from Akcea and CSL Behring. Lisa Butler has nothing to disclose. Todd Levine has received consulting fees/honoraria from Alexion and Grifols. Anne Haudrich is an employee of CSL Behring and holds stock in CSL Behring.
Compliance with Ethics Guidelines
The New England Independent Review Board (NEIRB) reviewed and deemed the study as exempt from NEIRB review. All respondents provided informed consent as part of the survey completion.
Data Availability
CSL will only consider requests to share Individual Patient Data (IPD) that are received from systematic review groups or bona-fide researchers. CSL will not process or act on IPD requests until 12 months after article publication on a public website. An IPD request will not be considered by CSL unless the proposed research question seeks to answer a significant and unknown medical science or patient care question. Applicable country specific privacy and other laws and regulations will be considered and may prevent sharing of IPD.
Requests for use of the IPD will be reviewed by an internal CSL review committee. If the request is approved, and the researcher agrees to the applicable terms and conditions in a data sharing agreement, IPD that has been appropriately anonymized will be made available. Supporting documents including study protocol and Statistical Analysis Plan will also be provided.
For information on the process and requirements for submitting a voluntary data sharing request for IPD, please contact CSL at clinicaltrials@cslbehring.com.
Author information
Authors and Affiliations
Corresponding author
Additional information
The original online version of this article was revised due to caption were missing for figure 2 and 3.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Allen, J.A., Butler, L., Levine, T. et al. A Global Survey of Disease Burden in Patients Who Carry a Diagnosis of Chronic Inflammatory Demyelinating Polyneuropathy. Adv Ther 38, 316–328 (2021). https://doi.org/10.1007/s12325-020-01540-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-020-01540-6