Abstract
Inhaled therapy is the cornerstone of asthma management in that it optimizes the delivery of the medication to the site of action. The effectiveness of inhaled therapy is affected by the correct choice of the device and proper inhalation technique. In fact, this influences the drug delivery and distribution along the bronchial tree, including the most peripheral airways. In this context, accumulating evidence supports the contribution of small airways in asthma, and these have become an important target of treatment. In reality, the “ideal inhaler” does not exist, and not all inhalers are the same. Advances in technology has highlighted these differences, and have led to the design of new devices and the development of formulations characterized by extrafine particles that facilitate the distribution and deposition of the drug particles along the respiratory tract. In addition, efforts have been made to implement adherence to chronic treatment, which translates into clinical benefit. Taken together, the optimal control of asthma depends on the drug that is selected, the device that is employed and the removal of factors that reduce patient’s adherence to therapy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction: Asthma Control and its Obstacles
Current asthma management guidelines emphasize the importance of disease control [1]. Despite the availability of effective drug therapies and relevant advances in inhaler technology, real-life data clearly show that the level of asthma control is still unacceptably low [2]. Asthma control consists of two domains: symptom control and management of future risk of adverse outcomes [1]. Inhaled therapy is the cornerstone of asthma management, in that it optimizes the delivery of the medication to the site of action as opposed to systemic administration of the drug [1]. In addition to the proper choice of the active drug, the selection of the correct device plays a crucial role in achieving optimal control and preventing future risk. The selection of the inhaler device should be primarily based on the specific needs of the individual patients, bearing in mind that the “ideal” inhaler does not exist in real life, but is rather the “right” inhaler for the “right” patient.
This review provides a critical analysis of factors related to both drug formulation and device features that can have an impact on asthma management. The efficacy of a treatment is linked also to patient adherence, and developments in drugs and devices over the past decades have consistently contributed to improve adherence to inhaled treatment and, possibly, drug delivery to the entire bronchial tree [3, 4]. It is known that low drug deposition and device mishandling are associated with poor disease control and increased consumption of healthcare resources [5]. All these aspects will be discussed to better understanding how these factors should be considered as part of the decision-making process, leading to the choice of an inhaler device.
The Patient and the Physician
Misuse of an inhaler is common in clinical practice, and proper training of patients and physicians is important to ensure the correct use of the device. Patient technique has a large impact on the amount of active drug delivered to the site of action and, therefore, on the efficacy and disease control [3]. A study by Harnett et al. [6] demonstrated the importance of educating and formally assessing inhaler technique in patients with asthma as part of their ongoing clinical review. The first step to ensure proper management of the disease is therefore to educate patients in inhaler technique. Patient satisfaction is also an important aspect, as it significantly correlates with better outcomes [3]. On the other hand, little attention is given to the training of the physicians, and to the patient-to-physician education interaction. Current guidelines recommend that physicians follow adherence management strategies; however, evidence for these interventions is weak [1]. It is estimated that 39–67% of nurses, doctors, and respiratory therapists are not able to adequately train patients on the correct use of the device [3]. Perhaps, physicians’ choice of the device is also based on how confident he/she feels in explaining the use of the inhaler to their patients. This has clinical implications in daily practice. In addition, the interaction between patients and physicians often does not incorporate any agreement on the choice of the device, and the level of compliance with the prescribed treatment is not routinely assessed, and this is becoming a challenge in primary care. Healthcare providers cannot assume that correct inhaler use is self-evident [7], and cannot rely on patients following instructions provided in inhaler packages [8]. Patients may misreport non-adherence, or avoid embarrassing questions on the use of the inhaler, to avoid disappointing their physicians who may not actively inquire about it [3]. It is logical to expect that this phenomenon would invariably lead to the progressive loss of treatment efficacy.
In this scenario, the value of other healthcare professionals has been increasingly recognized to support (or replace) physicians. Certified Respiratory Educators (CREs) have been demonstrated to properly manage chronic respiratory patients as effectively as specialists [9]. CREs such as nurses, respiratory therapists, physiotherapists and pharmacists supply services traditionally delivered by physicians. This may include patient education, with an emphasis on patient self-management and instruction in proper inhaler technique [10].
Drug Formulation and Device Characteristics
Inhalation technique is critical to achieve treatment success and differs substantially between inhaler devices [11]. Four different classes of device are currently available for pulmonary drug delivery: pressurized metered dose inhalers (pMDIs), dry powder inhalers (DPIs), soft mist inhalers (SMIs) and nebulizers. All these devices have both advantages and limitations [11]. As a general rule, pMDIs require good coordination, which is not always obtained in obstructed subjects. To overcome this limitation, the use of a spacer with pMDIs is often recommended, especially for patients with known or suspected poor coordination (e.g., children and the elderly). Spacers reduce the risk of cold-Freon effect and the need for perfect hand–breath coordination [11]. DPIs require a turbulent flow of air to transform the metered powder formulation combined with a lactose carrier into disaggregated particles. For DPIs, the inhalation flow must be maintained from the start of inhalation for an extended period of time, and patients are required to breathe as deeply and forcefully as possible to allow the coarse particles combined with a lactose carrier to disaggregate. In reality, patients have a variable degree of airflow limitation, with consequently lower inspiratory flows compared to healthy subjects [12]. This may affect the distribution of the active compounds in the lung [12]. As mentioned earlier, the “ideal inhaler” does not exist. Not all inhalers are the same, and advances in technology have highlighted their differences. This is a paradox, since the scopes of these devices are to simplify the steps of usage, and to increase the amount of drug deposited in the lungs. Nowadays, it is plausible to assume that switching from one device to another may have dramatic consequences on loss of asthma control.
Several factors play a primary role in determining the efficacy of the drug deposition in the large and small airways, and are related to the characteristics of both the drug formulation and the device [13]. The ideal device should allow high levels of drug deposition in the lung while minimizing the deposition outside the lung. To accomplish this, the inhaler should incorporate two main characteristics. First, ease of use and feedback systems to optimize the patient’s technique/coordination and to reassure them that the drug has been administered. Secondly, a uniform and consistent deposition of the drug between doses. A consensus statement by the task force of the European Respiratory Society (ERS) and the International Society for Aerosols in Medicine (ISAM) provides clear recommendations for choosing the best aerosol delivery device based on a patient’s actuation–inhalation coordination, level of inspiratory flow and clinical conditions [14]. For example, some inhalers require strong inspiratory force, which may not be possible in emergency situations or in children and elderly.
Are All Inhalers the Same?
Over the years, inhalers have undergone improvements in terms of technical design. Special efforts have been made to improve the relationship between inspiratory flow rate and actuation, as well as to the characteristics of propellant and reproducibility of the dose between inhalations. pMDIs are all similar in their mode of use, but they invariably differ in terms of homogeneity of the concentration of the drug and, consequently, the emission of a constant amount of drug between doses. In suspension formulations, the active drug is not soluble in the propellant, thus remaining in solid powder form in the container. It follows that formulations in suspension must be shaken until uniformity of distribution within the canister is reached before use. In clinical settings, this is one of the most common critical errors [5]. Switching to solution formulations overcomes the issue of non-uniform distribution and can allow extrafine drug delivery, reducing the particle size of emitted aerosol and, thereby, allowing deeper penetration in the bronchial tree [15]. Functional changes that occur in the smallest airways have been linked to features of severe uncontrolled disease [16, 17], so it is therefore plausible to assume that the extrafine formulations that act on peripheral airway abnormalities [18–20] could also improve asthma control, as demonstrated by randomized trials [21] and real-life observational studies [22, 23]. Breath-activated pMDI (BA-pMDI) devices are also available, which release the drug when inhalation triggers the metered dose inhaler. Currently, there is only one commercially available soft mist inhaler: Respimat® (Boehringer Ingelheim, Ingelheim am Rhein, Germany). This inhaler atomizes the drug solution using mechanical energy imparted by a spring, producing a fine, slow-moving mist, with less deposition in the mouth and throat and relatively higher lung deposition.
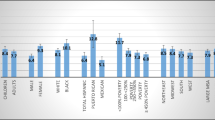
Three types of DPIs are available with different handling instructions: single dose [Breezhaler® (Novartis International AG, Basel, Switzerland), HandiHaler® (Boehringer Ingelheim GmbH, Ingelheim am Rhein, Germany), Aerolizer® (Novartis International AG, Basel, Switzerland)], multiple dose [Diskus® (GlaxoSmithKline, Uxbridge, UK. Durable Sidestream)] and reservoir [Turbuhaler® (AstraZeneca Plc, London UK), NEXThaler® (Chiesi Farmaceutica SPA, Parma, Italy)]. DPIs contain powdered drug that is dispersed into particles after activation by an inspiratory maneuver. Unlike pMDIs, DPIs differ from one to another, and the required steps to activate them may vary significantly [14]. The major advantage of DPIs is that since they are breath actuated, they do not need any coordination between activation and inhalation [14]. The major disadvantage is the fact that the powders contained in the DPI can be hygroscopic, and, for this reason, they can attract humidity and stick to the nozzle walls [14]. In addition, an adequate inspiratory flow is required to activate the device. Each DPI offers a different resistance to inhalation and needs a specific threshold inspiratory flow to accurately deliver the dose [14]. Recent devices have been designed with the aim of simplifying inhalation maneuvers. In this regard, the NEXThaler, Ellipta® (GlaxoSmithKline, Uxbridge, UK) and Spiromax® (Teva Pharma B.V., Utrecht, The Netherlands) devices are novel, intuitively designed DPIs conceived for straightforward open–inhale–close operation that ensures ease of use. In comparison to the other DPIs available, the NEXThaler is able to ensure a consistent full-dose delivery independently from the patient’s inhalation profile. Once the breath-actuated mechanism (BAM) is activated, the NEXThaler guarantees, at each inhalation, that the full therapeutic dose is delivered [24]. Therefore, by checking the dose counter both patients and doctors are able to understand if the drug has been effectively inhaled. The NEXThaler is the first DPI that has been developed to generate extrafine particles [4]. The currently available inhaled corticosteroid/long-acting beta agonist fixed combinations delivered through DPIs are not able to dispense extrafine shots with a mass median aerodynamic diameter (MMAD) lower than 2 μm [19]. The extrafine beclometasone dipropionate/formoterol (BDP/F) fixed combination therefore represents the only extrafine combination in both pMDI and DPI formulations developed so far (Fig. 1).
Dimensions of the particles of the inhaled fixed combination formulations in terms of mass median aerodynamic diameter (MMAD). BDP/F beclometasone dipropionate/formoterol, BUD/F budesonide/formoterol, DPI dry powder inhaler, FP/Salm fluticasone propionate/salmeterol, FP/F fluticasone propionate/formoterol, pMDI pressurized metered dose inhaler
Particle size is an important feature to be considered when comparing different devices since the particle dimension influences drug deposition in the lungs [4]. Bigger particles have a higher kinetic energy and, for this reason, they impact on the airway walls, while small particles can also reach the peripheral airways. Therefore, lung deposition of a drug formulation depends on the MMAD, which is the droplet size at which half of the mass of the aerosol is contained in smaller droplets and half in larger droplets. It also depends on the fine particle fraction (FPF), meaning the percentage of particles <5 μm in diameter [4]. Generally, FPF is proportional to the fraction of emitted dose reaching the lung. Particles with an MMAD >5 μm tend to accumulate in the oropharynx, while particles with an MMAD between 1 and 5 μm tend to reach the airways at different levels according to their dimensions [4]. Obviously, the smaller the particles (between 1 and 5 μm), the higher the proportion of particles reaching the peripheral airways [25]. The advantages in terms of asthma control with the use of extrafine formulations have been shown in real-life studies [22, 23, 26–29]. These findings obtained in real-life studies may benefit from further confirmation in randomized controlled trials.
Poor adherence with asthma management plans and treatment regimens has been associated with poor disease control [30] and increased risk of hospital admission [31]. Patient and parental reports of adherence are often inaccurate [32], and also physicians’ subjective impressions may be equally unreliable [33]. Monitoring devices characterized by smart technology should ideally be able to monitor adherence covertly, accurately record the time that each dose was taken, store data over a reasonable time period, detect multiple successive actuations and provide access to data that may be downloaded to a personal computer [34]. It is logical to imply that “smart technology” would improve patient adherence, with a possible positive impact on asthma control. This needs to be proven in large population-based studies.
How Does the Inhaler Improve Adherence to Treatment?
The challenge of patients not following medical advice dates back to the 4th century BC, when Hippocrates observed that some patients were not taking their prescribed treatments [35]. Adherence is usually dichotomized for research purposes and is often defined as missing at least 20% of the medication. This cutoff has been demonstrated to predict subsequent hospitalization across several chronic conditions [36]. Adherence to therapy is likely to be influenced by patients’ attitudes and their experience in using the device. If patients feel that treatment is not working, adherence is likely to be poor with consequently reduced efficacy of treatment [37]. The availability of several inhaler devices may also confuse the patient. Switching between different inhalers negatively affects care, as inhaler classes and brands differ in design (particularly DPIs) and each device has unique required steps and inhalation techniques [38]. Booker showed that half of asthmatic patients were reluctant to change their current DPI to an alternative device, and the majority reported concerns about being trained in the use of a new device or confusion around the need for change [39]. Regular training sessions should be provided at each visit and patients should be encouraged to bring their inhalers to provide demonstration of competence in inhalation technique.
The clinical variability of asthma makes the disease particularly prone to erratic adherence. Patients usually tend to reduce adherence during the asymptomatic phases of their disease. Unintentional adherence often occurs when, for example, a patient does not understand the proper technique for inhaler use. In this regard, improper use of inhaler devices has been shown to be associated with loss of asthma control [40, 41], presumably contributing to increased disease burden for caregivers and society. Customized patient-friendly treatment that anticipates and accommodates usual behavior and addresses conscious and unconscious medication beliefs is more likely to achieve the desired goal of disease control [42].
Conclusion
Optimal control of asthma depends on the drug that is selected, the device that is employed and the removal of factors that reduce patients’ adherence to therapy. In this regard, while international guidelines recommend careful attention to the choice of the device and the education to patients, a consensus is lacking on how to properly choose appropriate inhaler devices. This implies that factors that are not clinically relevant may, however, influence the choice of the optimal inhaler device. Simplistic “one size fits all” approaches are not the solution. Rather, a personalized selection of the device based on patient preferences and perceptions should be encouraged and implemented in clinical practice. Most probably, differences in efficacy become trivial, or even disappear, when the device is used with the proper inhalation technique. The key issue in asthma management is therefore to train patients and to verify the correct inhalation maneuver.
References
Global Initiative for Asthma (GINA) 2014. Global strategy for asthma management and prevention [online]. http://www.ginasthma.com. Accessed Dec 4, 2014.
Demoly P, Annunziata K, Gubba E, Adamek L. Repeated cross-sectional survey of patient-reported asthma control in Europe in the past 5 years. Eur Respir Rev. 2012;21(123):66–74.
Bjermer L. The importance of continuity in inhaler device choice for asthma and chronic obstructive pulmonary disease. Respiration. 2014;88(4):346–52.
Scichilone N, Spatafora M, Battaglia S, Arrigo R, Benfante A, Bellia V. Lung penetration and patient adherence considerations in the management of asthma: role of extra-fine formulations. J Asthma Allergy. 2013;6:11–21.
Melani AS, Bonavia M, Cilenti V, et al. Inhaler mishandling remains common in real life and is associated with reduced disease control. Respir Med. 2011;105(6):930–8.
Harnett CM, Hunt EB, Bowen BR, et al. A study to assess inhaler technique and its potential impact on asthma control in patients attending an asthma clinic. J Asthma. 2014;51(4):440–5.
Hanania NA, Wittman R, Kesten S, Chapman KR. Medical personnel’s knowledge of and ability to use inhaling devices. Metered-dose inhalers, spacing chambers, and breath-actuated dry powder inhalers. Chest. 1994;105(1):111–6.
Souza ML, Meneghini AC, Ferraz E, Vianna EO, Borges MC. Knowledge of and technique for using inhalation devices among asthma patients and COPD patients. J Bras Pneumol. 2009;35(9):824–31.
Field SK, Conley DP, Thawer AM, Leigh R, Cowie RL. Assessment and management of patients with chronic cough by Certified Respiratory Educators: a randomized controlled trial. Can Respir J. 2009;16(2):49–54.
Cicutto L. Supporting successful asthma management in schools: the role of asthma care providers. J Allergy Clin Immunol. 2009;124(2):390–3.
Lavorini F, Fontana GA, Usmani OS. New inhaler devices—the good, the bad and the ugly. Respiration. 2014;88(1):3–15.
Wieshammer S, Dreyhaupt J. Dry powder inhalers: which factors determine the frequency of handling errors? Respiration. 2008;75(1):18–25.
Labiris NR, Dolovich MB. Pulmonary drug delivery. Part II: the role of inhalant delivery devices and drug formulations in therapeutic effectiveness of aerosolized medications. Br J Clin Pharmacol. 2003;56(6):600–12.
Laube BL, Janssens HM, de Jongh FH, European Respiratory Society, International Society for Aerosols in Medicine, et al. What the pulmonary specialist should know about the new inhalation therapies. Eur Respir J. 2011;37(6):1308–31.
Nicolini G, Scichilone N, Bizzi A, Papi A, Fabbri LM. Beclomethasone/formoterol fixed combination for the management of asthma: patient considerations. Ther Clin Risk Manag. 2008;4(5):855–64.
Sorkness RL, Bleecker ER, Busse WW, et al. Lung function in adults with stable but severe asthma: air trapping and incomplete reversal of obstruction with bronchodilation. J Appl Physiol (1985). 2008;104(2):394–403.
Berry M, Hargadon B, Morgan A, et al. Alveolar nitric oxide in adults with asthma: evidence of distal lung inflammation in refractory asthma. Eur Respir J. 2005;25(6):986–91.
Tunon-de-Lara JM, Laurent F, Giraud V, et al. Air trapping in mild and moderate asthma: effect of inhaled corticosteroids. J Allergy Clin Immunol. 2007;119:583–90.
Fabbri LM, Nicolini G, Olivieri D, Papi A. Inhaled beclometasone dipropionate/formoterol extra-fine fixed combination in the treatment of asthma: evidence and future perspectives. Expert Opin Pharmacother. 2008;9(3):479–90.
Scichilone N, Battaglia S, Sorino C, et al. Effects of extra-fine inhaled beclomethasone/formoterol on large and small airways in asthma. Allergy. 2010;65(7):897–902.
Huchon G, Magnussen H, Chuchalin A, Dymek L, Gonod FB, Bousquet J. Lung function and asthma control with beclomethasone and formoterol in a single inhaler. Respir Med. 2009;103(1):41–9.
Müller V, Gálffy G, Eszes N, et al. Asthma control in patients receiving inhaled corticosteroid and long-acting beta2-agonist fixed combinations. A real-life study comparing dry powder inhalers and a pressurized metered dose inhaler extrafine formulation. BMC Pulm Med. 2011;11:40.
Allegra L, Cremonesi G, Girbino G, PRISMA (PRospectIve Study on asthMA control) Study Group, et al. Real-life prospective study on asthma control in Italy: cross-sectional phase results. Respir Med. 2012;106(2):205–14.
Corradi M, Chrystyn H, Cosio BG, et al. NEXThaler, an innovative dry powder inhaler delivering an extrafine fixed combination of beclometasone and formoterol to treat large and small airways in asthma. Expert Opin Drug Deliv. 2014;11(9):1497–506.
Usmani OS, Biddiscombe MF, Barnes PJ. Regional lung deposition and bronchodilator response as a function of beta2-agonist particle size. Am J Respir Crit Care Med. 2005;172(12):1497–504.
Barnes N, Price D, Colice G, et al. Asthma control with extrafine-particle hydrofluoroalkane-beclometasone vs. large-particle chlorofluorocarbon-beclometasone: a real-world observational study. Clin Exp Allergy. 2011;41(11):1521–32.
Martin RJ, Price D, Roche N, et al. Cost-effectiveness of initiating extrafine- or standard size-particle inhaled corticosteroid for asthma in two health-care systems: a retrospective matched cohort study. NPJ Prim Care Respir Med. 2014;9(24):14081.
Price D, Thomas M, Haughney J, et al. Real-life comparison of beclometasone dipropionate as an extrafine- or larger-particle formulation for asthma. Respir Med. 2013;107(7):987–1000.
Terzano C, Cremonesi G, Girbino G, PRISMA (PRospectIve Study on asthMA control) Study Group, et al. 1-year prospective real life monitoring of asthma control and quality of life in Italy. Respir Res. 2012;13:112.
Horn CR, Clark TJ, Cochrane GM. Compliance with inhaled therapy and morbidity from asthma. Respir Med. 1990;84(1):67–70.
Ordonez GA, Phelan PD, Olinsky A, et al. Preventable factors in hospital admissions for asthma. Arch Dis Child. 1998;78(2):143–7.
Coutts JA, Gibson NA, Paton JY. Measuring compliance with inhaled medication in asthma. Arch Dis Child. 1992;67(3):332–3.
Mushlin AI, Appel FA. Diagnosing potential noncompliance. Physicians’ ability in a behavioral dimension of medical care. Arch Intern Med. 1977;137(3):318–21.
Burgess SW, Wilson SS, Cooper DM, et al. In vitro evaluation of an asthma dosing device: the smart-inhaler. Respir Med. 2006;100(5):841–5.
Lerner BH. From careless consumptives to recalcitrant patients: the historical construction of noncompliance. Soc Sci Med. 1997;45(9):1423–31.
Karve S, Cleves MA, Helm M, Hudson TJ, West DS, Martin BC. Good and poor adherence: optimal cut-point for adherence measures using administrative claims data. Curr Med Res Opin. 2009;25(9):2303–10.
Lavorini F. Inhaled drug delivery in the hands of the patient. J Aerosol Med Pulm Drug Deliv. 2014;27(6):414–8.
Thomas M, Price D, Chrystyn H, Lloyd A, Williams AE, von Ziegenweidt J. Inhaled corticosteroids for asthma: impact of practice level device switching on asthma control. BMC Pulm Med. 2009;9:1.
Booker R. Do patients think that dry powder inhalers can be used interchangeably? Int J Clin Pract Suppl. 2005;149:30–2.
Lindgren S, Bake B, Larsson S. Clinical consequences of inadequate inhalation technique in asthma therapy. Eur J Respir Dis. 1987;70(2):93–8.
Giraud V, Roche N. Misuse of corticosteroid metered-dose inhaler is associated with decreased asthma stability. Eur Respir J. 2002;19(2):246–51.
Bateman ED. Treatment adherence in asthmatic patients: the last frontier? J Allergy Clin Immunol. 2014;134(6):1269–70.
Acknowledgments
No funding or sponsorship was received for this study or the publication of this article. The author meets the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this manuscript, takes responsibility for the integrity of the work as a whole, and has given final approval to the version to be published.
Conflict of interest
Nicola Scichilone has received financial support for research and for congress attendance from Boehringer Ingelheim, Novartis, Chiesi Farmaceutici, Mundipharma, Glaxo SmithKline, AstraZeneca, Zambon, Almirall.
Compliance with ethics guidelines
This article is based on previously conducted studies and does not involve any new studies of human or animal subjects performed by any of the authors.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Scichilone, N. Asthma Control: The Right Inhaler for the Right Patient. Adv Ther 32, 285–292 (2015). https://doi.org/10.1007/s12325-015-0201-9
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-015-0201-9