Abstract
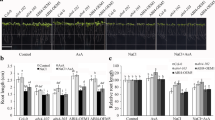
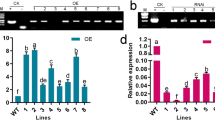
SOS5 locus, encodes cell wall adhesion protein under salt stress conditions in plants, and it is required for normal cell expansion as well as for sustaining cell wall integrity and structure. However, it is still unknown how this gene locus-ABA cross-talk and interacts with the antioxidant mechanism under salt stress conditions. For this purpose, the study focused on mutant sos5-1 plant treated with ABA under NaCl stress and observed its growth and development as well as stomatal aperture, lipid peroxidation, proline, hydrogen peroxide (H2O2) and ABA contents, and some antioxidant enzyme activities. In addition, the expression levels of ABA related genes have been analysed by RT-PCR after stress application. According to findings, sos5-1 mutant plants treated with ABA under salt stress resulted in eliminated cellular damage compared to those which are solely exposed to salt stress; other observations include closing of stomata, decreased H2O2 content, increased amount of proline, and similarity with the wild type due to induced antioxidant enzyme activities. Besides, both ABA biosynthetic and inducible gene expressions of the mutant plant under salt stress were lower compared to the control, and catabolism gene expression was higher. As a result, SOS5 gene in synergy with ABA, scavenge the ROS by stimulating antioxidant system, leads to an increase in stress related gene expressions and thus contributes to salinity tolerance. This study is significant in the way that it shows how SOS5 gene locus, under salt stress conditions, interacts with antioxidant system in sustaining cell wall integrity.









Similar content being viewed by others
References
Aebi H (1983) Catalase. In: Bergmeyer HU (ed) Methods of enzymatic analysis. Verlag Chemie, Weinhem, pp 273–286
Akyol TY, Yilmaz O, Uzilday B et al (2020) Plant response to salinity: an analysis of ROS formation, signaling, and antioxidant defense. Turk J Bot 44:1–13. https://doi.org/10.3906/bot-1911-15
Alazem M, Lin NS (2020) Interplay between ABA signaling and RNA silencing in plant viral resistance. Curr Opin Virol 42:1–7. https://doi.org/10.1016/j.coviro.2020.02.002
Bahin E, Bailly C, Sotta B et al (2011) Crosstalk between reactive oxygen species and hormonal signalling pathways regulates grain dormancy in barley. Plant Cell Env 34:980–993. https://doi.org/10.1111/j.1365-3040.2011.02298.x
Basu D, Tian L, DeBrosse T et al (2016) Glycosylation of a Fasciclin-Like Arabinogalactan-Protein (SOS5) mediates root growth and seed mucilage adherence via a cell wall receptor-like kinase (FEI1/FEI2) pathway in arabidopsis. PLoS ONE 11:1–27. https://doi.org/10.1371/journal.pone.0145092
Bates LS, Waldern RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:1276–1287
Boughalleb F, Abdellaoui R, Mahmoudi M, Bakhshandeh E (2020) Changes in phenolic profile, soluble sugar, proline, and antioxidant enzyme activities of polygonum equisetiforme in response to salinity. Turk J Bot 44:25–35. https://doi.org/10.3906/bot-1908-2
Brunetti C, Sebastiani F, Tattini M (2019) Review: ABA, flavonols, and the evolvability of land plants. Plant Sci 280:448–454. https://doi.org/10.1016/j.plantsci.2018.12.010
Bu Q, Lv T, Shen H et al (2014) Regulation of drought tolerance by the F-box protein MAX2 in Arabidopsis. Plant Physiol 164:424–439. https://doi.org/10.1104/pp.113.226837
Chen K, Song M, Guo Y et al (2019) MdMYB46 could enhance salt and osmotic stress tolerance in apple by directly activating stress-responsive signals. Plant Biotechnol J 17:2341–2355. https://doi.org/10.1111/pbi.13151
Chen F, Zhou W, Yin H et al (2020) Shading of the mother plant during seed development promotes subsequent seed germination in soybean. J Exp Bot 71:2072–2084. https://doi.org/10.1093/jxb/erz553
Cheng YW, Kong XW, Wang N et al (2020) Thymol confers tolerance to salt stress by activating anti-oxidative defense and modulating Na+ homeostasis in rice root. Ecotoxicol Environ Saf 188:109894. https://doi.org/10.1016/j.ecoenv.2019.109894
Claudia Blaukopf, Krol Matthaus SG (2011) New insights into the control of cell growth. Plant Cell Wall Methods Mol Biol 715:221–244
Clément M, Leonhardt N, Droillard M-J, Reiter I, Montillet J-L, Genty B, Laurière C, Nussaume L, Noël LD (2011) The cytosolic/nuclear HSC70 and HSP90 molecular chaperones are important for stomatal closure and modulate abscisic acid-dependent physiological responses in Arabidopsis. Plant Physiol 156(3):1481–1492
Daudi A, Cheng Z, O’Brien JA et al (2012) The apoplastic oxidative burst peroxidase in Arabidopsis is a major component of pattern-triggered immunity. Plant Cell 24:275–287. https://doi.org/10.1105/tpc.111.093039
Dhinsa SR, Matowe W (1981) Drought tolerance in two mosses: correlated with enzymatic defence against lipid peroxidation. J Exp Bot 32:79–91. https://doi.org/10.1093/jxb/32.1.79
Dong H, Ma X, Zhang P et al (2020) Characterization of Arabidopsis thaliana root-related mutants reveals aba regulation of plant development and drought resistance. J Plant Growth Regul. https://doi.org/10.1007/s00344-020-10076-6
Driouich A, Baskin TI (2008) Intercourse between cell wall and cytoplasm exemplified by arabinogalactan proteins and cortical microtubules. Am J Bot 95:1491–1497. https://doi.org/10.3732/ajb.0800277
FAO (2016) Extent of salt-affected soils. The Food and Agriculture Organization of the United Nations. https://www.fao.org/soils-portal/soil-management/management-of-some-problem-soils/salt-affected-soils/more-information-on-salt-affected-soils/en/. Accessed 18 Aug
Feng W, Kita D, Peaucelle A et al (2018) The FERONIA receptor kinase maintains cell-wall integrity during salt stress through Ca2+ Signaling. Curr Biol 28:666–675. https://doi.org/10.1016/j.cub.2018.01.023
Finkelstein R (2013) Abscisic acid synthesis and response. Arabidopsis Book 11:e0166. https://doi.org/10.1199/tab.0166
Fujii H, Verslues PE, Zhu JK (2011) Arabidopsis decuple mutant reveals the importance of SnRK2 kinases in osmotic stress responses in vivo. Proc Natl Acad Sci USA 108:1717–1722. https://doi.org/10.1073/pnas.1018367108
Fujita M, Fujita Y, Noutoshi Y et al (2006) Crosstalk between abiotic and biotic stress responses: a current view from the points of convergence in the stress signaling networks. Curr Opin Plant Biol 9:436–442
Han Y, Watanabe S, Shimada H, Sakamoto A (2020) Dynamics of the leaf endoplasmic reticulum modulate β-glucosidase-mediated stress-activated ABA production from its glucosyl ester. J Exp Bot 71:2058–2071. https://doi.org/10.1093/jxb/erz528
He JM, Xu H, She XP et al (2005) The role and the interrelationship of hydrogen peroxide and nitric oxide in the UV-B-induced stomatal closure in broad bean. Funct Plant Biol 32:237–247. https://doi.org/10.1071/FP04185
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Hong JH, Seah SW, Xu J (2013) The root of ABA action in environmental stress response. Plant Cell Rep 32:971–983. https://doi.org/10.1007/s00299-013-1439-9
Kadioglu A, Saruhan N, Sağlam A et al (2011) Exogenous salicylic acid alleviates effects of long term drought stress and delays leaf rolling by inducing antioxidant system. Plant Growth Regul 64:27–37. https://doi.org/10.1007/s10725-010-9532-3
Khan N, Bano A, Ali S, Babar MA (2020) Crosstalk amongst phytohormones from planta and PGPR under biotic and abiotic stresses. Plant Growth Regul 90:189–203. https://doi.org/10.1007/s10725-020-00571-x
Liu J, Xia Z, Wang M et al (2013) Overexpression of a maize E3 ubiquitin ligase gene enhances drought tolerance through regulating stomatal aperture and antioxidant system in transgenic tobacco. Plant Physiol Biochem 73:114–120. https://doi.org/10.1016/j.plaphy.2013.09.006
Lu S, Su W, Li H, Guo Z (2009) Abscisic acid improves drought tolerance of triploid bermudagrass and involves H2O2- and NO-induced antioxidant enzyme activities. Plant Physiol Biochem 47:132–138. https://doi.org/10.1016/j.plaphy.2008.10.006
Luo X, Li C, He X et al (2020) ABA signaling is negatively regulated by GbWRKY1 through JAZ1 and ABI1 to affect salt and drought tolerance. Plant Cell Rep 39:181–194. https://doi.org/10.1007/s00299-019-02480-4
Ma Y, Cao J, He J et al (2018) Molecular mechanism for the regulation of ABA homeostasis during plant development and stress responses. Int J Mol Sci 19:1–14. https://doi.org/10.3390/ijms19113643
Mahajan S, Pandey Girdhar K, Tuteja N (2008) Calcium- and salt-stress signaling in plants: shedding light on SOS pathway. Arch Biochem Biophys 471(2):146–158
Marco F, Busó E, Lafuente T, Carrasco P (2019) Spermine confers stress resilience by modulating abscisic acid biosynthesis and stress responses in arabidopsis plants. Front Plant Sci 10:972. https://doi.org/10.3389/fpls.2019.00972
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681. https://doi.org/10.1146/annurev.arplant.59.032607.092911
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nakano Y, Asada K (1987) Purification of ascorbate peroxidase in spinach chloroplasts; its inactivation in ascorbate-depleted medium and reactivation by monodehydroascorbate radical. Plant Cell Physiol 28:131–140. https://doi.org/10.1093/oxfordjournals.pcp.a077268
Ozfidan C, Turkan I, Sekmen AH, Seckin B (2012) Abscisic acid-regulated responses of aba2-1 under osmotic stress: the abscisic acid-inducible antioxidant defence system and reactive oxygen species production. Plant Bio 14:337–346
Ozfidan C, Turkan I, Sekmen AH, Seckin B (2013) Time course analysis of ABA and non-ionic osmotic stress-induced changes in water status, chlorophyll fluorescence and osmotic adjustment in Arabidopsis thaliana wild-type (Columbia) and ABA-deficient mutant (aba2). Environ Exp Bot 86:44–51. https://doi.org/10.1016/j.envexpbot.2010.09.008
Ozturk K, Saglam A, Kadioglu A (2020) Heliotropium thermophilum, an extreme heat tolerant species, promises plants about adaptation to high soil temperature conditions. Physiol Mol Biol Plants 26:525–535. https://doi.org/10.1007/s12298-020-00766-6
Pehlivan N, Sun L, Jarrett P et al (2016) Co-overexpressing a plasma membrane and a vacuolar membrane sodium/proton antiporter significantly improves salt tolerance in transgenic arabidopsis plants. Plant Cell Physiol 57:1069–1084. https://doi.org/10.1093/pcp/pcw055
Qi L, Liu S, Li C et al (2020) Phytochrome-interacting factors interact with the ABA receptors PYL8 and PYL9 to orchestrate ABA signaling in darkness. Mol Plant 13:414–430. https://doi.org/10.1016/j.molp.2020.02.001
Rui Y, Xiao C, Yi H et al (2017) Polygalacturonase involved in expansion3 functions in seedling development, rosette growth, and stomatal dynamics in Arabidopsis thaliana. Plant Cell 29:2413–2432. https://doi.org/10.1105/tpc.17.00568
Schroeder JI, Allen GJ, Hugouvieux V et al (2001) Guard cell signal transduction. Annu Rev Plant Physiol Plant Mol Biol 52:627–658
Seifert GJ, Roberts K (2007) The biology of arabinogalactan proteins. Annu Rev Plant Biol 58:137–161. https://doi.org/10.1146/annurev.arplant.58.032806.103801
Seifert GJ, Acet T, Xue H (2014) The Arabidopsis thaliana fasciclin like arabinogalactan protein 4 gene acts synergistically with abscisic acid signalling to control root growth. Ann Bot 114:1125–1133. https://doi.org/10.1093/aob/mcu010
Sezgin A, Altuntaş C, Sağlam A et al (2018) Abscisic acid cross-talking with hydrogen peroxide and osmolyte compounds may regulate the leaf rolling mechanism under drought. Acta Physiol Plant 40:1–12. https://doi.org/10.1007/s11738-018-2716-6
Shan H, Chen S, Jiang J et al (2012) Heterologous expression of the chrysanthemum R2R3-MYB transcription factor CmMYB2 enhances drought and salinity tolerance, increases hypersensitivity to ABA and delays flowering in Arabidopsis thaliana. Mol Biotechnol 51:160–173. https://doi.org/10.1007/s12033-011-9451-1
Shi H, Kim Y, Guo Y et al (2003) The arabidopsis SOS5 locus encodes a putative cell surface adhesion protein and is required for normal cell expansion. Plant Cell 15:19–32. https://doi.org/10.1038/nbt766
Shi CC, Feng CC, Yang MM et al (2014) Overexpression of the receptor-like protein kinase genes AtRPK1 and OsRPK1 reduces the salt tolerance of Arabidopsis thaliana. Plant Sci 217–218:63–70. https://doi.org/10.1016/j.plantsci.2013.12.002
Sun Y, Zhao J, Li X, Li Y (2020) E2 conjugases UBC1 and UBC2 regulate MYB42-mediated SOS pathway in response to salt stress in Arabidopsis. New Phytol. https://doi.org/10.1111/nph.16538
Teng K, Li J, Liu L et al (2014) Exogenous ABA induces drought tolerance in upland rice: the role of chloroplast and ABA biosynthesis-related gene expression on photosystem II during PEG stress. Acta Physiol Plant 36:2219–2227. https://doi.org/10.1007/s11738-014-1599-4
Turupcu A, Almohamed W, Oostenbrink C, Seifert GJ (2018) A speculation on the tandem fasciclin 1 repeat of FLA4 proteins in angiosperms. Plant Signal Behav 13:1–5. https://doi.org/10.1080/15592324.2018.1507403
Urbanek H, Kuzniak-Gebarowska E, Herka K (1991) Elicitation of defense responses in bean leave by botrytis cinerea polygalacturanase. Acta Physiol Plant 13:43–50
Van Hengel AJ, Roberts K (2003) AtAGP30, an arabinogalactan-protein in the cell walls of the primary root, plays a role in root regeneration and seed germination. Plant J 36:256–270. https://doi.org/10.1046/j.1365-313X.2003.01874.x
Van Zelm E, Zhang Y, Testerink C (2020) Salt tolerance mechanisms of plants. Annu Rev Plant Biol 71:1–31
Wang C, Chen S, Dong Y et al (2020) Chloroplastic Os3BGlu6 contributes significantly to cellular ABA pools and impacts drought tolerance and photosynthesis in rice. New Phytol 226:1042–1054. https://doi.org/10.1111/nph.16416
Wei M, Zhuang Y, Li H et al (2019) The cloning and characterization of hypersensitive to salt stress mutant, affected in quinolinate synthase, highlights the involvement of NAD in stress-induced accumulation of ABA and proline. Plant J 1:1–14. https://doi.org/10.1111/tpj.14613
Wu X, Qiao Z, Liu H et al (2017) CML20, an Arabidopsis calmodulin-like protein, negatively regulates guard cell ABA signaling and drought stress tolerance. Front Plant Sci 8:1–12. https://doi.org/10.3389/fpls.2017.00824
Yan J, Fang L, Yang L et al (2020) Abscisic acid positively regulates l-arabinose metabolism to inhibit seed germination through ABSCISIC ACID INSENSITIVE4-mediated transcriptional promotions of MUR4 in Arabidopsis thaliana. New Phytol 225:823–834. https://doi.org/10.1017/CBO9781107415324.004
Yang W, Chen S, Cheng Y et al (2020) Cell wall/vacuolar inhibitor of fructosidase 1 regulates ABA response and salt tolerance in Arabidopsis. Plant Signal Behav. https://doi.org/10.1080/15592324.2020.1744293
Yao XM, Ji J, Yue JY et al (2020) Exogenous abscisic acid modulates reactive oxygen metabolism and related gene expression in Platycladus orientalis under H2O2-induced stress. Russ J Plant Physiol 67:85–93. https://doi.org/10.1134/S1021443720010264
Yoshida K, Igarashi E, Wakatsuki E et al (2004) Mitigation of osmotic and salt stresses by abscisic acid through reduction of stress-derived oxidative damage in Chlamydomonas reinhardtii. Plant Sci 167:1335–1341. https://doi.org/10.1016/j.plantsci.2004.07.002
Zang L, Zheng T, Chu Y et al (2015) Genome-wide analysis of the fasciclin-like arabinogalactan protein gene family reveals differential expression patterns, localization, and salt stress response in populus. Front Plant Sci 6:1–16. https://doi.org/10.3389/fpls.2015.01140
Zhang X, Yang G, Shi R et al (2013) Arabidopsis cysteine-rich receptor-like kinase 45 functions in the responses to abscisic acid and abiotic stresses. Plant Physiol Biochem 67:189–198. https://doi.org/10.1016/j.plaphy.2013.03.013
Acknowledgements
We many thanks to Dr. Georg Seifert (University of Natural Resources and Life Science, Vienna, Austria) for all helps (I started to work on sos5 in his lab) especially supply seeds and for technical assistance with a confocal laser microscope photograph.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Acet, T., Kadıoğlu, A. SOS5 gene-abscisic acid crosstalk and their interaction with antioxidant system in Arabidopsis thaliana under salt stress. Physiol Mol Biol Plants 26, 1831–1845 (2020). https://doi.org/10.1007/s12298-020-00873-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12298-020-00873-4