Abstract
The relationship between Atlantic ribbed mussels (Geukensia demissa) and smooth cordgrass (Spartina alterniflora) represents a textbook example of a facultative mutualism. It is also foundational to wetland ecosystems along the Atlantic coast of North America. Spartina plantings play an essential role in marsh reconstruction projects, and Geukensia is rapidly gaining popularity as a tool in coastal restoration to help marshes resist erosion and to remove nutrient pollution. However, it remains unclear whether positive effects documented primarily in natural settings will translate to urban and constructed estuaries where they are most needed. Using systematic review and meta-analysis, we compared the influence of the Spartina-Geukensia mutualism on growth of the species and on nitrogen-cycling processes. Our review suggests the potential for context dependence in this mutualism, with substantial implications for coastal marsh resilience and restoration. Notably, the positive effect of the species on the growth of their mutualist found in natural marshes was absent in constructed marshes and negative in urban marshes. Encouragingly, the effect of each species on nitrogen cycling was similar regardless of context. We found that this mutualism remains severely understudied in urban and constructed marshes, and we suggest future directions to address the knowledge gaps identified in our analysis.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Species interactions mediate how organisms respond to and modify their environments, as well as the distribution and abundance of organisms within the environment. Positive interactions, which have been historically overlooked by ecologists (Bruno et al. 2003), may have particularly strong impacts on community diversity and ecosystem processes. Because they often confer greater resilience to abiotic stress, positive species interactions also have the potential to enhance the success of ecological restoration (Valdez et al. 2020). However, positive species interactions are also highly variable, and benefits received from mutualisms may differ as a result of changes in environmental context (Chamberlain et al. 2014). Rather than having a single linear relationship across all environments in which they are found, many mutualisms operate along gradients of abiotic and biotic stressors (He and Bertness 2014). A mutualism may be obligate in one site but facultative in another, and a relationship that is normally positive could become neutral or negative for one or more participants depending on environmental conditions (van der Heide et al. 2021). Moreover, asymmetries in positive species interactions (i.e., one species benefits more from the interaction than the other) are quite common and may even be critical in maintaining biodiversity (Bascompte et al. 2006). Context dependence in mutualisms, particularly facultative mutualisms, presents a substantial challenge for management of ecosystems facing an unprecedented rate of human-induced change (Gamfeldt et al. 2015; van der Heide et al. 2021).
Spartina alterniflora, smooth cordgrass, and Geukensia demissa, the Atlantic ribbed mussel, participate in a textbook example of a facultative mutualism. This relationship is foundational to the structure and function of coastal wetlands throughout the Atlantic coast of North America, and studies of this mutualism have significantly contributed to the ecological theory of positive species interactions (Bertness 1984; He et al. 2013). Spartina facilitates successful growth of Geukensia by providing structure that promotes recruitment of planktonic larvae and by forming a canopy that provides shelter from abiotic stressors (Bertness and Grosholz 1985). During the summer, Spartina protects Geukensia from heat stress that could lead to desiccation (Altieri et al. 2007). In the winter, Spartina protects Geukensia from ice shearing caused by wave action, which is often a major source of mortality (Franz 2001). Geukensia helps to stabilize Spartina’s root structure by attaching to its substrate with byssal threads (Bertness 1984). As a suspension-feeding bivalve, Geukensia provides additional inputs of nitrogen to the sediment surface in the form of feces and pseudofeces, which fertilize Spartina growth (Jordan and Valiela 1982).
Within their native range, Spartina and Geukensia contribute to the long-term resilience of marsh ecosystems and a host of ecosystem services that coastal marshes provide including the removal of nutrient pollution (Barbier et al. 2011; Bilkovic et al. 2017). Spartina assists in sediment construction by depositing slow-decaying biomass to the marsh benthos, promoting settlement of sediments from the water column, and stabilizing sediments through its root structure (Bertness 1984; Morris et al. 2016). Spartina also influences the rate of sediment microbial reactions by aerating the rhizosphere and releasing labile organic carbon from its roots (McClung et al. 1983). Spartina supports biodiversity within the marsh community by driving coastal primary production, providing structure for benthic species to attach and settle, and serving as refugia from predators (Gan et al. 2009; Chen et al. 2018). Geukensia increases the availability of nutrient-rich organic biodeposits in marsh sediments, providing greater energy for heterotrophic microbial reactions (Jordan and Valiela 1982). On the Pacific coast of North America, and in areas throughout Southeast Asia, the invasion of Spartina has led to rapid, irreversible changes to ecosystem structure, local biodiversity, and biogeochemical cycles (Li et al. 2009; Strong and Ayres 2013; Gao et al. 2019).
Foundational plant species like Spartina alterniflora are known to be an essential component of successful restorations. Geukensia is also rapidly gaining popularity as a tool in coastal restoration, particularly where the primary goal of restoration is nutrient remediation (Galimany et al. 2017; Bilkovic et al. 2021). Geukensia filter a wide diversity of particles from the water column, including bacteria < 1 μm in size, and are more tolerant of variation in salinity, temperature, and tidal inundation than other bivalves commonly used in nutrient-remediation projects like Crassotrea virginica (Kreeger et al. 2018). Recent work has also shown that the mutualism between these species can achieve even greater recovery of ecosystem function and greater resistance to abiotic stress (Derksen-Hooijberg et al. 2017).
On the Atlantic coast of North America, increasing rates of sea-level rise and eutrophication resulting from nutrient pollution near urban centers present two stressors that could drive context dependence in the Spartina-Geukensia mutualism (Kennish 2001; Gedan et al. 2009). Urban salt marshes are often lost at higher rates than their non-urban counterparts due to their proximity to multiple human disturbances including nutrient enrichment and altered hydrology due to coastal development (Zedler and Kercher 2005; Wigand et al. 2014). While urban managers can respond to marsh loss with restoration and reconstruction, constructed marshes may also face higher risk than their natural counterparts because they lack components and processes that contribute to long-term resilience, which only develop over decades of ecological succession (Gore and Shields 1995; Craft et al. 2003; Gutrich and Hitzhusen 2004; Herrick et al. 2006). Despite the obvious utility in identifying components and processes that contribute to resilience in highly stressed marsh ecosystems, urban and constructed marshes remain understudied relative to their natural counterparts (Grimm et al. 2008).
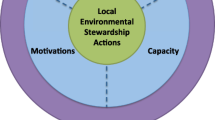
The mutualism between Spartina and Geukensia may confer greater resilience for marshes experiencing increasing rates of sea-level rise (Bertness 1984; Morris et al. 2016). Geukensia biodeposits fertilize both aboveground and belowground production of Spartina (Bertness 1984), both of which contribute to vertical marsh growth (Fig. 1A). Aboveground plant production increases sediment capture, and belowground production stabilizes sediments and contributes to the accumulation of organic material, both of which help the marsh to keep pace with sea-level rise and resist growing rates of tidal inundation (Cahoon et al. 2019). Enhanced belowground plant growth also aerates sediments, allowing for the oxidation of potentially harmful reduced substances like hydrogen sulfide and ammonium that often accumulate in flooded soils (Teal and Kanwisher 1966; Linthurst and Seneca 1981; Alldred et al. 2020). This plant-mediated aeration also promotes oxidation of ammonium to nitrate by nitrifying bacteria (Hamersley and Howes 2005), and subsequent removal of nitrate by denitrifying bacteria, thus promoting the ecosystem service of nitrogen removal (Fig. 1A). Spartina and Geukensia both produce labile organic matter, which provides an energy source for denitrifying bacteria (Sherr and Payne 1978; Bilkovic et al. 2017), further enhancing rates of microbial nitrogen removal. However, the outcome of this interaction may depend on the initial elevation of the marsh relative to local mean sea level, i.e., the marsh’s “elevation capital,” and delivery of sediment from the coastal watershed (Cole Ekberg et al. 2017; Gittman et al. 2018; Cahoon et al. 2019) (Fig. 1).
Hypothesized outcomes of the Geukensia-Spartina mutualism in marshes with A sufficient elevation capital and rates of sediment delivery, B sufficient elevation capital but insufficient sediment delivery, and C insufficient elevation capital and insufficient sediment delivery. The dashed line represents mean local sea level, and the solid line indicates the position of the sediment-water interface
Coastal urban marshes are subject to multiple stressors including eutrophication, rising sea levels, and land-use alterations that reduce inputs of sediment while increasing rates of erosion (Mudd 2011; Kirwan and Megonigal 2013; Wigand et al. 2014). Under these conditions, we hypothesize that the mutualism between Spartina and Geukensia could become decoupled (Fig. 1B, C). In a eutrophic system, regular inputs of nutrients from the watershed and from incoming tides may be sufficient, such that Spartina growth would not be limited by nutrient availability. Increased nutrient levels in the surrounding environment can also lead Spartina to develop less fine root structure (Darby and Turner 2008; Deegan et al. 2012; Alldred et al. 2017), leaving the marsh susceptible to erosion while simultaneously lowering rates of vertical accretion from the accumulation of belowground biomass (Turner 2011; Deegan et al. 2012). Under this scenario, additional nutrients provided by Geukensia’s biodeposits would no longer provide a positive subsidy for the plant and could potentially inhibit belowground plant growth.
If a marsh begins with sufficient elevation capital, and thus experiences shorter periods of daily tidal inundation, the plants may be less susceptible to increasing rates of inundation for some time (Cole Ekberg et al. 2017), continuing to accumulate sufficient belowground biomass and aerating surface sediments to promote nitrogen removal. However, marsh sediments below the zone of active plant growth and aeration could experience symptoms of eutrophication including increased rates of peat decomposition and prolonged anoxia that suppresses the formation of deep plant roots (Davey et al. 2011; Wigand et al. 2014). These conditions may result in a stable marsh platform but gradual loss of the marsh edge (Fig. 1B). In this scenario, we would expect to observe a decrease in the Geukensia population as its preferred edge habitat is lost (Franz 1996). In contrast, when a marsh with insufficient elevation capital and sediment delivery is exposed to sea-level rise and eutrophication, we expect that Geukensia would respond well in the short term, benefitting from longer periods of inundation and thus a greater amount of time to feed on organic material from the water column (Gittman et al. 2018). However, longer inundation periods would adversely affect plant growth by inhibiting root respiration and facilitating the buildup of harmful reduced substances (Alldred et al. 2020), and the loss of plant structure may eventually inhibit recruitment of juvenile Geukensia to the marsh (Fig. 1C). As sediments become inundated for longer periods of time, permanent anoxia would decrease the availability of nitrate, thereby decreasing rates of microbial nitrogen removal via denitrification and promoting accumulation of ammonium from dissimilatory nitrate reduction (Koop-Jakobsen and Giblin 2010; Giblin et al. 2013).
Here, we conducted a systematic review and meta-analysis to address the following questions and to identify any knowledge gaps in our understanding of the Spartina-Geukensia mutualism:
-
1.
How does the presence of Spartina alterniflora affect the growth of Geukensia demissa and vice versa, and are there asymmetries in benefits received from the mutualism?
-
2.
How does the presence of Spartina alterniflora and Geukensia demissa affect the components of the salt marsh nitrogen cycle?
-
3.
How do these trends change depending on environmental context, specifically in the cases of urban vs. non-urban wetlands and constructed vs. natural wetlands?
If the outcome of the Spartina-Geukensia mutualism does indeed differ in urban and/or constructed marshes, then much more information is needed to understand the long-term trajectory of marsh development in highly impacted coastal ecosystems. If the standard reinforced mutualism pathway occurs (Fig. 1A), then the mutualism could serve as a useful tool for conserving and restoring salt marsh ecosystems by regulating nitrogen and helping the marsh to keep pace with sea-level rise. However, if either decoupled mutualism pathway occurs (Fig. 1B or C), then the relationship between Spartina and Geukensia may not enhance long-term resilience, and may even accelerate rates of salt marsh loss. Investigating the outcome of the mutualism under differing salt marsh conditions is crucial to maintaining these highly valuable but highly threatened ecosystems.
Methods
Systematic Review Methods
To study the influence of the mutualism on species responses, we performed a search for records on 6/2/2020 using the database “Web of Science,” using eight search phrases (Table 1). We performed a similar search on 3/12/2020 to investigate the effect of each species on components of the marsh nitrogen cycle, using 14 search phrases (Table 1). Searches were limited to records published between 1970 and 2020 and sorted for relevance, using the first 40 records for each search phrase. Due to the low number of records returned for effects on species responses (n = 31), we performed a second search on 7/20/2020 with the search engine “Google Scholar,” using the same search phrases and search limitations. For all searches, if a record was returned that did not appear to correspond to any existing academic or scientific material (e.g., conference paper/abstract, advertisement, book review), we did not count it as part of the 40 records for that search phrase.
After compiling a list of records from our searches, we eliminated duplicates, including where the same data were presented in different sources (e.g., a graduate thesis and the papers published from that thesis) (Fig. 2). We then scanned abstracts for relevance and eliminated any record that featured neither of the two target species. Records were then read in full. For effects on species responses, we eliminated records in which both species were featured but the study focus was not on their interaction (Fig. 2A). For effects of species on the nitrogen cycle, we discarded studies that failed to include either of our species of interest and an estimate of its effect on a component of the nitrogen cycle (Fig. 2B). We also discarded measurements of the nitrogen cycle that were irrelevant to our study (e.g., identifying fraction of biologically incorporated nitrogen coming from specific sources). Records in which the published data were insufficient to perform meta-analysis (e.g., missing sample sizes, missing standard deviations or other estimates of error) were also eliminated. We identified 18 suitable publications for estimates of the species on the growth of their mutualist, from which we extracted 45 independent measurements. We identified 24 suitable publications for estimates of the effect of either species on the marsh nitrogen cycle, from which we extracted 49 independent measurements.
Meta-analysis Methods
Once a final list of records was identified, we extracted independent measurements from these records. For studies measuring species responses to the mutualism, the treatment group included the co-occurrence of both species, and the control group included the species in the absence of its mutualist. For studies of nitrogen cycling, we compared nitrogen processes or pools in the presence of either Spartina or Geukensia to the same process or pool in “control” measurements where the species was absent. For studies involving Spartina presence, it was not possible to have a standard definition for “control” sites across all studies. Whenever possible, bare sediments were used as controls, but in some studies Spartina presence could only be compared to the presence of other marsh plant communities. In all cases, the presence of Geukensia was compared to a control group in which marsh plants were present but Geukensia was not, and the treatment group included both Geukensia and Spartina. Studies of Geukensia in isolation were rare, and we found only one study that conducted a full factorial study that included both species, in combination and alone, relative to bare sediment controls (Bilkovic et al. 2017). We were therefore unable to include all of these combinations in our analysis. Responses of components of the nitrogen cycle to Geukensia are thus best understood as the added value of Geukensia to the marsh, relative to a vegetated marsh where Geukensia is absent. We extracted means, standard deviations, and sample sizes directly from the text of the records for treatment and control groups. In many cases, these data were not available in the text, and we used the image analysis program ImageJ (Schneider et al. 2012) to extract measurements from figures.
Information about the environmental category of each study site (i.e., urban vs non-urban and constructed vs natural) was obtained directly from the record. Sites were categorized as “urban” if they were identified as urban, highly impacted, or highly eutrophic in the site description. When this information was not available in the record, we looked at publicly available maps of the study site (using coordinates from the record if available) as well as photographs and geographic descriptions from official sources to characterize each site. In these cases, sites were considered “urban” if they were located within a densely populated or highly developed local watershed. Both artificial laboratory mesocosms and reconstructed or restored full-scale marshes were placed under the “constructed” category.
After all the data were extracted and compiled, we analyzed and visualized them using R version 4.1.1 (R Core Team 2021) and the packages meta, metafor, and dmetar (Viechtbauer 2010; Harrer et al. 2019; Balduzzi et al. 2019). We estimated mean effect sizes using the Hedges’ g estimator for species responses and nitrogen process/pool using a random-effects model. A random-effects model was used because we anticipated random variance in effect sizes among studies, both in terms biotic and abiotic characteristics of the study locations and the measurement techniques used to estimate effect sizes (Gurevitch and Hedges 2001). Hedges’ g was used as the measure of effect size due to the prevalence of small sample sizes in the analyzed studies (Hedges 1981). We performed subgroup analyses to compare the relative change in species growth variables when the species is in the presence of its mutualist. These responses included aboveground and belowground biomass and clonal outgrowth for Spartina alterniflora, and growth constant, population density, and shell length for Geukensia demissa. We then performed separate subgroup analyses to test whether the overall effect of the mutualism on species growth differed between urban and non-urban systems and between constructed and natural systems.
We performed subgroup analyses to compare the relative change in components of the nitrogen cycle in the presence of either of the species; these components included nitrogen fixation, nitrification, denitrification, nitrous oxide emissions, and Spartina tissue N concentrations. We subdivided studies of nitrogen cycling into urban vs. non-urban and constructed vs. natural systems and repeated the subgroup analysis to determine whether the effects of species on components of the nitrogen cycle differed depending on environmental context. For all subgroup analyses, effects were considered significant if the 95% confidence interval did not overlap zero. Subgroups are considered significantly different from each other in cases where the 95% confidence intervals of the subgroups did not overlap with each other. To investigate the possibility of publication bias within our literature sample, we generated a funnel plot and a p-curve for both the species-response and nitrogen-cycling data.
Data and metadata from our systematic review are publicly available (Alldred and Whaley 2022), and complete documentation of our analyses, including annotated R code, is available in the supplementary material (Online Resource 1). The published dataset includes categorizations that were applied to each measurement (e.g., urban vs. non-urban) and additional information including measurement techniques used in each study.
Results
Influence of Mutualism on Species Growth
While measurements were well distributed throughout the native range of the species (Fig. S1, Online Resource 1), many of the species-response data (33/45 measurement entries) come from two areas in the USA: New England and Georgia. All the Georgia studies (19/45 measurement entries) were performed on Sapelo Island. Natural and non-urban systems dominate the data, with a minority of data from urban systems (9/45 measurement entries) and an even smaller minority from constructed systems (4/45 measurement entries). Notably, all of the urban sites were located in the northern half of the species’ native range, including New York, NY, USA; Town Point, Nova Scotia, CA; Branford, CT, USA; and Arlington, NJ, USA (Fell et al. 1982; McClary 2004; Watt et al. 2011; Zhu et al. 2019). Likewise, the four studies in constructed systems were performed in Connecticut and New York, USA (Peck et al. 1994; Swamy et al. 2002; Zhu et al. 2019). Two of the studies in constructed marshes were performed at sites that had been wholly reconstructed using dredge material, and two involved impounded marshes that had natural tidal hydrology restored. Of the two species, Spartina was more commonly the focus of measurements (26/45 measurement entries) than Geukensia. In 6/19 measurement entries in which Geukensia growth was measured, the control for Spartina presence was a different plant species rather than bare sediment; these were either invasive Phragmites australis or other high marsh natives such as Spartina patens, Distichlis spicata, or Juncus gerardii (Fell et al. 1982; Peck et al. 1994; Swamy et al. 2002; McClary 2004). The funnel plot and p-curve for these data did not show any substantial evidence for publication bias (Online Resource 1).
Increases in aboveground Spartina biomass and belowground Spartina biomass from Geukensia presence were both significant, but aboveground biomass was more strongly affected (Fig. 3A). Clonal outgrowth showed a nonsignificant increase (Fig. 3A). Geukensia populations showed a significant increase in growth constant in the presence of Spartina, although the low number of observations makes it hard to draw meaningful conclusions for this response measure (Fig. 3B). Geukensia showed a nonsignificant increase in population density and a non-significant decrease in shell length due to Spartina presence (Fig. 3B). Removing the studies with non-sediment controls did not change results (Fig. S2, Online Resource 1).
Influence of Mutualism on Nitrogen Cycling
Most of the data for this analysis either come from the Atlantic coast of the USA (19/48 measurement entries) or the east coast of China (23/48 measurement entries) (Fig. S1, Online Resource 1). A small number of measurement entries (4/48) come from Brazil and Argentina. Many US studies were performed on Sapelo Island Reserve in Georgia (8/48 measurement entries), with the other 11 US measurement entries from Virginia, New York, and Rhode Island. These measurements represent fairly comprehensive coverage of Spartina’s native range. Many Chinese studies are from Dongtan Reserve in Shanghai (8/48 measurement entries). The majority of data come from natural (37/48 measurement entries) and non-urban systems (35/41 measurement entries), while constructed and urban systems are underrepresented in the literature. Urban sites were equally distributed between the native and invasive range of Spartina alterniflora, with three measurements in New York and Rhode Island, USA (Bertness 1984; Zhu et al. 2019), and four measurements in the Shandong and Zhejiang provinces in China (Zhang et al. 2019b, c). Most of the constructed studies come from laboratory mesocosms (7/11 constructed measurement entries) rather than field sites. All of the constructed field sites were located in urban watersheds in either New York, USA (Zhu et al. 2019) or Zhejiang Province, China (Zhang et al. 2019a). As in the species-response dataset, no urban or constructed sites were sampled within the southern half of the species’ native range. Of the two species, Spartina was the more-studied species (38/48 measurement entries) with regard to nitrogen processes. In 25/38 measurement entries focusing on Spartina addition, the grass is acting as an invasive. Eleven of 38 Spartina measurement entries, all of them when Spartina was an invasive, had other plant species as the controls rather than bare sediment; the control was commonly Phragmites australis, but Kandelia obovata and Cyperus malaccensis were also used as controls (Cheng et al. 2007; Zhang et al. 2013, 2016, 2019a; Jia et al. 2016; Huang et al. 2016; Yang and Silver 2016; Gao et al. 2017). The funnel plot and p-curve for these data did not show any substantial evidence for publication bias (Online Resource 1).
Denitrification and N2O emission increased significantly in the presence of Spartina (Fig. 4A). Spartina had no consistent effect on dissolved organic nitrogen, nitrogen fixation, or nitrification (Fig. 4A). Removing the non-sediment control studies led to similar results, with the exception of N2O emission, which became non-significant (Fig. S4, Online Resource 1). Geukensia presence showed little consistent effect on denitrification relative to vegetated marshes in which Geukensia was absent (Fig. 4B). Geukensia presence did correspond to a nonsignificant increase in nitrification and a significant increase in Spartina tissue nitrogen content (Fig. 4B).
A Influence of Spartina on pools and rates of the marsh nitrogen cycle. B Influence of Geukensia on rates and pools of the marsh nitrogen cycle. Mean effects are standardized mean differences; error bars represent 95% confidence intervals; and numeric labels indicate number of studies. Mean effect of Spartina on pools of dissolved organic nitrogen SMD = −10.46, 95% CI = [−34.10, 13.19], n = 3
Evidence of Context Dependence
The effect of the mutualism differed between natural and constructed marshes and between non-urban and urban marshes (Fig. 5). The mutualism had a significant positive effect on species growth in natural systems, while having no effect in constructed systems (Fig. 5A). In non-urban systems, the mutualism has significant positive effects for its component species, while having significant negative effects in urban systems (Fig. 5B). Bare-sediment-control-only results showed no significant differences from results with all control types included (Fig. S3, Online Resource 1).
We detected no significant differences in the effects of Spartina on components of the marsh nitrogen cycle when comparing natural and constructed marsh ecosystems (Fig. 6A). Results remain similar when non-sediment control studies are removed, with the exception that no studies have reported the influence of Spartina relative to bare sediment on nitrogen fixation or nitrification in constructed marshes (Fig. S5, Online Resource 1). The influences of Geukensia on nitrification and denitrification were not significantly different (Fig. 6B). However, in each case, only one study has estimated the effect of Geukensia in a constructed system, and no studies have estimated the effect of Geukensia on Spartina tissue N in a constructed marsh system (Fig. 6B).
A Overall effect of Spartina and B Geukensia on components of the marsh nitrogen cycle in natural vs. constructed marsh ecosystems. Mean effects are standardized mean differences; error bars represent 95% confidence intervals; and numeric labels indicate number of studies. All estimates of the effect of Spartina on dissolved organic nitrogen were conducted in natural marshes, SMD = −10.46, 95% CI = [−34.10, 13.19], n = 3
We detected no significant differences in the effect of Spartina on components of the marsh nitrogen cycle between non-urban and urban marsh ecosystems, with the exception of one study that found a very large effect of Spartina on denitrification at an urban site (Fig. 7A). However, our sample sizes for urban systems were extremely limited, with only one study estimating the effects of Spartina on nitrification, denitrification, and nitrous oxide emissions in urban systems, and no studies estimating the effect of Spartina on nitrogen fixation in urban systems (Fig. 7A). When non-sediment control studies were removed, no studies remained that allowed us to estimate the influence of Spartina in urban systems (Fig. S6, Online Resource 1). The influence of Geukensia on components of the marsh nitrogen cycle also did not differ between non-urban and urban marshes (Fig. 7B). However, our sample sizes for urban marshes were again limited, with only one estimate for each component of the nitrogen cycle.
A Effect of Spartina and B Geukensia on components of the marsh nitrogen cycle in non-urban vs. urban marsh ecosystems. Mean effects are standardized mean differences; error bars represent 95% confidence intervals; and numeric labels indicate number of studies. One study investigated the effect of Spartina on denitrification in an urban marsh and found a very large effect size, SMD = 22.40, 95% CI = [0.71, 44.09], n = 1
The influence of Spartina on components of the marsh nitrogen cycle did not differ between its native and invasive range (Fig. 8). The one study that estimated the effect of Spartina on nitrogen fixation in its invasive range did find a significant positive effect, which differs from the non-significant effect observed between the two studies conducted in its native range. Notably, no studies estimated the influence of Spartina on nitrous oxide emissions in its native range (Fig. 8). Results were similar when studies that did not use bare-sediment controls were removed, with the caveat that the one study that estimated the influence of Spartina on nitrogen fixation was eliminated from the analysis (Fig. S7, Online Resource 1).
Effect of Spartina on components of the marsh nitrogen cycle in its native and invasive range. Mean effects are standardized mean differences; error bars represent 95% confidence intervals; and numeric labels indicate number of studies. All estimates of the effect of Spartina on dissolved organic nitrogen were conducted in its invasive range, SMD = −10.46, 95% CI = [−34.10, 13.19], n = 3
Discussion
Though studies in urban and constructed marshes were extremely limited, our analysis provides compelling evidence that the mutualism between Spartina alterniflora and Geukensia demissa may function differently in highly impacted sites than in natural salt marshes. As expected (Fig. 1A), both species responded positively to the presence of their mutualist in studies conducted in natural and non-urban salt marshes. However, responses of the species to the presence of their mutualist were inconsistent in constructed marshes and significantly negative in urban marshes. Documentation of these benefits was stronger for Spartina, with significant increases in both aboveground and belowground growth in the presence of Geukensia. Responses of Geukensia to the presence of Spartina were more modest, with a positive effect on growth but no clear pattern in population density and a negative but nonsignificant trend in shell size. The difference in documented benefits for Spartina and Geukensia hints at a potential asymmetry in benefits derived from this mutualistic interaction, a common feature among mutualisms (Bascompte et al. 2006). However, the lack of clear trends in Geukensia could also result from complex interactions among recruitment of young mussels and growth and survivorship of adults (Franz and Tanacredi 1993; Franz 1996). Because Spartina is expected to facilitate recruitment of mussels, we may expect to observe a larger abundance of small juvenile mussels where Spartina is present compared to areas where it is absent. Size and age are directly related in mussels, but growth constant and age are inversely related (Borrero and Hilbish 1988). A greater abundance of juvenile mussels would therefore correspond to a smaller body size but faster growth, which is consistent with the results of our analysis.
Future studies that focus on juvenile recruitment and survival of Geukensia in the presence and absence of Spartina would allow us to clarify how, and in which contexts, Geukensia benefits from participating in this mutualism. Varying densities of Spartina among studies, which we were not able to document consistently enough to include in this analysis, may also have contributed to variance in the response of Geukensia to its mutualist. Because effects of Spartina may vary depending on plant density, future studies should also take care to document density of Spartina in field surveys or experiments. Notably, none of the studies included in our analysis measured adult survivorship for Geukensia in the presence and absence of their Spartina mutualist. Because enhanced adult survivorship is one of the primary benefits Spartina is thought to provide to its partner (Bertness 1984), it is possible that the absence of these measurements may have resulted in our underestimating the benefit of the mutualism to Geukensia.
We hypothesized that the positive effect of Geukensia in fertilizing Spartina may be absent or even negative in urban systems (Jin et al. 2005; Fig. 1B, C). Available data support this hypothesis. In urban systems, the presence of a mutualist had a significant negative effect on species growth (Fig. 5). However, a number of variables including the presence of predators (Hughes et al. 2014; Hensel et al. 2021), rates of sediment delivery and erosion, and extreme weather events (Derksen-Hooijberg et al. 2019) may differ between urban and non-urban systems. The lack of published studies in urban systems as of 2020 hampered our ability to investigate these variables in greater detail.
Studies of urban marshes in the southern half of the species native range were notably lacking, which is troubling given that these variables likely differ across the species’ native range. Likewise, we were unable to distinguish whether this potential decoupling in the mutualism we observed in urban systems was consistent with a scenario of sufficient or insufficient elevation capital (Fig. 1B or C). In a scenario of sufficient elevation capital, we would expect to observe negative effects on mussels consistent with the loss of edge habitat (Fig. 1B); whereas, in a scenario of insufficient elevation capital, we would expect to see negative effects on plant growth and a temporary increase in mussel growth consistent with increased rates of inundation (Fig. 1C). Our analysis revealed a modest but significant negative response of both species to the presence of their mutualist in urban systems, which may be consistent with either scenario (Fig. 5B). Given the stark difference in the outcome of the Spartina-Geukensia mutualism between urban and non-urban marshes, the precise mechanisms that may result in context dependence in urban marshes are worthy of investigation in future studies, with particular emphasis on urban areas in the southeastern Atlantic coast and the Gulf coast. Studies in constructed marshes were even more limited, and the variation in restoration techniques (e.g., reconstruction on dredged sediment vs. restoration of tidal regime to an impounded marsh) (Peck et al. 1994; Swamy et al. 2002; Zhu et al. 2019) likely resulted in the lack of a clear signal in species responses to the presence of their mutualist (Fig. 5A). The rapid increase in wetland construction and “living shoreline” projects (Zhu et al. 2019; Bilkovic et al. 2021) will provide exciting opportunities to study potential mechanisms of context dependence in this mutualism over the next decade.
While our analysis strongly suggests context dependence in the effect of the mutualism on responses of the species, we observed no such evidence of context dependence in the effect of the species on components of the marsh nitrogen cycle. Contrary to our predictions for an urban eutrophic marsh (Fig. 1C), the influence of the species on nitrogen cycling appears to be relatively consistent, even in cases where the species no longer have a positive influence on each other. While this result is encouraging for managers hoping to restore function to urban marshes, it should also be viewed with caution given the extremely limited number of studies that have been performed in constructed and urban systems. Moreover, the effects of either species on many processes of the nitrogen cycle, including nitrogen fixation and dissimilatory nitrate reduction to ammonia (DNRA), remain poorly quantified (Fig. 4). Our understanding of the effect of both species on the marsh nitrogen cycle would be greatly enhanced by a larger number of experimental field studies in a variety of newly constructed, stable, and degraded marsh sites throughout the species’ native range.
The overall effect of both species on the marsh nitrogen cycle among studies was largely consistent with our expectations for stable marshes with sufficient elevation capital (Fig. 1A or B). The increase in denitrification rates in the presence of Spartina is consistent with the commonly held consensus on Spartina’s effects on marsh sediments (Sherr and Payne 1978; Hamersley and Howes 2005; Alldred and Baines 2016). Nitrogen removal is often a management goal of marsh ecosystems, so these results are promising for management purposes (Cheng et al. 2020). As a byproduct of denitrification, it is likewise unsurprising that nitrous oxide emissions also increase in the presence of Spartina. However, because nitrous oxide is a potent greenhouse gas, increases in the local service of nitrogen removal may also contribute to global climate challenges.
Contrary to our expectations, we did not observe significant increases in sediment nitrification rates in the presence of Spartina (Sherr and Payne 1978; Hamersley and Howes 2005) relative to when Spartina was absent (Fig. 1C). As many of the Spartina-focused nitrogen cycling studies took place in Spartina’s invasive range, where it is often compared to Phragmites australis, another species that aerates the rhizosphere, differences in study design may have confounded results. However, removing the measurements where Phragmites and other plants were used as controls did not change the results of our analysis (Online Resource 1), nor did we observe any differences in the effect of Spartina on nitrogen cycling between its native and invasive range (Fig. 8).
The strongest effect of Geukensia on nitrogen cycling was an increase in Spartina tissue nitrogen content. This result was not surprising given that fertilization of Spartina should increase opportunistic uptake of nitrogen. Among all studies, Geukensia had a positive effect on nitrification, relative to vegetated marshes in which Geukensia was absent, likely as a result of increasing ammonium availability in sediments (Fig. 1A). However, contrary to our expectations, we observed considerable variation and no clear effect on denitrification rates, suggesting that Geukensia’s effects on marsh nitrogen cycling may be more variable than is commonly assumed (Kreeger et al. 2018). Studies of the influence of Geukensia on components of the marsh nitrogen cycle remain very limited. An additional challenge is that very few studies have quantified the effect of Geukensia on nitrogen-cycling processes in the absence of its Spartina mutualist (Fig. 1C), despite observations that Geukensia assemblages may persist in isolation (Watt et al. 2011). A greater number of full-factorial studies that address the effects of the species, alone and in combination, is needed to fully clarify the role of Geukensia in marsh nitrogen cycling, particularly within constructed and urban systems.
That the Spartina-Geukensia mutualism may vary in its effects depending on environmental context is a highly valuable piece of knowledge for land management and restoration projects. Although the mutualism could prove useful in restoring and maintaining naturally occurring salt marshes that receive little human influence, the mutualism may have less value and may become detrimental to long-term marsh stability in urban systems. More work is needed to identify variables within the urban environment that could decrease the value of the mutualism to the growth of Spartina and the overall stability of the marsh ecosystem. Urban and constructed systems require greater attention in the future, especially given the vulnerability of these systems to threats from human activities and their immense value to coastal communities.
References
Alldred, M., and S.B. Baines. 2016. Effects of wetland plants on denitrification rates: a meta-analysis. Ecological Applications 26: 676–685.
Alldred, M., A. Liberti, and S.B. Baines. 2017. Impact of salinity and nutrients on salt marsh stability. Ecosphere 8: e02010.
Alldred, M., J.J. Borrelli, T. Hoellein, D. Bruesewitz, and C. Zarnoch. 2020. Marsh plants enhance coastal marsh resilience by changing sediment oxygen and sulfide concentrations in an urban, eutrophic estuary. Estuaries and Coasts 43: 801–813.
Alldred, M., and T. Whaley. 2022. Systematic review of mutualistic interaction between Spartina alterniflora and Geukensia demissa. figshare. Dataset. https://doi.org/10.6084/m9.figshare.20113427.v1.
Altieri, A.H., B.R. Silliman, and M.D. Bertness. 2007. Hierarchical organization via a facilitation cascade in intertidal cordgrass bed communities. The American Naturalist 169: 195–206.
Balduzzi, S., G. Rücker, and G. Schwarzer. 2019. How to perform a meta-analysis with R: a practical tutorial. Evidence Based Mental Health 22: 153.
Barbier, E.B., S.D. Hacker, C. Kennedy, E.W. Koch, A.C. Stier, and B.R. Silliman. 2011. The value of estuarine and coastal ecosystem services. Ecological Monographs 81: 169–193.
Bascompte, J., P. Jordano, and J.M. Olesen. 2006. Asymmetric coevolutionary networks facilitate biodiversity maintenance. Science 312: 431–433.
Bertness, M.D. 1984. Ribbed mussels and Spartina alterniflora production in a New England salt marsh. Ecology 65: 1794–1807.
Bertness, M.D., and E. Grosholz. 1985. Population dynamics of the ribbed mussel, Geukensia demissa: the costs and benefits of an aggregated distribution. Oecologia 67: 192–204.
Bilkovic, D.M., M.M. Mitchell, R.E. Isdell, M. Schliep, and A.R. Smyth. 2017. Mutualism between ribbed mussels and cordgrass enhances salt marsh nitrogen removal. Ecosphere 8: e01795.
Bilkovic, D.M., R.E. Isdell, A.G. Guthrie, M.M. Mitchell, and R.M. Chambers. 2021. Ribbed mussel Geukensia demissa population response to living shoreline design and ecosystem development. Ecosphere 12: e03402.
Borrero, F., and T. Hilbish. 1988. Temporal variation in shell and soft tissue growth of the mussel Geukensia demissa. Marine Ecology Progress Series 42: 9–15.
Bruno, J.F., J.J. Stachowicz, and M.D. Bertness. 2003. Inclusion of facilitation into ecological theory. Trends in Ecology & Evolution 18: 119–125.
Cahoon, D.R., J.C. Lynch, C.T. Roman, J.P. Schmit, and D.E. Skidds. 2019. Evaluating the relationship among wetland vertical development, elevation capital, sea-level rise, and tidal marsh sustainability. Estuaries and Coasts 42: 1–15.
Chamberlain, S.A., J.L. Bronstein, and J.A. Rudgers. 2014. How context dependent are species interactions? Ecology Letters 17: 881–890.
Chen, Q., G. Xu, S. Zhang, and K. Ma. 2018. Consumption of an exotic plant (Spartina alterniflora) by the macrobenthic fauna in a mangrove wetland at Zhanjiang, China. Wetlands 38: 327–335.
Cheng, X., R. Peng, J. Chen, Y. Luo, Q. Zhang, S. An, J. Chen, and B. Li. 2007. CH4 and N2O emissions from Spartina alterniflora and Phragmites australis in experimental mesocosms. Chemosphere 68: 420–427.
Cheng, F.Y., K.J. Van Meter, D.K. Byrnes, and N.B. Basu. 2020. Maximizing US nitrate removal through wetland protection and restoration. Nature 588: 625–630.
Cole Ekberg, M.L., K.B. Raposa, W.S. Ferguson, K. Ruddock, and E.B. Watson. 2017. Development and application of a method to identify salt marsh vulnerability to sea level rise. Estuaries and Coasts 40: 694–710.
Craft, C., P. Megonigal, S. Broome, J. Stevenson, R. Freese, J. Cornell, L. Zheng, and J. Sacco. 2003. The pace of ecosystem development of constructed Spartina alterniflora marshes. Ecological Applications 13: 1417–1432.
Darby, F.A., and R.E. Turner. 2008. Below- and aboveground biomass of Spartina alterniflora: response to nutrient addition in a Louisiana salt marsh. Estuaries and Coasts 31: 326–334.
Davey, E., C. Wigand, R. Johnson, K. Sundberg, J. Morris, and C.T. Roman. 2011. Use of computed tomography imaging for quantifying coarse roots, rhizomes, peat, and particle densities in marsh soils. Ecological Applications 21: 2156–2171.
Deegan, L.A., D.S. Johnson, R.S. Warren, B.J. Peterson, J.W. Fleeger, S. Fagherazzi, and W.M. Wollheim. 2012. Coastal eutrophication as a driver of salt marsh loss. Nature 490: 388–392.
Derksen-Hooijberg, M., C. Angelini, L.P.M. Lamers, A. Borst, A. Smolders, J.R.H. Hoogveld, H. Paoli, J. Koppel, B.R. Silliman, and T. Heide. 2017. Mutualistic interactions amplify saltmarsh restoration success. Journal of Applied Ecology 55: 405–414.
Derksen-Hooijberg, M., C. Angelini, J.R.H. Hoogveld, L.P.M. Lamers, A. Borst, A. Smolders, S.F. Harpenslager, L.L. Govers, and T. Heide. 2019. Repetitive desiccation events weaken a salt marsh mutualism. Journal of Ecology 107: 2415–2426.
Fell, P.E., N.C. Olmstead, E. Carlson, W. Jacob, D. Hitchcock, and G. Silber. 1982. Distribution and abundance of macroinvertebrates on certain Connecticut tidal marshes, with emphasis on dominant molluscs. Estuaries 5: 234.
Franz, D.R. 1996. Size and age at first reproduction of the ribbed mussel Geukensia demissa (Dillwyn) in relation to shore level in a New York salt marsh. Journal of Experimental Marine Biology and Ecology 205: 1–13.
Franz, D.R. 2001. Recruitment, survivorship, and age structure of a New York ribbed mussel population (Geukensia demissa) in relation to shore level: a nine year study. Estuaries 24: 319.
Franz, D.R., and J.T. Tanacredi. 1993. Variability in growth and age structure among populations of ribbed mussels, Geukensia demissa (Dillwyn) (Bivalvia: Mytilidae), in Jamaica Bay, New York (Gateway NRA). The Veliger 36: 220–227.
Galimany, E., G.H. Wikfors, M.S. Dixon, C.R. Newell, S.L. Meseck, D. Henning, Y. Li, and J.M. Rose. 2017. Cultivation of the ribbed mussel (Geukensia demissa) for nutrient bioextraction in an urban estuary. Environmental Science & Technology 51: 13311–13318.
Gamfeldt, L., J.S. Lefcheck, J.E.K. Byrnes, B.J. Cardinale, J.E. Duffy, and J.N. Griffin. 2015. Marine biodiversity and ecosystem functioning: what’s known and what’s next? Oikos 124: 252–265.
Gan, X., Y. Cai, C. Choi, Z. Ma, J. Chen, and B. Li. 2009. Potential impacts of invasive Spartina alterniflora on spring bird communities at Chongming Dongtan, a Chinese wetland of international importance. Estuarine, Coastal and Shelf Science 83: 211–218.
Gao, D., X. Li, X. Lin, D. Wu, B. Jin, Y. Huang, M. Liu, and X. Chen. 2017. Soil dissimilatory nitrate reduction processes in the Spartina alterniflora invasion chronosequences of a coastal wetland of southeastern China: dynamics and environmental implications. Plant and Soil 421: 383–399.
Gao, G.-F., P.-F. Li, J.-X. Zhong, Z.-J. Shen, J. Chen, Y.-T. Li, A. Isabwe, X.-Y. Zhu, Q.-S. Ding, S. Zhang, C.-H. Gao, and H.-L. Zheng. 2019. Spartina alterniflora invasion alters soil bacterial communities and enhances soil N2O emissions by stimulating soil denitrification in mangrove wetland. Science of the Total Environment 653: 231–240.
Gedan, K.B., B.R. Silliman, and M.D. Bertness. 2009. Centuries of human-driven change in salt marsh ecosystems. Annual Review of Marine Science 1: 117–141.
Giblin, A., C. Tobias, B. Song, N. Weston, G. Banta, and V. Rivera-Monroy. 2013. The importance of dissimilatory nitrate reduction to ammonium (DNRA) in the nitrogen cycle of coastal ecosystems. Oceanography 26: 124–131.
Gittman, R.K., F.J. Fodrie, C.J. Baillie, M.C. Brodeur, C.A. Currin, D.A. Keller, M.D. Kenworthy, J.P. Morton, J.T. Ridge, and Y.S. Zhang. 2018. Living on the edge: increasing patch size enhances the resilience and community development of a restored salt marsh. Estuaries and Coasts 41: 884–895.
Gore, J.A., and F.D. Shields. 1995. Can large rivers be restored? BioScience 45: 142–152.
Grimm, N.B., S.H. Faeth, N.E. Golubiewski, C.L. Redman, J. Wu, X. Bai, and J.M. Briggs. 2008. Global change and the ecology of cities. Science 319: 756–760.
Gurevitch, J., and L.V. Hedges. 2001. Meta-analysis: combining the results of independent experiments. In Design and analysis of ecological experiments, 2nd ed., ed. S.M. Scheiner and J. Gurevitch, 378–398. New York, NY: Oxford University Press.
Gutrich, J.J., and F.J. Hitzhusen. 2004. Assessing the substitutability of mitigation wetlands for natural sites: estimating restoration lag costs of wetland mitigation. Ecological Economics 48: 409–424.
Hamersley, M., and B. Howes. 2005. Coupled nitrification-denitrification measured in situ in a Spartina alterniflora marsh with a 15NH4+ tracer. Marine Ecology Progress Series 299: 123–135.
Harrer, M., P. Cuijpers, T. Furukawa, and D.D. Ebert. 2019. dmetar: companion R package for the guide “Doing meta-analysis in R.” R.
He, Q., and M.D. Bertness. 2014. Extreme stresses, niches, and positive species interactions along stress gradients. Ecology 95: 1437–1443.
He, Q., M.D. Bertness, and A.H. Altieri. 2013. Global shifts towards positive species interactions with increasing environmental stress. Ecology Letters 16: 695–706.
Hedges, L.V. 1981. Distribution theory for Glass’s estimator of effect size and related estimators. Journal of Educational Statistics 6: 107–128.
Hensel, M.J.S., B.R. Silliman, J. van de Koppel, E. Hensel, S.J. Sharp, S.M. Crotty, and J.E.K. Byrnes. 2021. A large invasive consumer reduces coastal ecosystem resilience by disabling positive species interactions. Nature Communications 12: 6290.
Herrick, J.E., G.E. Schuman, and A. Rango. 2006. Monitoring ecological processes for restoration projects. Journal for Nature Conservation 14: 161–171.
Huang, J., X. Xu, M. Wang, M. Nie, S. Qiu, Q. Wang, Z. Quan, M. Xiao, and B. Li. 2016. Responses of soil nitrogen fixation to Spartina alterniflora invasion and nitrogen addition in a Chinese salt marsh. Scientific Reports 6: 20384.
Hughes, A.R., A.F.P. Moore, and M.F. Piehler. 2014. Independent and interactive effects of two facilitators on their habitat-providing host plant, Spartina alterniflora. Oikos 123: 488–499.
Jia, D., F. Qi, X. Xu, J. Feng, H. Wu, J. Guo, W. Lu, R. Peng, X. Zhu, Y. Luo, and G. Lin. 2016. Co-regulations of Spartina alterniflora invasion and exogenous nitrogen loading on soil N2O efflux in subtropical mangrove mesocosms. PLoS ONE 11: e0146199.
Jin, X., Q. Xu, and C. Huang. 2005. Current status and future tendency of lake eutrophication in China. Science in China Series C, Life Sciences 48: 948–954.
Jordan, T.E., and I. Valiela. 1982. A nitrogen budget of the ribbed mussel, Geukensia demissa, and its significance in nitrogen flow in a New England salt marsh: Nitrogen budget for G. demissa. Limnology and Oceanography 27: 75–90.
Kennish, M.J. 2001. Coastal salt marsh systems in the U.S.: a review of anthropogenic impacts. Journal of Coastal Research 17: 731–748.
Kirwan, M.L., and J.P. Megonigal. 2013. Tidal wetland stability in the face of human impacts and sea-level rise. Nature 504: 53–60.
Koop-Jakobsen, K., and A.E. Giblin. 2010. The effect of increased nitrate loading on nitrate reduction via denitrification and DNRA in salt marsh sediments. Limnology and Oceanography 55: 789–802.
Kreeger, D.A., C.M. Gatenby, and P.W. Bergstrom. 2018. Restoration potential of several native species of bivalve molluscs for water quality improvement in Mid-Atlantic watersheds. Journal of Shellfish Research 37: 1121.
Li, B., C. Liao, X. Zhang, H. Chen, Q. Wang, Z. Chen, X. Gan, J. Wu, B. Zhao, Z. Ma, X. Cheng, L. Jiang, and J. Chen. 2009. Spartina alterniflora invasions in the Yangtze River estuary, China: an overview of current status and ecosystem effects. Ecological Engineering 35: 511–520.
Linthurst, R.A., and E.D. Seneca. 1981. Aeration, nitrogen and salinity as determinants of Spartina alterniflora Loisel. growth response. Estuaries 4: 53–63.
McClary, M., Jr. 2004. Spartina alterniflora and Phragmites australis as habitat for the ribbed mussel, Geukensia demissa (Dillwyn), in Saw Mill Creek of New Jersey’s Hackensack Meadowlands. Urban Habitats 2: 83–90.
McClung, C.R., P. van Berkum, R.E. Davis, and C. Sloger. 1983. Enumeration and localization of N 2-fixing bacteria associated with roots of Spartina alterniflora Loisel. Applied and Environmental Microbiology 45: 1914–1920.
Morris, J.T., D.C. Barber, J.C. Callaway, R. Chambers, S.C. Hagen, C.S. Hopkinson, B.J. Johnson, P. Megonigal, S.C. Neubauer, T. Troxler, and C. Wigand. 2016. Contributions of organic and inorganic matter to sediment volume and accretion in tidal wetlands at steady state. Earth’s Future 4: 110–121.
Mudd, S.M. 2011. The life and death of salt marshes in response to anthropogenic disturbance of sediment supply. Geology 39: 511–512.
Peck, M.A., P.E. Fell, E.A. Allen, J.A. Gieg, C.R. Guthke, and M.D. Newkirk. 1994. Evaluation of tidal marsh restoration: comparison of selected macroinvertebrate populations on a restored impounded valley marsh and an unimpounded valley marsh within the same salt marsh system in Connecticut, USA. Environmental Management 18: 283–293.
R Core Team. 2021. R: a language and environment for statistical computing. R Foundation for Statistical Computing.
Schneider, C.A., W.S. Rasband, and K.W. Eliceiri. 2012. NIH Image to ImageJ: 25 years of image analysis. Nature Methods 9: 671–675.
Sherr, B.F., and W.J. Payne. 1978. Effect of the Spartina alterniflora root-rhizome system on salt marsh soil denitrifying bacteria. Applied and Environmental Microbiology 35: 724–729.
Strong, D.R., and D.R. Ayres. 2013. Ecological and evolutionary misadventures of Spartina. Annual Review of Ecology, Evolution, and Systematics 44: 389–410.
Swamy, V., P.E. Fell, M. Body, M.B. Keaney, M.K. Nyaku, E.C. Mcilvain, and A.L. Keen. 2002. Macroinvertebrate and fish populations in a restored impounded salt marsh 21 years after the reestablishment of tidal flooding. Environmental Management 29: 516–530.
Teal, J.M., and J.W. Kanwisher. 1966. Gas transport in the marsh grass, Spartina alterniflora. Journal of Experimental Botany 17: 355–361.
Turner, R.E. 2011. Beneath the salt marsh canopy: loss of soil strength with increasing nutrient loads. Estuaries and Coasts 34: 1084–1093.
Valdez, S.R., Y.S. Zhang, T. van der Heide, M.A. Vanderklift, F. Tarquinio, R.J. Orth, and B.R. Silliman. 2020. Positive ecological interactions and the success of seagrass restoration. Frontiers in Marine Science 7: 91.
van der Heide, T., C. Angelini, J. Fouw, and J.S. Eklöf. 2021. Facultative mutualisms: a double-edged sword for foundation species in the face of anthropogenic global change. Ecology and Evolution 11: 29–44.
Viechtbauer, W. 2010. Conducting meta-analyses in R with the metafor package. Journal of Statistical Software 36: 1–48.
Watt, C., D.J. Garbary, and C. Longtin. 2011. Population structure of the ribbed mussel Geukensia demissa in salt marshes in the southern Gulf of St. Lawrence. Canada. Helgoland Marine Research 65: 275–283.
Wigand, C., C.T. Roman, E. Davey, M. Stolt, R. Johnson, A. Hanson, E.B. Watson, S.B. Moran, D.R. Cahoon, J.C. Lynch, and P. Rafferty. 2014. Below the disappearing marshes of an urban estuary: historic nitrogen trends and soil structure. Ecological Applications 24: 633–649.
Yang, W.H., and W.L. Silver. 2016. Gross nitrous oxide production drives net nitrous oxide fluxes across a salt marsh landscape. Global Change Biology 22: 2228–2237.
Zedler, J.B., and S. Kercher. 2005. Wetland resources: status, trends, ecosystem services, and restorability. Annual Review of Environment and Resources 30: 39–74.
Zhang, Y., L. Wang, X. Xie, L. Huang, and Y. Wu. 2013. Effects of invasion of Spartina alterniflora and exogenous N deposition on N2O emissions in a coastal salt marsh. Ecological Engineering 58: 77–83.
Zhang, Y., X. Xu, Y. Li, L. Huang, X. Xie, J. Dong, and S. Yang. 2016. Effects of Spartina alterniflora invasion and exogenous nitrogen on soil nitrogen mineralization in the coastal salt marshes. Ecological Engineering 87: 281–287.
Zhang, C., W. Liu, M. Guan, J. Wang, X. Pan, Y. Ge, and J. Chang. 2019a. Nitrous oxide emission rate in response to plant, soil and microbial properties in marshes impacted by alien Spartina alterniflora. Biologia 74: 1087–1097.
Zhang, H., Y. Li, M. Pang, M. Xi, and F. Kong. 2019b. Responses of contents and structure of DOM to Spartina alterniflora invasion in Yanghe Estuary wetland of Jiaozhou Bay, China. Wetlands 39: 729–741.
Zhang, X., L. Cui, M. Nie, J. Shao, S. Wei, X. Kang, and B. Li. 2019c. Macrophytes and crabs affect nitrogen transformations in salt marshes of the Yangtze River Estuary. Estuarine, Coastal and Shelf Science 225: 106242.
Zhu, J., C. Zarnoch, J. Gosnell, M. Alldred, and T. Hoellein. 2019. Ribbed mussels Geukensia demissa enhance nitrogen-removal services but not plant growth in restored eutrophic salt marshes. Marine Ecology Progress Series 631: 67–80.
Acknowledgements
We thank Eileen Allen and Mark Lesser in the Center for Earth and Environmental Sciences at SUNY Plattsburgh for helpful feedback on an earlier draft of this manuscript. Jonathan J. Borrelli provided line drawings used in Fig. 1 as well as helpful feedback. Thanks also to Chester Zarnoch at Baruch College CUNY for many helpful discussions, and to Review Editor Ken Heck and two anonymous reviewers whose helpful feedback improved the quality of our review. We would also like to thank the researchers whose published data we used in this meta-analysis.
Funding
This project was funded by the Hudson River Foundation (Award 001-19A).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Kenneth L. Heck
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Whaley, T., Alldred, M. A Meta-analysis Reveals Knowledge Gaps in Our Understanding of the Spartina-Geukensia Mutualism. Estuaries and Coasts 46, 1021–1034 (2023). https://doi.org/10.1007/s12237-023-01194-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12237-023-01194-x