Abstract
Infectious diseases have for centuries been the leading causes of death and disability worldwide and the environmental fluctuation is a crucial part of an ecosystem in the natural world. In this paper, we proposed and discussed a stochastic SIRI epidemic model incorporating double saturated incidence rates and relapse. The dynamical properties of the model were analyzed. The existence and uniqueness of a global positive solution were proven. Sufficient conditions were derived to guarantee the extinction and persistence in mean of the epidemic model. Additionally, ergodic stationary distribution of the stochastic SIRI model was discussed. Our results indicated that the intensity of relapse and stochastic perturbations greatly affected the dynamics of epidemic systems and if the random fluctuations were large enough, the disease could be accelerated to extinction while the stronger relapse rate were detrimental to the control of the disease.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The control and eradication of infectious diseases has been an urgent problems in mathematical epidemiology and it is attracting the interest of researchers from many fields. In the past few decades, various epidemiological models which can analyze the underlying mechanisms have been put forward and discussed extensively, such as SIS, SIR, SIER, SIRS [1,2,3,4].
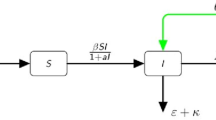
Recently, some researchers have paid attention to an appealing phenomenon that in the transmission and control of some diseases, the recovered populations may relapse with reactivation of latent infection and then they revert back to the infective one, such as tuberculosis including human and bovine and herpes [5,6,7]. Then, in the past few years, SIRI epidemic models are proposed and investigated by many authors [7,8,9,10]. In Liu et al. [7], discussed the impact of relapse to the transmission of Tuberculosis in Beijing, China and derived that relapse of TB in Beijing was more common than reinfection with a new strain. In Driessche and Zou [8], considered a more general SIRI model and threshold property were obtained. In Fatini [9], proposed an epidemic model with stochastic perturbations and relapse. By virtue of Lyapunov functions, the persistence and extinction of the diseases were analysed. However, they only used the bilinear incidence rate for the proposed model, not considering the effect of the behavioral change and crowding effect of the infected population, where a saturation incidence rate may be properer and prevent the unboundedness of the contact rate [11,12,13,14,15]. As far as we know, there are few epidemic models considering the effects of saturated incidence and relapse rates together and in this paper, we try to fill the gap. In the paper, considering the effects of saturated incidence rate and the saturation phenomenon of the developed medical techniques and conditions, an SIRI epidemic model with double saturated rates is formulated:
where the parameters are supposed to be positive constants. And \(\mu \) is the recruitment rate due to births and immigration and it is also supposed to be equal to the natural death rate. \(\frac{\beta I}{1+\alpha I}\) represents the disease transmission rate, e is the recovery rate and \(\frac{\gamma }{1+kR(t)}\) denotes the rate by which no-infectious individuals are reverted to the infectious state [9]. \(\alpha \), k are the so-called half-saturation constants, respectively. All the coefficients of system (1.1) are assumed to be nonnegative, and \(\mu >0\). \(N(t)=S(t)+I(t)+R(t)\) denotes the total population size. The parameter
is the basic reproduction number of model (1.1). Moreover, we can obtain that
-
(i)
if \(R_{0}<1\), model (1.1) has a unique disease-free equilibrium \(E_{0}(1,0,0)\) and it is globally asymptotically stable;
-
(ii)
if \(R_{0}>1\), model (1.1) has positive equilibrium point \(E^{*}\).
In this paper, we assume that both the saturated incidence rate and relapse rate of the disease are affected by environmental perturbations as follows:
\({\dot{B}}_{i}(t)\) \((i=1,2)\) stands for the white noise, that is, \(B_{i}(t)\) is a standard Brownian motion defined on a complete probability space \((\Omega ,{\mathcal {F}},{\mathbb {P}})\) with respect to a filtration \(\{{\mathcal {F}}_{t}\}_{t\in {\mathbb {R}}_{+}}\) satisfying the usual conditions (i.e. it is increasing and right continuous while \({\mathcal {F}}_{0}\) contains all \({\mathbb {P}}\)-null sets). \(B_{1}(0)=0\) and \(B_{2}(0)=0\). \(\sigma _{i}(i=1,2)\) are the standard deviation of the white noise and \(\sigma ^{2}_{i}>0\) represents the white noises intensity [16, 17]. Motivated by the aforementioned factors, we propose the resulting model as follows:
The environmental fluctuation is a crucial part of an ecosystem in the natural world, including temperature, the immunological state of the host, incubation periods, and so on. Sometimes small noise can suppress explosions in population dynamics. The theory of stochastic differential equations (SDE) is a better tool in representing such influencing factors as some additional degree of realism can be provided by SDE compared to the deterministic systems [18]. Numerous scholars introduce the effects of stochastic perturbations in their models both from a biological and mathematical perspective [19,20,21,22,23,24,25,26]. However, to the best of our knowledge, there are no results about the stochastic SIRI system with saturated incidence and relapse.
The remaining of the paper is organized as follows. In Sect. 2, the proof for the existence of a unique positive solution is derived. And also, the threshold dynamics are discussed to govern the persistence and extinction of the stochastic SIRI model. Section 3 presents sufficient conditions to guarantee that the system has an ergodic stationary distribution based on the method of Khasminskii [27]. Numerical simulations are presented in Sect. 4 and finally some conclusions are obtained in Sect. 5. Several numerical simulations were carried out to support theoretical results.
2 Some results for extinction and persistence in the mean
In this part, we will discuss the threshold dynamics to govern the persistence and extinction of the stochastic SIRI model. Considering the characteristics of equation (1.2), we firstly list the following lemma.
Lemma 2.1
([28]) For the solution (S(t), I(t), R(t)) of model (1.1) with any initial value \((S(0), I(0), R(0))\in {\mathbb {R}}^{3}_{+}\), we obtain
By virtue of the above lemma, we denote
is a positively invariant region for model (1.1). In the following, we will discuss the existence and uniqueness of the global positive solution.
Theorem 2.1
There exists a unique solution (S(t), I(t), R(t)) of model (1.2) on \(t\ge 0\) with any initial value \((S(0), I(0), R(0))\in {\mathbb {R}}^{3}_{+}\) and the solution will remain in \({\mathbb {R}}^{3}_{+}\) with probability one.
Proof
According to the local Lipschitz continuity of the coefficients of system (1.2), it can be achieved that there exists a unique local solution (S(t), I(t), R(t)) on \([0, \tau _{e})\) with any initial value \((S(0), I(0), R(0))\in {\mathbb {R}}^{3}_{+}\), where \(\tau _{e}\) represents the explosion time. To prove the globality of the solution, we have to show that \(\tau _{e}=\infty \) a.s. We suppose that \(k_{0}\ge 1\) is sufficiently large such that S(0), I(0) and R(0) lie within the interval \([1/k_{0},k_{0}]\). For each integer \(k>k_{0}\), we define the stopping time \(\tau _{k}\) \(=\inf \{t \in [0,\tau _{e}): \min \{S(t),I(t),R(t)\}\le 1/k \ or \ \max \{S(t),I(t),R(t)\}\ge k\}\). Then, \(\tau _{k}\) increases as \(k\rightarrow \infty \). Denote \(\tau _{\infty }={\lim \limits _{k\rightarrow +\infty }}\tau _{k}\), then \(\tau _{\infty }\le \tau _{e}\). Next, we prove that \(\tau _{\infty }=\infty \) almost surely. If that is not true, constants \(T>0\) and \(\varepsilon \in (0,1)\) exists such that \(P \{\tau _{\infty }<\infty \}>\varepsilon \). Thus, there exists an integer \(k_{1}\ge k_{0}\) satisfying
for all \(k>k_{1}\). Define a \(C^{2}\)-function \(V: {\mathbb {R}}_{+}^{3}\rightarrow {\mathbb {R}}_{+}\) by
where a is a constant that will be given later. By Itô’s formula, we have
Here, \({\mathcal {L}}V: {\mathbb {R}}^{3}_{+}\rightarrow {\mathbb {R}}_{+}\) and choose \(a=\frac{\mu +e}{\beta }\), then we achieve
Herein, K is a constant. Hence,
Taking integral on the above inequality from 0 to \(\tau _{k}\wedge T\), we obtain
where \(\tau _{k}\wedge T =\min \{\tau _{k}, T\}\). Consequently,
Let \(\Omega _{k}=\{\tau _{k}\le T\}\), then we have \(P (\Omega _{k})\ge \varepsilon \). For each \(\omega \in \Omega _{k}\), \(S(\tau _{k},\omega )\) or \(I(\tau _{k},\omega )\) equals either k or 1/k, and
Therefore,
where \(1_{\Omega _{k}}\) is the indicator function of \(\Omega _{k}\). Letting \(k \rightarrow \infty \), we obtain the contradiction.
This completes the proof. \(\square \)
Now we are in the position to give some sufficient conditions for the extinction and persistence in the mean of the disease by constructing suitable Lyapunov functions. Above all, we will discuss the globally asymptotically stable in probability and exponentially stable a.s. of solutions to the equilibrium \(E_{0}\).
Theorem 2.2
Suppose (S(t), I(t), R(t)) be the solution of model (1.2) with any initial value \((S(0), I(0), R(0))\in {\mathbb {R}}^{3}_{+}\). If \(R_{0*}={\tilde{R}}_{0}(1+\frac{\sigma _{1}^{2}}{2\beta })<1\), where \({\tilde{R}}_{0}=\frac{\beta }{( \mu +e)-\frac{e\gamma }{\mu +\frac{\gamma }{1+k}}}\) hold, then the trivial solution of system (1.2) is globally asymptotically stable in probability.
Proof
Define \(V_{1}(S,I,R)=a(1-S)^{2}+bI^{2}+R^{2}\), here \(a,b>0\) will be chosen later. Then applying Itô’s formula, we get
where
The discriminant of H(b) is
We let \((2(\mu +e-\beta )-\sigma _{1}^{2}-\frac{2e\gamma }{\mu +\frac{\gamma }{1+k}})>0\) and a sufficiently small such that \(\Delta >0\), then we can derive that \(H(b)<0\) for every \(b\in [b_{1}, b_{2}]\) where \(b_{1}\) and \(b_{2}\) are distinct positive roots for H(b). Then \({\mathcal {L}}V_{1}\) is negative-definite if \(\left( 2(\mu +e-\beta )-\sigma _{1}^{2}-\frac{2e\gamma }{\mu +\frac{\gamma }{1+k}}\right) >0\). That is,
Thus, \({\mathcal {L}}V_{1}\) is negative-definite if \(R_{0*}={\tilde{R}}_{0}\left( 1+\frac{\sigma _{1}^{2}}{2\beta }\right) <1\).
The proof is completed. \(\square \)
Remark 1
For any \((S(0), I(0), R(0))\in A\), if \(R_{0*}={\tilde{R}}_{0}\left( 1+\frac{\sigma _{1}^{2}}{2\beta }\right) <1\), then the solution of (1.2) satisfies: \(\lim \limits _{t\rightarrow +\infty } S(t)=1,\ \lim \limits _{t\rightarrow +\infty } I(t)=0,\ \lim \limits _{t\rightarrow +\infty } R(t)=0 \quad a.s.\)
Theorem 2.3
Suppose (S(t), I(t), R(t)) be the solution of model (1.2) with any initial value \((S(0), I(0), R(0))\in {\mathbb {R}}^{3}_{+}\).
-
(i)
If \(\beta <\frac{\sigma _{1}^{2}}{2}\), then the solution of system (1.2) obeys
$$\begin{aligned} \limsup \limits _{t\rightarrow +\infty }\frac{1}{t}\mathrm{ln}[(1-S)+I+R]\le \frac{\beta ^{2}-\mu \sigma _{1}^{2}}{\sigma _{1}^{2}}. \end{aligned}$$ -
(ii)
If \(\beta \ge \frac{\sigma _{1}^{2}}{2}\), then the solution of system (1.2) obeys
$$\begin{aligned} \limsup \limits _{t\rightarrow +\infty }\frac{1}{t}\mathrm{ln}[(1-S)+I+R]\le \beta -\mu -\frac{\sigma _{1}^{2}}{4}. \end{aligned}$$
Proof
Define \(V_{2}(S,I,R)=ln[(1-S)+I+R]\), then we have that
Let \(X=\frac{SI}{[(1-S)+I+R]`(1+\alpha I)}\) and \(f(X)=-\sigma _{1}^{2}X^{2}+2\beta X-\mu \), by virtue of Itô’s formula, we obtain that
where \(\nu =\sup \limits _{X\in (0,\frac{1}{2})}f(X)\) obtained from the fact that \(X=\frac{SI}{(1-S)+I+R}=\frac{SI}{2(1-S)(1+\alpha I)}\le \frac{S}{2(1+\alpha I)}\le \frac{1}{2}\). Taking integral on both sides of (2.9) and divided by t, we have that
Notice the fact that \(M(t)=\int _{0}^{t}2\sigma _{1}X(s)dB(s)\) is a continuous local martingale, then by the strong law of large number for local martingales and (2.10), we have that
For the quadratic function f(X), we have that it attains its maximum value \(\nu =f({\bar{X}})=\frac{\beta ^{2}-\mu \sigma _{1}^{2}}{\sigma _{1}^{2}}\) at \(X={\bar{X}}=\frac{\beta }{\sigma _{1}^{2}}\) if \(\beta <\frac{\sigma _{1}^{2}}{2}\) while it reaches its maximum value \(\nu =f({\bar{X}})=\beta -\mu -\frac{\sigma _{1}^{2}}{4}\) at \(X={\bar{X}}=\frac{1}{2}\) if \(\beta \ge \frac{\sigma _{1}^{2}}{2}\).
This completes the proof. \(\square \)
Remark 2
The trivial solution for stochastic model (1.2) is exponentially stable a.s. in A, if one of the following conditions hold:
-
(1)
\(\beta <\min \{\frac{\sigma _{1}^{2}}{2}, \sigma _{1}\sqrt{\mu }\}\);
-
(2)
\(\frac{\sigma _{1}^{2}}{2}\le \beta <\mu +\frac{\sigma _{1}^{2}}{4}\).
As the positive invariance of set A, system (1.2) can be reduced to the following form:
Theorem 2.4
Suppose (S(t), I(t)) be the solution of model (2.11) with any initial value \((S(0), I(0))\in {\mathbb {R}}^{2}_{+}\). If
-
(i)
\(R_{1}=\frac{2\gamma ^{2}}{\sigma _{2}^{2}(\mu +e)}+\frac{2\beta ^{2}(1+\alpha )^{2}}{\sigma _{1}^{2}(\mu +e)} <1\), or
-
(ii)
\(2\beta (1+\alpha )^{2}\ge \sigma _{1}^{2}\) and \(R_{2}=\frac{1}{\mu +e}[2\beta +\frac{2\gamma ^{2}}{\sigma _{2}^{2}}-\frac{\sigma _{1}^{2}}{2(1+\alpha )^{2}}] <1\)
hold, then the infected population go to extinction.
Proof
By virtue of Itô’s formula, we have
Case (i). If \(R_{1}<1\), then we have
where \(M_{1}(t)=\int _{0}^{t}\frac{\sigma _{1}S(\theta )}{1+\alpha I(\theta )}\mathrm{d}B_{1}(\theta )\) and \(M_{2}(t)=\int _{0}^{t}\frac{\sigma _{2}(1-S(\theta )-I(\theta ))}{I(\theta )(1+k(1-S(\theta )-I(\theta )))}\mathrm{d}B_{2}(\theta )\). Thus
Noticing that \(M_{i}(t)\) \((i=1,2)\) are local continuous martingale which satisfy \(M_{i}(0)=0\) and \(\lim _{t\rightarrow +\infty }\frac{M_{i}(t)}{t}=0\quad a.s.,\) by the condition \(R_{1}<1\) and taking the limit superior on both sides of expression (2.14), we derive
Thus, we have that \(\lim _{t\rightarrow +\infty } I(t)=0\).
Case (ii). If \(2\beta (1+\alpha )^{2}\ge \sigma _{1}^{2}\) and \(R_{2}=\frac{1}{\mu +e}[2\beta +\frac{2\gamma ^{2}}{\sigma _{2}^{2}}-\frac{\sigma _{1}^{2}}{2(1+\alpha )^{2}}] <1\), then by (2.12), we obtain
The last inequality is obtained as S(t) in (2.15) takes its maximum value at 1 on the interval [0, 1]. Then
Similarly to the proof of Case (i), we can derive that
which implies \(\lim \limits _{t\rightarrow +\infty } I(t)=0\).
Remark 3
For the reduced system (2.11), if
-
(i)
\(R_{1}=\frac{2\gamma ^{2}}{\sigma _{2}^{2}(\mu +e)}+\frac{2\beta ^{2}(1+\alpha )^{2}}{\sigma _{1}^{2}(\mu +e)} <1\), or
-
(ii)
\(2\beta (1+\alpha )^{2}\ge \sigma _{1}^{2}\) and \(R_{2}=\frac{1}{\mu +e}\left[ 2\beta +\frac{2\gamma ^{2}}{\sigma _{2}^{2}}-\frac{\sigma _{1}^{2}}{2(1+\alpha )^{2}}\right] <1\), then \(\lim \limits _{t\rightarrow +\infty } S(t)=1,\ \lim \limits _{t\rightarrow +\infty } I(t)=0,\ \lim \limits _{t\rightarrow +\infty } R(t)=0 \quad a.s.\)
Theorem 2.5
Suppose (S(t), I(t)) be the solution of model (2.11) with any initial value \((S(0), I(0))\in {\mathbb {R}}^{2}_{+}\). If \(\bar{R_{0}}=\frac{1}{2\mu +e+\gamma }\left[ \frac{\beta }{1+\alpha }+\frac{\sigma _{2}^{2}}{2(1+\gamma )^{2}}-\frac{\sigma _{1}^{2}}{2}\right] >1\) hold, then the infected population I(t) is persistent in the mean,
Proof
For system (2.11), we have
Then
here, \(M_{3}(t)=\int _{0}^{t}\frac{\sigma _{2}(1-S(\theta )-I(\theta ))}{1+k (1-S(\theta )-I(\theta ))}\mathrm{d}B_{2}(\theta )\). Taking limit on both sides of (2.19), we derive that
In the following, define a \(C^{2}-\) function \(V_{3}: {\mathbb {R}}_{+}^{2}\rightarrow \mathbb {R_{+}}\)
Then by Itô’s formula, we get
where
Taking integration on both sides of (2.22) from 0 to t and dividing by t, we obtain
where \(M_{3}(t)=\int _{0}^{t}\frac{\sigma _{2}(S(\theta )+I(\theta ))}{1+k(1-S(\theta )-I(\theta ))}\mathrm{d}B_{2}(\theta )\). Then,
Thus by (2.20), we have
Taking the inferior limit of (2.25), we have
According to Lemma 2.1, we have
The proof is completed. \(\square \)
3 Ergodic stationary distribution of system (1.2)
In this section, we give the ergodic stationary distribution of the SDE model (1.2) as follows:
Theorem 3.1
If \({\widehat{R}}_{0}=\frac{\beta }{\left( \mu +\frac{\beta }{\alpha }+\frac{\sigma ^{2}_{1}}{2(1+\alpha )^{2}}\right) \left( e+\mu +\frac{\sigma ^{2}_{1}}{2}+\frac{\sigma ^{2}_{2}}{2{\bar{m}}^{2}(1+k)^{2}}\right) }>1\), then model (1.2) has a unique stationary distribution \(\pi (\cdot )\).
Proof
First, the validity of the condition (B2) of Lemma 2.1 in Ref. [27] will be verified. Consider a \(C^{2}-\)function \({\widehat{V}}(S,I,R):\) \({\mathbb {R}}_{+}^{3}\rightarrow {\mathbb {R}}\) as follows
here m and M are positive constants satisfying
and
herein,
and
Then it is obvious that
here, \(U_{k}=(\frac{1}{k}, k)\times (\frac{1}{k}, k)\times (\frac{1}{k}, k)\). Thus, \({\widehat{V}}(S,I,R)\) is a continuous function with a minimum point \(({\bar{S}}_{0}, {\bar{I}}_{0}, {\bar{R}}_{0})\) in the interior of \({\mathbb {R}}^{3}_{+}\) and then a nonnegative \(C^{2}-\)function \(V: {\mathbb {R}}_{+}^{3}\rightarrow {\mathbb {R}}\) can be defined by
Making use of Itô’s formula,
Moreover, we derive that
and
Consequently,
In the following, a bounded closed set is defined
where \(\varepsilon >0\) is sufficiently small such that in the set \({\mathbb {R}}_{+}^{3}\setminus D_{\varepsilon }\), it satisfies
where constants \(D, F, G, H>0\) and the expressions (3.10), (3.14), (3.16), and (3.18) are satisfied. Hence, \({\mathbb {R}}_{+}^{3}\setminus D_{\varepsilon }\) can be divided into six domains,
then we prove that \(LV(S,I,R)\le -1\) on \({\mathbb {R}}_{+}^{3}\setminus D_{\varepsilon }\).
Case 1. If \((S,I,R)\in D_{1}\),
where
Making use of (3.3), we have that \(LV\le -1\) for all \((S,I,R)\in D_{1}\).
Case 2. If \((S,I,R)\in D_{2}\),
By inequality (3.4), we obtain \(LV\le -1\) for all \((S,I,R)\in D_{2}\).
Case 3. If \((S,I,R)\in D_{3}\),
Applying (3.5), we achieve \(LV\le -1\) for all \((S,I,R)\in D_{3}\).
Case 4. If \((S,I,R)\in D_{4}\),
where
Combining with (3.6), it can be derived that \(LV\le -1\) for all \((S,I,R)\in D_{4}\).
Case 5. If \((S,I,R)\in D_{5}\),
where
According to the inequality (3.7), it can be got that \(LV\le -1\) for all \((S,I,R)\in D_{5}\).
Case 6. If \((S,I,R)\in D_{6}\),
where
By the inequality (3.8), we have \(LV\le -1\) for all \((S,I,R)\in D_{6}\).
Therefore, according to (3.9), (3.11), (3.12), (3.13), (3.15), and (3.17), we derive \(LV\le -1\), for all \((S,I,R)\in {\mathbb {R}}_{+}^{3}\setminus D_{\varepsilon }\) with sufficiently small constant \(\varepsilon >0\).
Next, the diffusion matrix of system (1.1) can be defined as
Suppose \(M=\min _{(S,I,R)\in {\bar{D}}_{\varepsilon }}\{ \frac{\sigma _{1}^{2}S^{2}I^{2}}{(1+\alpha I)^{2}},\frac{\sigma _{2}^{2}R^{2}}{(1+k R)^{2}}+\frac{\sigma _{1}^{2}S^{2}I^{2}}{(1+\alpha I)^{2}}, \frac{\sigma _{2}^{2}R^{2}}{(1+k R)^{2}}\}\), then we achieve
\( (S,I,R)\in {\bar{D}}_{\varepsilon }, \xi \in {\mathbb {R}}^{3}\). Hence, the condition (B1) of Lemma 2.1 in Ref. [27] is proven.
Therefore, it follows from Lemma 2.1 in Ref. [27] that system (1.1) has a unique stationary distribution and the ergodicity holds. This completes the proof. \(\square \)
4 Numerical simulations
In this part, we will introduce the Milstein method [29] to simulate our main theoretical findings for stochastic system (1.2) and the reduced system (2.11). And we will discuss the effect of environmental fluctuations and relapse to the transmission dynamics of the disease.
4.1 Case 1: for the system (1.2)
Example 1
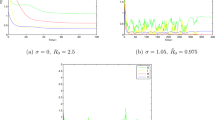
Consider \(\beta =0.2\), \(\mu =0.2\), \(\alpha =2.7\), \(k=0.8\), \(e=1.6\), \((S(0),I(0),R(0))=(0.7,1.8,1.6)\). Furthermore, we let \(\gamma =0.2\), \(\sigma _{1}=0.9\) and \(\sigma _{2}=1.2\). We have that \(R_{0*}=0.9787<1\) and it supports the theoretical result in Theorem 2.2. The disease dies out with probability 1. (See Fig. 1).
Example 2
We keep the parameters the same as in Example 1 except \(\gamma =3.2\). Then we calculate that \(R_{0}=1.19>1\) and \(\beta =0.35<\min \{\frac{\sigma _{1}^{2}}{2},\sigma _{1}\sqrt{\mu }\}=0.4025\). By virtue of Remark 2, the solutions of stochastic model (1.2) will tend exponentially to \(E_{0}(1,0,0)\) a.s. while the disease-free equilibrium of the corresponding deterministic model (1.1) is unstable. Thus, the density of noises can greatly change the disease dynamics (See Fig. 2).
4.2 Case 2: for the reduced system (2.11)
Example 3
Consider \(\beta =0.2\), \(\mu =0.2\), \(\alpha =0.7\), \(k=1.8\), \(\gamma =1.2\), \(e=2.6\), \(\sigma _{1}=0.8\), \(\sigma _{2}=1.2\), and \((S(0),I(0))=(0.7,1.8)\). We can obtain that \(R_{1}=0.8433<1\) which satisfy case (i) of Theorem 2.4 (See Fig. 3a). For Fig. 3b, we choose \(\sigma _{1}=0.95\), \(\sigma _{2}=1.1\), we have that \(2\beta (1+\alpha )^{2}-\sigma _{1}^{2}=0.2535>0, R_{2}=0.9372<1\), which satisfy case (ii) of Theorem 2.4. The disease is extinct. Moreover, from the expression of \(R_{1}\) and \(R_{2}\) and Fig. 3, we can conclude that \(R_{1}>R_{2}\).
Time sequence diagram of stochastic system (2.11)
Example 4
The effect of relapse rate \(\gamma \) to the dynamics of the system is investigated. Fix \(\gamma =1.2,3.5\) and 6, respectively and the other parameters are the same as in Example 3. It can be shown that the relapse rate do greatly effect the disease dynamics and the stronger relapse rate will be not beneficial to the extinction of the disease and Fig. 4 clearly support this result.
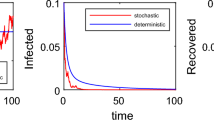
Example 5
Choose \(\beta =0.7\), \(\mu =0.05\), \(\alpha =0.9\), \(k=1.4\), \(\gamma =0.1\), \(e=0.1\), \(\sigma _{1}=0.3\), \(\sigma _{2}=0.3\), and we derive \(\bar{R_{0}}=1.202>1\). By Theorem 2.5, we can observe that the stochastic system (2.11) is persistent in mean when the random fluctuations are small (Fig. 5). In addition, comparing Figs. 3a, b and 6, where the only difference is the density of white noises, we obtain that if the random fluctuations are large enough, the disease can be led to extinction.
5 Conclusion
This paper discusses a Susceptible-Infected-Recovered-Infected epidemic model with random disturbances. The advantage of it is introducing double saturated incidence rate and relapse. The existence and uniqueness of the global solution for model (1.2) is obtained, and the stochastic endemic dynamics of this model is analyzed. Persistence and extinction of the epidemic diseases are derived. Moreover, by constructing some appropriate Lyapunov functions and applying the method of Khasminskii, we derive sufficient conditions to guarantee the existence of an ergodic stationary distribution.
In addition, we obtain some thresholds \(R_{0*}\), \(\bar{R_{0}}\), and \(R_{i}\) \((i=1,2)\) for system (1.2) and (2.11). We conclude that
- (a):
-
for system (1.2),(i)if \(R_{0*}={\tilde{R}}_{0}(1+\frac{\sigma _{1}^{2}}{2\beta })<1\), then the solution is globally asymptotically stable in probability; (ii) if \(\beta <\min \{\frac{\sigma _{1}^{2}}{2}, \sigma _{1}\sqrt{\mu }\}\), or \(\frac{\sigma _{1}^{2}}{2}\le \beta <\mu +\frac{\sigma _{1}^{2}}{4}\) hold, the solution is exponentially stable a.s. in A.
- (b):
-
for the reduced system (2.11), if (i) \(R_{1}=\frac{2\gamma ^{2}}{\sigma _{2}^{2}(\mu +e)}+\frac{2\beta ^{2}(1+\alpha )^{2})}{\sigma _{1}^{2}(\mu +e)} <1\), or \(2\beta (1+\alpha )^{2}\ge \sigma _{1}^{2}\) and \(R_{2}=\frac{1}{\mu +e}[2\beta +\frac{2\gamma ^{2}}{\sigma _{2}^{2}}-\frac{\sigma _{1}^{2}}{2(1+\alpha )^{2}}] <1\) hold, then the infected population go to extinction; (ii)if \(\bar{R_{0}}=\frac{1}{2\mu +e+\gamma }[\frac{\beta }{1+\alpha }+\frac{\sigma _{2}^{2}}{2(1+\gamma )^{2}}-\frac{\sigma _{1}^{2}}{2}]>1\) hold, then the infected population I(t) is persistent in the mean.
Moreover, from the expression and numerical simulations, we could conclude that \(R_{0*}>\tilde{R_{0}}>R_{0}\) and \(R_{1}>R_{2}\). Also, according to the above discussion, we can achieve that the intensity of noises and relapse do greatly affect the dynamics of epidemic systems and if the random fluctuations are large enough, the disease can be led to extinction while the stronger relapse rate will be not beneficial to the extinction of the disease.
Finally, we present an interesting question. If the effects of time delays are also considered in the formulation of the stochastic SIRI model, what will happen for the dynamics of the model? Moreover, in recent years, some authors have introduced some more appropriate definitions of permanence for stochastic population model, for example, Ref. [30, 31]. We left these for future study.
References
Anderson, R., May, R.: Population biology of infectious diseases: Part I. Nature 280, 361–367 (1979)
Ruan, S., Wang, W.: Dynamical behavior of an epidemic model with a nonlinear incidence rate. J. Differ. Equ. 188, 135–163 (2003)
Meng, X., Chen, L., Wu, B.: A delay SIR epidemic model with pulse vaccination and incubation times. Nonlinear Anal. Real. 11(1), 88–98 (2010)
Wang, W.: Global behavior of an SEIRS epidemic model with time delays. Appl. Math. Lett. 15(4), 423–428 (2002)
Blower, S.: Modelling the genital herpes epidemic. Herpes 3(Suppl 3), 138A-146A (2004)
Wildy, P., Field, H., Nash, A.: Classical herpes latency revisited. In: Mahy, B.W., Minson, A.C., Darby, G.K. (eds.) Virus Persistence Symposium, 33, pp. 133–168. Cambridge University Press, Cambridge (1982)
Liu, Y., Zhang, X., et al.: Tuberculosis relapse is more common than reinfection in Beijing, China. Infect. Dis. Nor. 52, 858–865 (2020). https://doi.org/10.1080/23744235.2020.1794027
Driessche, P.V.D., Zou, X.: Modeling relapse in infectious diseases. Math. Biosci. 207, 89–103 (2007)
Fatini, M., Lahrouz, A., Pettersson, R., Settati, A., Taki, R.: Stochastic stability and instability of an epidemic model with relapse. Appl. Math. Comput. 316, 326–341 (2018)
Georgescu, P., Zhang, H.: A Lyapunov functional for a SIRI model with nonlinear incidence of infection and relapse. Appl. Math. Comput. 219(16), 8496–8507 (2013)
Capasso, V., Serio, G.: A generalization of the Kermack–McKendrick deterministic epidemic model. Math. Bios. 42, 41–61 (1978)
Liu, W.M., Levin, S.A., Iwasa, Y.: Influence of nonlinear incidence rates upon the behavior of SIRS epidemiological models. J. Math. Biol. 23, 187–204 (1986)
Xu, R., Ma, Z.: Global stability of a delayed SEIRS epidemic model with saturation incidence rate. Nonlinear Dyn. 61, 229–239 (2010)
Xu, R., Ma, Z.: Stability of a delayed SIRS epidemic model with a nonlinear incidence rate. Chaos Soliton Fract. 41(5), 2319–2325 (2009)
Zhang, T., Teng, Z.: Global asymptotic stability of a delayed SEIRS epidemic model with saturation incidence. Chaos Soliton Fract. 37, 1456–1468 (2008)
Mao, X.: Stochastic Differential Equations and Applications. Horwood Publishing, Sawston (2007)
Liu, M., Wang, K., Wu, Q.: Survival analysis of stochastic competitive models in a polluted environment and stochastic competitive exclusion principle. Bull. Math. Biol. 73, 1969–2012 (2011)
Liu, Q., Jiang, D.: Stationary distribution of a stochastic SIS epidemic model with double diseases and the Beddington–DeAngelis incidence. Chaos 27, 083126 (2017)
Ball, F., Neal, P.: A general model for stochastic SIR epidemics with two levels of mixing. Math. Biosci. 180, 73–102 (2002)
Zhang, Y., Fan, K., Gao, S., Chen, S.: A remark on stationary distribution of a stochastic SIR epidemic model with double saturated rates. Appl. Math. Lett. 76, 46–52 (2018)
Zhang, Y., Chen, S., Gao, S., Wei, X.: Stochastic periodic solution for a perturbed non-autonomous predator-prey model with generalized nonlinear harvesting and impulses. Phys. A 486, 347–366 (2017)
Liu, M., Bai, C.: Optimal harvesting of a stochastic mutualism model with lévy jumps. Appl. Math. Comput. 276, 301–309 (2016)
Sardar, M., Khajanchi, S.: Is the Allee effect relevant to stochastic cancer model? J. Appl. Math. Comput. (2021). https://doi.org/10.1007/s12190-021-01618-6
Teng, Z., Wang, L.: Persistence and extinction for a class of stochastic SIS epidemic models with nonlinear incidence rate. Phys. A 451, 507–518 (2016)
Zhao, Y., Jiang, D.: The threshold of a stochastic sis epidemic model with vaccination. Appl. Math. Comput. 243, 718–727 (2014)
Zhang, S., Meng, X., Feng, T., Zhang, T.: Dynamics analysis and numerical simulations of a stochastic non-autonomous predator-prey system with impulsive effects. Nonlinear Anal. Hybri. 26, 19–37 (2017)
Khasminskii, R.: Stochastic Stability of Differential Equations, 2nd edn. Springer, Berlin, Heidelberg (2012)
Zhang, S., Meng, X., Wang, X.: Application of stochastic inequalities to global analysis of a nonlinear stochastic SIRS epidemic model with saturated treatment function. Adv. Differ. Equ. N. Y. 2018, 50 (2018)
Higham, D.: An algorithmic introduction to numerical simulation of stochastic differential equations. SIAM Rev. 43, 525–546 (2001)
Schreiber, S., Benaïm, M., Atchadé, K.: Persistence in fluctuating environments. J. Math. Biol. 62, 655–683 (2011)
Liu, M., Bai, C.: Analysis of a stochastic tri-trophic food-chain model with harvesting. J. Math. Biol. 73, 597–625 (2016)
Acknowledgements
This work is supported by The National Natural Science Foundation of China (11901110,11961003), and the Natural Science Foundation of Jiangxi (20192BAB211003).
Author information
Authors and Affiliations
Contributions
All authors contributed substantially to this paper, participated in drafting and checking the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no competing of interests regarding the publication of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, Y., Gao, S. & Chen, S. Stochastic analysis of a SIRI epidemic model with double saturated rates and relapse. J. Appl. Math. Comput. 68, 2887–2912 (2022). https://doi.org/10.1007/s12190-021-01646-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12190-021-01646-2