Abstract
Background
Eradication of micrometastases present in lymph nodes of cancer patients improves their prognosis significantly. Radionuclide therapy possesses the potential to eliminate such metastases.
Objective
This study was performed to evaluate the efficacy and safety of 32P-nanocolloid therapy in the treatment of distant carcinoma cell metastases in lymph nodes of VX2 tumor-bearing rabbits.
Methods
To obtain VX2 tumor micrometastases in right armpit lymph nodes of 12 male New Zealand white rabbits, VX2 tumors were implanted by hypodermal inoculation into the right anterior limb. Animals were randomly divided into therapy (n = 6) and control (n = 6) groups. 32P-nanocolloid (0.5 mCi), 95% of which was >50 nm in diameter, was administered to the therapy group, and saline was administered to the control group. Injections were given once weekly for 4 weeks.
Results
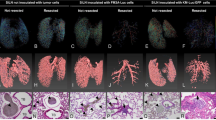
2-Deoxy-2[18F]-fluoro-d-glucose positron emission tomography revealed that the number of involved lymph nodes and the maximum standardized uptake value decreased in the 32P-nanocolloid therapy group as compared with the baseline or saline control grou0 (P < 0.05). The expression of the lymphangiogenesis factors vascular endothelial growth factors (VEGF)-C and VEGF-D by VX2 tumor cells present in lymph nodes was significantly lower in the therapy group as compared with the control group. Additionally, apoptotic VX2 tumor cell death was significantly greater in lymph nodes of the therapy as compared with the control group (P < 0.01). With the exception of a decrease in white blood cells of peripheral blood (P < 0.05), standard laboratory values were unaffected throughout the course of therapy with 32P-nanocolloid.
Conclusions
These findings support treatment with 32P-nanocolloid as a safe and effective approach for eradication of lymph node micrometastases.
Similar content being viewed by others
References
Pantel K, Woelfle U. Micrometastasis in breast cancer and other solid tumors. J Biol Regul Homeost Agents 2004;18:120–125.
Ji RC. Lymphatic endothelial cells, tumor lymphangiogenesis and metastasis: new insights into intratumoral and peritumoral lymphatics. Cancer Metastasis Rev 2006;25:677–694.
Harisinghani MG, Weissleder R. Sensitive, noninvasive detection of lymph node metastases. PloS Med 2004;1:e66.
Wissmann C, Detmar M. Pathways targeting tumor lymphangiogenesis. Clin Cancer Res 2006;12:6865–6868.
Stacker SA, Baldwin ME, Achen MG. The role of tumor lymphangiogenesis in metastatic spread. FASEB J 2002;16:922–934.
Schoppmann SF, Birner P, Stockl J, Kalt R, Ullrich R, Caucig C, et al. Tumor-associated macrophages express lymphatic endothelial growth factors and are related to peritumoral lymphangiogenesis. Am J Pathol 2002;161:947–956.
Pepper MS, Tille JC, Nisato R, Skobe M. Lymphangiogenesis and tumor metastasis. Cell Tissue Res 2003;314:167–177.
Byrne AM, Bouchier-Hayes DJ, Harmey JH. Angiogenic and cell survival functions of vascular endothelial growth factor (VEGF). J Cell Mol Med 2005;9:777–794.
Syme A, McQuarrie S, Fallone BG. Beta dose-rate distributions in microscopic spherical tumors for intraperitoneal radioimmunotherapy. Int J Radiat Oncol Biol Phys 2003;56:1495–1506.
Brans B, Linden O, Giammarile F, Tennvall J, Punt C. Clinical applications of newer radionuclide therapies. Eur J Cancer 2006;42:994–1003.
Sullivan DC, Harris CC, Currie JL, Wilkinson RH, Creasman WT. Observations on the intraperitoneal distribution of chromic phosphate (32P) suspension for intraperitoneal therapy. Radiology 1983;146:539–541.
Spencer TR Jr, Marks RD Jr, Fenn JO, Jenrette JM III, Lutz MH. Intraperitoneal P-32 after negative second-look laparotomy in ovarian carcinoma. Cancer 1989;63:2434–2437.
Meredith RF, Buchsbaum DJ, Alvarez RD, LoBuglio AF. Brief overview of preclinical and clinical studies in the development of intraperitoneal radioimmunotherapy for ovarian cancer. Clin Cancer Res 2007;13:5643s–5645s.
Goh AS, Chung AY, Lo RH, Lau TN, Yu SW, Chng M, et al. A novel approach to brachytherapy in hepatocellular carcinoma using a phosphorous32 (32P) brachytherapy delivery device: a first-in-man study. Int J Radiat Oncol Biol Phys 2007;67:786–792.
Izbicki R, Weyhing BT III, Baker L, Caoili EM, Vaitkevicius VK. Pleural effusion in cancer patients: a prospective randomized study of pleural drainage with the addition of radioactive phosphorous to the pleural space vs. pleural drainage alone. Cancer 1975;36:1511–1518.
Jackson GL, Blosser NM. Intracavitary chromic phosphate (32P) colloidal suspension therapy. Cancer 1981;48:2596–2598.
Rogoff EE, Hilaris BS, Huvos AG. Long-term survival in patients with malignant peritoneal mesothelioma treated with irradiation. Cancer 1973;32:656–664.
Alimi KA, Firusian N, Dempke W. Effects of intralesional 32-P chromic phosphate in refractory patients with head and neck tumors. Anticancer Res 2007;27:2997–3000.
Westlin JE, Andersson-Forsman C, Garske U, Linne T, Aas M, Glimelius B, et al. Objective responses after fractionated infusional brachytherapy of unresectable pancreatic adenocarcinomas. Cancer 1997;80:2743–2748.
DeNittis AS, Stambaugh MD, Lang P, Wallner PE, Lustig RA, Dillman RO, et al. Complete remission of nonresectable pancreatic cancer after infusional colloidal phosphorus-32 brachytherapy, external beam radiation therapy, and 5- fluorouracil: a preliminary report. Am J Clin Oncol 1999;22:355–360.
Lord RS, Kinmonth JB. Histologic effects of endolymphatic radiotherapy. Cancer 1969;23:440–450.
Kassis AI, Adelstein SJ. Radiobiologic principles in radionuclide therapy. J Nucl Med 2005;46:4S–12S.
Ikomi F, Hanna GK, Schmid-Schonbein GW. Mechanism of colloidal particle uptake into the lymphatic system: basic study with percutaneous lymphography. Radiology 1995;196:107–113.
Uenosono Y, Natsugoe S, Higashi H, Ehi K, Miyazono F, Ishigami S, et al. Evaluation of colloid size for sentinel nodes detection using radioisotope in early gastric cancer. Cancer Lett 2003;200:19–24.
Tsopelas C. Particle size analysis of (99m)Tc-labeled and unlabeled antimony trisulfide and rhenium sulfide colloids intended for lymphoscintigraphic application. J Nucl Med 2001;42:460–466.
Pandey U, Sarma HD, Ingle AD, Kulloli BS, Samuel G, Venkatesh M. Radioactive skin bandages incorporating 32P for treatment of superficial tumors. Cancer Biother Radiopharm 2006;21:257–262.
Sapundzhiev NR, Dunne AA, Ramaswamy A, Sitter H, Davis RK, Werner JA. Lymph node metastasis in an animal model: effect of piecemeal laser surgical resection. Lasers Surg Med 2005;36:371–376.
Ege GN. Radiocolloid lymphoscintigraphy in neoplastic disease. Cancer Res 1980;40:3065–3071.
Jansen L, Nieweg OE, Kapteijn AE, Valdes Olmos RA, Muller SH, Hoefnagel CA, et al. Reliability of lymphoscintigraphy in indicating the number of sentinel nodes in melanoma patients. Ann Surg Oncol 2000;7:624–630.
Mariani G, Moresco L, Viale G, Villa G, Bagnasco M, Canavese G, et al. Radioguided sentinel lymph node biopsy in breast cancer surgery. J Nucl Med 2001;42:1198–1215.
Kitagawa Y, Fujii H, Mukai M, Kubo A, Kitajima M. Current status and future prospects of sentinel node navigational surgery for gastrointestinal cancers. Ann Surg Oncol 2004;11:242S–244S.
Leichner PK, Rosenshein NB, Leibel SA, Order SE. Distribution and tissue dose of intraperitoneally administered radioactive chronic phosphate (32P) in New Zealand white rabbits. Radiology 1980;134:729–734.
Kaplan WD, Zimmerman RE, Bloomer WD, Knapp RC, Adelstein SJ. Therapeutic intraperitoneal 32P: a clinical assessment of the dynamics of distribution. Radiology 1981;138:683–688.
Daozhen C, Lu L, Guansheng T, Zhiyong L, Xudong L, Ying H. Preventing and treating lymphatic minute metastasis with (32)p-chromic phosphate during an operation. Cancer Biother Radiopharm 2007;22:24–32.
Verheij M, Bartelink H. Radiation-induced apoptosis. Cell Tissue Res 2000;301:133–142.
Lee I. Enhanced tumor targeting by an intratumoral injection of colloidal chromic 32P in two human tumors (AsPC-1 pancreas and Ls174T colon) in nude mice. J Surg Oncol 1999;70:161–166.
Akine Y, Tokita N, Tokuuye K, Satoh M, Churei H, Le Pechoux C, et al. Suppression of rabbit VX-2 subcutaneous tumor growth by gadolinium neutron capture therapy. Jpn J Cancer Res 1993;84:841–843.
Lee I, Wallner PE. Evaluation of cellular uptake, tumor retention, radiation response, and tumor pathophysiology in experimental solid tumors after an intratumoral infusion of colloidal 32P. Cancer 1997;80:2611–2617.
Liu L, Feng GS, Gao H, Tong GS, Wang Y, Gao W, et al. Chromic-P32 phosphate treatment of implanted pancreatic carcinoma: mechanism involved. World J Gastroenterol 2005;11:2101–2108.
Kaluzova M, Kaluz S, Lerman MI, Stanbridge EJ. DNA damage is a prerequisite for p53-mediated proteasomal degradation of HIF-1alpha in hypoxic cells and downregulation of the hypoxia marker carbonic anhydrase IX. Mol Cell Biol 2004;24:5757–5766.
Moreno-Sanchez R, Rodriguez-Enriquez S, Marin-Hernandez A, Saavedra E. Energy metabolism in tumor cells. FEBS J 2007;274:1393–1418.
Weber WA. Use of PET for monitoring cancer therapy and for predicting outcome. J Nucl Med 2005;46:983–995.
Oya N, Nagata Y, Tamaki N, Takagi T, Murata R, Magata Y, et al. FDG-PET evaluation of therapeutic effects on VX2 liver tumor. J Nucl Med 1996;37:296–302.
Basu S, Zaidi H, Houseni M, Bural G, Udupa J, Acton P, et al. Novel quantitative techniques for assessing regional and global function and structure based on modern imaging modalities: implications for normal variation, aging and diseased states. Semin Nucl Med 2007;37:223–239.
Zhuang H, Pourdehnad M, Lambright ES, Yamamoto AJ, Lanuti M, Li P, et al. Dual time point 18F-FDG PET imaging for differentiating malignant from inflammatory processes. J Nucl Med 2001;42:1412–1417.
Ishii K, Hosono MN, Wada Y, Maeda M, Kondo S, Takada Y, et al. Usefulness of FDG-microPET for early evaluation of therapeutic effects on VX2 rabbit carcinoma. Ann Nucl Med 2006;20:123–130.
Bhana N. Granulocyte colony-stimulating factors in the management of chemotherapy-induced neutropenia: evidence based review. Curr Opin Oncol 2007;19:328–335.
Leichner PK, Rosenshein NB, Leibel SA, Order SE. Distribution and tissue dose of intraperitoneally administered radioactive chromic phosphate (32P) in New Zealand white rabbits. Radiology 1980;134:729–734.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dong, S., Huang, G., Liu, P. et al. Efficacy and safety of 32P-nanocolloid for treatment of distant lymph node metastasis in VX2 tumor-bearing rabbits. Ann Nucl Med 22, 849–858 (2008). https://doi.org/10.1007/s12149-008-0188-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-008-0188-2