Abstract
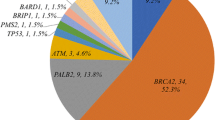
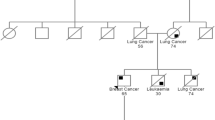
In this study, we evaluated the incidence of pathogenic germline mutations in 30 breast cancer susceptibility genes in breast cancer patients. Our aim was to understand the involvement of the inherited mutations in these genes in a breast cancer cohort. Two hundred ninety-six female breast cancer patients including 4.5% of familial breast cancer cases were included in the study. 200 ng of genomic DNA was used to evaluate the pathogenic mutations, detected using Global Screening Array (GSA) microchip (Illumina Inc.) according to the manufacturer’s instructions. The pathogenic frameshift and nonsense mutations were observed in BRCA2 (10.9%), MLH1 (58.6%), MTHFR (50%), MSH2 (14.2%), and CYTB (52%) genes. Familial breast cancer patients (4.5%) had variations in BRCA2, MLH1, MSH2, and CYTB genes. 28% of patients with metastasis, recurrence, and death harbored mono/biallelic alterations in MSH2, MLH1, and BRCA2 genes. The results of this study can guide to develop a panel to test the breast cancer patients for pathogenic mutations, from Malwa region of Punjab. The screening of MSH2, MLH1, and BRCA2 should be carried in individuals with or without family history of breast cancer as these genes have been reported to increase the cancer risk by tenfold.

Similar content being viewed by others
References
Mavaddat N, Antoniou AC, Easton DF, Garcia-Closas M. Genetic susceptibility to breast cancer. Mol Oncol. 2010;4(3):174–91.
Lalloo F, Evans DG. Familial breast cancer. Clin Genet. 2012;82(2):105–14.
Saxena S, Chakraborty A, Kaushal M, Kotwal S, Bhatanager D, Mohil RS. Contribution of germline BRCA1 and BRCA2 sequence alterations to breast cancer in Northern India. BMC Med Genet. 2006;4:75.
Blaurock-Busch E, Busch YM, Friedle A, Buerner H, Parkash C, Kaur A. Comparing the metal concentration in the hair of cancer patients and healthy people living in the Malwa region of Punjab, India. Clin Med Insights Oncol. 2014;8:1.
Thakur J, Rao B, Rajwanshi A, Parwana H, Kumar R. Epidemiological study of high cancer among rural agricultural community of Punjab in Northern India. Int J Environ Res Public Health. 2008;5(5):399–407.
Ali I, Wani WA, Saleem K. Cancer scenario in India with future perspectives. Cancer Ther. 2011;8(1):56–70.
Bedi J, Gill J, Aulakh R, Kaur P, Sharma A, Pooni P. Pesticide residues in human breast milk: risk assessment for infants from Punjab, India. Sci Total Environ. 2013;463:720–26.
Berzina D, Nakazawa-Miklasevica M, Zestkova J, Aksenoka K, Irmejs A, Gardovskis A, Kalniete D, Gardovskis J, Miklasevics E. BRCA1/2 mutation screening in high-risk breast/ovarian cancer families and sporadic cancer patient surveilling for hidden high-risk families. BMC Med Genet. 2013;14(1):61.
Ripperger T, Gadzicki D, Meindl A, Schlegelberger B. Breast cancer susceptibility: current knowledge and implications for genetic counselling. Eur J Hum Genet. 2009;17(6):722–31.
Levy-Lahad E, Friedman E. Cancer risks among BRCA1 and BRCA2 mutation carriers. Br J Cancer. 2007;96(1):11–5.
Shailani A, Kaur RP, Munshi A. A comprehensive analysis of BRCA2 gene: focus on mechanistic aspects of its functions, spectrum of deleterious mutations, and therapeutic strategies targeting BRCA2-deficient tumors. Med Oncol. 2018;35(3):18.
Vaidyanathan K, Lakhotia S, Ravishankar HM, Tabassum U, Mukherjee G, Somasundaram K. BRCA1 and BRCA2 germline mutation analysis among Indian women from south India: identification of four novel mutations and high-frequency occurrence of 185delAG mutation. J Biosci. 2009;34(3):415–22.
Kim H, Choi DH. Distribution of BRCA1 and BRCA2 mutations in Asian patients with breast cancer. J Breast Cancer. 2013;16(4):357–65.
Peshkin BN, Alabek ML, Isaacs C. BRCA1/2 mutations and triple negative breast cancers. Breast Dis. 2011;32(1–2):25–33.
Meyer P, Landgraf K, Högel B, Eiermann W, Ataseven B. BRCA2 mutations and triple-negative breast cancer. PLoS ONE. 2012;7(5):e38361.
Tung N, Battelli C, Allen B, Kaldate R, Bhatnagar S, Bowles K, Timms K, Garber JE, Herold C, Ellisen L, Krejdovsky J. Frequency of mutations in individuals with breast cancer referred for BRCA1 and BRCA2 testing using next-generation sequencing with a 25-gene panel. Cancer. 2015;121(1):25–33.
Dedes KJ, Wilkerson PM, Wetterskog D, Weigelt B, Ashworth A, Reis-Filho JS. Synthetic lethality of PARP inhibition in cancers lacking BRCA1 and BRCA2 mutations. Cell Cycle. 2011;10:1192–9.
Rennert G, Lejbkowicz F, Cohen I, Pinchev M, Rennert HS, Barnett-Griness O. MutYH mutation carriers have increased breast cancer risk. Cancer. 2012;118(8):1989–93.
Vogt S, Jones N, Christian D, Engel C, Nielsen M, Kaufmann A, Steinke V, Vasen HF, Propping P, Sampson JR, Hes FJ. Expanded extracolonic tumor spectrum in MUTYH-associated polyposis. Gastroenterology. 2009;137(6):1976–85.
Wasielewski M, Out AA, Vermeulen J, Nielsen M, van den Ouweland A, Tops CM, Wijnen JT, Vasen HF, Weiss MM, Klijn JG, Devilee P. Increased MUTYH mutation frequency among Dutch families with breast cancer and colorectal cancer. Breast Cancer Res Treat. 2010;124(3):635–41.
Lo YL, Hsiao CF, Jou YS, Chang GC, Tsai YH, Su WC, Chen KY, Chen YM, Huang MS, Hsieh WS, Chen CJ. Polymorphisms of MLH1 and MSH2 genes and the risk of lung cancer among never smokers. Lung Cancer. 2011;72(3):280–6.
Harkness EF, Barrow E, Newton K, Green K, Clancy T, Lalloo F, Hill J, Evans DG. Lynch syndrome caused by MLH1 mutations is associated with an increased risk of breast cancer: a cohort study. J Med Genet. 2015;52(8):553–6.
Cybulski C, Wokołorczyk D, Jakubowska A, Huzarski T, Byrski T, Gronwald J, Masojć B, Dębniak T, Górski B, Blecharz P, Narod SA. Risk of breast cancer in women with a CHEK2 mutation with and without a family history of breast cancer. J Clin Oncol. 2011;29(28):3747–52.
Thompson D, Duedal S, Kirner J, McGuffog L, Last J, Reiman A, Byrd P, Taylor M, Easton DF. Cancer risks and mortality in heterozygous ATM mutation carriers. J Natl Cancer Inst. 2005;97(11):813–22.
Weischer M, Bojesen SE, Ellervik C, Tybjaerg-Hansen A, Nordestgaard BG. CHEK2*1100delC genotyping for clinical assessment of breast cancer risk: meta-analyses of 26,000 patient cases and 27,000 controls. J Clin Oncol. 2008;26:542–8.
Podralska MJ, Stembalska A, Ślęzak R, Lewandowicz-Uszyńska A, Pietrucha B, Kołtan S, Wigowska-Sowińska J, Pilch J, Mosor M, Ziółkowska-Suchanek I, Dzikiewicz-Krawczyk A. Ten new ATM alterations in Polish patients with ataxia-telangiectasia. Mol Genet Genomic Med. 2014;2(6):504–11.
Jalilvand M, Oloomi M, Najafipour R, Alizadeh SA, Saki N, Rad FS, Shekari M. An association study between CHEK2 gene mutations and susceptibility to breast cancer. Comp Clin Pathol. 2017;26:1–9.
Apostolou P, Papasotiriou I. Current perspectives on CHEK2 mutations in breast cancer. Breast Cancer: Targets Ther. 2017;9:331.
Kim YI. Folate and carcinogenesis: evidence, mechanisms, and implications. J Nutr Biochem. 1999;10(2):66–88.
Mason JB, Choi SW. Folate and carcinogenesis: developing a unifying hypothesis. Adv Enzyme Regul. 2000;40(1):127–41.
Ma LM, Ruan LH, Yang HP. Meta-analysis of the association of MTHFR polymorphisms with multiple myeloma risk. Sci Rep 2015; 5.
Beetstra S, Thomas P, Salisbury C, Turner J, Fenech M. Folic acid deficiency increases chromosomal instability, chromosome 21 aneuploidy and sensitivity to radiation-induced micronuclei. Mutat Res. 2005;578(1):317 – 26.
Ergul E, Sazci A, Utkan Z, Canturk NZ. Polymorphisms in the MTHFR gene are associated with breast cancer. Tumor Biol. 2003;24(6):286 – 90.
Tan DJ, Bai RK, Wong LJ. Comprehensive scanning of somatic mitochondrial DNA mutations in breast cancer. Cancer Res. 2002;62(4):972–6.
Zhu W, Qin W, Bradley P, Wessel A, Puckett CL, Sauter ER. Mitochondrial DNA mutations in breast cancer tissue and in matched nipple aspirate fluid. Carcinogenesis. 2005;26(1):145–52.
Ratajska M, Antoszewska E, Piskorz A, Brozek I, Borg Å, Kusmierek H, Biernat W, Limon J. Cancer predisposing BARD1 mutations in breast–ovarian cancer families. Breast Cancer Res Treat. 2012;131(1):89–97.
Klonowska K, Ratajska M, Czubak K, Kuzniacka A, Brozek I, Koczkowska M, Sniadecki M, Debniak J, Wydra D, Balut M, Stukan M. Analysis of large mutations in BARD1 in patients with breast and/or ovarian cancer: the Polish population as an example. Sci Rep. 2015; 5.
Michailidou K, Lindström S, Dennis J, Beesley J, Hui S, Kar S, Lemaçon A, Soucy P, Glubb D, Rostamianfar A, Bolla MK. Association analysis identifies 65 new breast cancer risk loci. Nature. 2017;551(7678):92–4.
Funding
The authors are thankful to the Central University of Punjab (CUPB) for providing financial assistance for the present study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors do not hold any conflict of interest.
Ethical approval
The study was carried out only after the ethical clearance by Institutional Ethics Committee (IES) of CUPB and the study hospital.
Informed consent
The samples and information were collected from the participants only after their written informed consent.
Rights and permissions
About this article
Cite this article
Kaur, R.P., Shafi, G., Benipal, R.P.S. et al. Frequency of pathogenic germline mutations in cancer susceptibility genes in breast cancer patients. Med Oncol 35, 81 (2018). https://doi.org/10.1007/s12032-018-1143-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-018-1143-2