Abstract
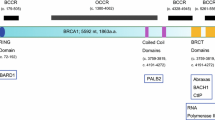
BRCA2is the main susceptibility gene known to be involved in the pathogenesis of breast cancer. It plays an important role in maintaining the genome stability by homologous recombination through DNA double-strand breaks repairing, by interacting with various other proteins including RAD51, DSS1, RPA, MRE11, PALB2, and p53. BRCA2-deficient cells show the abnormalities of chromosome number. BRCA2 is also found to be involved in centrosome duplication specifically in the metaphase to anaphase transition. Inactivation or depletion of BRCA2 leads to centrosome amplification that results in unequal separation of chromosomes. BRCA2 localizes with central spindle and midbody during telophase and cytokinesis. Inactivation or depletion of BRCA2 leads to multinucleation of cell. Around 2000 mutations have been reported in BRCA2 gene. BRCA2-deficient tumors are being taking into consideration for targeted cancer therapy by using different inhibitors like poly ADP-ribose polymerase and thymidylate synthase. The present review focusses on the role of BRCA2 in various critical cellular processes based on the mechanistic approaches. Mutations reported in the BRCA2 gene in various ethnic groups till date have also been compiled with an insight into the functional aspects of these alterations. The therapeutic strategies for targeting BRCA2-deficient tumors have also been targeted.



Similar content being viewed by others
Abbreviations
- BRCA1:
-
Breast cancer type 1
- BRCA2:
-
Breast cancer type 2
- RPA:
-
Replication protein A
- MRE11:
-
Meiotic recombination 11
- PALB2:
-
Partner and localizer of BRCA2
- SSB:
-
Single-strand binding protein
- HR:
-
Homologous recombination
- DDR:
-
DNA damage response
- ATR:
-
Ataxia telangiectasia and Rsd3-related
- ATM:
-
Ataxia telangiectasia mutated
- EMSY:
-
BRCA2 interacting transcriptional repressor
- AURKA:
-
Serine/threonine kinase Aurora-A
- UCV:
-
Unknown classified variants
- LOH:
-
Loss of heterozygosity
- NLS:
-
Nuclear localization signal
- DBD:
-
Double-strand break domain
- TS:
-
Thymidylate synthase
- PARP:
-
Poly ADP-ribose polymerase
- FU:
-
Fluoropyrimidines
References
Lo PK, Wolfson B, Zhou X, Duru N, Gernapudi R, Zhou Q. Noncoding RNAs in breast cancer. Brief Funct Genom. 2016;15(3):200–21.
Wasson MK, Chauhan PS, Singh LC, Katara D, Sharma JD, Zomawia E, Kataki A, Kapur S, Saxena S. Association of DNA repair and cell cycle gene variations with breast cancer risk in Northeast Indian population: a multiple interaction analysis. Tumor Biol. 2014;35(6):5885–94.
Shiovitz S, Korde LA. Genetics of breast cancer: a topic in evolution. Ann Oncol. 2015;26(7):1291–9.
Huang Z, Wen W, Zheng Y, Gao YT, Wu C, Bao P, Zhang M. Breast cancer incidence and mortality: trends over 40 years among women in Shanghai, China. Ann Oncol. 2016;27(6):1129–34.
Spugnesi L, Balia C, Collavoli A, Falaschi E, Quercioli V, Caligo MA, Galli A. Effect of the expression of BRCA2 on spontaneous homologous recombination and DNA damage-induced nuclear foci in Saccharomyces cerevisiae. Mutagenenesis. 2013;28:187–95.
Venkitaraman AR. Chromosome stability, DNA recombination and the BRCA2 tumour suppressor. Curr Opin Cell Biol. 2001;13(3):338–43.
Gochhait S, Bukhari SIA, Bairwa N, Vadhera S, Darvishi K, Raish M, Bamezai RN. Implication of BRCA2-26G>A 5′ untranslated region polymorphism in susceptibility to sporadic breast cancer and its modulation by p53 codon 72 Arg> Pro polymorphism. Breast Cancer Res. 2007;9(5):1.
Rudkin TM, Foulkes WD. BRCA2: breaks, mistakes and failed separations. Trends Mol Med. 2005;11(4):145–8.
Schlacher K, Christ N, Siaud N, Egashira A, Wu H, Jasin M. Double-strand break repair-independent role for BRCA2 in blocking stalled replication fork degradation by MRE11. Cell. 2011;145(4):529–42.
Jonsdottir AB, Vreeswijk MP, Wolterbeek R, Devilee P, Tanke HJ, Eyfjörd JE, Szuhai K. BRCA2 heterozygosity delays cytokinesis in primary human fibroblasts. Anal Cell Pathol. 2009;31(3):191–201.
Zhao W, Vaithiyalingam S, San Filippo J, Maranon DG, Jimenez-Sainz J, Fontenay GV, Chazin WJ. Promotion of BRCA2-dependent homologous recombination by DSS1 via RPA targeting and DNA mimicry. Mol Cell. 2015;59(2):176–87.
Zhang F, Shi J, Bian C, Yu X. Poly (ADP-Ribose) mediates the BRCA2-dependent early DNA damage response. Cell Rep. 2015;13(4):678–89.
Zhang F, Ma J, Wu J, Ye L, Cai H, Xia B, Yu X. PALB2 links BRCA1 and BRCA2 in the DNA-damage response. Curr Biol. 2009;19(6):524–9.
Buisson R, Niraj J, Pauty J, Maity R, Zhao W, Coulombe Y, Masson JY. Breast cancer proteins PALB2 and BRCA2 stimulate polymerase η in recombination-associated DNA synthesis at blocked replication forks. Cell Rep. 2014;6(3):553–64.
Krejčí L, Altmannová V, Špírek M, Zhao X, Krejci L, Altmannova V, Spirek M, Zhao X. Homologous recombination and its regulation. Nucleic Acids Res. 2012;40(13):5795–818.
Verma S, Rao BJ. p53 suppresses BRCA2-stimulated ATPase and strand exchange functions of human RAD51. J Biochem. 2013;154(3):237–48.
O’Connor MJ. Targeting the DNA damage response in cancer. Mol Cell. 2015;60(4):547–60.
Song L, Dai T, Xie Y, Wang C, Lin C, Wu Z, Li J. Up-regulation of miR-1245 by c-myc targets BRCA2 and impairs DNA repair. J Mol Cell Biol. 2012;4(2):108–17.
Haber DA. The BRCA2-EMSY connection: implications for breast and ovarian tumorigenesis. Cell. 2003;115(5):507–8.
Xia B, Sheng Q, Nakanishi K, Ohashi A, Wu J, Christ N, Livingston DM. Control of BRCA2 cellular and clinical functions by a nuclear partner, PALB2. Mol Cell. 2006;22(6):719–29.
Lekomtsev S, Guizetti J, Pozniakovsky A, Gerlich DW, Petronczki M. Evidence that the tumor-suppressor protein BRCA2 does not regulate cytokinesis in human cells. J Cell Sci. 2010;123(9):1395–400.
Mondal G, Rowley M, Guidugli L, Wu J, Pankratz VS, Couch FJ. BRCA2 localization to the midbody by filamin A regulates cep55 signaling and completion of cytokinesis. Dev Cell. 2012;23(1):137–52.
Daniels MJ, Wang Y, Lee M, Venkitaraman AR. Abnormal cytokinesis in cells deficient in the breast cancer susceptibility protein BRCA2. Science. 2004;306(5697):876–9.
Gudmundsdottir K, Ashworth A. BRCA2 in meiosis: turning over a new leaf. Trends Cell Biol. 2004;14(8):401–4.
Lee M, Daniels MJ, Garnett MJ, Venkitaraman AR. A mitotic function for the high-mobility group protein HMG20b regulated by its interaction with the BRC repeats of the BRCA2 tumor suppressor. Oncogene. 2011;30(30):3360–9.
Yang G, Mercado-Uribe I, Multani AS, Sen S, Shih IE, Wong KK, Liu J. Erratum: RAS promotes tumorigenesis through genomic instability induced by imbalanced expression of aurora-A and BRCA2. Cancer. 2013;133(2):275–85.
Ryser S, Dizin E, Jefford CE, Delaval B, Gagos S, Christodoulidou A, Irminger-Finger I. Distinct roles of BARD1 isoforms in mitosis: full-length BARD1 mediates Aurora B degradation, cancer-associated BARD1β scaffolds Aurora B and BRCA2. Can Res. 2009;69(3):1125–34.
Thorslund T, Esashi F, West SC. Interactions between human BRCA2 protein and the meiosis-specific recombinase DMC1. EMBO J. 2007;26(12):2915–22.
Park I, Lee HO, Choi E, Lee YK, Kwon MS, Min J, Lee H. Loss of BubR1 acetylation causes defects in spindle assembly checkpoint signaling and promotes tumor formation. J Cell Biol. 2013;202(2):295–309.
Welcsh PL, King MC. BRCA1 and BRCA2 and the genetics of breast and ovarian cancer. Hum Mol Genet. 2001;10(7):705–13.
Johansson I, Killander F, Linderholm B, Hedenfalk I. Molecular profiling of male breast cancer–lost in translation? Int J Biochem Cell Biol. 2014;53:526–35.
Zahra AZ, Kadkhoda S, Behjati F, Moghaddam FA, Badiei A, Sirati F, Keyhani E. Mutation screening of BRCA genes in 10 Iranian males with breast cancer. Int J Mol Cell Med. 2016;5(2):114.
Ruddy KJ, Winer EP. Male breast cancer: risk factors, biology, diagnosis, treatment, and survivorship. Ann Oncol. 2013;24(6):1434–43.
Liede A, Karlan BY, Narod SA. Cancer risks for male carriers of germline mutations in BRCA1 or BRCA2: a review of the literature. J Clin Oncol. 2004;22(4):735–42.
Hilton JL, Geisler JP, Rathe JA, Hattermann-Zogg MA, DeYoung B, Buller RE. Inactivation of BRCA1 and BRCA2 in ovarian cancer. J Natl Cancer Inst. 2002;94(18):1396–406.
Lancaster JM, Wooster R, Mangion J, Phelan CM, Cochran C, Gumbs C, Patel S. BRCA2 mutations in primary breast and ovarian cancers. Nat Genet. 1996;13(2):238–40.
Edwards SM, Kote-Jarai Z, Meitz J, Hamoudi R, Hope Q, Osin P, Dearnaley DP. Two percent of men with early-onset prostate cancer harbor germline mutations in the BRCA2 gene. Am J Hum Genet. 2003;72(1):1–12.
Goggins M, Schutte M, Lu J, Moskaluk CA, Weinstein CL, Petersen GM, Kern SE. Germline BRCA2 gene mutations in patients with apparently sporadic pancreatic carcinomas. Can Res. 1996;56(23):5360–4.
Karami F, Mehdipour PA. Comprehensive focus on global spectrum of BRCA1 and BRCA2 mutations in breast cancer. BioMed Res Int. 2013;201:1–21.
Biswas K, Das R, Eggington JM, Qiao H, North SL, Stauffer S, Pruss D. Functional evaluation of BRCA2 variants mapping to the PALB2 binding and C-terminal DNA binding domains using a mouse ES cell-based assay. Hum Mol Genet. 2012;21:3993–4006.
Antoniou A, Pharoah PDP, Narod S, Risch HA, Eyfjord JE, Hopper JL, Pasini B. Average risks of breast and ovarian cancer associated with BRCA1 or BRCA2 mutations detected in case series unselected for family history: a combined analysis of 22 studies. Am J Hum Genet. 2003;72(5):1117–30.
Ferla R, Calo V, Cascio S, Rinaldi G, Badalamenti G, Carreca I, Russo A. Founder mutations in BRCA1 and BRCA2 genes. Ann Oncol. 2007;18(6):93–8.
Neuhausen SL, Godwin AK, Gershoni-Baruch R, Schubert E, Garber J, Stoppa-Lyonnet D, Osorio A. Haplotype and phenotype analysis of nine recurrent BRCA2 mutations in 111 families: results of an international study. Am J Hum Genet. 1998;62(6):1381–8.
Tonin PN, Mes-Masson AM, Futreal PA, Morgan K, Mahon M, Foulkes WD, Narod SA. Founder BRCA1 and BRCA2 mutations in French Canadian breast and ovarian cancer families. Am J Hum Genet. 1998;63(5):1341–51.
Campos B, Diez O, Domenech M, Baena M, Pericay C, Balmana J, Baiget M. BRCA2 mutation analysis of 87 Spanish breast/ovarian cancer families. Ann Oncol. 2001;12(12):1699–703.
Liede A, Narod SA. Hereditary breast and ovarian cancer in Asia: genetic epidemiology of BRCA1 and BRCA2. Hum Mutat. 2002;20(6):413–24.
Ang P, Lim A, Lee TC, Luo JT, Ong DC, Tan PH, Lee AS. BRCA1 and BRCA2 mutations in an Asian clinic-based population detected using a comprehensive strategy. Cancer Epidemiol Prev Biomark. 2007;16(11):2276–84.
Zhi X, Szabo C, Chopin S, Suter N, Wang QS, Ostrander EA, Shi YR. BRCA1 and BRCA2 sequence variants in Chinese breast cancer families. Hum Mutat. 2002;20(6):474.
Liede A, Malik IA, Aziz Z, De los Rios P, Kwan E, Narod SA. Contribution of BRCA1 and BRCA2 mutations to breast and ovarian cancer in Pakistan. Am J Hum Genet. 2002;71(3):595–606.
Farooq A, Naveed AK, Azeem Z, Ahmad T. Breast and ovarian cancer risk due to prevalence of BRCA1 and BRCA2 variants in Pakistani population: a Pakistani database report. J Oncol. 2011; 2011:1–8.
Seo JH, Cho DY, Ahn SH, Yoon KS, Kang CS, Cho HM, Shin SW. BRCA1 and BRCA2 germline mutations in Korean patients with sporadic breast cancer. Hum Mutat. 2004;24(4):350.
Pietschmann A, Mehdipour P, Atri M, Hofmann W, Hosseini-Asl SS, Scherneck S, Peters H. Mutation analysis of BRCA1 and BRCA2 genes in Iranian high risk breast cancer families. J Cancer Res Clin Oncol. 2005;131(8):552–8.
Rytelewski M, Ferguson PJ, Vareki SM, Figueredo R, Vincent M, Koropatnick J. Inhibition of BRCA2 and thymidylate synthase creates multidrug sensitive tumor cells via the induction of combined “complementary lethality”. Mol Ther Nucleic Acids. 2013;2(3):e78.
Sessa C. Update on PARP1 inhibitors in ovarian cancer. Ann Oncol. 2011;22(8):viii72–6.
Rouleau M, Patel A, Hendzel MJ, Kaufmann SH, Poirier GG. PARP inhibition: PARP1 and beyond. Nat Rev Cancer. 2010;10(4):293–301.
Bryant HE, Schultz N, Thomas HD, Parker KM, Flower D, Lopez E, Helleday T. Specific killing of BRCA2-deficient tumours with inhibitors of poly (ADP-ribose) polymerase. Nature. 2005;434(7035):913–7.
Venkitaraman AR. Targeting the molecular defect in BRCA-deficient tumors for cancer therapy. Cancer Cell. 2009;16(2):89–90.
Evers B, Drost R, Schut E, de Bruin M, van der Burg E, Derksen PW, Smith GC. Selective inhibition of BRCA2-deficient mammary tumor cell growth by AZD2281 and cisplatin. Clin Cancer Res. 2008;14(12):3916–25.
Zimmer J, Tacconi EM, Folio C, Badie S, Porru M, Klare K, Jackson SP. Targeting BRCA1 and BRCA2 deficiencies with G-quadruplex-interacting compounds. Mol Cell. 2016;61(3):449–60.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Shailani, A., Kaur, R.P. & Munshi, A. A comprehensive analysis of BRCA2 gene: focus on mechanistic aspects of its functions, spectrum of deleterious mutations, and therapeutic strategies targeting BRCA2-deficient tumors. Med Oncol 35, 18 (2018). https://doi.org/10.1007/s12032-018-1085-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12032-018-1085-8