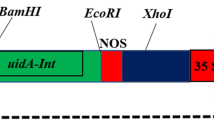
Abstract
A simple and efficient protocol for Agrobacterium-mediated genetic transformation of tomato was developed using combination of non-tissue culture and micropropagation systems. Initially, ESAM region of 1-day-old germinated tomato seeds were microinjected for one to five times with Agrobacterium inoculums (OD600 = 0.2–1.0). The germinated seeds were cocultivated in the MS medium fortified with (0–200 mM) acetosyringone and minimal concentrations of (0–20 mg L−1) kanamycin, and the antibiotic concentration was doubled during the second round of selection. Bacterial concentration of OD600 = 0.6 served as an optimal concentration for infection and the transformation efficiency was significantly higher of about 46.28 %. In another set of experiment, an improved and stable regeneration system was adapted for the explants from the selection medium. Four-day-old double cotyledonary nodal explants were excised from the microinjected seedlings and cultured onto the MS medium supplemented with 1.5 mg L−1 thidiazuron, 1.5 mg L−1 indole-3-butyric acid, 30 mg L−1 kanamycin, and 0–1.5 mg L−1 adenine sulphate. Maximum of 9 out of 13 micropropagated shoots were shown positive to GUS assay. By this technique, the transformation efficiency was increased from 46.28 to 65.90 %. Thus, this paper reports the successful protocol for the mass production of transformants using microinjection and micropropagation techniques.






Similar content being viewed by others
References
Bhatia, P., Ashwath, N., Senaratna, T., & Midmore, D. (2004). Plant Cell Tissue and Organ Culture, 78, 1–21.
Jia, G. X., Zhu, Z. Q., Chang, F. Q., & Li, Y. (2002). Plant Cell Reports, 21, 141–146.
Wang, J., Sun, Y., & Li, Y. (2007). Biotechnololgy and Applied Biochemistry, 46, 51–55.
Van, D. T., Ferro, N., & Jacobsen, H.-J. (2010). GM Crops, 1–5, 312–321.
McCormick, S., Niedermeyer, J., Fry, J., Barnason, A., Horsch, R., & Fraley, R. (1986). Plant Cell Reports, 5, 81–84.
Ellul, P., Garcia-Sogo, B., Pineda, B., Rios, G., Roig, L. A., & Moreno, V. (2003). Theoretical and Applied Genetics, 106, 231–238.
Ling, H. Q., Kriseleit, D., & Ganal, M. W. (1998). Plant Cell Reports, 17, 843–847.
Fillati, J. J., Kiser, J., Ronald, R., & Comai, L. (1987). Biotechnology, 5, 726–730.
Hamza, S., & Chupeau, Y. (1993). Journal of Experimental Botany, 44, 1837–1845.
Pfitzner, A. J. P. (1998). Methods in Molecular Biology, 81, 359–363.
Shivakumar, B., Mythili, J. B., Anand, L., & Saiprasad, G. V. S. (2007). Indian Journal of Horticulture, 64, 251–257.
Gao, N., Shen, W., Cao, Y., Su, Y., & Shi, W. (2009). Plant Cell Tissue and Organ Culture, 98, 321–330.
Ohki, S., Bigot, C., & Mousseau, J. (1978). Plant & Cell Physiology, 19, 27–42.
Chyi, Y. S., & Phillips, G. C. (1987). Plant Cell Reports, 6, 105–108.
Bird, C. R., Smith, C. J. S., Ray, J. A., Moureau, P., Bevan, M. J., Birds, A. S., Hughes, S., Morris, P. C., Grierson, D., & Schuch, W. (1988). Plant Molecular Biology, 11, 651–662.
Davis, M. R., & Miller, L. D. (1991). Journal of Experimental Botany, 42, 359–364.
Vidya, C. S. S., Manoharan, M., Kumar, C. T. R., Savithri, H. S., & Sita, G. L. (2000). Journal of Plant Physiology, 156, 106–110.
Roekel, J. S. C., Damm, B., Melchers, L. S., & Hoekema, A. (1993). Plant Cell Reports, 12, 644–647.
Frary, A., & Earle, E. D. (1996). Plant Cell Reports, 6, 235–240.
Park, S. H., Morris, J. L., Park, J. E., Hirschi, K. D., & Smith, R. H. (2003). Journal of Plant Physiology, 160, 1253–1257.
Raj, S. K., Singh, R., Pandey, S. K., & Singh, B. P. (2005). Current Science, 88, 1674–1679.
Qiu, D., Diretoo, G., Tavarza, R., & Giuliano, G. (2007). Scientia Horticulturae, 112, 172–175.
Hu, W., & Phillips, G. C. (2001). In Vitro Cellular & Developmental Biology-Plant, 37, 12–18.
Sun, H. J., Uchii, S., Watanabe, S., & Ezira, H. (2006). Plant & Cell Physiology, 47, 426–431.
Velcheva, M., Faltin, Z., Flaishman, M., Eshdat, Y., & Perl, A. (2005). Plant Science, 168, 121–130.
Feldmann, K. A. (1991). The Plant Journal, 1, 71–82.
Grob-Hardt, R., & Laux, T. (2003). Journal of Cell Science, 116, 1659–1666.
Sujatha, M., & Reddy, T. P. (1998). Plant Cell Reports, 17, 561–566.
Pavingerova, D., & Ond ej, M. (1995). Biologia Plantarum, 37, 467–471.
Baskaran, P., & Dasgupta, I. (2012). Journal of Plant Biochemistry and Biotechnology, 21(2), 268–274.
Murashige, T., & Skoog, F. (1962). Physiologia Plantarum, 15, 473–497.
Vinoth, S., Gurusaravanan, P., & Jayabalan, N. (2012). Journal of Applied Phycology, 24(5), 1329–1337.
Jefferson, R. A., Burgees, S. M., & Hirsh, D. (1986). Proceedings of the National Academy of Sciences, 83, 8447–8451.
Sailaja, M., Tarakeswari, M., & Sujatha, M. (2008). Plant Cell Reports, 27, 1509–1519.
Carolina, C., & Francisco, A. C. M. (2004). Plant Cell Tissue and Organ culture, 76, 269–275.
Andreoli, C., & Khan, A. (1999). Pesquisa Agropecuária Brasileira, 34, 1953–1958.
Stachel, S. E., Nester, E. W., & Zambryski, P. C. (1986). Proceedings of the National Academy of Sciences, 83, 379–383.
Sujatha, M., & Sailaja, M. (2005). Plant Cell Reports, 23, 803–810.
Lin, J. J., Assad-Garcia, N., & Kuo, J. (1995). Plant Science, 109, 171–177.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Vinoth, S., Gurusaravanan, P. & Jayabalan, N. Optimization of Factors Influencing Microinjection Method for Agrobacterium tumefaciens-Mediated Transformation of Tomato. Appl Biochem Biotechnol 169, 1173–1187 (2013). https://doi.org/10.1007/s12010-012-0006-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12010-012-0006-0