Abstract
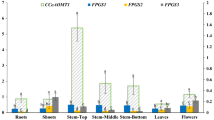
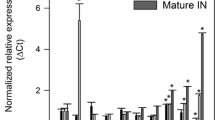
Rice straw is one of the largest biomasses in the world that can potentially be exploited for biofuel. Nevertheless, the association of lignin with cellulose and hemicellulose has hindered the efficient utilization of rice straw for cellulosic biofuel. The objective of this study was to down-regulate key genes involved in lignin biosynthesis pathway, such as hydroxycinnamoyl CoA: shikimate hydroxycinnamoyl transferase (HCT), cinnamoyl CoA reductase (CCR), and cinnamyl alcohol dehydrogenase (CAD), through “terminator-less” constructs to reduce lignin in transgenic rice. Real-time qPCR analyses of the selected T1 transgenic rice plants indicated 36–86% transcript reduction in HCT lines, 75–94% in CCR lines, and 10–85% in CAD lines. Of the nine down-regulated lines (three lines from each genes) subjected to lignin analysis, seven showed significant reduction in total lignin content (HCT-4, HCT-7, CAD-1, CAD-7, CCR-3, CCR-7, and CCR-12) with lignin reduction ranging from 4.6 to 10.8%. The results from this study indicated that truncated gene fragments lacking transcription termination sequence can be used for down-regulation of lignin genes in rice, and the rice straw from these transgenic lines could be useful as feedstock for cellulosic biofuel.



Similar content being viewed by others
References
Abbott JC, Barakate A, Pincon G, Legrand M, Lapierre C, Mila I, Schuch W, Halpin C (2002) Simultaneous suppression of multiple genes by single transgenes. Down-regulation of three unrelated lignin biosynthetic genes in tobacco. Plant Physiol 128:844–853
Akbudak MA, Nicholson SJ, Srivastava V (2013) Suppression of Arabidopsis genes by terminator-less transgene constructs. Plant Biotechnol Rep 7:415–424
Bai Y, Gong W, Liu T, and Zhu Y (2003) Cloning and expression analyses of a cinnamoyl CoA resductase cDNA from rice seeelings. Chin Sci Bull 48:2221–2225
Barakat A, Yassin NB, Park JS, Choi A, Herr J, Carlson JE (2011) Comparative and phylogenomic analyses of cinnamoyl-CoA reductase and cinnamoyl-CoA-reductase-like gene family in land plants. Plant Sci 181:249–257
Baxter HL, Mazarei M, Labbe N, Kline LM, Cheng Q, Windham MT, Mann DGJ, Fu C, Ziebell A, Sykes RW, Rodriguez M Jr, Davis MF, Mielenz JR, Dixon RA, Wang Z-Y, Stewart CN Jr (2014) Two-year field analysis of reduced recalcitrance transgenic switchgrass. Plant Biotechnol J 12:914–924
Besseau S, Hoffmann L, Geoffroy P, Lapierre C, Pollet B, Legrand M (2007) Flavonoid accumulation in Arabidopsis repressed in lignin synthesis affects auxin transport and plant growth. Plant Cell 9:148–162
Binod P, Sindhu R, Singhania R, Vikram S, Devi L, Nagalakashmi S, Kurien N, Sukumaran R, Pandey A (2010) Bioethanol production from rice straw: an overview. Bioresour Technol 101:4767–4774
Blee K, Choi JW, O’Connell AP, Jupe SC, Schuch W, Lewis NG, Bolwell GP (2001) Antisense and sense expression of cDNA coding for CYP73A15, a class II cinnamate 4-hydroxylase, leads to a delayed and reduced production of lignin in tobacco. Phytochemistry 57:1159–1166
Bouvier d’Yvoire M, Bouchabke-Coussa O, Voorend W, Antelme S, Cezard L, Leg_ee F, Lebris P, Legay S, Whitehead C, McQueen-Mason SJ, Gomez LD, Jouanin L, Lapierre C, Sibout R (2013) Disrupting the cinnamyl alcohol dehydrogenase 1 gene (BdCAD1) leads to altered lignification and improved saccharification in Brachypodium distachyon. Plant J 73:496–508
Carpita N, McCann M (2000) The cell wall. In: Buchanan BB, Wilhelm G, Jones RL (eds) Biochemistry and molecular biology of plants. American Society of Plant Physiologists, Rockville, pp 52–108
Carroll A, Somerville C (2009) Cellulosic biofuels. Annu Rev Plant Biol 60:165–182
Chabannes M, Barakate A, Lapierre C, Marita JM, Ralph J, Pean M, Danoun S, Halpin C, Grima-Pettenati J, Boudet AM (2001) Strong decrease in lignin content without significant alteration of plant development is induced by simultaneous down-regulation of cinnamoyl CoA reductase (CCR) and cinnamyl alcohol dehydrogenase (CAD) in tobacco plants. Plant J 28:257–270
Chen F, Dixon RA (2007) Lignin modification improves fermentable sugar yields for biofuel production. Nat Biotechnol 25:759–761
Chen L, Auh CK, Dowling P, Bell J, Chen F, Hopkins A, Dixon RA, Wang ZY (2003) Improved forage digestibility of tall fescue (Festuca arundinacea) by transgenic down-regulation of cinnamyl alcohol dehydrogenase. Plant Biotech J 1:437–449
Chen L, Auh C, Dowling P, Bell J, Lehmann D, Wang ZY (2004) Transgenic down regulation of caffeic acid o-methyltransferase (COMT) led to improved digestibility in tall fescue (Festuca arundinacea). Funct Plant Biol 31:235–245
Dauwe R, Morreel K, Goeminne G, Gielen B, Rohde A, Van Beeumen J, Ralph J, Boudet AM, Kopka J, Rochange SF, Halpin C, Messens E, Boerjan W (2007) Molecular phenotyping of lignin-modified tobacco reveals associated changes in cell-wall metabolism, primary metabolism, stress metabolism and photorespiration. Plant J 52:263–285
Dellaporta S, Wood J, Hicks J (1983) Isolation of DNA from higher plants. Plant Mol Biol Rep 1:19–21
Dien B, Sarath G, Pedersen J, Sattler S, Chen H, Funnell-Harris D, Nichols N, Cotta M (2009) Improved sugar conversion and ethanol yield for forage sorghum (Sorghum bicolor L. Moench) lines with reduced lignin contents. Bioenerg Res 2:153–164
Doorsselaere J, Baucher M, Chognot E, Chabbert B, Tollier MT, Petit-Conil M, Leplé JC, Pilate G, Cornu D, Monties B (2003) A novel lignin in poplar trees with a reduced caffeic acid/5 hydroxyferulic acid O-methyltransferase activity. Plant J 8:855–864
Evans RJ, Milne TA (1987) Molecular characterization of the pyrolysis of biomass. Energy Fuels 1:123–137
Fornalé S, Capellades M, Encina A, Wang K, Irar S, Lapierre C, Ruel K, Joseleau JP, Berenguer, J, Puigdomenech P, Rigau J, Caparros-Ruiz D (2012) Altered lignin biosynthesis improves cellulosic bioethanol production in transgenic maize plants down-regulated for cinnamyl alcohol dehydrogenase. Mol Plant 5:817–830
Fornalé S, Rencoret J, Garcia-Calvo L, Capellades M, Encina A, Santiago R, Rigau J, Gutiérrez A, del Río JC, Caparros-Ruiz D (2015) Cell wall modifications triggered by the down-regulation of Coumarate 3-hydroxylase-1 in maize. Plant Sci 236:272–282
Fu C, Mielenz JR, Xiao X, Ge Y, Hamilton CY, Rodriguez M Jr, Chen F, Foston M, Ragauskas A, Bouton J, Dixon RA, Wang ZY (2011a) Genetic manipulation of lignin reduces recalcitrance and improves ethanol production from switchgrass. Proc Natl Acad Sci USA 108:3803–3808
Fu C, Xiao X, Xi Y, Ge Y, Chen F, Bouton J, Dixon RA, Wang ZY (2011b) Downregulation of cinnamyl alcohol dehydrogenase (CAD) leads to improved saccharification efficiency in switchgrass. Bioenerg Res 4:153–164
Gallego-Giraldo L, Jikumaru Y, Kamiya Y, Tang Y, Dixon RA (2011) Selective lignin down-regulation leads to constitutive defense response expression in alfalfa (Medicago sativa L.). New Phytol 190:627–639
Garrote G, Dominguez H, Parajo JC (2002) Autohydrolysis of corncob: study of non-isothermal operation for xylooligosaccharide production. J Food Eng 52:211–218
Giordano A, Liu Z, Panter SN, Dimech AM, Shang Y, Wijesinghe H, Fulgueras K, Ran Y, Mouradov A, Rochfort S, Patron NJ, Spangenberg GC (2014) Reduced lignin content and altered lignin composition in the warm season forage grass Paspalum dilatatum by down-regulation of a Cinnamoyl CoA reductase gene. Transgenic Res 23:503–517
Goldemberg J (2007) Ethanol for a sustainable energy future. Science 315:808–810
Gondolf VM, Stoppel R, Ebert B, Rautengarten C, Liwanag AJM, Loqué D, Scheller HV (2014) A gene stacking approach leads to engineered plants with highly increased galactan levels in Arabidopsis. BMC Plant Biol 14:344
Goujon T, Ferret V, Mila I, Pollet B, Ruel K, Burlat V, Joseleau J, Barriere Y, Lapierre C, Jouanin L (2003) Down-regulation of the AtCCR1 gene in Arabidopsis thaliana: effects on phenotype, lignins and cell wall degradability. Planta 217:218–228
Guo D, Chen F, Inoue K, Blount JW, Dixon RA (2001) Downregulation of caffeic acid 3-O-methyltransferase and caffeoyl CoA 3-O-methyltransferase in transgenic alfalfa. Impacts on lignin structure and implications for the biosynthesis of G and S lignin. Plant Cell 13:73–88
Hirano K, Aya K, Kondo M, Okuno A, Morinaka Y, Matsuoka M (2012) OsCAD2 is the major CAD gene responsible for monolignol biosynthesis in rice culm. Plant Cell Rep 31:91–101
Hoffmann L, Besseau S, Geoffroy P, Ritzenthaler C, Meyer D, Lapierre C, Pollet B, Legrand M (2004) Silencing of hydroxycinnamoyl-coenzyme A shikimate/quinate hydroxycinnamoyltransferase affects phenylpropanoid biosynthesis. Plant Cell 16:1446–1465
Jackson L, Shadle G, Zhou R, Nakashima J, Chen F, Dixon R (2008) Improving saccharification efficiency of alfalfa stems through modification of the terminal stages of monolignol biosynthesis. BioEnergy Res 1:180–192
Jones L, Ennos AR, Turner SR (2001) Cloning and characterization of irregular xylem4 (irx4): a severely lignin-deficient mutant of Arabidopsis. Plant J 26:205–216
Jung HG, Deetz DA (1993) Cell wall lignification and degradability. In: Jung HG, Buxton DR, Hatfield RD, Ralph J (eds) Forage cell wall structure and digestibility). ASA-CSSA-SSSA, Madison, pp. 315–346
Jung JH, Fouad WM, Vermerris W, Gallo M, Altpeter F (2012) RNAi suppression of lignin biosynthesis in sugarcane reduces recalcitrance for biofuel production from lignocellulosic biomass. Plant Biotechnol J 10:1067–1076
Jung JH, Vermerris W, Gallo M, Fedenko JR, Erickson JE, Altpeter F (2013) RNA interference suppression of lignin biosynthesis increases fermentable sugar yields for biofuel production from field-grown sugarcane. Plant Biotechnol J 11:709–716
Kawasaki T, Koita H, Nakatsubo T, Hasegawa K, Wakabayashi K, Takahashi H, Umemura K, Umezawa T, Shimamoto K (2006) Cinnamoyl-CoA reductase, a key enzyme in lignin biosynthesis, is an effector of small GTPase Rac in defense signaling in rice. Proc Natl Acad Sci USA 103:230–235
Kim IA, Kim B-G, Kim M, Ahn J-H (2012) Charactrization of hydroxycinnamoyltransferase from rice and its application for biological synthesis of hydroxycinnamoyl glycerols. Phytochemistry 76:25–31
Lapierre C, Pollet B, Petit-Conil M, Toval G, Romero J, Pilate G, Leple JC, Boerjan W, Ferret VV, De Nadai V, Jouanin L (1999) Structural alterations of lignins in transgenic poplars with depressed cinnamoyl alcohol dehydrogenase or caffeic acid O-methyltransferase activity have opposite impact on the efficiency of industrial Kraft pulping. Plant Physiol 119:153–163
Lapierre C, Pilate G, Pollet B, Mila I, Leple J-C, Jouanin L, Kim H, Ralph J (2004) Signatures of cinnamyl alcohol dehydrogenase deficiency in poplar lignins. Phytochemistry 65:313–321
Leplé J-C, Dauwe R, Morreel K, Storme V, Lapierre C, Pollet B, Naumann A, Kang K-Y, Kim H, Ruel K, Lefebvre A, Joseleau J-P, Grima-Pettenati J, De Rycke R, Andersson-Gunneras S, Erban A, Fehrle I, Petit-Conil M, Kopka J, Polle A, Messens E, Sundberg B, Mansfield SD, Ralph J, Pilate G, Boerjan W (2007) Downregulation of cinnamoyl-Coenzyme A reductase in poplar: multiple-level phenotyping reveals effects on cell wallpolymer metabolism and structure. Plant Cell 19:3669–3691
Liu CJ, Cai Y, Zhang X, Gou M, Yang H (2014) Tailoring lignin biosynthesis for efficient and sustainable biofuel production. Plant Biotechnol J 12:1154–1162
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25:402–408
Luo Z, Chen Z (2007) Improperly terminated, unpolyadenylated mRNA of sense transgenes is targeted by RDR6-mediated RNA silencing in Arabidopsis. Plant Cell 19:943–958
Maiorella BL (1983) Ethanol. In: Young M (ed) Industrial chemicals, biochemicals and Fuels, V.3, comprehensive biotechnology. Pergamon Press, UK, pp 861–914
Mir Derikvand M, Sierra JB, Ruel K, Pollet B, Do CT, Thevenin J, Buffard D, Jouanin L, Lapierre C (2008) Redirection of the phenylpropanoid pathway to feruloyl malate in Arabidopsis mutants deficient for cinnamoyl-CoA reductase 1. Planta 227:943–956
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497
Nakashima J, Chen F, Jackson, L, Shadle G, Dixon RA (2008) Multisite genetic modification of monolignol biosynthesis in alfalfa (Medicago sativa): effects on lignin composition in specific cell types. New Phytol 179:738–750
Nicholson SJ, Srivastava V (2009) Transgene constructs lacking transcription termination signal induce efficient silencing of endogenous targets in Arabidopsis. Mol Genet Genom 282:319–328
Piquemal J, Lapierre C, Myton K, O’Connell A, Schuch W, Grima-Pettenati J, Boudet AM (1998) Down-regulation of cinnamoyl-CoA reductase induces significant changes of lignin profiles in transgenic tobacco plants. Plant J 13:71–83
Poovaiah CR, Nageswara-Rao M, Soneji JR, Baxter HL, Stewart CN Jr (2014) Altered lignin biosynthesis using biotechnology to improve lignocellulosic biofuel feedstocks. Plant Biotechnol J 12:1163–1173
Prashant S, Srilakshmi Sunita M, Pramod S, Ranadheer K, Gupta K, Anil Kumar S, Rao Karumanchi S, Rawal SK, Kavi Kishor PS (2011) Downregulation of Leucaena leucocephala cinnamoyl CoA reductase (LiCCR) gene induces significant changes in phenotype, soluble phenolic pools and lignin in transgenic tobacco. Plant Cell Rep 30:2215–2231
Ralph J, Hatfield RD, Piquemal J, Yahiaoui N, Pean M (1998) NMR characterization of altered lignins extracted from tobacco plants downregulated for lignification enzymes cinnamylalcohol dehydrogenase and cinnamoyl-CoA reductase. Proc Natl Acad Sci USA 95:12803–12808
Ralph J, Akiyama T, Kim H, Lu FC, Schatz PF, Marita JM, Ralph SA, Reddy MSS, Chen FDixon RA (2006) Effects of coumarate 3-hydroxylase down-regulation on lignin structure. J Biol Chem 281:8843–8853
Ralph J, Akiyama T, Coleman HD, Mansfield SD (2012) Effects on lignin structure of coumarate 3-hydroxylase downregulation in poplar. Bioenergy Res 5:1009–1019
Reddy MS, Chen F, Shadle G, Jackson L, Aljoe H, Dixon RA (2005) Targeted down-regulation of cytochrome P450 enzymes for forage quality improvement in alfalfa (Medicago sativa L.). Proc Natl Acad Sci USA 102:16573–16578
Ruel K, Berrio-Sierra J, Mir Derikvand M, Pollet B, Thévenin J, Lapierre C, Jouanin L, Joseleau J-P (2009) Impact of CCR1 silencing on the assembly of lignified secondary walls in Arabidopsis thaliana. New Phytol 184:99–113
Saathoff AJ, Sarath G, Chow EK, Dien BS, Tobias CM (2011) Downregulation of cinnamyl-alcohol dehydrogenase in switchgrass by RNA silencing results in enhanced glucose release after cellulase treatment. PLoS One 6:e16416
Saha BC (2003) Hemicellulose bioconversion. J Ind Microbiol Biotechnol 30:279–291
SAS Institute (2008) Inc. SAS Software version 9.2. SAS Institute, Cary, NC
Sewalt V, Ni W, Blount JW, Jung HG, Masoud SA, Howles PA, Lamb C, Dixon RA (1997) Reduced lignin content and altered lignin composition in transgenic tobacco down-regulated in expression of l-phenylalanine ammonia-lyase or Cinnamate 4-hydroxylase. Plant Physiol 115:41–50
Shadle G, Chen F, Srinivasa Reddy MS, Jackson L, Nakashima J, Dixon RA (2007) Down-regulation of hydroxycinnamoyl CoA: shikimate hydroxycinnamoyl transferase in transgenic alfalfa affects lignification, development and forage quality. Phytochemistry 68:1521–1529
Simmons BA, Loque D, Ralph J (2010) Advances in modifying lignin for enhanced biofuel production. Curr Opin Plant Biol 13:313–320
Singleton VL, Rossi JA (1965) Colorimetry of total phenolics with phosphomolybdic-phosphotungstic acid reagents. Am J Enol Vitic 16:144–158
Studer M, DeMartini J, Davis M, Sykes R, Davison B, Keller M, Tuskan G, Wyman C (2011) Lignin content in natural Populus variants affects sugar release. Proc Natl Acad Sci USA 108:6300–6305
Sykes R, Yung M, Novaes E, Kirst M, Peter G, Davis M (2009) High-throughput screening of plant cell-wall composition using pyrolysis molecular beam mass spectroscopy. Biofuels: Methods Protoc Methods Mol Bio 581:169–183
Tamasloukht B, Wong Quai Lam MS, Martinez Y, Tozo K, Barbier O, Jourda C, Jauneau A, Borderies G, Balzergue S, Renou JP, Huguet S, Martinant JP, Tatout C, Lapierre C, Barriere Y, Goffner D, Pichon M (2011) Characterization of a cinnamoyl-CoA reductase 1 (CCR1) mutant in maize: effects on lignification, fibre development, and global gene expression. J Exp Bot 62:3837–3848
Toki S, Hara N, Ono K, Onodera H, Tagiri A, Oka S, Tanaka H (2006) Early infection of scutellum tissue with Agrobacterium allows high-speed transformation of rice. Plant J 47:969–976
Tsai CJ, Popko JL, Mielke MR, Hu WJ, Podila GK, Chiang VL (1998) Suppression of O-methyltransferase gene by homologous sense transgene in quaking aspen causes red-brown wood phenotypes. Plant Physiol 117:101–112
Tu Y, Rochfort S, Liu Z, Ran Y, Griffith M, Badenhorst P, Louie GV, Bowman ME, Smith KF, Noel JP, Mouradov A, Spangenberg G (2010) Functional analyses of caffeic acid O-methyltransferase and cinnamoyl-CoA-reductase genes from perennial ryegrass (Lolium perenne). Plant Cell 22:3357–3373
Van Acker R, Leplé J-C, Aerts D, Storme V, Goeminne G, Ivens B, Légée F, Lapierre C, Piens K, Van Montagu MCE, Santoro N, Foster CE, Ralph J, Soetaert W, Pilate G, Boerjan W (2014) Improved saccharification and ethanol yield from field-grown transgenic poplar deficient in cinnamoyl-CoA reductase. Proc Natl Acad Sci USA 111:845–850
Van der Rest B, Danoun S, Boudet AM, Rochange SF (2006) Downregulation of cinnamoyl-CoA reductase in tomato (Solanum lycopersicum L.) induces dramatic changes in soluble phenolic pools. J Exp Bot 57:1399–1411
Vanholme R, Demedts B, Morreel K, Ralph J, Boerjan W (2010) Lignin biosynthesis and structure. Plant Physiol 153:895–905
Voelker SL, Lachenbruch B, Meinzer FC, Jourdes M, Ki C, Patten AM, Davin LB, Lewis NG, Tuskan GA, Gunter L, Decker SR, Selig MJ, Sykes R, Himmel ME, Kitin P, Shevchenko O, Strauss SH (2010) Antisense down-regulation of 4CL expression alters lignification, tree growth, and saccharification potential of field-grown poplar. Plant Physiol 154:874–886
Wadenbäck J, von Arnold S, Egertsdotter U, Walter MH, Grima-Pettenati J, Goffner D, Gellerstedt G, Gullion T, Clapham D (2008) Lignin biosynthesis in transgenic Norway spruce plants harboring an antisense construct for cinnamoyl CoA reductase (CCR). Transgenic Res 17:379–392
Wagner A, Ralph J, Akiyama T, Flint H, Phillips L, Torr K, Nanayakkara B, Te Kiri L (2007) Exploring lignification in conifers by silencing hydroxycinnamoyl-CoA: shikimate hydroxycinnamoyltransferase in Pinus radiata. Proc Natl Acad Sci USA 104:11856–11861
Wagner A, Tobimatsu Y, Goeminne G, Phillips L, Flint H, Steward D, Torr K, Donaldson L, Boerjan W, Ralph J (2013) Suppression of CCR impacts metabolite profile and cell wall composition in Pinus radiate tracheary elements. Plant Mol Biol 81:105–117
Waterhouse PM, Graham HW, Wang MB (1998) Virus resistance and gene silencing in plants can be induced by simultaneous expression of sense and antisense RNA. Proc Natl Acad Sci USA 95:13959–13964
Wesley SV, Helliwell CA, Smith NA, Wang MB, Rouse DT, Liu Q, Gooding PS, Singh SP, Abbott D, Stoutjesdijk PA, Robinson SP, Gleave AP, Green AG, Waterhouse PM (2001) Construct design for efficient, effective and high throughput gene silencing in plants. Plant J 27:581–590
Xu B, Escamilla-Trevino LL, Sathitsuksanoh N, Shen Z, Shen H, Zhang YHP, Dixon RA, Zhao B (2011) Silencing of 4-coumarate:coenzyme A ligase in switchgrass leads to reduced lignin content and improved fermentable sugar yields for biofuel production. New Phytol 192:611–625
Xu N, Zhang W, Ren S, Liu F, Zhao C, Liao H, Xu Z, Huang J, Li Q, Tu Y, Bin Yu, Wang Y, Jiang J, Qin J, Peng L (2012) Hemicelluloses negatively affect lignocellulose crystallinity for high biomass digestibility under NaOH and H2SO4 pretreatments in Miscanthus. Biotech Biofuel 5:58
Yang B, Wyman CE (2004) Effect of xylan and lignin removal by batch and flowthrough pretreatment on the enzymatic digestibility of corn stover cellulose. Biotechnol Bioeng 86:88–95
Zamora R, Crispin JAS (1995) Production of an acid extract of rice straw. Acta Cient Venez 46:135–139
Zhang K, Qian Q, Huang Z, Wang Y, Li M, Hong L, Zeng D, Gu M, Chu C, Cheng Z (2006) GOLD HULL AND INTERNODE2 encodes a primarily multifunctional cinnamyl-alcohol dehydrogenase in rice. Plant Physiol 140:972–83
Zhong R, Lee C, McCarthy RL, Reeves CK, Jones EG, Ye ZH (2011) Transcriptional activation of secondary wall biosynthesis by rice maize NAC and MYB transcription factors. Plant Cell Physiol 52:1856–1871
Ziebell A, Gracom K, Katahira R, Chen F, Pu YQ, Ragauskas A, Dixon RA, Davis M (2010) Increase in 4-coumaryl alcohol units during lignification in alfalfa (Medicago sativa) alters the extractability and molecular weight of lignin. J Biol Chem 285:38961–38968
Acknowledgements
This work was supported by USDA-NIFA Capacity Building Grant # 2010-38821-21540. Authors thank Dr. Parastoo Azadi, Complex Carbohydrate Research Center, University of Georgia, Athens, GA, USA, for lignin analysis. The cDNA clones used in this study were obtained from Rice Genome Resource Center, National Institute of Agrobiological Sciences, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ponniah, S.K., Shang, Z., Akbudak, M.A. et al. Down-regulation of hydroxycinnamoyl CoA: shikimate hydroxycinnamoyl transferase, cinnamoyl CoA reductase, and cinnamyl alcohol dehydrogenase leads to lignin reduction in rice (Oryza sativa L. ssp. japonica cv. Nipponbare). Plant Biotechnol Rep 11, 17–27 (2017). https://doi.org/10.1007/s11816-017-0426-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11816-017-0426-y