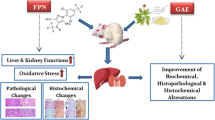
Abstract
Zearalenone (ZEN) is a mycotoxin that contaminates crops worldwide and whose toxic adverse effects are well documented. This study aims to evaluate the protective effect of gallic acid (GA) against biochemical, oxidative, inflammatory, and pathological changes in ZEN treated rats’ hepatorenal system. Wistar rats (n = 50; 150 ± 30 g) were randomly grouped into five cohorts (= 10) specifically: Control (rat chow); ZEN alone (100 µg/kg; per os), GA alone (40 mg/kg; per os), ZEN + GA1 (100 µg/kg + 20 mg/kg per os) and ZEN + GA2 (100 µg/kg + 40 mg/kg per os) and the study was for 28 successive days. Upon terminal sacrifice, biomarkers of hepatorenal function and oxidative stress were analyzed. An assessment of cytokine levels (IL-1β, IL-10) and histopathology of the liver and kidneys was also performed. Relative to the control, serum levels of urea, creatinine, and hepatic transaminases increased significantly (p < 0.05) in the ZEN alone group and reduced in groups co-treated with GA. ZEN treatment further resulted in decreases in the rat’s antioxidant status. The increase in the reactive oxygen and nitrogen species (RONS) and lipid peroxidation (LPO) levels caused by ZEN exposure was reduced by GA in a dose-dependent manner (p < 0.05). Furthermore, ZEN-mediated increase in nitric oxide (NO), xanthine oxidase (XO), IL-1β, and myeloperoxidase (MPO) levels and suppression of IL-10 levels were reversed in the liver and kidney of GA co-treated rats. The extent of ZEN-mediated hepatorenal lesions was reduced in rats co-treated with GA. Our findings suggest that GA effectively abated biochemical, oxido-inflammatory and histological alteration caused by ZEN exposure, limiting ZEN toxicity and cellular damage in rats’ hepatic and renal tissues.









Similar content being viewed by others
References
Aiko V, Mehta A (2015) Occurrence, detection and detoxification of mycotoxins. J Biosci 40:943–954. https://doi.org/10.1007/s12038-015-9569-6
Appell M, Wang LC, Bosma WB (2017) Analysis of the photophysical properties of zearalenone using density functional theory. J Lumines 188:551–557
Badhani B, Sharma N, Kakkar R (2015) Gallic acid: a versatile antioxidant with promising therapeutic and industrial applications. RSC Adv 5:27540–27557. https://doi.org/10.1039/c5ra01911g
Bai X, Sun C, Xu J, Liu D, Han Y, Wu S, Luo X (2018) Detoxification of zearalenone from corn oil by adsorption of functionalized GO systems. Appl Surf Sci 430:198–207. https://doi.org/10.1016/j.apsusc.2017.06.055
Bancroft JD, Gamble M (2008) Theory and practise of histological techniques, 6th edn. Churchill Livingstone Elsevier, Philadelphia
Banerjee R, Mukherjee G, Patra KC (2005) Microbial transformation of tannin-rich substrate to gallic acid through co-culture method. Bioresour Technol 96:949–953. https://doi.org/10.1016/j.biortech.2004.08.004
Banerjee N, Kim H, Krenek K, Talcott ST, Mertens-Talcott SU (2015) Mango polyphenolics suppressed tumor growth in breast cancer xenografts in mice: role of the PI3K/AKT pathway and associated microRNAs. Nutr Res 35:744–751. https://doi.org/10.1016/j.nutres.2015.06.002
Battelli MG, Musiani S, Valgimigli M, Gramantieri L, Tomassoni F, Bolondi L, Stirpe F (2001) Serum xanthine oxidase in human liver disease. Am J Gastroenterol 96:1194–1199. https://doi.org/10.1111/j.1572-0241.2001.03700.x
Ben Salah-Abbes J, Abbes S, Abdel-Wahhab MA, Oueslati R (2009) Raphanus sativus extract protects against Zearalenone induced reproductive toxicity, oxidative stress and mutagenic alterations in male Balb/c mice. Toxicon 53:525–533. https://doi.org/10.1016/j.toxicon.2009.01.013
Ben Salem I, Boussabbeh M, Helali S, Abid-Essefi S, Bacha H (2015) Protective effect of Crocin against zearalenone-induced oxidative stress in liver and kidney of Balb/c mice . Environ Sci Pollut Res Int 22:19069–19076. https://doi.org/10.1007/s11356-015-5086-2
Ben Salem I, Boussabbeh M, Prola A, Guilbert A, Bacha H, Lemaire C, Abid-Essefi S (2016) Crocin protects human embryonic kidney cells (HEK293) from alpha- and beta-Zearalenol-induced ER stress and apoptosis . Environ Sci Pollut Res Int 23:15504–15514. https://doi.org/10.1007/s11356-016-6741-y
Bennett JW, Klich M (2003) Mycotoxins. Clin Microbiol Rev 16:497–516. https://doi.org/10.1128/cmr.16.3.497-516.2003
Berthiller F et al (2013) Masked mycotoxins: a review . Mol Nutr Food Res 57:165–186. https://doi.org/10.1002/mnfr.201100764
Bidani A, Churchill PC (1989) Acute renal failure. Dis Mon 35:63–132. https://doi.org/10.1016/0011-5029(89)90017-5
Biehl ML, Prelusky DB, Koritz GD, Hartin KE, Buck WB, Trenholm HL (1993) Biliary excretion and enterohepatic cycling of zearalenone in immature pigs. Toxicol Appl Pharmacol 121:152–159. https://doi.org/10.1006/taap.1993.1140
Binder SB, Schwartz-Zimmermann HE, Varga E, Bichl G, Michlmayr H, Adam G, Berthiller F (2017) Metabolism of zearalenone and its major modified forms in pigs. Toxins (Basel) 9. https://doi.org/10.3390/toxins9020056
Brodehl A, Moller A, Kunte HJ, Koch M, Maul R (2014) Biotransformation of the mycotoxin zearalenone by fungi of the genera Rhizopus and Aspergillus FEMS. Microbiol Lett 359:124–130. https://doi.org/10.1111/1574-6968.12586
Carr AC, McCall MR, Frei B (2000) Oxidation of LDL by myeloperoxidase and reactive nitrogen species: reaction pathways and antioxidant protection Arterioscler. Thromb Vasc Biol 20:1716–1723. https://doi.org/10.1161/01.atv.20.7.1716
Choubey S, Goyal S, Varughese LR, Kumar V, Sharma AK, Beniwal V (2018) Probing gallic acid for its broad spectrum applications. Mini Rev Med Chem 18:1283–1293. https://doi.org/10.2174/1389557518666180330114010
Clairborne A (1995) Catalase activity. Handbook of Methods for Oxygen Radical Research. CRC Press, Boca Raton
Constant J (1997) Alcohol, ischemic heart disease, and the French paradox. Clin Cardiol 20:420–424. https://doi.org/10.1002/clc.4960200504
Cressey P, Thomson B (2006) Risk profile: Mycotoxins in the New Zealand food supply, in The New Zealand Mycotoxin Surveillance Program 06-14 Report Series, R. Vannoort, Editor. 2014, Institute of Environmental Sciences & Research Limited: Christchurch, p 177
Darwish WS, Ikenaka Y, Nakayama SM, Ishizuka M (2014) An overview on mycotoxin contamination of foods in Africa. J Vet Med Sci 76:789–797. https://doi.org/10.1292/jvms.13-0563
El Golli-Bennour E, Timoumi R, Koroit M, Bacha H, Abid-Essefi S (2019) Protective effects of kefir against zearalenone toxicity mediated by oxidative stress in cultured HCT-116cells. Toxicon 157:25–34. https://doi.org/10.1016/j.toxicon.2018.11.296
Ellman GL (1959) Tissue sulfhydryl groups. Arch Biochem Biophys 82:70–77. https://doi.org/10.1016/0003-9861(59)90090-6
Gao X et al (2018) Dietary silymarin supplementation alleviates zearalenone-induced hepatotoxicity and reproductive toxicity in rats. J Nutr 148:1209–1216. https://doi.org/10.1093/jn/nxy114
Garud MS, Kulkarni YA (2018) Gallic acid attenuates type I diabetic nephropathy in rats. Chem Biol Interact 282:69–76. https://doi.org/10.1016/j.cbi.2018.01.010
Granell S et al (2003) Heparin mobilizes xanthine oxidase and induces lung inflammation in acute pancreatitis. Crit Care Med 31:525–530. https://doi.org/10.1097/01.CCM.0000049948.64660.06
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15 N]nitrate in biological fluids. Anal Biochem 126:131–138. https://doi.org/10.1016/0003-2697(82)90118-x
Gutierrez-Murgas YM, Skar G, Ramirez D, Beaver M, Snowden JN (2016) IL-10 plays an important role in the control of inflammation but not in the bacterial burden in S. epidermidis CNS catheter infection. J Neuroinflammation 13:271. https://doi.org/10.1186/s12974-016-0741-1
Gzyl-Malcher B, Rudolphi-Skorska E, Sieprawska A, Filek M (2019) Manganese protects wheat from the mycotoxin zearalenone and its derivatives. Sci Rep 9:14214. https://doi.org/10.1038/s41598-019-50664-5
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139. https://doi.org/10.1016/S0021-9258(19)42083-8
Hejazy M, Koohi MK (2017) Effects of Nano-zinc on Biochemical Parameters in Cadmium-Exposed Rats . Biol Trace Elem Res 180:265–274. https://doi.org/10.1007/s12011-017-1008-0
Jia Z, Liu M, Qu Z, Zhang Y, Yin S, Shan A (2014) Toxic effects of zearalenone on oxidative stress, inflammatory cytokines, biochemical and pathological changes induced by this toxin in the kidney of pregnant rats. Environ Toxicol Pharmacol 37:580–591. https://doi.org/10.1016/j.etap.2014.01.010
Jiang SZ et al (2012) Effect on hepatonephric organs, serum metabolites and oxidative stress in post-weaning piglets fed purified zearalenone-contaminated diets with or without calibrin-Z. J Anim Physiol Anim Nutr (Berl) 96:1147–1156. https://doi.org/10.1111/j.1439-0396.2011.01233.x
Joslyn MA, Glick (1969) Comparative effects of gallotannic acid and related phenolics on the growth of rats. J Nutr 98:119–126. https://doi.org/10.1093/jn/98.1.119
Kaplan MM (1993) Laboratory tests, vol 1. Diseases of the liver, 7th edn. Lippincott, Philadelphia
Katari R et al (2017) Tissue engineering and regenerative medicine solutions for the abdominal organs. In: Thomaz-Soccol V, Pandey A, Resende RR (eds) Current developments in biotechnology and bioengineering. Elsevier, Amsterdam, pp 325–347. https://doi.org/10.1016/B978-0-444-63660-7.00013-9
Koraichi F, Videmann B, Mazallon M, Benahmed M, Prouillac C, Lecoeur S (2012) Zearalenone exposure modulates the expression of ABC transporters and nuclear receptors in pregnant rats and fetal liver. Toxicol Lett 211:246–256. https://doi.org/10.1016/j.toxlet.2012.04.001
Kowalska K, Habrowska-Gorczynska DE, Piastowska-Ciesielska AW (2016) Zearalenone as an endocrine disruptor in humans. Environ Toxicol Pharmacol 48:141–149. https://doi.org/10.1016/j.etap.2016.10.015
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Malekinejad H, Maas-Bakker R, Fink-Gremmels J (2006) Species differences in the hepatic biotransformation of zearalenone. Vet J 172:96–102. https://doi.org/10.1016/j.tvjl.2005.03.004
Mansouri MT, Naghizadeh B, Ghorbanzadeh B, Farbood Y, Sarkaki A, Bavarsad K (2013) Gallic acid prevents memory deficits and oxidative stress induced by intracerebroventricular injection of streptozotocin in rats. Pharmacol Biochem Behav 111:90–96. https://doi.org/10.1016/j.pbb.2013.09.002
Marin DE, Pistol GC, Neagoe IV, Calin L, Taranu I (2013) Effects of zearalenone on oxidative stress and inflammation in weanling piglets. Food Chem Toxicol 58:408–415. https://doi.org/10.1016/j.fct.2013.05.033
Misra HP, Fridovich I (1972) The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247:3170–3175
Monod J, Wyman J, Changeux J-P (1965) On the nature of allosteric transitions: A plausible model. J Mol Biol 12:88–118. https://doi.org/10.1016/s0022-2836(65)80285-6
Monteiro E et al (2017) Gallic acid-loaded gel formulation combats skin oxidative stress: Development, characterization and ex vivo biological assays. Polymers (Basel) 9. https://doi.org/10.3390/polym9090391
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Olsen M, Pettersson H, Sandholm K, Visconti A, Kiessling KH (1987) Metabolism of zearalenone by sow intestinal mucosa in vitro. Food Chem Toxicol 25:681–683. https://doi.org/10.1016/0278-6915(87)90101-3
Ow YY, Stupans I (2003) Gallic acid and gallic acid derivatives: effects on drug metabolizing enzymes. Curr Drug Metab 4:241–248. https://doi.org/10.2174/1389200033489479
Owumi SE, Dim UJ (2019) Manganese suppresses oxidative stress, inflammation and caspase-3 activation in rats exposed to chlorpyrifos. Toxicol Rep 6:202–209. https://doi.org/10.1016/j.toxrep.2019.02.007
Owumi SE, Ajijola IJ, Agbeti OM (2019a) Hepatorenal protective effects of protocatechuic acid in rats administered with anticancer drug methotrexate. Hum Exp Toxicol 38:1254–1265. https://doi.org/10.1177/0960327119871095
Owumi SE, Dim UJ, Najophe ES (2019b) Diethylnitrosamine aggravates cadmium-induced hepatorenal oxidative damage in prepubertal rats. Toxicol Ind Health 35:537–547. https://doi.org/10.1177/0748233719863287
Owumi S, Najophe ES, Farombi EO, Oyelere AK (2020a) Gallic acid protects against Aflatoxin B1 -induced oxidative and inflammatory stress damage in rats kidneys and liver. J Food Biochem 44:e13316. https://doi.org/10.1111/jfbc.13316
Owumi SE, Najophe SE, Farombi OE, Oyelere AK (2020b) Gallic Acid protects against Aflatoxin B1-induced oxidative and inflammatory stress damage in rats kidneys and liver. J Food Biochem. https://doi.org/10.1111/jfbc.13316
Owumi SE, Olayiwola YO, Alao GE, Gbadegesin MA, Odunola OA (2020) Cadmium and nickel co-exposure exacerbates genotoxicity and not oxido-inflammatory stress in liver and kidney of rats: Protective role of omega-3 fatty acid. Environ Toxicol 35:231–241. https://doi.org/10.1002/tox.22860
Perez-Severiano F, Santamaria A, Pedraza-Chaverri J, Medina-Campos ON, Rios C, Segovia J (2004) Increased formation of reactive oxygen species, but no changes in glutathione peroxidase activity, in striata of mice transgenic for the Huntington’s disease mutation. Neurochem Res 29:729–733. https://doi.org/10.1023/b:nere.0000018843.83770.4b
Pfeiffer E, Hildebrand A, Mikula H, Metzler M (2010) Glucuronidation of zearalenone, zeranol and four metabolites in vitro: formation of glucuronides by various microsomes and human UDP-glucuronosyltransferase isoforms. Mol Nutr Food Res 54:1468–1476. https://doi.org/10.1002/mnfr.200900524
Prakash M, Upadhya S, Prabhu R (2004) Protein thiol oxidation and lipid peroxidation in patients with uraemia. Scand J Clin Lab Invest 64:599–604. https://doi.org/10.1080/00365510410002869
Rai A, Das M, Tripathi A (2019) Occurrence and toxicity of a fusarium mycotoxin, zearalenone. Crit Rev Food Sci Nutr:1–20 https://doi.org/10.1080/10408398.2019.1655388
Rai A, Das M, Tripathi A (2020) Occurrence and toxicity of a fusarium mycotoxin zearalenone. Crit Rev Food Sci Nutr 60:2710–2729. https://doi.org/10.1080/10408398.2019.1655388
Ramaiah SK (2007) A toxicologist guide to the diagnostic interpretation of hepatic biochemical parameters. Food Chem Toxicol 45:1551–1557. https://doi.org/10.1016/j.fct.2007.06.007
Roberts AT et al (2007) The safety and efficacy of a dietary herbal supplement and gallic acid for weight loss. J Med Food 10:184–188. https://doi.org/10.1089/jmf.2006.272
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179:588–590. https://doi.org/10.1126/science.179.4073.588
Rubenstein DA, Yin W, Frame MD (2012) Chap. 12 - flow through the kidney. In: Rubenstein DA, Yin W, Frame MD (eds) Biofluid Mechanics. Academic, Boston, pp 325–345. https://doi.org/10.1016/B978-0-12-381383-1.00012-6
Shahrzad S, Aoyagi K, Winter A, Koyama A, Bitsch I (2001) Pharmacokinetics of gallic acid and its relative bioavailability from tea in healthy humans. J Nutr 131:1207–1210. https://doi.org/10.1093/jn/131.4.1207
Sharma V, McNeill JH, Verma S (2006) Diabetic cardiomyopathy: Where are we 40 years later? Can J Cardiol 22:305–308. https://doi.org/10.1016/s0828-282x(06)70914-x
Shin BS et al (2009) Physiologically based pharmacokinetics of zearalenone. J Toxicol Environ Health A 72:1395–1405. https://doi.org/10.1080/15287390903212741
Steen EH, Wang X, Balaji S, Butte MJ, Bollyky PL, Keswani SG (2020) The role of the anti-inflammatory cytokine interleukin-10 in tissue fibrosis . Adv Wound Care (New Rochelle) 9:184–198. https://doi.org/10.1089/wound.2019.1032
Totani N, Tateishi S, Takimoto T, Maeda Y, Sasaki H (2011) Gallic acid glycerol ester promotes weight-loss in rats. J Oleo Sci 60:457–462. https://doi.org/10.5650/jos.60.457
Usha T, Middha SK, Bhattacharya M, Lokesh P, Goyal AK (2014) Rosmarinic acid, a new polyphenol from Baccaurea ramiflora Lour. Leaf: A probable compound for its anti-inflammatory activity . Antioxidants (Basel) 3:830–842. https://doi.org/10.3390/antiox3040830
Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M (2006) Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact 160:1–40. https://doi.org/10.1016/j.cbi.2005.12.009
Videmann B, Mazallon M, Tep J, Lecoeur S (2008) Metabolism and transfer of the mycotoxin zearalenone in human intestinal Caco-2 cells. Food Chem Toxicol 46:3279–3286. https://doi.org/10.1016/j.fct.2008.07.011
Wang N et al (2018a) The protective role of Bacillus velezensis A2 on the biochemical and hepatic toxicity of zearalenone in mice. Toxins (Basel) 10. https://doi.org/10.3390/toxins10110449
Wang X et al (2018) Toxic effects of Zearalenone on intestinal microflora and intestinal mucosal immunity in mice. Food Agric Immunol 29:1002–1011. https://doi.org/10.1080/09540105.2018.1503233
Wang JP, Chi F, Kim IH (2012) Effects of montmorillonite clay on growth performance, nutrient digestibility, vulva size, faecal microflora, and oxidative stress in weaning gilts challenged with zearalenone. Anim Feed Sci Technol 178:158–166. https://doi.org/10.1016/j.anifeedsci.2012.09.004
Wang Y, Zhang J, Wang Y, Wang K, Wei H, Shen L (2018) Isolation and characterization of the Bacillus cereus BC7 strain, which is capable of zearalenone removal and intestinal flora modulation in mice. Toxicon 155:9–20. https://doi.org/10.1016/j.toxicon.2018.09.005
Wang N, Wu W, Pan J, Long M (2019) Detoxification strategies for zearalenone using microorganisms: a review. Microorganisms 7. https://doi.org/10.3390/microorganisms7070208
Wang M, Yang S, Cai J, Yan R, Meng L, Long M, Zhang Y (2020) Proteomic analysis using iTRAQ technology reveals the toxic effects of zearalenone on the leydig cells of rats. Food Chem Toxicol:111405. https://doi.org/10.1016/j.fct.2020.111405
Zhang H, Dong M, Yang Q, Apaliya MT, Li J, Zhang X (2016) Biodegradation of zearalenone by Saccharomyces cerevisiae: Possible involvement of ZEN responsive proteins of the yeast. J Proteomics 143:416–423. https://doi.org/10.1016/j.jprot.2016.04.017
Zhou C, Zhang Y, Yin S, Jia Z, Shan A (2015) Biochemical changes and oxidative stress induced by zearalenone in the liver of pregnant rats. Hum Exp Toxicol 34:65–73. https://doi.org/10.1177/0960327113504972
Zinedine A, Soriano JM, Molto JC, Manes J (2007) Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: an oestrogenic mycotoxin. Food Chem Toxicol 45:1–18. https://doi.org/10.1016/j.fct.2006.07.030
Zourgui L, Golli EE, Bouaziz C, Bacha H, Hassen W (2008) Cactus (Opuntia ficus-indica) cladodes prevent oxidative damage induced by the mycotoxin zearalenone in Balb/C mice. Food Chem Toxicol 46:1817–1824. https://doi.org/10.1016/j.fct.2008.01.023
Acknowledgements
This research was done without any specific grant from a funding agency in the public, commercial, or not-for-profit sector.
Author information
Authors and Affiliations
Contributions
All authors partook in the design, interpretation, and analysis of the study’s data. SEO and SON conceptualized the experiments; SN and TI: carried out the research and preliminary data analysis. SEO, SON, supervised the investigation, SEO, SON proof check data for error. The manuscript was written and revised by SEO, SON, SN and TI.
Corresponding author
Ethics declarations
Ethical standards
All experimentation results reported in this manuscript comply with all laws on using laboratory animals in Nigeria’s experimentation and according to global best practices.
Conflict of interest
The authors have no conflict of interest to declare.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Owumi, S.E., Najophe, S.E., Idowu, T.B. et al. Protective mechanisms of gallic acid on hepatorenal dysfunction of zearalenone treated rat. Biologia 76, 3123–3135 (2021). https://doi.org/10.1007/s11756-021-00828-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-021-00828-4