Abstract
Background
Intrahepatic cholangiocarcinoma (ICC) is the second most common type of primary hepatic malignancy. Aim of this work is to analyse the features of ICC and its differential diagnosis at MRI, assessing two categories intraparenchymal and peribiliary lesions.
Methods
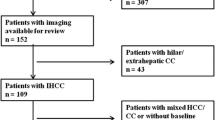
The study population included 88 patients with histological diagnosis of ICCs: 61 with mass-forming type, 23 with periductal-infiltrating tumours and 4 with intraductal-growing type. As a control study groups, we identified: 86 consecutive patients with liver colorectal intrahepatic metastases (mCRC) (groups A); 35 consecutive patients with peribiliary metastases (groups B); 62 consecutive patients (groups C) with hepatocellular carcinoma (HCC); 18 consecutive patients (groups D) with combined hepatocellular cholangiocarcinoma (cHCC-CCA); and 26 consecutive patients (groups E) with hepatic hemangioma. For all lesions, magnetic resonance (MR) features were assessed according to Liver Imaging Reporting and Data System (LI-RADS) version 2018. The liver-specific gadolinium ethoxybenzyl dimeglumine—EOB (Primovist, Bayer Schering Pharma, Germany), was employed. Chi-square test was employed to analyse differences in percentage values of categorical variable, while the nonparametric Kruskal–Wallis test was used to test for statistically significant differences between the median values of the continuous variables. However, false discovery rate adjustment according to Benjamin and Hochberg for multiple testing was considered.
Results
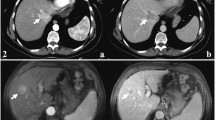
T1- and T2-weighted signal intensity (SI), restricted diffusion, transitional phase (TP) and hepatobiliary phase (HP) aspects allowed the differentiation between study group (mass-forming ICCs) and each other control group (A, C, D, E) with statistical significance, while arterial phase (AP) appearance allowed the differentiation between study group and the control groups C and D with statistical significance and PP appearance allowed the differentiation between study group and the control groups A, C and D with statistical significance. Instead, no MR feature allowed the differentiation between study group (periductal-infiltrating type) and control group B.
Conclusion
T1 and T2 W SI, restricted diffusion, TP and HP appearance allowed the differentiation between mass-forming ICCs and mimickers with statistical significance, while AP appearance allowed the differentiation between study group and the control groups C and D with statistical significance and PP appearance allowed the differentiation between study group and the control groups A, C and D.









Similar content being viewed by others
Abbreviations
- ADC:
-
Apparent diffusion coefficient
- APHE:
-
Arterial phase hyperenhancement
- AP:
-
Arterial phase
- CC:
-
Cholangiocarcinoma
- cHCC-CCA:
-
Combined hepatocellular cholangiocarcinoma
- DWI:
-
Diffusion-weighted imaging
- FLASH:
-
Fast low angle shot magnetic resonance imaging
- FISP:
-
Fast imaging with steady-state free precession
- HASTE:
-
Half-Fourier-acquired single-shot turbo spin echo
- HCC:
-
Hepatocellular carcinoma
- HP:
-
Hepatobiliary phase
- Hyper:
-
Hyperintense
- Hypo:
-
Hypointense
- ICC:
-
Intrahepatic cholangiocarcinoma
- IN:
-
Inhomogeneous
- Iso:
-
Isointense
- LI-RADS:
-
Liver Imaging Reporting and Data System
- mCRC:
-
Colorectal intrahepatic metastases
- MR:
-
Magnetic resonance
- NA:
-
Not assessed
- NPW:
-
Non-peripheral washout appearance
- PCE:
-
Progressive contrast enhancement
- PP:
-
Portal phase
- ROI:
-
Region of interest
- SPAIR:
-
Spectral attenuated inversion recovery
- SI:
-
Signal intensity
- TA:
-
Targetoid appearance
- TP:
-
Transitional phase
- VIBE:
-
Volumetric interpolated breath-hold examination
- W:
-
Weighted
- WA:
-
Washout appearance
References
Cholangiocarcinoma Working Group (2020) Italian clinical practice guidelines on cholangiocarcinoma—part I: classification, diagnosis and staging. Dig Liver Dis 52(11):1282–1293. https://doi.org/10.1016/j.dld.2020.06.045
Cholangiocarcinoma Working Group (2020) Italian clinical practice guidelines on cholangiocarcinoma—part II: treatment. Dig Liver Dis 52(12):1430–1442. https://doi.org/10.1016/j.dld.2020.08.030
Nakanuma Y, Sato Y, Harada K, Sasaki M, Xu J, Ikeda H (2010) Pathological classification of intrahepatic cholangiocarcinoma based on a new concept. World J Hepatol 2:419–427
Aishima S, Oda Y (2015) Pathogenesis and classification of intrahepatic cholangiocarcinoma: different characters of perihilar large duct type versus peripheral small duct type. J Hepatobiliary Pancreat Sci 22:94–100
Nakanuma Y, Kakuda Y (2015) Pathologic classification of cholangiocarcinoma: new concepts. Best Pract Res Clin Gastroenterol 29:277–293
Seo N, Kim DY, Choi JY (2017) Cross-sectional imaging of intrahepatic cholangiocarcinoma: development, growth, spread, and prognosis. AJR Am J Roentgenol 209(2):W64–W75. https://doi.org/10.2214/AJR.16.16923
Granata V, Fusco R, Setola SV, Avallone A, Palaia R, Grassi R, Izzo F, Petrillo A (2020) Radiological assessment of secondary biliary tree lesions: an update. J Int Med Res 48(6):300060519850398. https://doi.org/10.1177/0300060519850398
Granata V, Fusco R, Catalano O, Avallone A, Palaia R, Botti G, Tatangelo F, Granata F, Cascella M, Izzo F, Petrillo A (2017) Diagnostic accuracy of magnetic resonance, computed tomography and contrast enhanced ultrasound in radiological multimodality assessment of peribiliary liver metastases. PLoS ONE 12(6):e0179951. https://doi.org/10.1371/journal.pone.0179951
Granata V, Fusco R, Catalano O, Avallone A, Leongito M, Izzo F, Petrillo A (2017) Peribiliary liver metastases MR findings. Med Oncol 34(7):124. https://doi.org/10.1007/s12032-017-0981-7
American College of Radiology (2018) CT/MRI LI-RADS version 2018. https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/LI-RADS/CT-MRI-LI-RADS-v2018. Accessed 1 Nov 2019
Bosman FT, Carneiro F, Hruban RH, Theise ND (2010) WHO classification of tumours of the digestive system. World Health Organization, Geneva
Inchingolo R, Maino C, Gatti M, Tricarico E, Nardella M, Grazioli L, Sironi S, Ippolito D, Faletti R (2020) Gadoxetic acid magnetic-enhanced resonance imaging in the diagnosis of cholangiocarcinoma. World J Gastroenterol 26(29):4261–4271. https://doi.org/10.3748/wjg.v26.i29.4261
Maetani Y, Itoh K, Watanabe C et al (2001) MR imaging of intrahepatic cholangiocarcinoma with pathologic correlation. AJR 176:1499–1507
Shin N, Choi JA, Choi JM, Cho ES, Kim JH, Chung JJ, Yu JS (2020) Sclerotic changes of cavernous hemangioma in the cirrhotic liver: long-term follow-up using dynamic contrast-enhanced computed tomography. Radiol Med 125(12):1225–1232. https://doi.org/10.1007/s11547-020-01221-y (Epub 2020 May 15 PMID: 32415477)
Jeong HT, Kim MJ, Chung YE, Choi JY, Park YN, Kim KW (2013) Gadoxetate disodium-enhanced MRI of mass-forming intrahepatic cholangiocarcinomas: imaging-histologic correlation. AJR 201:W603–W611
Granata V, Fusco R, Venanzio Setola S, Mattace Raso M, Avallone A, De Stefano A, Nasti G, Palaia R, Delrio P, Petrillo A, Izzo F (2019) Liver radiologic findings of chemotherapy-induced toxicity in liver colorectal metastases patients. Eur Rev Med Pharmacol Sci 23(22):9697–9706. https://doi.org/10.26355/eurrev_201911_19531
Granata V, Fusco R, Avallone A, Catalano O, Filice F, Leongito M, Palaia R, Izzo F, Petrillo A (2017) Major and ancillary magnetic resonance features of LI-RADS to assess HCC: an overview and update. Infect Agent Cancer 28(12):23. https://doi.org/10.1186/s13027-017-0132-y
Granata V, Cascella M, Fusco R, dell’Aprovitola N, Catalano O, Filice S, Schiavone V, Izzo F, Cuomo A, Petrillo A (2016) Immediate adverse reactions to gadolinium-based MR contrast media: a retrospective analysis on 10,608 examinations. Biomed Res Int 2016:3918292. https://doi.org/10.1155/2016/3918292
Kang Y, Lee JM, Kim SH, Han JK, Choi BI (2012) Intrahepatic mass-forming cholangiocarcinoma: enhancement patterns on gadoxetic acid-enhanced MR images. Radiology 264:751–760
Esposito A, Buscarino V, Raciti D, Casiraghi E, Manini M, Biondetti P, Forzenigo L (2020) Characterization of liver nodules in patients with chronic liver disease by MRI: performance of the Liver Imaging Reporting and Data System (LI-RADS vol 2018) scale and its comparison with the Likert scale. Radiol Med 125:15–23
Granata V, Catalano O, Fusco R, Tatangelo F, Rega D, Nasti G, Avallone A, Piccirillo M, Izzo F, Petrillo A (2015) The target sign in colorectal liver metastases: an atypical Gd-EOB-DTPA “uptake” on the hepatobiliary phase of MR imaging. Abdom Imaging 40(7):2364–2371. https://doi.org/10.1007/s00261-015-0488-7
Granata V, Fusco R, Setola SV, Picone C, Vallone P, Belli A, Incollingo P, Albino V, Tatangelo F, Izzo F, Petrillo A (2019) Microvascular invasion and grading in hepatocellular carcinoma: correlation with major and ancillary features according to LIRADS. Abdom Radiol (N Y) 44(8):2788–2800. https://doi.org/10.1007/s00261-019-02056-6
Granata V, Fusco R, Avallone A, Filice F, Tatangelo F, Piccirillo M, Grassi R, Izzo F, Petrillo A (2017) Critical analysis of the major and ancillary imaging features of LI-RADS on 127 proven HCCs evaluated with functional and morphological MRI: Lights and shadows. Oncotarget 8(31):51224–51237. https://doi.org/10.18632/oncotarget.17227
Granata V, Fusco R, Maio F, Avallone A, Nasti G, Palaia R, Albino V, Grassi R, Izzo F, Petrillo A (2019) Qualitative assessment of EOB-GD-DTPA and Gd-BT-DO3A MR contrast studies in HCC patients and colorectal liver metastases. Infect Agent Cancer 27(14):40. https://doi.org/10.1186/s13027-019-0264-3
Granata V, Fusco R, Venanzio Setola S, Sandomenico F, Luisa Barretta M, Belli A, Palaia R, Tatangelo F, Grassi R, Izzo F, Petrillo A (2020) Major and ancillary features according to LI-RADS in the assessment of combined hepatocellular-cholangiocarcinoma. Radiol Oncol 54(2):149–158. https://doi.org/10.2478/raon-2020-0029
Lee HS, Kim MJ, An C (2018) How to utilize LR-M features of the LI-RADS to improve the diagnosis of combined hepatocellular-cholangiocarcinoma on gadoxetate-enhanced MRI? Eur Radiol. https://doi.org/10.1007/s00330-018-5893-1
Potretzke TA, Tan BR, Doyle MB, Brunt EM, Heiken JP, Fowler KJ (2016) Imaging features of biphenotypic primary liver carcinoma (hepatocholangiocarcinoma) and the potential to mimic hepato- cellular carcinoma: LI-RADS analysis of CT and MRI features in 61 cases. AJR Am J Roentgenol 207:25–31
Park HJ, Kim YK, Park MJ, Lee WJ (2013) Small intra- hepatic mass-forming cholangiocarcinoma: target sign on diffusion-weighted imaging for differentiation from hepatocellular carcinoma. Abdom Imaging 38:793–801
Park MJ, Kim YK, Park HJ, Hwang J, Lee WJ (2013) Scirrhous hepatocellular carcinoma on gadoxetic acid-enhanced magnetic resonance imaging and diffusion-weighted imaging: emphasis on the differentiation of intrahepatic cholangiocarcinoma. J Comput Assist Tomogr 37:872–881
Mathew RP, Sam M, Raubenheimer M, Patel V, Low G (2020) Hepatic hemangiomas: the various imaging avatars and its mimickers. Radiol Med 125(9):801–815. https://doi.org/10.1007/s11547-020-01185-z
Gabelloni M, Di Nasso M, Morganti R, Faggioni L, Masi G, Falcone A, Neri E (2020) Application of the ESR iGuide clinical decision support system to the imaging pathway of patients with hepatocellular carcinoma and cholangiocarcinoma: preliminary findings. Radiol Med 125(6):531–537. https://doi.org/10.1007/s11547-020-01142-w
Bozkurt M, Eldem G, Bozbulut UB, Bozkurt MF, Kılıçkap S, Peynircioğlu B, Çil B, Lay Ergün E, Volkan-Salanci B (2020) Factors affecting the response to Y-90 microsphere therapy in the cholangiocarcinoma patients. Radiol Med. https://doi.org/10.1007/s11547-020-01240-9
Gatti M, Calandri M, Bergamasco L, Darvizeh F, Grazioli L, Inchingolo R, Ippolito D, Rousset S, Veltri A, Fonio P et al (2020) Characterization of the arterial enhancement pattern of focal liver lesions by multiple arterial phase magnetic resonance imaging: Comparison between hepatocellular carcinoma and focal nodular hyperplasia. Radiol Med 125:348–355
Orlacchio A, Chegai F, Roma S, Merolla S, Bosa A, Francioso S (2020) Degradable starch microspheres transarterial chemoembolization (DSMs-TACE) in patients with unresectable hepatocellular carcinoma (HCC): long-term results from a single-center 137-patient cohort prospective study. Radiol Med 125:98–106
Barabino M, Gurgitano M, Fochesato C, Angileri SA, Franceschelli G, Santambrogio R, Mariani NM, Opocher E, Carrafiello G (2021) LI-RADS to categorize liver nodules in patients at risk of HCC: tool or a gadget in daily practice? Radiol Med 126(1):5–13. https://doi.org/10.1007/s11547-020-01225-8 (Epub 2020 May 26 PMID: 32458272)
Schicchi N, Fogante M, Palumbo P, Agliata G, Esposto Pirani P, Di Cesare E, Giovagnoni A (2020) The sub-millisievert era in CTCA: the technical basis of the new radiation dose approach. Radiol Med 125(11):1024–1039. https://doi.org/10.1007/s11547-020-01280-1 (Epub 2020 Sep 15 PMID: 32930945)
Bottari A, Silipigni S, Carerj ML, Cattafi A, Maimone S, Marino MA, Mazziotti S, Pitrone A, Squadrito G, Ascenti G (2020) Dual-source dual-energy CT in the evaluation of hepatic fractional extracellular space in cirrhosis. Radiol Med 125:7–14. https://doi.org/10.1007/s11547-019-01089-7
Crimi F, Capelli G, Spolverato G, Bao QR, Florio A, Milite Rossi S, Cecchin D, Albertoni L, Campi C, Pucciarelli S et al (2020) MRI T2-weighted sequences-based texture analysis (TA) as a predictor of response to neoadjuvant chemo-radiotherapy (nCRT) in patients with locally advanced rectal cancer (LARC). Radiol Med 125:1216–1224. https://doi.org/10.1007/s11547-020-01215-w
Danti G, Berti V, Abenavoli E, Briganti V, Linguanti F, Mungai F, Pradella S, Miele V (2020) Diagnostic imaging of typical lung carcinoids: relationship between MDCT, (111)In-Octreoscan and (18)F-FDG-PET imaging features with Ki-67 index. Radiol Med 125:715–729. https://doi.org/10.1007/s11547-020-01172-4
Koc A, Sezgin OS, Kayipmaz S (2020) Comparing different planimetric methods on volumetric estimations by using cone beam computed tomography. Radiol Med 125(4):398–405. https://doi.org/10.1007/s11547-019-01131-8
Neri E, Miele V, Coppola F, Grassi R (2020) Use of CT and artificial intelligence in suspected or COVID-19 positive patients: statement of the Italian Society of Medical and Interventional Radiology. Radiol Med 125:505–508
Farchione A, Larici AR, Masciocchi C, Cicchetti G, Congedo MT, Franchi P, Gatta R, Lo Cicero S, Valentini V, Bonomo L et al (2020) Exploring technical issues in personalized medicine: NSCLC survival prediction by quantitative image analysis-usefulness of density correction of volumetric CT data. Radiol Med 125:625–635. https://doi.org/10.1007/s11547-020-01157-3
Fornell-Perez R, Vivas-Escalona V, Aranda-Sanchez J, Gonzalez-Dominguez MC, Rubio-Garcia J, Aleman-Flores P, Lozano-Rodriguez A, Porcel-de-Peralta G, Loro-Ferrer JF (2020) Primary and post-chemoradiotherapy MRI detection of extramural venous invasion in rectal cancer: the role of diffusion-weighted imaging. Radiol Med 125:522–530. https://doi.org/10.1007/s11547-020-01137-7
Caruso D, Polici M, Zerunian M, Pucciarelli F, Polidori T, Guido G, Rucci C, Bracci B, Muscogiuri E, De Dominicis C, Laghi A (2021) Quantitative Chest CT analysis in discriminating COVID-19 from non-COVID-19 patients. Radiol Med 126(2):243–249
Grassi R, Belfiore MP, Montanelli A, Patelli G, Urraro F, Giacobbe G, Fusco R, Granata V, Petrillo A, Sacco P, Mazzei MA, Feragalli B, Reginelli A, Cappabianca S (2021) COVID-19 pneumonia: computer-aided quantification of healthy lung parenchyma, emphysema, ground glass and consolidation on chest computed tomography (CT). Radiol Med 126(4):553–560
van Assen M, Muscogiuri G, Caruso D, Lee SJ, Laghi A, De Cecco CN (2020) Artificial intelligence in cardiac radiology. Radiol Med 125(11):1186–1199. https://doi.org/10.1007/s11547-020-01277-w
Zhang L, Kang L, Li G, Zhang X, Ren J, Shi Z, Li J, Yu S (2020) Computed tomography-based radiomics model for discriminating the risk stratification of gastrointestinal stromal tumors. Radiol Med 125(5):465–473. https://doi.org/10.1007/s11547-020-01138-6
Hu HT, Shan QY, Chen SL, Li B, Feng ST, Xu EJ, Li X, Long JY, Xie XY, Lu MD, Kuang M, Shen JX, Wang W (2020) CT-based radiomics for preoperative prediction of early recurrent hepatocellular carcinoma: technical reproducibility of acquisition and scanners. Radiol Med 125(8):697–705. https://doi.org/10.1007/s11547-020-01174-2
D’Agostino V, Caranci F, Negro A, Piscitelli V, Tuccillo B, Fasano F, Sirabella G, Marano I, Granata V, Grassi R, Pupo D, Grassi R (2021) A rare case of cerebral venous thrombosis and disseminated intravascular coagulation temporally associated to the COVID-19 vaccine administration. j pers med 11(4):285. https://doi.org/10.3390/jpm11040285.PMID:33917902;PMCID:PMC8068274
La Seta F, Buccellato A, Tesè L, Biscaldi E, Rollandi GA, Barbiera F, Cappabianca S, Di Mizio R, Grassi R (2006) Multidetector-row CT enteroclysis: indications and clinical applications. Radiol Med 111(2):141–158. https://doi.org/10.1007/s11547-006-0016-5 (English. Italian PMID: 16671373)
Cantisani V, Iannetti G, Miele V, Grassi R, Karaboue M, Cesarano E, Vimercati F, Calliada F (2021) Addendum to the sonographic medical act. J Ultrasoun 24(3):229–230. https://doi.org/10.1007/s40477-021-00603-w (Epub 2021 Jul 9. PMID: 34241829; PMCID: PMC8363678)
Granata V, Fusco R, Costa M, Picone C, Cozzi D, Moroni C, La Casella GV, Montanino A, Monti R, Mazzoni F, Grassi R, Malagnino VG, Cappabianca S, Grassi R, Miele V, Petrillo A (2021) Preliminary report on computed tomography radiomics features as biomarkers to immunotherapy selection in lung adenocarcinoma patients. Cancers (Basel) 13(16):3992. https://doi.org/10.3390/cancers13163992.PMID:34439148;PMCID:PMC8393664
Nardone V, Boldrini L, Grassi R, Franceschini D, Morelli I, Becherini C, Loi M, Greto D, Desideri I (2021) Radiomics in the setting of neoadjuvant radiotherapy: a new approach for tailored treatment. Cancers (Basel) 13(14):3590. https://doi.org/10.3390/cancers13143590.PMID:34298803;PMCID:PMC8303203
Fiorini F, Granata A, Battaglia Y, Karaboue MAA (2019) Talking about medicine through mass media. G Ital Nefrol 36(1):1 (Italian, PMID: 30758148)
Gundogdu E, Emekli E, Kebapci M (2020) Evaluation of relationships between the final Gleason score, PI-RADS v2 score, ADC value, PSA level, and tumor diameter in patients that underwent radical prostatectomy due to prostate cancer. Radiol Med 125:827–837. https://doi.org/10.1007/s11547-020-01183-1
Han JH, Ahn JH, Kim JS (2020) Magnetic resonance elastography for evaluation of renal parenchyma in chronic kidney disease: a pilot study. Radiol Med 125:1209–1215. https://doi.org/10.1007/s11547-020-01210-1
Higashi M, Tanabe M, Okada M, Furukawa M, Iida E, Ito K (2020) Influence of fat deposition on T1 mapping of the pancreas: evaluation by dual-flip-angle MR imaging with and without fat suppression. Radiol Med 125:1–6. https://doi.org/10.1007/s11547-019-01087-9
Hu HT, Shan QY, Chen SL, Li B, Feng ST, Xu EJ, Li X, Long JY, Xie XY, Lu MD et al (2020) CT-based radiomics for preoperative prediction of early recurrent hepatocellular carcinoma: technical reproducibility of acquisition and scanners. Radiol Med 125:697–705. https://doi.org/10.1007/s11547-020-01174-2
Kirienko M, Ninatti G, Cozzi L, Voulaz E, Gennaro N, Barajon I, Ricci F, Carlo-Stella C, Zucali P, Sollini M et al (2020) Computed tomography (CT)-derived radiomic features differentiate prevascular mediastinum masses as thymic neoplasms versus lymphomas. Radiol Med 125:951–960. https://doi.org/10.1007/s11547-020-01188-w
Lian S, Zhang C, Chi J, Huang Y, Shi F, Xie C (2020) Differentiation between nasopharyngeal carcinoma and lymphoma at the primary site using whole-tumor histogram analysis of apparent diffusion coefficient maps. Radiol Med 125:647–653. https://doi.org/10.1007/s11547-020-01152-8
Liguori C, Farina D, Vaccher F, Ferrandino G, Bellini D, Carbone I (2020) Myocarditis: imaging up to date. Radiol Med 125:1124–1134. https://doi.org/10.1007/s11547-020-01279-8
Maddalo M, Buglione M, Pasinetti N, Triggiani L, Costa L, Magrini SM, Murphy BA (2020) The linguistic validation process of the Vanderbilt Head and Neck Symptom Survey—Italian Version (VHNSS-IT). Radiol Med 125:228–235. https://doi.org/10.1007/s11547-019-01105-w
Maddalo M, Buglione M, Pasinetti N, Triggiani L, Pegurri L, Magli A, Magrini SM, Murphy BA (2020) A pilot study on the Vanderbilt head and neck symptom survey Italian version (VHNSS-IT) to test its feasibility and utility in routine clinical practice. Radiol Med 125:423–431. https://doi.org/10.1007/s11547-019-01125-6
Nazari M, Shiri I, Hajianfar G, Oveisi N, Abdollahi H, Deevband MR, Oveisi M, Zaidi H (2020) Noninvasive Fuhrman grading of clear cell renal cell carcinoma using computed tomography radiomic features and machine learning. Radiol Med 125:754–762. https://doi.org/10.1007/s11547-020-01169-z
Acknowledgements
The authors are grateful to Alessandra Trocino, librarian at the National Cancer Institute of Naples, Italy. Moreover, for the collaboration, authors are grateful to Paolo Pariante and Paola Gargiulo (research support) and Dr Ivano Rossi (TSRM) of Radiology Division, “Istituto Nazionale Tumori IRCCS Fondazione Pascale-IRCCS di Napoli,” Naples, I-80131, Italy
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to be disclosed. The authors confirm that the article is not under consideration for publication elsewhere. Each author has participated sufficiently to take public responsibility for the manuscript content.
Research involving human participants and/or animals
Institutional review board approval was obtained for this retrospective study.
Informed consent
Informed consent was waived for the retrospective nature of the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Granata, V., Grassi, R., Fusco, R. et al. Intrahepatic cholangiocarcinoma and its differential diagnosis at MRI: how radiologist should assess MR features. Radiol med 126, 1584–1600 (2021). https://doi.org/10.1007/s11547-021-01428-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11547-021-01428-7