Abstract
Four commercially available disinfection products were tested for their efficacy against Clavibacter michiganensis subsp. sepedonicus (Cms), causative agent of bacterial ring rot, on wooden potato storage crates. Each of these products represented a different class of biocide, i.e. organic acids (benzoic acid), peroxygenous compounds (potassium peroxysulfate), quaternary ammonium compounds (didecyldimethylammoniumchloride) and hypochlorite-generating compounds (sodium-p-toluenesulfochloramide). The target objects consisted of small wooden panels that were smeared with potato tuber pulp homogenized with a high inoculum of Cms. By dipping the inoculated panels into the product solutions and subsequent determination of the densities of colony-forming units, the relative efficacy of the products was established. The two products that exhibited the highest relative efficacy were studied for their disinfection performance when applied as cleaning agent in a conventional automated crate washer. The results showed that jet cleaning in a crate washer for 2 min using the authorised dose of the product containing sodium-p-toluenesulfochloramide is an effective method for disinfecting Cms-contaminated wooden potato crates.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Clavibacter michiganensis subsp. sepedonicus (Cms) can cause bacterial ring rot in potato. Since effective chemical control agents are not available, the control of the disease is highly reliant on strict hygienic practices. Cms is a hemi-biotrophic bacterium and for growth and reproduction, it therefore essentially depends on its host plants. The bacterium nevertheless is able to survive on contaminated surfaces for many years, in particular in a non-competitive environment at conditions of low humidity and low temperature (Nelson 1978, 1979, 1980; Nelson and Kozub 1990; Ward et al. 2001). Being mainly seed-borne, Cms thus can be introduced in seed potatoes from contaminated soil, organic matter, equipment and storage facilities (Abdel-Kader et al. 2004; De Boer and Boucher 2011). The viability in water is reported to be relatively short, extending only to several weeks (Van der Wolf and Van Beckhoven 2004).
Seed potato tubers generally are stored and transported in open wooden crates which are re-used year after year. It is a common practice to clean the crates after usage by mechanical removal of dirt, including potato debris. Generally, this is done with water by means of a water jet apparatus. Water jet apparatuses can be simple hand-held cleaners or more sophisticated automated equipment provided with conveyor belts, crate rotors and multiple nozzles. Although mechanical removal of dirt is expected to substantially reduce the number of microbes attached to the wood, it will not eradicate bacterial life present on the wood surface. Hygiene protocols intended to manage bacterial potato diseases therefore comprise a chemical disinfection step, which either is incorporated in or chronologically added to the cleaning process. The efficacy of chemicals against Cms on wooden surfaces depends on various factors. Among them may be the exposure time, the moist content of the target, the temperature, the stability of the active substance, the mechanism of action, the Cms matrix and in relation to that, the presence of organic material that may act as scavenger for the disinfectant.
Similar to other vascular plant pathogenic bacteria, Cms forms a self-produced extracellular matrix of polysaccharides inside the host plant that protects the bacterium against unfavourable conditions (Marques et al. 2003; Ramey et al. 2004). Bacterial cells of microbial biofilms generally exhibit a significantly higher tolerance towards antimicrobials than their planktonic analogues (Howard et al. 2015). Cms contamination on surfaces of potato tuber crates generally originates from tuber material and therefore mainly constitutes Cms biofilms containing tuber debris, in particular starch. To enable the employment of disinfectants against Cms at their full potential rates, disintegration of the protective biofilm matrix can be considered as a prerequisite for effective disinfection of the crate surfaces. The common practice of water jet cleaning is an obvious method to disrupt microbial biofilms by suspending the particles and dissolving the extracellular polysaccharides in water.
In the present study, four commercially available disinfection products were tested against Cms. Each of these products represented a different class of biocide, i.e. organic acids (benzoic acid), peroxygenous compounds (potassium peroxysulfate), quaternary ammonium compounds (didecyldimethylammoniumchloride) and hypochlorite-generating compounds (sodium-p-toluenesulfochloramide). In order to mimic the conditions at the crate surfaces after jet cleaning, the target objects consisted of small wooden panels that were smeared with potato tuber pulp homogenized with a high inoculum of Cms prepared from suspended biofilms freshly harvested from agar medium. By dipping the inoculated panels into the product solutions and subsequent determination of remaining Cms viability by a cultivation-based method, the relative efficacy of the products was established. The two products that exhibited the highest relative efficacy were studied for their disinfection performance when applied as medium for mechanical cleaning in a conventional automated crate washer. The results and the factors affecting the effectiveness of use are discussed in view of the need for a reliable and practical disinfection procedure.
Materials and Methods
Disinfection Products
The trade names of the disinfectants used (with proportion and name of the active substance between brackets) are as follows: Menno Clean (90 g/l benzoic acid), Virkon S (45.3% pentapotassium bis(peroxymonosulphate) bis(sulphate)), Bardac 22 (450 g/l didecyldimethylammoniumchloride) and Halamid-d (81% sodium-p-toluenesulfochloramide). They are here referred to as products A, B, C and D, respectively. Foam of product C was prepared (experiment 2) by extruding an aqueous solution of the product (2% w/v) under high air pressure.
Preparation of the Inoculum
Experiments 1 to 5 were performed with Cms strain IPO 1873, which is a streptomycin-resistant mutant of NCPB 4053. Harvested from a solid agar plate culture, Cms-biofilm was suspended in tap water to a final density of ca. 108–1010 cfu/ml. This suspension was homogenized with unpeeled (experiment 1) or peeled (experiments 2 to 5) potato tubers (suspension to tuber ratio = 1:9 w/w; cultivar Frieslander), resulting in a fine pulp, which in the following is referred to as the inoculum. Pulp for mock inoculations was prepared in the same way with tap water instead of suspended Cms. For experiment 6, inoculum was prepared from ring rot-diseased potato tubers that were harvested from plants grown from seed potatoes injected with Cms strain LMG2889. Heavily infected vascular tuber tissue was carefully isolated and subsequently homogenized with water using a blender (water to tuber ratio = 1:9 w/w).
Preparation of the Experimental Target Objects for Disinfection
Dry, undamaged planks (untreated whitewood from spruce tree, Picea abies), which were derived from original potato storage crates, were cut into rectangular panels with the following dimensions: 9.5 cm × 5 cm × 2 cm (length × width × depth). The direction of the wood veins was in parallel with the length of the panels. Each panel was provided with (a) one perforation (diameter 3 mm), centred at a distance of ca. 0.7 cm from one edge of the flat and long side, and (b) one pencil-drawn semi-circle (diameter 6 cm) with the diameter of the semi-circle centred and on top of the opposite long side panel edge. The area inside the semi-circle was covered with 0.4–0.9 g of inoculum or mock inoculum, which was firmly smeared onto the wood surface using a spoon. The applied inoculum was allowed to dry under moderate laminar airflow for ca. 65 h at room temperature before the target objects were subjected to the disinfection treatments.
Disinfection Treatments
The target objects were treated either by dipping them into the product solution or by exposing them in a crate washer to pressure sprayed product solution. In the latter case, the panels were mounted at four different positions inside an original potato storage crate, which subsequently was subjected in the crate washer to a conventional cleaning procedure using disinfectant solution instead of water as cleaning medium. Pre-moistening of the inoculum (experiments 2 to 5) was done immediately before application of the disinfection treatments by moderately spraying a fine dust of water over the inoculum. For the dip treatments, panels belonging to the same group of replicates (n = 3 or n = 5) were hung side by side to a saté skewer, using the perforations of the panels as mounting point. Each string of panels was individually sunk into a plastic container (volume 1 l) filled with 700–800 ml of freshly prepared disinfectant solution; care was taken that the semi-circles were completely submerged in the liquid by firmly pressing both ends of the saté skewer on the rim of the container. The wood volume submerged was ca. 81 ml per panel and the wood area submerged was ca. 77 cm2 per panel. After the treatments were applied and excess of liquid was dripped off, the panels were allowed to air-dry at room temperature. Finally, the dry panels were incubated at 20 °C in dark, aerated and dry conditions until sampling. The crate washer used for the automated spray treatments was a standard Boxer box cleaner (Mechatec BV, Tollebeek, The Netherlands), provided with a flat jet spray nozzle and a cylindrical jet spray nozzle that is centrally positioned in the inner crate space exposing the inner surfaces of the crate to ca. 167 l of liquid per minute with a jet power of 15 kg cm−2 while the crate was turning with two rotations per minute; in the meantime, the outer surfaces of the crate were exposed to ca. 83 l of liquid per minute applied under the same pressure by 32 flat jet nozzles.
Sampling, Plating and Colony Counting
About 7 days after the disinfection treatments of the panels, the organic material remaining within the semi-circles was removed from the wood surface using a cotton swab moistened with 0.01% Tween 20 in quarter strength Ringers solution (RT, Sigma). Each sample was individually collected and suspended in 1 ml (experiment 1) or 3 ml (experiments 2–6) of the RT. The sample suspensions were plated undiluted and diluted (dilution factor 100, and in experiment 6, dilution factor 100 and 1000; prepared in 0.85% NaCl) in duplicate on agar medium YGM containing 100 mg/l of streptomycin and 100 mg/ml of cycloheximide (Van der Wolf and Van Beckhoven 2004), except for the samples derived from the naturally infected inoculum (experiment 6), which were plated on agar medium YGM containing no antibiotic but 100 mg/ml of cycloheximide. After 1–2 weeks of incubation of plates at 20–23 °C, the Cms colonies formed were counted.
TaqMan Protocol
Crude DNA samples were prepared by suspending individual Cms colonies in 50 μl of Milli-Q water and subsequent cooking of the mixtures for 5 min at 95 °C. Two microlitres of these DNA preparations was added to a mix of ROXII (1×), premix ex Taq (1×) from Takara (Westburg, Leusden, The Netherlands), forward primer Cms 50-2F and reverse primer Cms 133-R (0.3 μM each) and probe FAM-Cms 50–53 T (0.1 μM) to a total volume of 25 μl (Schaad et al. 1999). The real-time PCR amplification was performed using a real-time PCR system ABI7500 (applied Biosystems BV) with the following cycling conditions: initial denaturation for 2 min at 95 °C; then 40 cycles of 15 s at 95 °C and 1 min at 60 C.
Statistical Analysis
All experiments were laid out as randomized block designs with two or three influential factors. The number of colony-forming units per gramme inoculum was the response variate. Analysis of variance was performed on 10log (cfu/g inoculum) using Genstat (Anonymous 2015) for experiments 1, 2, 4 and 5. The probability for interaction according to the F test is reported for experiments 1 and 2 and for object in experiments 4 and 5. The data from experiments 1 and 2 were analysed as randomized block designs and the data from experiments 4 and 5 were analysed as complete randomize designs, because data from lab and crates were analysed simultaneously. Results of the pairwise t test are presented with letters. Means without a common letter differ significantly according to Student’s t test (P = 0.05). Also, the least significant differences (lsd) are reported according to Student’s t test at probability 0.05. In experiments 3 and 6, an exponential curve was fitted to the number of colony-forming units per gramme inoculum and 10log (cfu/g inoculum +1), respectively, using a generalized linear model (GLM) with distribution = Poisson, link function = logarithm and dispersion = *.
Results and Discussion
The effectiveness of use of a disinfection product largely depends on the actual physical and chemical conditions on the application site. These conditions control the chemical activity of the product and determine to what extent the active substance can make contact with the target microorganism. The environment may also affect the biological sensitivity of the microorganism for the product. The experiments therefore were designed to meet as much as possible the conditions encountered in practice. Cms contamination of potato storage crates generally concerns Cms-infected tuber material smeared on the wood surfaces of the crates. The preferred inoculum to start with therefore was Cms-infected tuber material. However, various attempts to directly initiate decay in harvested potato tubers with our streptomycin-resistant Cms mutant were unsuccessful (data not shown). In order to mimic the biological matrix of Cms contamination in disrupted biofilm, artificial inoculum was prepared by mixing a thick suspension of the Cms mutant with freshly homogenized potato tuber material. The thick suspension was freshly prepared from Cms biofilms harvested from Cms cultures on agar medium. This inoculum was firmly applied to the surface of small panels derived from original wooden potato storage crates, and subsequently air-dried at room temperature, resulting in the experimental target objects for testing the disinfectants in a series of six experiments.
Experiment 1: Product Concentration
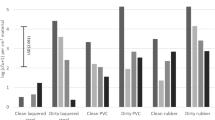
The four disinfection products A, B, C and D each were tested in three concentrations, i.e. in the authorized concentration, recommended by the supplier and in one lower and one higher concentration (Table 1). The inoculated panels were dipped into the freshly prepared solutions for 1 s, which means that the panels were extracted from the liquid immediately after submersion. As expected, treatment with increased concentration of disinfectant led to decreased numbers of viable Cms recovered from the wood surface. Comparison of the number of Cms colonies that developed from 100 times diluted samples indicated that the four products slightly differed with respect to their Cms killing capacity under the experimental conditions applied (Fig. 1). However, as concluded from the number of Cms colonies grown from the undiluted samples, none of the concentrations of any of the four products resulted in complete eradication of Cms (data not shown). Further increase of the concentration of A, B and C is constrained by the limited solubility of these products and additional registration requirements. Application of increased concentration of product D is discouraged by economic and environmental costs and additional registration requirements. It was therefore decided to continue the study with the authorized concentration of A, B, C and D (for each product, the second listed concentration in Table 1), which obviously is not always the maximum biocidal capacity.
Number of cfu of Cms per gramme of inoculum, recovered from the wood panels after a single dip of 1 s in solutions of products A–D. Means denoted by the same letter did not significantly differ. LSD = 1.9; P < 0.05; n = 3. The average number of cfu of Cms per grammes of inoculum recovered from the water-treated controls was 5.0 on the 10log scale. The cfu were established from 100 times diluted swab samples. It should be noted that none of the undiluted swab samples were free of Cms
Experiment 2: Duration of Exposure and Effect of Pre-moistening the Target Object
In addition to product concentration and temperature, the duration of exposure of the inoculum to the active substance is a determining factor for successful disinfection. Since the chemical activity is deployed in solution, it might seem that the period of exposure after an instant dip lasts until the solvent has evaporated from the wood surface. However, in reality, the exposure time might be shorter, because an instant dip provides only a limited amount of reactant, which may be dissipated completely before the wood surface runs dry. In contrast to an instant dip application, submersion for a prolonged period of time allows the reactant for that period of time access from the bulk solution to Cms at a constant rate. An alternative method to increase the exposure time is covering the wood surface with a layer of foam prepared from the disinfection solution. In this experiment, the disinfecting activity of products A, B, C and D was compared after application by a single dip, by a repeated dip (with 2 h of incubation in between each dip) and by submersion for 10 min. A foam treatment with product C was also included, which comprised the covering of horizontally oriented panels with 1.20 g of foam freshly prepared from 2% (w/v) of product C (ca 25 mg of foam per cm2) and which slowly collapsed over a period of ca. 20 min, leaving a wet surface which was allowed to dry. The amount of moisture that on average was taken up by the panels during the single dip, the repeated dip and the 10 min of submersion was 1.20, 1.98 and 4.54 g, respectively. The treatments were applied to dry panels and to panels that were moderately pre-moistened with a fine dust of water.
Submersion for a period of 10 min appeared to be very effective, since no Cms colonies did develop from the samples obtained after treatment with all products, both with and without pre-moistening of the panels (results not shown). The samples obtained from control panels that were submerged for 10 min in water showed more than 106 cfu/g of inoculum.
As shown in Fig. 2, the repeated dip of the pre-moistened panels did not appear to be significantly more effective than the single dip, irrespective of the product applied. Furthermore, the efficacy of the foam application with product C did not differ from the efficacy of the other treatments. Interestingly, after the repeated dip application of products A, B and C, the panels that had not been pre-moistened presented a substantial (although not in every case significant) lower average recovery of Cms than the panels that had been pre-moistened. In contrast, the single dip and the repeated dip application with product D were significantly more effective on pre-moistened panels than on panels that were not pre-moistened (Fig. 2). The observation that the efficacy of product D depends on pre-moistening of the target object may be explained by the fact that product D, contrary to products A, B and C, does not contain any surfactant. Presumably, the organic surface of the target object shows in dry condition a relatively hydrophobic nature, which reduces efficient attachment of the aqueous solution of product D during the short dip exposure. The surfactants present in products A, B and C enhance the wetting ability of the dissolved products. Even without the presence of a surfactant, the efficacy of product D at least equalled the efficacy of the other disinfectants. These results confirm that a dip application, or a repeated dip application, is by far a too short exposure to obtain total Cms eradication using authorized concentrations of any of the four products.
Number of cfu of Cms per gramme of inoculum, recovered from the wood panels after a single dip (“1× dip”) or a repeated dip (“2× dip”) in product A (1% w/v), product B (0.3% w/v), product C (2% w/v), product D (1% w/v) or a foam treatment with product C (“foam”; 2% w/v). The panels were treated either without pre-moistening (“dry”; white bars) or with pre-moistening (“moist”; black bars). Means denoted by the same letter did not significantly differ. LSD = 2.331; P < 0.05; n = 3. The cfu were established from 100 times diluted swab samples. The average number of Cms freshly applied was ca. 107 cfu/g of inoculum
Experiment 3: Disinfection by Submersion as a Function of the Exposure Time
The results of experiment 2 indicated for all products tested that the minimum exposure time to obtain total Cms disinfection of the target objects must be somewhere between 1 s and 10 min. In experiment 3, the disinfection efficacy as a function of the exposure time was therefore established by submerging the pre-moistened target objects for 0, 1, 2, 5 and 10 min in aqueous solutions of products A, B, C and D at their authorized concentrations. Pre-moistened controls were exposed to water for the same periods of time. Per exposure period, five panels were included in the experiment for each tested chemical. The exposure time required for total Cms eradication lies between 2 and 5 min in the case of product A, between 1 and 2 min in case of product D and between 5 and 10 min in case of products B and C (n = 5). None of the periods of exposure to water resulted in a substantial reduction of Cms. Figure 3 shows the colony-forming units per gramme of applied inoculum that was determined after application of the various treatments.
The number of viable Cms remaining on the inoculated wooden target objects as a function of the period of exposure to product A (1% w/v), product B (0.3% w/v), product C (2% w/v) and product D (1% w/v). The symbols represent the experimentally recovered average cfu number. The curves show the exponential Cms decrease and were obtained by fitting a GLM to the experimental data
Based on these experimental data, the exponential decrease of cfu as a function of the exposure time was fitted using regression analysis. The resulting curves are presented in Fig. 3. They can be described by the equation:
where N represents the predicted number of Cms remaining on the target object, T represents the exposure time (in min) and constant C (expressed in min−1) represents the disinfection power of the product. In this experiment, C was found to be 5.73 for product A, 1.935 for product B, 1.245 for product C, 11.5 for product D and 0.0340 for water. Total eradication of Cms is characterized by N < 1. The predicted minimum incubation time required for N < 1 is 1.2 min for product A, 3.6 min for product B, 5.5 min for product C and 0.6 min for product D. These results show that product D exhibited the best disinfection performance, followed by product A, product B and product C, respectively.
Experiment 4: Efficacy of Products A and D When Applied by Jet Cleaning in a Crate Washer
Submersion is not the usual technique to treat potato crates with disinfecting solution. Normally, this is done by jet cleaning inside a crate washer, or by a hand-held jet apparatus. Submersion ensures continuous availability of the disinfecting reactant to the wooden surface of the crate, which is not necessarily the case during jet cleaning. A more obvious difference is that jet cleaning, contrary to submersion, mechanically removes debris from the infected object, which may increase the total disinfection rate. The objective of the present experiment was to determine the efficacy of the two best performing disinfectants (i.e. products A and D) when applied in the authorized concentration (1% w/v) by means of jet cleaning inside a conventional crate washer and to compare this with the efficacy by submersion. A batch of inoculated wooden target objects was prepared. One part of this batch was mounted at four positions (1, 2, 3 and 4) inside original potato storage crates (Fig. 4), which after pre-moistening were disinfected using the standard cleaning procedure in the crate washer for 2 min or for 5 min. In parallel, a second part of the inoculated batch of target objects after pre-moistening was treated by submersion for 2 min or for 5 min. Controls were either not treated or treated with water. The remaining viable Cms bacteria per grammes of inoculum were calculated from the cfu established in the samples taken from the wooden target objects. The experiment was performed in three-fold, except for the 5 min of jet cleaning with product A, which was performed only once.
Schematic representation of a potato storage crate, showing the four positions of the experimental target objects inside the storage crates during the processing procedure in the crate washer, i.e. 1 central on the crate bottom, 2 beneath the diagonal buttress near the crate bottom, 3 in a box corner near the crate bottom and 4 central beneath the upper rim of the crate
The average 10log (number of cfu) recovered from the untreated controls was 6.38 per grammes of inoculum (Fig. 5). In none of the experiments, submersion did lead to any visible loss of the inoculum layer from the wooden panels into the disinfectant solution. Apparently, submersion provided disinfection without substantial cleaning. In contrast, treatment in the crate washer (2 and 5 min) resulted in shiny wood surfaces on which no organic material could be detected by eye; apparently, jet cleaning led to efficient mechanical removal of the organic inoculum. Comparison of the number of cfu that remained after jet cleaning with water and the number of cfu that remained after submersion in water revealed that the mechanical removal of organic material contributed to a significant decline of Cms (Fig. 5). The cleaning efficiency did not significantly differ between the target objects mounted on different positions inside the crate, except for the objects mounted central on the crate bottom (position 1), which showed a significantly lower number of viable Cms after 5 min of cleaning with water than the other objects (Fig. 5).
Number of viable Cms per gramme of inoculum, recovered from the wood panels after 2 (black bars) and 5 min (white bars) jet cleaning with water inside a potato crate on positions 1, 2, 3 and 4 (see Fig. 4), and after submersion in water for 2 and 5 min. The hatched column at the right shows the average densities on the untreated control panels. Means denoted by the same letter did not significantly differ. Figure 5. LSD = 1.12; P not significant, ≥0.05; n = 3
No Cms was detected in any of the samples recovered after jet cleaning for 2 min and for 5 min with either product A or product D. The same was found after submersion for 2 min and for 5 min in product D. Product A was less effective, since 2 min of submersion in product A appeared insufficient for eradication of Cms. The average number of cfu recovered after submersion in product A was ca. 200 cfu/g of inoculum. No viable Cms was recovered after submersion in product A for 5 min.
Experiment 5: Disinfection by Jet Cleaning with Product D as a Function of the Exposure Time
Disinfection by means of jet cleaning inside the crate washer with 1% of product D was investigated in more detail using exposure times of 0, 0.5, 1, 2 and 5 min. The target objects were mounted inside the crates at four positions (position 1, 2, 3 and 4; Fig. 4). Controls were treated with water. In parallel, inoculated panels were subjected to submersion in 1% of product D for 0, 0.5, 1, 2 and 5 min. All target objects were prepared at the same moment from the same batch of inoculum and were pre-moistened before treatment. The experiment was performed in three-fold.
The target samples were supplied with circa 10 times less Cms than in experiment 4; the average number of cfu recovered from the untreated controls was 105 per grammes of inoculum (Fig. 6). The results obtained with the control jet treatments using water confirmed that the cleaning procedure by itself leads to a significant removal of Cms at all four positions tested (Fig. 6). As was observed in experiment 4, the central position at the crate bottom (position 1) is cleaned significantly more efficient than the three other positions. The results also show that total eradication of Cms at least requires disinfection. None of the samples recovered from the target objects that were subjected to 1% of product D in the crate washer exhibited any viable Cms. These results confirm the observations of experiment 4 that 2 and 5 min of exposure is adequate for complete eradication of Cms, and indicate that even 0.5 and 1 min may be sufficient.
Number of viable Cms per gramme of inoculum, recovered from the wood panels after 0.5-, 1-, 2- and 5-min jet cleaning with water inside a potato crate on positions 1, 2, 3 and 4 (see Fig. 4), and after submersion in water for 0.5, 1, 2 and 5 min. The hatched column at the right shows the average Cms densities on the untreated control panels. Means denoted by the same letter did not significantly differ. LSD = 1.56, P ≥ 0.05, not significant; n = 3
Submersion for only 0.5 min in 1% of product D appeared to be sufficient for total eradication of Cms. This exposure period is relatively short as compared to the exposure time predicted by regression analysis in experiment 3 (being 0.6 min). This can be explained by the fact that the starting density of Cms was circa 10 times lower than the starting density in experiments 3 and 4. Apart from the presence of Cms, also the presence of other bacteria was monitored after plating the undiluted sample suspensions on the selective medium. No or only some sporadic other bacterial colonies than Cms were found.
Experiment 6: Disinfection by Submersion in 1% of Product D of Panels Inoculated with Natural Infected Tuber Material
The experiments described above were performed with artificial inoculum freshly prepared by homogenisation of tuber pulp with a streptomycin-resistant Cms mutant. To verify whether this artificial inoculum constituted a realistic representation of naturally infected tuber material with regard to its susceptibility to 1% of product D, a submersion experiment was performed using panels smeared with material derived from tubers showing bacterial ring rot. The natural inoculum was derived from the area near the vascular tuber tissue and contained high densities of Cms (ca. 107 cfu/g of inoculum). The target objects were exposed to 1% of product D or water for 0, 0.5, 1, 2 and 5 min. Surviving Cms was detected on agar plates devoid of streptomycin; colony identification was done by eye, supported by TaqMan PCR. The panels were not pre-moistened before immersion, since experiment 2 showed that pre-moistening promotes the efficacy of product D.
The relation between exposure time and the cfu densities of Cms recovered is depicted in Fig. 7. No viable Cms was recovered after submersion for 2 min in 1% of product D. These results show that product D exhibited the same disinfection efficacy towards the artificial Cms inoculum as towards the Cms inoculum prepared from bacterial ring rot-infected tubers.
The average number of viable Cms recovered from the inoculated wooden target objects as a function of the time of exposure to submersion in water (open symbols and dotted line) and to 1% of product D (closed symbols and solid line). n = 3. The curves show the exponential Cms decrease and were obtained by fitting a GLM to the experimental data
Conclusion
A standard cleaning procedure inside a conventional crate washer using water as washing medium can be an adequate method for mechanical disruption and removal of Cms-infected potato tuber debris that is stuck at the inside surfaces of wooden potato storage crates. However, the procedure is definitively insufficient to obtain crates that are devoid of viable Cms. For total eradication of Cms, the procedure requires application of a disinfectant solution instead of water. Since Cms biofilms generally exhibit a significantly higher tolerance towards antimicrobials than their planktonic analogues (Howard et al. 2015), disinfection of crate surfaces should be preceded or accompanied by disruption of the biofilm matrix. This study shows that Cms originating from disrupted biofilms, mixed with tuber material and present on wooden crate surfaces can be eradicated within 2 min by the exposure to conventional disinfectants. Application of the disinfectant by means of a crate washer is preferable to submersion, because the treatment inside the crate washer provides the strong combination of mechanical jet cleaning and chemical disinfection.
Among the four chemical disinfectants tested, the product based on the active substance sodium-p-toluenesulfochloramide (product D) appeared to be the most effective. Since 2 min of mere submersion into the authorized concentration of product D did not leave any detectable viable Cms, exposure to this solution for 2 min by means of jet cleaning inside a conventional crate washer is considered as a reliable method for disinfection of Cms-contaminated wooden potato crates.
All experiments described here were performed with freshly prepared product solutions. Under practical conditions, conventional crate washers generally recirculate the cleaning solution that has been applied to the target objects. During this process, the active substance is inevitably dissipated by reaction with organic material that is washed from the wooden crates during jet cleaning. It is therefore recommended to provide the crate washer with an installation for filtering off organic material and for monitoring and supplementing the concentration of active substance.
References
Abdel-Kader D, Kakau J, Müller P, Pastrik K-H, Seigner L (2004) Verbreitung der Bakteriellen Ringfäule der Kartoffel (Clavibacter michiganensis ssp. sepedonicus) durch kontaminierte Lagerkisten. Gesunde Pflanzen 56:116–121
Anonymous (2015) Genstat for windows 18th edition. VSN International, Hemel Hempstead, UK. Web page: Genstat.co.uk
De Boer SH, Boucher A (2011) Minireview/Minisynthèse prospect for functional eradication of the bacterial ring rot disease of potato. Can J Plant Pathol 33:297–307
Howard RJ, Harding MW, Daniels GC, Mobbs SL, Lisowski SLI, De Boer SH (2015) Efficacy of agricultural disinfectants on biofilms of the bacterial ring rot pathogen, Clavibacter michiganensis subsp sepedonicus. Can J Plant Pathol 37:273–284
Marques LLR, De Boer SH, Ceri H, Olsen ME (2003) Evaluation of biofilms formed by Clavibacter michiganensis subsp. sepedonicus. Phytopathology 93:S57
Nelson GA (1978) Survival of Corynebacterium sepedonicum on contaminated surfaces. Am Potato J 55:449–453
Nelson GA (1979) Persistence of Corynebacterium sepedonicum in soil and in buried potato stems. Am Potato J 56:71–77
Nelson GA (1980) Long-term survival of Corynebacterium sepedonicum on contaminated surfaces and in infected potato stems. Am Potato J 57:595–599
Nelson GA, Kozub GC (1990) Survival of Corynebacterium sepedonicum at freezing and at wide fluctuations between freezing and above-freezing temperatures. Am Potato J 67:625–631
Ramey BE, Koutsoudis M, Von Bodman SB, Fuqua C (2004) Biofilm formation in plant-microbe associations. Curr Opin Microbiol 7:602–609
Schaad NW, Berthier-Schaad Y, Sechler A, Knorr D (1999) Detection of Clavibacter michiganensis subsp. sepedonicus in potato tubers by BIO-PCR and an automated real-time fluorescence detection system. Plant Dis 83:1095–1100
Van der Wolf JM, Van Beckhoven JRCM (2004) Factors affecting survival of Clavibacter michiganensis subsp. sepedonicus in water. J Phytopathol 152:161–168
Ward L, D’Aubin J, De Boer SH (2001) Persistence of Clavibacter michiganensis subsp. sepedonicus in sterilized soil but failure to confirm its survival overwinter in field soil. In: De Boer SH (ed) Plant pathogenic bacteria. Kluwer Academic Press, Dordrecht, pp 375–378
Acknowledgments
This study was financially supported by the Dutch Ministry of Economic Affairs. The authors thank Dr. J. van Vaerenbergh (ILVO, Merelbeke, Belgium) for providing bacterial ring rot-affected potato tubers.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Stevens, L.H., Lamers, J.G., van der Zouwen, P.S. et al. Chemical Eradication of the Ring Rot Bacterium Clavibacter michiganensis subsp. sepedonicus on Potato Storage Crates. Potato Res. 60, 145–158 (2017). https://doi.org/10.1007/s11540-017-9342-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11540-017-9342-3