Abstract
We consider a two-patches SIR model where communication occurs through commuters, distinguishing explicitly permanently resident populations from commuters populations. We give an explicit formula of the reproduction number and show how the proportions of permanently resident populations impact it. We exhibit nonintuitive situations for which allowing commuting from a safe territory to another one where the transmission rate is higher can reduce the overall epidemic threshold and avoid an outbreak.
Similar content being viewed by others
1 Introduction
Since the pioneer work of Kermack and McKendrick (1927), the SIR model has been very popular in epidemiology, as the basic model for infectious diseases with direct transmission (e.g., Anderson and May 1991). It retakes great importance nowadays due to the recent coronavirus pandemic. While early models were not spatialized, the importance of accounting for spatial heterogeneity has been often reported in the literature (see, e.g., Angulo et al. 1979; Sattenspiel and Dietz 1995; Keeling et al. 2004; Keeling and Rohani 2007; Kelly et al. 2016; Li et al. 2021). However, different mechanisms come into play to explain the spatial spreading of a disease. Although diffusion appears to be a natural process to describe the local propagation of an infectious agent among a population, which leads to models with partial differential equations (Murray 2003), it appears to be not well suited for describing long distance spreading. In particular, transportation between cities comes into the picture as a major source of rapid spreading among nonhomogeneous populations (Arino and van den Driessche 2003; Arino et al. 2007; Takeuchi et al. 2007; Liu and Stechlinski 2013; Mpolya et al. 2014; Chen et al. 2014; Yin et al. 2020; Tocto-Erazo et al. 2021; Lipshtat et al. 2021). Meta-populations or multi-patches models are then more appropriate to describe the spatial characteristics of the propagation (Wang and Mulone 2003; Wang and Zhao 2004; Arino and van den Driessche 2006; Gao 2007; Arino 2009), as already well considered in ecology (Hanski 1999; MacArthur 2001). These models require a precise description of the movements between patches, which are most of the time assumed to be linear and thus encoded into a connection matrix (Arino and van den Driessche 2006; Arino 2009). Typically one obtains a system of ordinary differential equations on a graph, which couples the communication dynamics with the epidemiological one.
For diseases spreading among human populations living in different cities, commuters (individuals housing in a city, traveling regularly for short periods in a neighboring city, and coming back to their home city) play a crucial role in the disease propagation among territories (Keeling and Rohani 2002; Keeling et al. 2004; Keeling and Rohani 2007; Mpolya et al. 2014; Yin et al. 2020). Such coupling between patches have been already considered in the literature, distinguishing among populations \(N_i\) attached to a city i the subpopulation \(N_{ii}\) present in its permanent housing from other subpopulations \(N_{ij}\) temporary present in another city \(j \ne i\) [it can be also seen as multi-groups models as in Clancy (1996), Guo et al. (2006), Iggidr et al. (2012)]. However, such models explicitly assume that the whole population housing in a given city can potentially commute to another one. We believe that this is not always fully realistic and that a subpopulation that never (or very rarely) moves to another city should be distinguished from the subpopulation that visits at a regular basis another city. Therefore, we consider an extension of such models, which explicitly takes into consideration two kinds of movement: an Eulerian one which describes the flow between patches that mixes populations, and an Lagrangian one which assigns home locations of individuals, as described in the more general framework (Citron et al. 2021). The study of this extension, which has not yet been analyzed analytically in the literature, to our knowledge, and how it impacts the disease spreading, is the primary objective of the present work. For this purpose, we establish an analytical expression of the reproduction number [as the epidemic threshold formerly introduced and analyzed in Diekmann et al. (1990), van den Driessche and Watmough (2002), Diekmann et al. (2007), Dhirasakdanon et al. (2007)] for the two patches case (that is also valid for the particular case when the whole populations travel, for which the exact expression of the reproduction number has not been yet provided in the literature).
We also had in mind to consider heterogeneity among territories when disease transmission differs from one city to another one. Typically, non-pharmaceutical interventions (such as reducing physical distance in the population) could be applied with different strength in each city, providing distinct transmission rates. When one territory being isolated presents a higher reproduction number than the other territory, it can be considered as a core group in the epidemiological terminology (Hadeler and Castillo-Chavez 1995; Brunham 1997), and commuters contribute then to spread the epidemics in both territories. We aim at analyzing more precisely how the proportions of commuters in each city can increase or decrease the overall reproduction number. Intuitively, one may believe that the best way to reduce the spreading is to encourage commuters from the city with the lowest transmission rate not to travel to the other city, and on the opposite to encourage as much as possible commuters from the other city to spend time in the safer city. Indeed, we shall see that this is not always true... The second objective of the present work is thus to study the minimization of the epidemic threshold of the two-patches model with respect to these proportions, depending on the commuting rates. This analysis can potentially serve for decisions making to prevent epidemic outbreak [as in Knipl (2016), for instance].
The paper is organized as follows. In the next section, we present the complete model in dimension 18 and give some preliminaries. Sect. 3 is devoted to the analysis of the asymptotic behavior of the solutions of the model. We give and demonstrate an explicit expression of the reproduction number, introducing four relevant quantities \(q_{ij}\) (\(i, j = 1,2\)). In a corollary, we also give an alternative way of computation, which is useful in the following. In Sect. 4, we study the minimization of the reproduction number with respect to the proportions of commuters in each patch. Finally, Sect. 5 gives a numerical illustration of the results, considering two territories with intrinsic basic reproduction numbers lower and higher than one. We depict the relative sizes of the permanently resident populations that can avoid the outbreak of the epidemic depending on the commuting rates, and discuss the various cases. We end with a conclusion.
2 The Model
We follow the modeling of commuters proposed in Keeling and Rohani (2002) between two patches (such as cities or territories), but here we consider in addition that a part of the population in each patch do not commute (the permanently resident subpopulation). We consider populations of size \(N_i\) whose home belongs to a patch \(i \in \{1,2\}\), structured in three groups:
-
1.
permanently resident, being all the time in patch i, whose population size is denoted \(N_{ir}\),
-
2.
commuters to patch j, but located in patch i at time t, of population size denoted \(N_{ii}\),
-
3.
commuters to patch j and located in patch j at time t, of population size denoted \(N_{ij}\).
We shall denote \(N_{ic}=N_{ii}+N_{ij}\) the size of the total population of commuters with their home in patch i. The individuals commutes to patch j at a rate \(\lambda _i\) with a return rate \(\mu _i\). For each group \(g \in \{ir,ii,ij\}\) we denote by \(S_g\), \(I_g\), \(R_g\) the sizes of susceptible, infected and recovered subpopulations. This modeling implicitly assumes that at any time there is no individual out the territories, that is traveling time is negligible. This assumption is therefore only valid for adjoining territories with short transportation times (by train, road, etc.). It would not be valid between distant territories connected for example by boat or plane with non-negligible crossing times. In this case, it would be necessary to consider additional nodes of in-transit populations, as it has been considered for example in Colizza et al. (2006), Patil et al. (2021) or Ruan et al. (2015) where distance between nodes are explicitly taken into consideration. This would of course complicates the model and its study.
We consider the SIR model assuming that the recovery parameter \(\gamma \) is identical everywhere while the transmission rate \(\beta _i\) depends on the patch i but is identical among each group. Typically lifestyle and hygienic measures may differ between two cities, implying different values of \(\beta \). Moreover, if two cities are on both sides of the border between two countries, the strength of non-pharmaceutical interventions are likely to be different, as is was, for instance, the case between European countries during the SARS-2 outbreak. The model is written as follows (with \(i \ne j\) in \(\{1,2\}\)).
Parameters \(\lambda _i\), \(\mu _i\) represent switching rates of populations i, leaving home and returning. This modeling implicitly assumes that movements between territories are not synchronized, as often considered in multi-city models (see, e.g., Sattenspiel and Dietz 1995; Keeling and Rohani 2002; Arino and van den Driessche 2003; Wang and Mulone 2003; Wang and Zhao 2004; Keeling et al. 2004; Arino and van den Driessche 2006; Takeuchi et al. 2007; Keeling and Rohani 2007; Liu and Stechlinski 2013; Chen et al. 2014). Note that we also consider, in all generality, that commuting is asymmetrical (i.e., \(\lambda _1\) and \(\lambda _2\) may be different, as well as \(\mu _1\), \(\mu _2\)). Typically, each territory may offer different activities that attract commuters from the other territory and thus different mean sojourn times. One can check that the population sizes \(N_{ir}\) and \(N_{ic}\) are constant. Moreover \(N_{ii}\), \(N_{ij}\) fulfill the system of equations
whose solutions verify
We shall assume that populations are already balanced at initial time, i.e., that one has \(N_{ii}={\bar{N}}_{ii}\), \(N_{ij}={\bar{N}}_{ij}\) (constant). For simplicity, we shall drop the notation \(\bar{ }\,\) in the following and denote
which represents the (constant) size of the total population present in patch i.
3 The Epidemic Threshold
We denote the vectors
and consider the state vector
which belongs to the invariant domain
where \({\mathbb {N}}\) is the vector
and \({\mathbb {M}}\) the \(6\times 12\) matrix which consists in the concatenation of the identity matrix \({\mathbb {I}}_6\) of dimension \(6\times 6\)
The disease free equilibrium is defined as
Let \({{\mathcal {R}}}_i\) be the intrinsic reproduction number in the patch i (i.e., when there is no connection between patches), that is
We give now an explicit expression of the epidemic threshold when the two patches communicates via commuters.
Proposition 1
Let
where
Then, one has the following properties.
-
1.
If \({{\mathcal {R}}}_{1,2}>1\), then \(X^\star \) is unstable.
-
2.
If \({{\mathcal {R}}}_{1,2}<1\), then \(X^\star \) is exponentially stable with respect to the variableFootnote 1I.
-
3.
If \({{\mathcal {R}}}_1={{\mathcal {R}}}_2:={{\mathcal {R}}}\), then \({{\mathcal {R}}}_{1,2}={{\mathcal {R}}}\).
Proof
Write the dynamics of X as \(\dot{X}=f(X)\). The Jacobian matrix J of f at \(X^\star \) is of the form
where
and
Note that F is a nonnegative matrix and V is a non-singular M-matrix. We recall [see, for instance, from van den Driessche and Watmough (2002)] that one has the property
The computation of the matrix \(M:=FV^{-1}\) gives the following expression
Let us consider the diagonal matrix
and the matrix \(Q=D^{-1} M D\), whose computation gives the expression
The matrix Q is nonnegative and irreducible. By Perron–Frobenius theorem [see, for instance, Berman and Plemmons (1994)], this matrix admits a unique positive eigenvector (up to a scalar multiplication) that corresponds to the simple (positive) eigenvalue \(\ell =\rho (Q)=\rho (M)\).
Note that the rank of Q is two. We posit
and define \(Q_Y\), \(Q_Z\) the first and third lines, respectively, of the matrix Q. Then, for any vector \(X \in {\mathbb {R}}^6\), one has \(QX=(Q_Y X)Y+(Q_Z X)Z\). We look for an positive eigenvector X of the form \(X=\alpha Y + (1-\alpha ) Z\) with \(\alpha \in (0,1)\). One has then
On the other hand, as X is an eigenvector, one has
The vectors Y and Z being orthogonal, one obtains from (4)–(5) the conditions
Let \(r=\frac{1-\alpha }{\alpha }\). Eliminating \(\ell \) in the two previous equations, r is the positive solution of the polynomial
and \(\ell =Q_Y Y + r Q_Y Z\). One obtains the expression of the eigenvalue
Finally, from the expression of Q, one gets
and thus \(\ell ={{\mathcal {R}}}_{1,2}\), which is exactly \(\rho (M)\).
i. When \({{\mathcal {R}}}_{1,2}>1\), the matrix A has at least one eigenvalue with positive real part and the matrix J as well. The equilibrium \(X^\star \) is thus unstable on \({{\mathcal {D}}}\).
ii. When \({{\mathcal {R}}}_{1,2}<1\), the matrix A is Hurwitz, but \(X^\star \) is not an hyperbolic equilibrium. However, on can write the dynamics of the vector I as an non-autonomous system
Note that this dynamics is cooperative and as for any \(t\ge 0\) one has \(S_{ij}(t)\le N_{ij}\) for \(ij \in \{1r,11,12,2r,22,21\}\), one gets
Therefore, any solution \(I(\cdot )\) of \(\dot{I}=g(t,I)\) with \(I(0)=I_0\ge 0\) verifies \(0\le I(t) \le {\bar{I}}(t)\) for any \(t\ge 0\), where \({\bar{I}}(\cdot )\) is solution of the linear dynamics \(\dot{{\bar{I}}}={\bar{g}}({\bar{I}})\) with \({\bar{I}}(0)=I_0\). As A is Hurwitz, we conclude that \(X^\star \) is exponentially stable with respect to I, which proves point ii.
iii. For the particular case \({{\mathcal {R}}}_1={{\mathcal {R}}}_2:={{\mathcal {R}}}\), the transpose of the matrix M writes
One can check that one has \(M^\top U={{\mathcal {R}}}U\) where \(U=(1,1,1,1,1,1)^\top \). As U is a positive vector, we deduce from the Perron–Frobenius theorem that one has \(\rho (M)=\rho (M^T)=\mathcal{R}\), which ends the proof. \(\square \)
Remark 1
More generally, the next-generation matrix \(M=FV^{-1}\) can be shown to have a rank equal to the number n of patches and that its Perron vector can be expressed as a convex combination of a family of orthogonal vectors in the image of M. This implies that the positive eigenvalue of M (i.e., the reproduction number) is also the positive eigenvalue of the n-dimensional positive matrix given by the decomposition of the image of this vectors by the matrix M.
Alternatively, one may consider the epidemic spread in a virgin population as a Markov process, to determine the expected numbers of secondary cases in each patch, and obtain this \(n\times n\) matrix, as described in Diekmann et al. (2013). This method consists in a first-step analysis by determining the mean residence times of an infected individual of each group in each of the patches. Then, for a given patch the expected numbers of new infected present in each path are given by the products of the mean residence times by the transmission rate, averaged by the constant distribution given in (1).
This explains why the formula (2) takes the expression of a root of the characteristic polynomial of a 2 by 2 matrix.
Remark 2
The explicit expression (2) of the epidemic threshold given in Proposition 1 is also relevant in absence of permanently resident populations, which has not been yet provided explicitly in the literature (up to our knowledge).
Corollary 2
One has
Proof
Denote by \(M({{\mathcal {R}}}_1,{{\mathcal {R}}}_2)\) the matrix \(FV^{-1}\) for the parameters \({{\mathcal {R}}}_1\), \({{\mathcal {R}}}_2\), and let \(\mathcal{R}_-:=\min \left( {{\mathcal {R}}}_1,{{\mathcal {R}}}_2\right) \), \(\mathcal{R}_+:=\max \left( {{\mathcal {R}}}_1,{{\mathcal {R}}}_2\right) \). From the expression of the nonnegative matrices M, one gets
which implies [see, for instance, Berman and Plemmons (1994)] the inequalities
and thus
\(\square \)
Alternatively, the number \({{\mathcal {R}}}_{1,2}\) can be determined as follows.
Corollary 3
Assume \({{\mathcal {R}}}_2>{{\mathcal {R}}}_1\). Then, one has
where \(\alpha \in [0,1)\) is the smallest root of the polynomial
Proof
One can check, from expressions (3), that one has \(q_{11}+q_{21}={{\mathcal {R}}}_1\) and \(q_{22}+q_{12}={{\mathcal {R}}}_2\). Then, from (6), one get
where \(\alpha \) is a root of the polynomial P obtained from (6) by eliminating l, that is
From Corollary 2, we know that \(\alpha \) belongs to [0, 1].
Note that one has \(P(0)=q_{12}\ge 0\) and \(P(1)=-q_{21}\le 0\). Therefore, when \({{\mathcal {R}}}_2-{{\mathcal {R}}}_1>0\), P admits exactly one root in [0, 1) and another one in \([1,\rightarrow )\). However, if \(\alpha =1\) one should have \(q_{21}=0\) and thus \(\lambda _1=0\), \(\mu _2=0\), which implies \(N_{11}=N_{1c}\), \(N_{12}=0\), \(N_{22}=0\), \(N_{21}=N_{2c}\). Then, one obtains \(q_{11}={{\mathcal {R}}}_1\), \(q_{22}={{\mathcal {R}}}_2\) and from the expression (2) on gets \({{\mathcal {R}}}_{1,2}=\max (\mathcal{R}_1,{{\mathcal {R}}}_2)={{\mathcal {R}}}_2\) which contradicts \(\alpha =1\). We conclude that \(\alpha \) belongs to [0, 1) and is thus the smallest root of P. \(\square \)
Remark 3
When there is no communication between patches (that is \(N_{1r}=N_{1p}=N_1\), \(N_{2r}=N_{2p}=N_2\)), one has \(q_{21}=0\) and \(q_{12}=0\). If \({{\mathcal {R}}}_2>{{\mathcal {R}}}_1\), resp. \({{\mathcal {R}}}_1>\mathcal{R}_2\), one has \(\alpha =0\), resp. \(\alpha =1\), which gives
We look now for a characterization of the minimum value of the threshold \({{\mathcal {R}}}_{1,2}\).
4 Minimization of the Epidemic Threshold
In this section, we assume that the mixing is fast compared to the recovery rate (as its is often considered in the literature), which amounts to have numbers \(\lambda _i\), \(\mu _i\) large compared to \(\gamma \). Our objective is to study how the proportions of commuters in the populations impact the value of \({{\mathcal {R}}}_{1,2}\).
Given \({{\mathcal {R}}}_1\), \({{\mathcal {R}}}_2\), we consider the approximation \(\tilde{{\mathcal {R}}}_{1,2}\) of the threshold \({{\mathcal {R}}}_{1,2}\) which consists in keeping \(\gamma =0\) in the expressions (3). For convenience, we posit the numbers
One has a first result about the variations of \(\tilde{\mathcal{R}}_{1,2}\) with respect to \(N_{1c}\), \(N_{2c}\).
Proposition 4
Fix parameters \(N_i\), \(\beta _i\), \(\gamma \), \(\lambda _i\), \(\mu _i\) (\(i=1,2\)) such that \({{\mathcal {R}}}_2>{{\mathcal {R}}}_1\).
-
1.
For any \(N_{1c} \in (0,N_1)\), the map \(N_{2c} \mapsto \tilde{{\mathcal {R}}}_{1,2}(N_{1c},N_{2c})\) is decreasing.
-
2.
The map \(N_{1c} \mapsto \tilde{{\mathcal {R}}}_{1,2}(N_{1c},N_{2c})\) is increasing at \((N_{1c},N_{2c})\) when
$$\begin{aligned} \eta _2(1-\eta _2)N_{2c} > (1-\eta _1)(N_2-\eta _2N_{2c}) \end{aligned}$$(9) -
3.
The map \(N_{1c} \mapsto \tilde{{\mathcal {R}}}_{1,2}(N_{1c},N_{2c})\) is increasing, resp. decreasing, at \((N_{1c},N_{2c})\) if the numbers A and B are negative, resp. positive, where
$$\begin{aligned}&A:={{\mathcal {R}}}_2 \frac{\frac{N_2}{2}-\eta _1(\frac{1}{2}-\eta _1)N_{1c}-(\frac{3}{2}-\eta _2)\eta _2N_{2c}}{N_2-\eta _2N_{2c}+\eta _1N_{1c}}\\&\qquad -{{\mathcal {R}}}_1 \frac{\frac{N_1}{2}-(\frac{3}{2}-\eta _1)\eta _1N_{1c}-\eta _2(\frac{1}{2}-\eta _2)N_{2c}}{N_1-\eta _1N_{1c}+\eta _2N_{2c}} , \\&B := {{\mathcal {R}}}_2 \frac{(1-\eta _1)(N_2-\eta _2N_{2c})-\eta _2(1-\eta _2)N_{2c}}{(N_2-\eta _2N_{2c}+\eta _1N_{1c})^2}\\&\qquad -\mathcal{R}_1\frac{(1-\eta _1)(N_1+\eta _2N_{2c})+\eta _2(1-\eta _2)N_{2c}}{(N_1-\eta _1N_{1c}+\eta _2N_{2c})^2} \end{aligned}$$
Proof
Following Corollary 3, one has
where \({\tilde{\alpha }}\) is the smallest root of the polynomial
where \({\tilde{q}}_{12}\), \({\tilde{q}}_{21}\) are the approximations of \(q_{12}\), \(q_{21}\) defined in (3). Let us note that one can write \(N_{ii}=(1-\eta _i)N_{ic}\), \(N_{ij}=\eta _i N_{ic}\) (for \(j \ne i\)) and also \(N_{ip}=N_i-\eta _i N_{ic}+\eta _j N_{jc}\), which leads to the following expressions of \({\tilde{q}}_{12}\), \({\tilde{q}}_{21}\)
For simplicity, we shall drop the notation \(\tilde{ }\;\) in the rest of the proof. Note than \(\alpha \) being the smallest root of P, it verifies
Let us differentiate the equality \(P(\alpha )=0\) with respect to \(q_{12}\) and \(q_{21}\):
which gives
Then, one can write
and from inequality (12), we obtain that the signs of the derivatives \(\frac{\partial \alpha }{\partial N_{ic}}\) are given by the sign of the numbers
We begin by the dependency with respect to \(N_{2c}\). One has first
Note that one has
and thus
Then, one gets the inequality
On another hand, one gets from \(P(\alpha )=0\) the inequality
and with (14)
We then conclude that \(\sigma _2\) is positive, and from (10) we deduce that the map \(N_{2c} \mapsto \mathcal{R}_{1,2}\) is decreasing. This proves the point i.
We study now the dependency with respect to \(N_{1c}\). A calculation of the partial derivative gives
and
When \(\frac{\partial q_{12}}{\partial N_{1c}}<0\), we can conclude that \(\sigma _1\) is negative and \( {{\mathcal {R}}}_{1,2}\) is thus increasing with respect to \(N_{1c}\). This condition is equivalent to (9). This proves the point ii. When this last condition is not satisfied, having \(\frac{\partial q_{12}}{\partial N_{1c}}<\frac{\partial q_{21}}{\partial N_{1c}}\) with \(\alpha >\frac{1}{2}\) is another sufficient condition to obtain \(\sigma _1<0\) from expression (13). However, having \(\alpha >\frac{1}{2}\) amounts to have \(P(\frac{1}{2})>0\), that is
or equivalently
One can check that this last condition is equivalent to \(A<0\) and that the condition \(\frac{\partial q_{12}}{\partial N_{1c}}<\frac{\partial q_{21}}{\partial N_{1c}}\) is equivalent to \(B<0\). In the same manner, having \(A>0\) and \(B>0\) implies \(\alpha <\frac{1}{2}\) and \(\frac{\partial q_{12}}{\partial N_{1c}}>\frac{\partial q_{21}}{\partial N_{1c}}\), which is a sufficient condition to have \(\sigma _1>0\), and thus \( \mathcal{R}_{1,2}\) increasing with respect to \(N_{1c}\). This proves the point iii. \(\square \)
This result suggests that the map \(N_{1c} \mapsto \tilde{\mathcal{R}}_{1,2}(N_{1c},N_{2c})\) is not necessarily monotonic, differently to the map \(N_{2c} \mapsto \tilde{{\mathcal {R}}}_{1,2}(N_{1c},N_{2c})\). We show now that the possibilities of its variations are limited.
Proposition 5
Under hypotheses of Proposition 4, for each \(N_{2c} \in (0,N_2)\) the map \(N_{1c} \mapsto \tilde{{\mathcal {R}}}_{1,2}(N_{1c},N_{2c})\) possesses one of the three properties
-
1.
it is decreasing on \((0,N_1)\),
-
2.
it is increasing on \((0,N_1)\),
-
3.
there exists \(N_{1c}^\star \in (0,N_1)\) such that it is decreasing on \((0,N_{1c}^\star )\) and increasing on \((N_{1c}^\star ,N_1)\).
Proof
Fix \(N_{2c} \in (0,N_2)\). If the map \(N_{1c} \mapsto \tilde{\mathcal{R}}_{1,2}(N_{1c},N_{2c})\) is not monotonic, there exists \({\hat{N}}_{1c} \in (0,N_1)\) such that \(\frac{\partial \tilde{\mathcal{R}}_{1,2}}{\partial N_{1c}}({\hat{N}}_{1c},N_{2c})=0\). For simplicity, we shall drop the notation \(\tilde{ }\;\) in the rest of the proof. Following the proof of Proposition 4, one has \(\mathcal{R}_{1,2}=\alpha {{\mathcal {R}}}_1 + (1-\alpha ){{\mathcal {R}}}_2\) with
where \(\nu >0\). Therefore, one has \(\frac{\partial \alpha }{\partial N_{1c}}=0\) and \(\sigma _1=0\) at \(N_{1c}={\hat{N}}_{1c}\), and thus
From expressions (15) and (16), a calculation of the partial derivatives gives
where \(\frac{\partial q_{21}}{\partial N_{1c}}>0\) and from \(\sigma _1=0\) one gets \(\frac{\partial q_{12}}{\partial N_{1c}}>0\) for \(N_{1c}={\hat{N}}_{1c}\). Finally, one obtains
Consequently, any extremum of the map \(N_{1c} \mapsto \mathcal{R}_{1,2}(N_{1c},N_{2c})\) is a local minimizer, which implies that this map has at most one local minimizer. \(\square \)
Finally, we give conditions for which the minimization of the threshold \({{\mathcal {R}}}_{1,2}\) presents a trichotomy.
Proposition 6
Let parameters \(\beta _i\), \(\gamma \) be such that \({{\mathcal {R}}}_2>{{\mathcal {R}}}_1\) and assume that \(N_1\), \(N_2\) satisfy \(N_1{{\mathcal {R}}}_2>N_2{{\mathcal {R}}}_1\). Then, provided that \(\gamma \) is small enough compared to \(\lambda _i\) and \(\mu _i\), the function \((N_{1c},N_{2c}) \mapsto {{\mathcal {R}}}_{1,2}(N_{1c},N_{2c})\) admits an unique minimum at \((N_{1c}^\star ,N_{2c}^\star )\) with \(N_{2c}^\star =N_2\). Moreover, one has the following properties.
-
1.
\(N_{1c}^\star =0\) if \(\eta _2 > 1-\eta _1\),
-
2.
\(N_{1c}^\star =N_1\) if \(\eta _1\) and \(\eta _2\) are sufficiently small,
-
3.
there exists \(\eta _1\), \(\eta _2\) for which \(N_{1c}^\star \in (0,N_1)\).
Proof
We first show that the announced properties are satisfied for the approximate function \(\tilde{{\mathcal {R}}}_{1,2}\).
From Propositions 4 and 5 , we know that \(\tilde{{\mathcal {R}}}_{1,2}\) admits an unique minimum at \(({\hat{N}}_{1c},{\hat{N}}_{2c})\) with \({\hat{N}}_{2c}=N_2\). For \(N_{2c}=N_2\), the condition (9) simply writes \(\eta _2 > 1-\eta _1\) which implies from point ii. of Proposition 4 that one has \({\hat{N}}_{1c}=0\) when this condition is fulfilled. This shows that point 1 is verified for the function \(\tilde{{\mathcal {R}}}_{1,2}\).
One obtains the limits
which show that numbers A and B are positive when \(\eta _1\), \(\eta _2\) are small, and thus one has \({\hat{N}}_{1c}=N_1\) from point iii of Proposition 4. This shows that point 2 is verified for the function \(\tilde{{\mathcal {R}}}_{1,2}\).
Take now any \(N_{1c} \in (0,N_1)\). When \(\eta _2 > 1-\eta _1\), one has \(\frac{\partial \tilde{{\mathcal {R}}}_{1,2}}{\partial N_{1c}}(N_{1c},N_2)>0\), and for \(\eta _1\), \(\eta _2\) small, \(\frac{\partial \tilde{{\mathcal {R}}}_{1,2}}{\partial N_{1c}}(N_{1c},N_2)<0\) is verified. Then, by continuity of the function \(\tilde{{\mathcal {R}}}_{1,2}\) with respect to parameters \(\eta _1\), \(\eta _2\), one deduce that the existence of values \(\hat{\eta }_1\), \({\hat{\eta }}_2\) for which \(\frac{\partial \tilde{\mathcal{R}}_{1,2}}{\partial N_1}(N_{1c},N_2)=0\). As the function \(\tilde{{\mathcal {R}}}_{1,2}\) cannot have more than a local extremum (see Proposition 5), we deduce that \(N_{1c}\) realizes the minimum of the function \(N_{1c} \mapsto \tilde{\mathcal{R}}_{1,2}(N_{1c},N_2)\) when \(\eta _1={\hat{\eta }}_1\) and \(\eta _2=\hat{\eta }_2\). This shows that point 3 is verified for the function \(\tilde{{\mathcal {R}}}_{1,2}\).
Finally, note that the exact threshold \({{\mathcal {R}}}_{1,2}\) amounts to replace in the expression of \({\tilde{q}}_{12}\), \({\tilde{q}}_{21}\) the numbers \(\eta _i\) by \(\frac{\lambda _i+\gamma }{\lambda _i+\mu _i+\gamma }\) , which is continuous with respect to \(\gamma \) and equal to \(\eta _i\) for \(\gamma =0\). By continuity of \(\tilde{{\mathcal {R}}}_{1,2}\) with respect to \({\tilde{q}}_{12}\), \({\tilde{q}}_{21}\) , we deduce that uniqueness of the minimizer of \({{\mathcal {R}}}_{1,2}\) and properties 1. to 3. are also fulfilled by the function \((N_{1c},N_{2c})\mapsto {{\mathcal {R}}}_{1,2}\), provided that \(\gamma \) is small enough. \(\square \)
5 Numerical Illustration
We consider two territories of same population size \(N=N_1=N_2\) with different transmission rates such that one has \({{\mathcal {R}}}_1< 1 < {{\mathcal {R}}}_2\) (values are given in Table 1). Typically, some precautionary measures (such as social distance) are taken in the first territory so that the disease cannot spread in this territory if it is closed, while the epidemic can spread in the second territory in absence of communication with territory 1. We aim at studying how the epidemic can die out when commuting occur between territories, depending on the proportions of resident in each population, denoted
(in other words, how to obtain \({{\mathcal {R}}}_{1,2}<1\) playing with \(p_1\), \(p_2\)). Note that when \(N_1=N_2\), the threshold \(\mathcal{R}_{1,2}\) depends on the proportions \(p_1\), \(p_2\) independently of N.
Conditions of Proposition 6 are satisfied provided that commuting parameters \(\lambda _i\), \(\mu _i\) are large enough. We have considered three sets of these parameters, given in Table 2, that correspond to the three possible situations depicted in Proposition 6.
The approximate expression \(\tilde{{\mathcal {R}}}_{1,2}\) turns out to be a very good approximation of the exact value \({{\mathcal {R}}}_{1,2}\), even in case A for which \(\gamma \) is not so small compared to \(\mu _2\) (see Table 3).
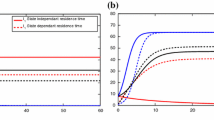
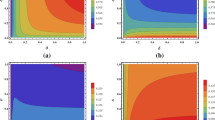
Figures 1, 2 and 3 show families of curves \(p_1 \mapsto {{\mathcal {R}}}_{1,2}\) for different values of \(p_2 \in [0,1]\). One can observe that theses curves possess the properties given by Propositions 4 and 5:
-
they are either decreasing, increasing or decreasing down to a minimum and then increasing,
-
they are ordered and the lower one is obtained for \(p_2=0\) (i.e., \(N_{2c}=N_2\)).
This last feature is intuitive: the more there are commuters from territory 2 (that spend time in territory 1 where the conditions of transmission disease is lower), the less the epidemic spreads. A way to reduce the value of \({{\mathcal {R}}}_{1,2}\) is thus to encourage commuting toward territory 1 (whatever are the commuting rates). However, the role of the resident population in territory 1 is far less intuitive because it does depends on the commuting rates.
-
1.
In case A, commuters from territory 2 return more rarely to home than commuters from territory 1 do. The condition of point 1 of Proposition 6 is fulfilled. Then, the threshold \({{\mathcal {R}}}_{1,2}\) can be made small (and below 1) when the proportion of resident in territory 1 is high, i.e., when the inhabitants of territory 1 are encouraged not to commute.
-
2.
In case B, both commuters return rapidly to their home. This means that the numbers of commuters from one territory present in the other one at a given time is low. Then the condition of point 2 of Proposition 6 is fulfilled. Here, it is better to encourage inhabitants of territory 1 to commute to the other territory where the disease spreads yet more easily which is counterintuitive at first sight. Indeed, commuters do not spend much time in the other territory and therefore heuristically have less time to encounter and transmit the disease...
-
3.
In case C, commuters from territory 2 return more rapidly to home than commuters from territory 1 do, on the opposite of case A. Conditions of points 1 and 2 of Proposition 6 are not fulfilled here, and we are in an intermediate situation for which point 3 of Proposition 6 occurs. It is theoretically possible to have \({{\mathcal {R}}}_{1,2}<1\) on the condition that the proportion of commuters of territory 1 is well balanced.
Finally, this example shows that changing only the return rates \(\mu _1\), \(\mu _2\) allows to obtain the three possible scenarios, but other changes could also exhibit them.
6 Conclusion
In this work, we have been able to provide an explicit expression of the reproduction number, although the model is in dimension 18. This expression has allowed us to study its minimization with respect to the proportions of permanently resident populations in each patch. We discovered a trichotomy of cases, with some counterintuitive situations. In each case, it is always beneficial to have commuters traveling to a safer city where the transmission rate is lower. However, for the safer city, three situations occur:
-
either it is better to avoid commuting to the other city,
-
or on the opposite encouraging commuting to the more risky city reduces the reproduction number,
-
and in a third case there exists an optimal intermediate proportion of commuters of the safer city which minimizes the epidemic threshold.
In some sense, the permanently resident populations, which have been ignored in former modeling, can play an hidden role in an epidemic outbreak. This is illustrated on an example for which only right proportions of commuters (or permanently resident) avoid the outbreak. This suggests that counterintuitive situations may also occur when considering networks with more than two nodes. The present study focuses on the reproduction number and how it can be reduced. The impacts of resident proportions on other epidemiological characteristics, such as the peak level or the finite size, may be the matter a future work. The extension of the present results to more general networks is also a future perspective.
Change history
04 January 2023
Tables 1,2,3 and Figure 1 placement were corrected. It has been placed in the appropriate place.
Notes
We refer to Vorotnikov (1998) for the definition of partial stability.
References
Anderson RM, May RM (1991) Infectious diseases of humans. Oxford University, Oxford
Angulo JJ, Takiguti CK, Pederneiras CA, Carvalho-de-Souza AM, Oliveira-de-Souza MC, Megale P (1979) Identification of pattern and process in the spread of a contagious disease. Soc Sci Med 13D(3):183–9
Arino J (2009) Diseases in metapopulations. In: Modeling and dynamics of infectious diseases. World Scientific, pp 64–122
Arino J, van den Driessche P (2003) A multi-city epidemic model. Math Popul Stud 10(3):175–193
Arino J, Jordan R, van den Driessche P (2007) Quarantine in a multi-species epidemic model with spatial dynamics. Math Biosci 206(1):46–60
Berman A, Plemmons RJ (1994) Nonnegative matrices in the mathematical sciences. SIAM, Philadelphia
Brunham R (1997) Core group theory: a central concept in STD epidemiology. Venereology 10(1):34–35, 38–39
Chen Y, Yan M, Xiang Z (2014) Transmission dynamics of a two-city SIR epidemic model with transport-related infections. J Appl Math ID 764278
Citron DT, Guerra CA, Dolgert AJ, Wu SL, Henry JM, Sánchez CHM, Smith DL (2021) Comparing metapopulation dynamics of infectious diseases under different models of human movement. Proc Natl Acad Sci 118(18):e2007488118
Clancy D (1996) Carrier-borne epidemic models incorporating population mobility. Math Biosci 132(2):185–204
Colizza V, Barrat A, Barthelemy M, Vespignani A (2006) The role of the airline transportation network in the prediction and predictability of global epidemics. Proc Natl Acad Sci USA 103:2015–2020
Dhirasakdanon T, Thieme HR, Van Den Driessche P (2007) A sharp threshold for disease persistence in host metapopulations. J Biol Dyn 1(4):363–378
Diekmann O, Heesterbeek JAP, Metz JAJ (1990) On the definition and computation of the basic reproduction ratio R0 in models for infectious diseases in heterogeneous populations. J Math Biol 28:365–382
Diekmann O, Heesterbeek JAP, Robert MG (2007) The construction of next-generation matrices for compartmental epidemic models. J R Soc Interface 7:873–885
Diekmann O, Heesterbeek H, Britton T (2013) Mathematical tools for understanding infectious disease dynamics. Princeton University Press, Princeton
Gao D (2007) How does dispersal affect the infection size? SIAM J Appl Math 80:2144–2169
Guo H, Li M, Shuai Z (2006) Global stability of the endemic equilibrium of multigroup SIR epidemic models. Can Appl Math Q 14(3):259–284
Hadeler KP, Castillo-Chavez C (1995) A core group model for disease transmission. Math Biosci 128(1–2):41–55
Hanski I (1999) Metapopulation ecology. Oxford University Press, Oxford
Iggidr A, Sallet G, Tsanou B (2012) Global stability analysis of a metapopulation SIS epidemic model. Math Popul Stud 19(3):115–129
Keeling MJ (2007) Modeling infectious diseases in humans and animals. Princeton University Press, Princeton
Keeling MJ, Rohani P (2002) Estimating spatial coupling in epidemiological systems: a mechanistic approach. Ecol Lett 5:20–29
Keeling MJ, Bjørnstad ON, Grenfell BT (2004) Metapopulation dynamics of infectious diseases. In: Hanski I, Gaggiotti O (eds) Ecology, genetics, and evolution of metapopulations, chapter 17. Elsevier, Amsterdam, pp 415–445
Kelly MR, Tien JH, Eisenberg MC, Lenhart S (2016) The impact of spatial arrangements on epidemic disease dynamics and intervention strategies. J Biol Dyn 10:222–249
Kermack W, McKendrick A (1927) A contribution to the mathematical theory of epidemics. Proc R Soc A 115:700–721
Knipl D (2016) A new approach for designing disease intervention strategies in metapopulation models. J Biol Dyn 10(1):71–94
Li J, Xiang T, He L (2021) Modeling epidemic spread in transportation networks: a review. J Traffic Transp Eng 8(2):139–152
Lipshtat A, Alimi R, Ben-Horin Y (2021) Commuting in metapopulation epidemic modeling. Sci Rep 11:15198
Liu X, Stechlinski P (2013) Transmission dynamics of a switched multi-city model with transport-related infections. Nonlinear Anal Real World Appl 14(1):264–279
MacArthur RH, Wilson EO (2001) The theory of island biogeography. Princeton University Press, Princeton
Mpolya EA, Yashima K, Ohtsuki H, Sasaki A (2014) Epidemic dynamics of a vector-borne disease on a villages-and-city star network with commuters. J Theor Biol 343:120–26
Murray JD (2003) Mathematical biology II: spatial models and biomedical applications. Springer, New York
Patil R, Dave R, Patel H, Shah VM, Chakrabarti D, Bhatia U (2021) Assessing the interplay between travel patterns and SARS-CoV-2 outbreak in realistic urban setting. Appl Netw Sci 6:4
Ruan Z, Wang C, Ming Hui P, Liu Z (2015) Integrated travel network model for studying epidemics: interplay between journeys and epidemic. Sci Rep 5:11401
Sattenspiel L, Dietz K (1995) A structured epidemic model incorporating geographic mobility among regions. Math Biosci 128(1–2):71–91
Takeuchi Y, Liu X, Cui J (2007) Global dynamics of SIS models with transport-related infection. J Math Anal Appl 329(2):1460–71
Tocto-Erazo MR, Olmos-Liceaga D, Montoya-Laos JA (2021) Effect of daily human movement on some characteristics of dengue dynamics. Math Biosci 332:108531
van den Arino J, Driessche P (2006) Disease spread in metapopulations. Fields Inst Commun 48:1–13
van den Driessche P, Watmough J (2002) Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math Biosci 180(1–2):29–48
Vorotnikov VI (1998) Partial stability and control. Birkhäuser, Basel
Wang W, Mulone G (2003) Threshold of disease transmission in a patch environment. J Math Anal Appl 285(1):321–335
Wang W, Zhao XQ (2004) An epidemic model in a patchy environment. Math Biosci 190(1):97–112
Yin Q, Wang Z, Xia C, Dehmer M, Emmert-Streib F, Jin Z (2020) A novel epidemic model considering demographics and intercity commuting on complex dynamical networks. Appl Math Comput 386, N. 125517
Acknowledgements
The authors are grateful for the support of the French platform MODCOV19, and the Algerian Government for the PhD grant of Ismail Mimouni. The authors thank the anonymous referee to let us know the alternative approach to obtain the expression of the reproduction number, mentioned in Remark 1.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rapaport, A., Mimouni, I. The Role of Permanently Resident Populations in the Two-Patches SIR Model with Commuters. Bull Math Biol 85, 3 (2023). https://doi.org/10.1007/s11538-022-01111-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11538-022-01111-6