Abstract
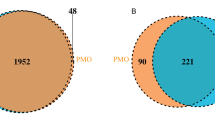
Few studies have focused on the effects of multiple metal mixtures on bone health and the underlying mechanisms related to alterations in the gut microbiota. This study aimed to examine the potential roles of gut microbiota alterations in metal mixtures and their association with osteoporosis traits. Adults aged ≥ 55 years were recruited from two community healthcare centers in Wuhan City during 2016–2019. The plasma concentrations of six metals (zinc, iron, selenium, lead, cadmium, and arsenic) were measured using an inductively coupled plasma mass spectrometer. The k-means clustering method was employed to explore the exposure profiles of metal mixtures for all participants. 16S rRNA gene sequencing was used to profile the gut microbiota of participants. Combining these results with those of our previous study, we identified overlapping taxa and evaluated their potential roles. A total of 806 participants (516 females), with an average age of 67.36 years were included. The participants were grouped into three clusters using k-means clustering: Cluster 1 (n = 458), Cluster 2 (n = 199), and Cluster 3 (n = 149). The high-exposure group for iron, zinc, lead, and cadmium (Cluster 3) showed a negative association with lumbar spine 1–4 bone mineral density (BMD). A total of 201 individuals (121 females) underwent sequencing of the gut microbiota. Both alpha and beta diversities were statistically different among the three groups. Bacteroidaceae, Lachnospiraceae, Bifidobacteriaceae, Bacteroides, and Lachnospiraceae_incertae_sedis were identified as overlapping taxa associated with the metal mixtures and BMD. Interaction analysis revealed that Cluster 3 interacted with Bacteroidaceae/Bacteroides, resulting in a positive effect on LS1-4 BMD (β = 0.358 g/cm2, 95% CI: 0.047 to 0.669, P = 0.025). Our findings indicate associations between multiple metal mixtures and BMD as well as gut microbiota alterations. Exploring the interaction between metal mixtures and the gut microbiota provides new perspectives for the precise prevention and treatment of osteoporosis.






Similar content being viewed by others
Data Availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request. The data are not publicly available due to privacy or ethical restrictions.
References
Al EAA, Parsian H, Fathi M et al (2018) ALOX12 gene polymorphisms and serum selenium status in elderly osteoporotic patients. Adv Clin Exp Med 27:1717–1722. https://doi.org/10.17219/acem/75689
Anam AK, Insogna K (2021) Update on osteoporosis screening and management. Med Clin North Am 105:1117–1134. https://doi.org/10.1016/j.mcna.2021.05.016
Anttila MA, Tammi RH, Tammi MI et al (2000) High levels of stromal hyaluronan predict poor disease outcome in epithelial ovarian cancer. Cancer Res 60(1):150–155
Arikan DC, Coskun A, Ozer A et al (2011) Plasma selenium, zinc, copper and lipid levels in postmenopausal Turkish women and their relation with osteoporosis. Biol Trace Elem Res 144:407–417. https://doi.org/10.1007/s12011-011-9109-7
Auvinen P, Tammi R, Parkkinen J et al (2000) Hyaluronan in peritumoral stroma and malignant cells associates with breast cancer spreading and predicts survival. Am J Pathol 156(2):529–536. https://doi.org/10.1016/S0002-9440(10)64757-8
Brabec JL, Wright J, Ly T et al (2020) Arsenic disturbs the gut microbiome of individuals in a disadvantaged community in Nepal. Heliyon 6:e03313. https://doi.org/10.1016/j.heliyon.2020.e03313
Buha A, Jugdaohsingh R, Matovic V et al (2019) Bone mineral health is sensitively related to environmental cadmium exposure- experimental and human data. Environ Res 176:108539. https://doi.org/10.1016/j.envres.2019.108539
Callan AC, Devine A, Qi L et al (2015) Investigation of the relationship between low environmental exposure to metals and bone mineral density, bone resorption and renal function. Int J Hyg Environ Health 218:444–451. https://doi.org/10.1016/j.ijheh.2015.03.010
Cao JJ, Singleton PA, Majumdar S et al (2005) Hyaluronan increases RANKL expression in bone marrow stromal cells through CD44. J Bone Miner Res 20(1):30–40. https://doi.org/10.1359/JBMR.041014
Carpenter DO, Arcaro K, Spink DC (2002) Understanding the human health effects of chemical mixtures. Environ Health Perspect 110(Suppl 1):25–42. https://doi.org/10.1289/ehp.02110s125
Cerovic A, Miletic I, Sobajic S et al (2007) Effects of zinc on the mineralization of bone nodules from human osteoblast-like cells. Biol Trace Elem Res 116:61–71. https://doi.org/10.1007/BF02685919
Chen QY, DesMarais T, Costa M (2019) Metals and mechanisms of carcinogenesis. Annu Rev Pharmacol Toxicol 59:537–554. https://doi.org/10.1146/annurev-pharmtox-010818-021031
Chi X, Liu Z, Wang H et al (2022) Regulation of a new type of selenium-rich royal jelly on gut microbiota profile in mice. Biol Trace Elem Res 200(4):1763–1775. https://doi.org/10.1007/s12011-021-02800-4
Ciosek Z, Kot K, Kosik-Bogacka D et al (2021) The effects of calcium, magnesium, phosphorus, fluoride, and lead on bone tissue. Biomolecules 11(4):506. https://doi.org/10.3390/biom11040506
Compston JE, McClung MR, Leslie WD (2019) Osteoporosis. Lancet 393:364–376. https://doi.org/10.1016/S0140-6736(18)32112-3
Duan H, Yu L, Tian F et al (2020) Gut microbiota: A target for heavy metal toxicity and a probiotic protective strategy. Sci Total Environ 742:140429. https://doi.org/10.1016/j.scitotenv.2020.140429
Dubey AK, Gupta U, Jain S (2016) Analysis of k-means clustering approach on the breast cancer Wisconsin dataset. Int J Comput Assist Radiol Surg 11:2033–2047. https://doi.org/10.1007/s11548-016-1437-9
Eggers S, Safdar N, Sethi AK et al (2019) Urinary lead concentration and composition of the adult gut microbiota in a cross-sectional population-based sample. Environ Int 133:105122. https://doi.org/10.1016/j.envint.2019.105122
Ensrud KE, Crandall CJ (2017) Osteoporosis. Ann Intern Med 167:Itc17-itc32. https://doi.org/10.7326/AITC201708010
Gaffney-Stomberg E (2019) The impact of trace minerals on bone metabolism. Biol Trace Elem Res 188:26–34. https://doi.org/10.1007/s12011-018-1583-8
Galvez-Fernandez M, Grau-Perez M, Garcia-Barrera T et al (2021) Arsenic, cadmium, and selenium exposures and bone mineral density-related endpoints: The HORTEGA study. Free Radic Biol Med 162:392–400. https://doi.org/10.1016/j.freeradbiomed.2020.10.318
Gaulke CA, Rolshoven J, Wong CP et al (2018) Marginal zinc deficiency and environmentally relevant concentrations of arsenic elicit combined effects on the gut microbiome. mSphere 3(6):e00521-18. https://doi.org/10.1128/mSphere.00521-18
He J, Xu S, Zhang B et al (2020) Gut microbiota and metabolite alterations associated with reduced bone mineral density or bone metabolic indexes in postmenopausal osteoporosis. Aging (Albany NY) 12:8583–8604. https://doi.org/10.18632/aging.103168
Inskip MJ, Franklin CA, Subramanian KS et al (1992) Sampling of cortical and trabecular bone for lead analysis: method development in a study of lead mobilization during pregnancy. Neurotoxicology 13(4):825–834
Jung TH, Han KS (2021) Imbalanced dietary intake alters the colonic microbial profile in growing rats. PLoS One 16(6):e0253959. https://doi.org/10.1371/journal.pone.0253959
Laue HE, Moroishi Y, Jackson BP et al (2020) Nutrient-toxic element mixtures and the early postnatal gut microbiome in a United States longitudinal birth cohort. Environ Int 138:105613. https://doi.org/10.1016/j.envint.2020.105613
Li C, Huang Q, Yang R et al (2019) Gut microbiota composition and bone mineral loss-epidemiologic evidence from individuals in Wuhan, China. Osteoporos Int 30:1003–1013. https://doi.org/10.1007/s00198-019-04855-5
Li GH, Robinson-Cohen C, Sahni S et al (2020) Association of genetic variants related to serum calcium levels with reduced bone mineral density. J Clin Endocrinol Metab 105(3):e328–e336. https://doi.org/10.1210/clinem/dgz088
Li T, Xie Y, Wang L et al (2022a) The Association between lead exposure and bone mineral density in childhood and ddolescence: results from NHANES 1999–2006 and 2011–2018. Nutrients 4(7):1523. https://doi.org/10.3390/nu14071523
Li LP, Qu L, Li T (2022b) Supplemental dietary Selenohomolanthionine affects growth and rumen bacterial population of Shaanbei white cashmere wether goats. Front Microbiol 13:942848. https://doi.org/10.3389/fmicb.2022.942848
Lin SL, Chang D, Ying SY (2007) Hyaluronan stimulates transformation of androgen-independent prostate cancer. Carcinogenesis 28(2):310–320. https://doi.org/10.1093/carcin/bgl134
Liu Y, Li Y, Xia Y et al (2020) The dysbiosis of gut microbiota caused by low-dose cadmium aggravate the injury of mice liver through increasing intestinal permeability. Microorganisms 8(2):211. https://doi.org/10.3390/microorganisms8020211
Lu K, Abo RP, Schlieper KA et al (2014) Arsenic exposure perturbs the gut microbiome and its metabolic profile in mice: an integrated metagenomics and metabolomics analysis. Environ Health Perspect 122:284–291. https://doi.org/10.1289/ehp.1307429
Ma Y, Ran D, Shi X et al (2021) Cadmium toxicity: A role in bone cell function and teeth development. Sci Total Environ 769:144646. https://doi.org/10.1016/j.scitotenv.2020.144646
Magoč T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27(21):2957–2963. https://doi.org/10.1093/bioinformatics/btr507
Messer JG, Kilbarger AK, Erikson KM et al (2009) Iron overload alters iron-regulatory genes and proteins, down-regulates osteoblastic phenotype, and is associated with apoptosis in fetal rat calvaria cultures. Bone 45:972–979. https://doi.org/10.1016/j.bone.2009.07.073
Nedyalkova M, Madurga S, Simeonov V (2021) Combinatorial k-means clustering as a machine learning tool applied to diabetes mellitus type 2. Int J Environ Res Public Health 18:1919. https://doi.org/10.3390/ijerph18041919
Osteoporosis prevention, diagnosis, and therapy (2001) JAMA 285: 785–795. https://doi.org/10.1001/jama.285.6.785
Parvaneh K, Ebrahimi M, Sabran MR et al (2015) Probiotics (Bifidobacterium longum) increase bone mass density and upregulate sparc and Bmp-2 genes in rats with bone loss resulting from ovariectomy. Biomed Res Int 2015:897639. https://doi.org/10.1155/2015/897639
Qu Z, Yang F, Yan Y et al (2021) Relationship between serum nutritional factors and bone mineral density: A Mendelian randomization study. J Clin Endocrinol Metab 106:e2434–e2443. https://doi.org/10.1210/clinem/dgab085
Schloss PD, Westcott SL, Ryabin T et al (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75(23):7537–7541. https://doi.org/10.1128/AEM.01541-09
Sommer F, Bäckhed F (2013) The gut microbiota–masters of host development and physiology. Nat Rev Microbiol 11(4):227–238. https://doi.org/10.1038/nrmicro2974
Song SY, Kim Y, Park H et al (2017) Effect of parity on bone mineral density: A systematic review and meta-analysis. Bone 101:70–76. https://doi.org/10.1016/j.bone.2017.04.013
Spicer AP, Kaback LA, Smith TJ et al (1998) Molecular cloning and characterization of the human and mouse UDP-glucose dehydrogenase genes. J Biol Chem 273(39):25117–25124. https://doi.org/10.1074/jbc.273.39.25117
Tsai TL, Pan WH, Chung YT et al (2016) Association between urinary lead and bone health in a general population from Taiwan. J Expo Sci Environ Epidemiol 26:481–487. https://doi.org/10.1038/jes.2015.30
Vanderlinde EM, Magnus SA, Tambalo DD et al (2011) Mutation of a broadly conserved operon (RL3499-RL3502) from Rhizobium leguminosarum biovar viciae causes defects in cell morphology and envelope integrity. J Bacteriol 193(11):2684–2694. https://doi.org/10.1128/JB.01456-10
Wallin M, Barregard L, Sallsten G et al (2016) Low-level cadmium exposure is associated with decreased bone mineral density and increased risk of incident fractures in elderly men: The MrOS Sweden Study. J Bone Miner Res 31:732–741. https://doi.org/10.1002/jbmr.2743
Wang X, Liu R, Zhu W et al (2019) UDP-glucose accelerates SNAI1 mRNA decay and impairs lung cancer metastasis. Nature 571(7763):127–131. https://doi.org/10.1038/s41586-019-1340-y
Wang X, Karvonen-Gutierrez CA, Herman WH et al (2020) Urinary metals and incident diabetes in midlife women: study of Women’s Health across the Nation (SWAN). BMJ Open Diabetes Res Care 8(1):e001233. https://doi.org/10.1136/bmjdrc-2020-001233
Wei M, Li C, Dai Y et al (2021) High-throughput absolute quantification sequencing revealed osteoporosis-related gut microbiota alterations in Han Chinese elderly. Front Cell Infect Microbiol 11:630372. https://doi.org/10.3389/fcimb.2021.630372
Yang J, Li J, Zhang X et al (2023) Effects of ecologically relevant concentrations of cadmium on the microbiota, short-chain fatty acids, and FFAR(2) expression in Zebrafish. Metabolites 13(5):657. https://doi.org/10.3390/metabo13050657
Yao X, Steven XuX, Yang Y et al (2021) Stratification of population in NHANES 2009–2014 based on exposure pattern of lead, cadmium, mercury, and arsenic and their association with cardiovascular, renal and respiratory outcomes. Environ Int 149:106410. https://doi.org/10.1016/j.envint.2021.106410
Zeng QB, Xu YY, Yu X et al (2014) Arsenic may be involved in fluoride-induced bone toxicity through PTH/PKA/AP1 signaling pathway. Environ Toxicol Pharmacol 37:228–233. https://doi.org/10.1016/j.etap.2013.11.027
Acknowledgements
We thank all participants for their contributions to this work. We would also like to thank the Wuhan Center for Disease Control and Prevention for their technology assistance.
Funding
This work was supported by the National Natural Science Foundation of China (Grant no.82273711) and Wuhan Municipal Health Commission (Grant no.WY22B06).
Author information
Authors and Affiliations
Contributions
Jianli Zhang: Develop the research plan, detection of metals in plasma samples, application statistical techniques to analyze study data, write the initial draft and revision. Qi Mai: Detection of metals in plasma samples, write the initial draft and revision. Dongsheng Di, Haolong Zhou, Ruyi Zhang: Data collection, development of the methodology and participant in the article revision. Qi Wang: Coming up with ideas, obtaining financial support for the project that led to this publication, supervision, project management, and writing-review and editing.
Corresponding author
Ethics declarations
Ethics statement
This study was approved by the Ethics Committee of Tongji Medical College of Huazhong University of Science and Technology. Each participant had signed an informed consent form.
Consent to participate
The manuscript has obtained the informed consent of all participants.
Consent to publish
The manuscript is approved by all authors for publication.
Competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Responsible Editor: Lotfi Aleya
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Jianli Zhang and Qi Mai contributed equally to this work.
Supplementary Information
Below is the link to the electronic supplementary material.
11356_2023_30388_MOESM1_ESM.tif
Fig. S1 Heatmap illustration of pairwise correlations in plasma concentrations of Fe, Zn, Se, As, Cd, and Pb. Red × indicated there was no significant correlation between the two metals (TIF 40495 KB)
11356_2023_30388_MOESM2_ESM.tif
Fig. S2 Violin plot of the six metals in plasma (Fe, Zn, Se, As, Cd, and Pb) grouped by the three clusters observed by k-means clustering method among 806 participants. Cluster 1, low-exposure group for all six metals; Cluster 2, high-exposure group for Se and As; Cluster 3, high-exposure group for Fe, Zn, Pb, and Cd (TIF 44488 KB)
11356_2023_30388_MOESM3_ESM.tif
Fig. S3 Bacterial community abundance barplot of each group: (A) Bacterial community abundance barplot at phylum level, (B) Bacterial community abundance barplot at family level. Cluster 1, low-exposure group for all six metals; Cluster 2, high-exposure group for Se and As; Cluster 3, high-exposure group for Fe, Zn, Pb, and Cd (TIF 13365 KB)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, J., Mai, Q., Di, D. et al. Potential roles of gut microbiota in metal mixture and bone mineral density and osteoporosis risk association: an epidemiologic study in Wuhan. Environ Sci Pollut Res 30, 117201–117213 (2023). https://doi.org/10.1007/s11356-023-30388-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-023-30388-w