Abstract
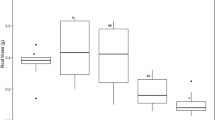
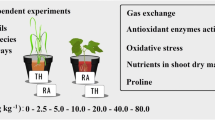
Phytoremediation represents a natural method to remove contaminants from soil. The goal of this study was to investigate the potential of phosphate-assisted phytoremediation by two energy crops, Cannabis sativa L. and Brassica juncea L., for the sustainable remediation of heavily arsenic-contaminated industrial soil. The two species were investigated for uptake, translocation, and physiological effects of arsenic and phosphate in a microcosm test. Although C. sativa and B. juncea were symptomless when grown in arsenic-contaminated soil, an important reduction of biomass (50 and 25%, respectively) was observed as a stress marker. Phytotoxicity and cytotoxicity effects promoted by contaminated soils were investigated in both the species and a model plant for ecotoxicity studies, Vicia faba L., which is the most developed model to test genotoxicity effects in terms of chromosomal aberration and micronuclei presence. The higher amount of arsenic was found in C. sativa and B. juncea roots (on average 1473 and 778 mg kg−1, respectively), but both species were able to uptake and translocate arsenic in leaves and stems, up to 47.0 and 189 mg kg−1, respectively. Phosphate treatment had no effect on arsenic uptake in none of the crop, but significantly improved the plant performance. Biomass production resulted similar to that of B. juncea control plants. Antioxidant enzymatic activities and photosynthetic performance responded differently in the two crops. The present investigation provides new insight for a proficient selection of the most suitable crop species for sustainable phytomanagement of a highly polluted As-contaminated site by coupled phytoremediation-bioenergy approach.
Graphical abstract







Similar content being viewed by others
Data availability
All data generated and analyzed during this study are included in the manuscript.
References
Aebi H (1974) Catalase. In: Bergmeyer H (ed) Methods of Enzymatic Analysis, Second Edi. Academic Press, pp 673–684
Ahmad MA, Gupta M (2013) Exposure of Brassica juncea(L) to arsenic species in hydroponic medium: comparative analysis in accumulation and biochemical and transcriptional alterations. Environ Sci Pollut Res 20:8141–8150. https://doi.org/10.1007/s11356-013-1632-y
Ahmad R, Tehsin Z, Malik ST, Asad SA, Shahzad M, Bilal M, Shah MM, Khan SA (2016) Phytoremediation potential of hemp (Cannabis sativa L.): identification and characterization of heavy metals responsive genes. CLEAN - Soil, Air, Water 44:195–201. https://doi.org/10.1002/clen.201500117
Ahmed S, Hassan MH, Kalam MA, Ashrafur Rahman SM, Abedin MJ, Shahir A (2014) An experimental investigation of biodiesel production, characterization, engine performance, emission and noise of Brassica juncea methyl ester and its blends. J Clean Prod 79:74–81. https://doi.org/10.1016/j.jclepro.2014.05.019
Amaducci S, Errani M, Venturi G (2002) Response of hemp to plant population and nitrogen fertilisation. Ital J Agron 6:103–111
Anawar HM, Rengel Z, Damon P, Tibbett M (2018)Arsenic-phosphorus interactions in the soil-plant-microbe system: dynamics of uptake, suppression and toxicity to plants. Environ Pollut 233:1003–1012. https://doi.org/10.1016/J.ENVPOL.2017.09.098
Ashraf S, Ali Q, Zahir ZA, Ashraf S, Asghar HN (2019) Phytoremediation: environmentally sustainable way for reclamation of heavy metal polluted soils. Ecotoxicol. Environ. Saf. 174:714–727
Aubin M-P, Seguin P, Vanasse A, Tremblay GF, Mustafa AF, Charron JB (2015) Industrial hemp response to nitrogen, phosphorus, and potassium fertilization. Crop Forage Turfgrass Manag 1:0. https://doi.org/10.2134/cftm2015.0159, 1, 10
Austruy A, Wanat N, Moussard C, Vernay P, Joussein E, Ledoigt G, Hitmi A (2013) Physiological impacts of soil pollution and arsenic uptake in three plant species: Agrostis capillaris, Solanum nigrum and Vicia faba. Ecotoxicol Environ Saf 90:28–34. https://doi.org/10.1016/J.ECOENV.2012.12.008
Barbafieri M, Giorgetti L (2016) Contaminant bioavailability in soil and phytotoxicity/genotoxicity tests in Vicia faba L.: A case study of boron contamination. Environ Sci Pollut Res 23:24327–24336. https://doi.org/10.1007/s11356-016-7653-6
Barbafieri M, Giansoldati V, Pedron F et al (2012) Arsenic mobilization and plant uptake from an industrial contaminated soil in tuscany (Italy). Crop plants vs Pteris vittata. In: Sorial G, Jihua H (eds) Environmental Science & Technology. American Science Press, Houston, pp 343–346
Barbafieri M, Pedron F, Petruzzelli G, Rosellini I, Franchi E, Bagatin R, Vocciante M (2017) Assisted phytoremediation of a multi-contaminated soil: investigation on arsenic and lead combined mobilization and removal. J Environ Manage 203:316–329
Barbafieri M, Morelli E, Tassi E, Pedron F, Remorini D, Petruzzelli G (2018) Overcoming limitation of “recalcitrant areas” to phytoextraction process: the synergistic effects of exogenous cytokinins and nitrogen treatments. Sci Total Environ 639:1520–1529. https://doi.org/10.1016/J.SCITOTENV.2018.05.175
Beyer WF, Fridovich I (1987) Assaying for superoxide dismutase activity: some large consequences of minor changes in conditions. Anal Biochem 161:559–566. https://doi.org/10.1016/0003-2697(87)90489-1
Bolan N, Mahimairaja S, Kunhikrishnan A, Choppala G (2013)Phosphorus–arsenic interactions in variable-charge soils in relation to arsenic mobility and bioavailability. Sci Total Environ 463–464:1154–1162. https://doi.org/10.1016/j.scitotenv.2013.04.016
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Campos NV, Loureiro ME, Azevedo AA (2014) Differences in phosphorus translocation contributes to differential arsenic tolerance between plants of Borreria verticillata(Rubiaceae) from mine and non-mine sites. Environ Sci Pollut Res 21:5586–5596. https://doi.org/10.1007/s11356-013-2444-9
Cao X, Ma LQ, Tu C (2004) Antioxidative responses to arsenic in the arsenic-hyperaccumulator Chinese brake fern (Pteris vittata L.). Environ Pollut 128:317–325. https://doi.org/10.1016/J.ENVPOL.2003.09.018
Choudhury B, Chowdhury S, Biswas AK (2011) Regulation of growth and metabolism in rice (Oryza sativa L.) by arsenic and its possible reversal by phosphate. J Plant Interact 6:15–24. https://doi.org/10.1080/17429140903487552
Choudhury FK, Rivero RM, Blumwald E, Mittler R (2017) Reactive oxygen species, abiotic stress and stress combination. Plant J 90:856–867. https://doi.org/10.1111/tpj.13299
Cotelle S, Dhyèvre A, Muller S, Chenon P, Manier N, Pandard P, Echairi A, Silvestre J, Guiresse M, Pinelli E, Giorgetti L, Barbafieri M, Silva VC, Engel F, Radetski CM (2015) Soil genotoxicity assessment—results of an interlaboratory study on the Vicia micronucleus assay in the context of ISO standardization. Environ Sci Pollut Res 22:988–995. https://doi.org/10.1007/s11356-014-3495-2
Das L, Liu E, Saeed A, Williams DW, Hu H, Li C, Ray AE, Shi J (2017) Industrial hemp as a potential bioenergy crop in comparison with kenaf, switchgrass and biomass sorghum. Bioresour Technol 244:641–649. https://doi.org/10.1016/j.biortech.2017.08.008
de Freitas-Silva L, de Araújo TO, da Silva LC et al (2016) Arsenic accumulation in Brassicaceae seedlings and its effects on growth and plant anatomy. Ecotoxicol Environ Saf 124:1–9. https://doi.org/10.1016/j.ecoenv.2015.09.028
Duquesnoy I, Champeau GM, Evray G, Ledoigt G, Piquet-Pissaloux A (2010) Enzymatic adaptations to arsenic-induced oxidative stress in Zea mays and genotoxic effect of arsenic in root tips of Vicia faba and Zea mays. C R Biol 333:814–824. https://doi.org/10.1016/j.crvi.2010.07.004
EPA - United State Environmental Protection Agency (1995a) Method 3051A Microwave assisted acid digestion of sediments, sludges, soils, and oils. In: U.S. Environmental Protection Agency (ed) Test Methods for Evaluating Solid Waste: Physical/Chemical Methods (SW-846), 3rd edn. Washington D.C., pp 3051A-1 3051A-30
EPA - United State Environmental Protection Agency (1995b) Method 3052 Microwave assisted acid digestion of siliceous and organically based matrices. In: U.S. Environmental Protection Agency (ed) Test Methods for Evaluating Soild Waste: Physical/Chemical Methods (SW-846), 3rd edn. Washington D.C., pp 3052–1 3052–21
Feng H, Li X, Sun D, Chen Y, Xu G, Cao Y, Ma LQ (2021) Expressing phosphate transporter PvPht2;1 enhances p transport to the chloroplasts and increases arsenic tolerance in Arabidopsis thaliana. Environ Sci Technol 55:2276–2284. https://doi.org/10.1021/acs.est.0c03316
Finnegan PM, Chen W (2012) Arsenic toxicity: the effects on plant metabolism. Front Physiol 182. https://doi.org/10.3389/fphys.2012.00182
Foyer CH, Halliwell B (1976) The presence of glutathione and glutathione reductase in chloroplasts: a proposed role in ascorbic acid metabolism. Planta 133:21–25. https://doi.org/10.1007/BF00386001
Franchi E, Rolli E, Marasco R, Agazzi G, Borin S, Cosmina P, Pedron F, Rosellini I, Barbafieri M, Petruzzelli G (2017) Phytoremediation of a multi contaminated soil: mercury and arsenic phytoextraction assisted by mobilizing agent and plant growth promoting bacteria. J Soils Sediments 17:1224–1236. https://doi.org/10.1007/s11368-015-1346-5
Franchi E, Cosmina P, Pedron F, Rosellini I, Barbafieri M, Petruzzelli G, Vocciante M (2019) Improved arsenic phytoextraction by combined use of mobilizing chemicals and autochthonous soil bacteria. Sci Total Environ 655:328–336. https://doi.org/10.1016/j.scitotenv.2018.11.242
Garg N, Singla P (2011) Arsenic toxicity in crop plants: physiological effects and tolerance mechanisms. Environ Chem Lett 9:303–321. https://doi.org/10.1007/s10311-011-0313-7
Gerhardt KE, Gerwing PD, Greenberg BM (2017) Opinion: taking phytoremediation from proven technology to accepted practice. Plant Sci. 256:170–185
Giansoldati V, Tassi E, Morelli E, Gabellieri E, Pedron F, Barbafieri M (2012) Nitrogen fertilizer improves boron phytoextraction by Brassica juncea grown in contaminated sediments and alleviates plant stress. Chemosphere 87:1119–1125. https://doi.org/10.1016/J.CHEMOSPHERE.2012.02.005
Giorgetti L, Talouizte H, Merzouki M, Caltavuturo L, Geri C, Frassinetti S (2011) Genotoxicity evaluation of effluents from textile industries of the region Fez-Boulmane, Morocco: a case study. Ecotoxicol Environ Saf 74:2275–2283. https://doi.org/10.1016/J.ECOENV.2011.08.002
Grifoni M, Schiavon M, Pezzarossa B, Petruzzelli G, Malagoli M (2015) Effects of phosphate and thiosulphate on arsenic accumulation in the species Brassica juncea. Environ Sci Pollut Res 22:2423–2433. https://doi.org/10.1007/s11356-014-2811-1
Grifoni M, Pedron F, Petruzzelli G, Rosellini I, Barbafieri M, Franchi E, Bagatin R, 1 Institute of Ecosystem Studies, National Council of Research, Pisa, Italy (2017a) Assessment of repeated harvests on mercury and arsenic phytoextraction in a multi-contaminated industrial soil. AIMS Environ Sci 4:187–205. https://doi.org/10.3934/environsci.2017.2.187
Grifoni M, Petruzzelli G, Barbafieri M, et al (2017b) Soil quality protection at heavy metal-contaminated manufactured gas plant sites: Role of biological remediation
Grifoni M, Pedron F, Barbafieri M, Rosellini I, Petruzzelli G, Franchi E (2020) Sustainable valorization of biomass: from assisted phytoremediation to green energy production. In: Prasad MNV (ed) Handbook on Assisted and Amendments Enhanced Sustainable Remediation Technology, In press. John Wiley & Sons, UK
Gunes A, Pilbeam DJ, Inal A (2009) Effect of arsenic–phosphorus interaction on arsenic-induced oxidative stress in chickpea plants. Plant Soil 314:211–220. https://doi.org/10.1007/s11104-008-9719-9
Gupta K, Srivastava A, Srivastava S, Kumar A (2020)Phyto-genotoxicity of arsenic contaminated soil from Lakhimpur Kheri, India on Vicia faba L. Chemosphere 241:125063. https://doi.org/10.1016/j.chemosphere.2019.125063
ISO - International Standardization Organization (2013) Soil quality—assessment of genotoxic effects on higher plants—Vicia faba micronucleus test. ISO 29200
Kabata-Pendias A (2010) Trace elements in soils and plants, 4th edn. CRC Press, Taylor & Francis Group, Boca Raton
Karimi N, Souri Z (2015) Effect of phosphorus on arsenic accumulation and detoxification in arsenic hyperaccumulator, Isatis cappadocica. J Plant Growth Regul 34:88–95. https://doi.org/10.1007/s00344-014-9445-x
Kumar S, Singh R, Kumar V, Rani A, Jain R (2017)Cannabis sativa: a plant suitable for phytoremediation and bioenergy production. In: Bauddh K, Singh B, Korstad J (eds) Phytoremediation Potential of Bioenergy Plants. Springer, Cham, pp 269–285
Leme DM, Marin-Morales MA (2009)Allium cepa test in environmental monitoring: a review on its application. Mutat Res 682:71–81. https://doi.org/10.1016/J.MRREV.2009.06.002
Lou LQ, Ye ZH, Lin AJ, Wong MH (2010) Interaction of arsenic and phosphate on their uptake and accumulation in Chinese brake fern. Int J Phytoremediation 12:487–502. https://doi.org/10.1080/15226510903051732
Mandal A, Purakayastha TJ, Patra AK, Sarkar B (2018) Arsenic phytoextraction by Pteris vittata improves microbial properties in contaminated soil under various phosphate fertilizations. Appl Geochemistry 88:258–266. https://doi.org/10.1016/J.APGEOCHEM.2017.04.008
Manzano R, Moreno-Jiménez E, Esteban E (2015) Arsenic in the soil-plant system: phytotoxicity and phytoremediation. In: Chakrabarty N (ed) Arsenic Toxicity: Prevention and Treatment, 1st Editio. Boca Raton, Florida, pp 219–234
Mascher R, Lippmann B, Holzinger S, Bergmann H (2002) Arsenate toxicity: effects on oxidative stress response molecules and enzymes in red clover plants. Plant Sci 163:961–969. https://doi.org/10.1016/S0168-9452(02)00245-5
Mehmood T, Bibi I, Shahid M, Niazi NK, Murtaza B, Wang H, Ok YS, Sarkar B, Javed MT, Murtaza G (2017) Effect of compost addition on arsenic uptake, morphological and physiological attributes of maize plants grown in contrasting soils. J Geochemical Explor 178:83–91. https://doi.org/10.1016/j.gexplo.2017.03.018
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Niazi NK, Bibi I, Fatimah A, Shahid M, Javed MT, Wang H, Ok YS, Bashir S, Murtaza B, Saqib ZA, Shakoor MB (2017)Phosphate-assisted phytoremediation of arsenic by Brassica napus and Brassica juncea: Morphological and physiological response. Int J Phytoremediation 19:670–678. https://doi.org/10.1080/15226514.2016.1278427
O’Connor D, Zheng X, Hou D et al (2019) Phytoremediation: climate change resilience and sustainability assessment at a coastal brownfield redevelopment. Environ Int 130:104945. https://doi.org/10.1016/j.envint.2019.104945
Pandey VC, Bajpai O, Singh N (2016) Energy crops in sustainable phytoremediation. Renew. Sustain. Energy Rev. 54:58–73
Pant S, Ritika KA et al (2021) NaOH pretreatment and enzymatic hydrolysis of Brassica juncea using mixture of cellulases. Environ Technol Innov 21:101324. https://doi.org/10.1016/j.eti.2020.101324
Parvez AM, Lewis JD, Afzal MT (2021) Potential of industrial hemp (Cannabis sativa L.) for bioenergy production in Canada: Status, challenges and outlook. Renew. Sustain. Energy Rev 141:110784
Patra M, Bhowmik N, Bandopadhyay B, Sharma A (2004) Comparison of mercury, lead and arsenic with respect to genotoxic effects on plant systems and the development of genetic tolerance. Environ Exp Bot 52:199–223. https://doi.org/10.1016/j.envexpbot.2004.02.009
Pedron F, Grifoni M, Barbafieri M, Petruzzelli G, Rosellini I, Franchi E, Bagatin R, Vocciante M (2017) Applicability of a Freundlich-like model for plant uptake at an industrial contaminated site with a high variable arsenic concentration. Environments 4:67. https://doi.org/10.3390/environments4040067
Petrová Š, Benešová D, Soudek P, Vanĕk T (2012) Enhancement of metal(loid)s phytoextraction by Cannabis sativa L. J Food, Agric Environ 10:631–641
Petruzzelli G, Pedron F, Rosellini I, Barbafieri M (2015) The bioavailability processes as a key to evaluate phytoremediation efficiency. In: Ansari AA, Gill SS, Gill R, Lanza GRNL (eds) Phytoremediation. Springer International Publishing, Cham, pp 31–43
Pigna M, Cozzolino V, Violante A, Meharg AA (2009) Influence of phosphate on the arsenic uptake by wheat (Triticum durum L.) irrigated with arsenic solutions at three different concentrations. Water Air Soil Pollut 197:371–380. https://doi.org/10.1007/s11270-008-9818-5
Pigna M, Caporale A, Cozzolino V et al (2012) Influence of phosphorus on the arsenic uptake by tomato (Solanum lycopersicum L) irrigated with arsenic solutions at four different concentrations. J Soil Sci plant Nutr 12. https://doi.org/10.4067/S0718-95162012005000031
Prasad MNV, De Oliveira Freitas HM (2003) Metal hyperaccumulation in plants - biodiversity prospecting for phytoremediation technology. Electron J Biotechnol 6. https://doi.org/10.2225/vol6-issue3-fulltext-6
Pudełko K, Majchrzak L, Narożna D (2014) Allelopathic effect of fibre hemp (Cannabis sativa L.) on monocot and dicot plant species. Ind Crops Prod 56:191–199. https://doi.org/10.1016/J.INDCROP.2014.02.028
Rathore SS, Shekhawat K, Dass A, Kandpal BK, Singh VK (2019) Phytoremediation mechanism in Indian mustard (Brassica juncea) and its enhancement through agronomicinterventions. Proc. Natl. Acad. Sci. India Sect. B - Biol. Sci. 89:419–427
Rheay HT, Omondi EC, Brewer CE (2021) Potential of hemp (Cannabis sativa L.) for paired phytoremediation and bioenergy production. GCB Bioenergy 13:525–536
Shelmerdine PA, Black CR, McGrath SP, Young SD (2009) Modelling phytoremediation by the hyperaccumulating fern, Pteris vittata, of soils historically contaminated with arsenic. Environ Pollut 157:1589–1596. https://doi.org/10.1016/j.envpol.2008.12.029
Shikha D, Singh PK (2021) In situ phytoremediation of heavy metal–contaminated soil and groundwater: a green inventive approach. Environ Sci Pollut Res 28:4104–4124. https://doi.org/10.1007/s11356-020-11600-7
Singh KP, Kumari R, Treas J, Dumond JW (2011) Chronic exposure to arsenic causes increased cell survival, DNA damage, and increased expression of mitochondrial transcription factor A (mtTFA) in human prostate epithelial cells. Chem Res Toxicol 24:340–349. https://doi.org/10.1021/tx1003112
Singh S, Sounderajan S, Kumar K, Fulzele DP (2017) Investigation of arsenic accumulation and biochemical response of in vitro developed Vetiveria zizanoides plants. Ecotoxicol Environ Saf 145:50–56. https://doi.org/10.1016/J.ECOENV.2017.07.013
Song Y, Kirkwood N, Maksimović Č, Zheng X, O'Connor D, Jin Y, Hou D (2019) Nature based solutions for contaminated land remediation and brownfield redevelopment in cities: a review. Sci Total Environ 663:568–579. https://doi.org/10.1016/j.scitotenv.2019.01.347
Sparks DL, Page AL, Helmke PA, Loeppert RH (1996) Methods of soil analysis part 3—chemical methods. Soil Science Ssociety of American, Madison
Srivastava S, Sharma YK (2013) Impact of asenic toxicity on black gram and its amelioration using phosphate. ISRN Toxicol
Srivastava M, Ma LQ, Singh N, Singh S (2005) Antioxidant responses of hyper-accumulator and sensitive fern species to arsenic. J Exp Bot 56:1335–1342. https://doi.org/10.1093/jxb/eri134
Stoeva N, Berova M, Zlatev Z (2005) Effect of arsenic on some physiological parameters in bean plants. Biol Plant 49:293–296. https://doi.org/10.1007/s10535-005-3296-z
Szczygłowska M, Piekarska A, Konieczka P, Namieśnik J (2011) Use of brassica plants in the phytoremediation and biofumigation processes. Int J Mol Sci 12:7760–7771. https://doi.org/10.3390/ijms12117760
Tiwari S, Sarangi BK (2017) Comparative analysis of antioxidant response by Pteris vittata and Vetiveria zizanioides towards arsenic stress. Ecol Eng 100:211–218. https://doi.org/10.1016/J.ECOLENG.2016.12.007
Tripathi V, Edrisi SA, Abhilash PC (2016) Towards the coupling of phytoremediation with bioenergy production. Renew. Sustain. Energy Rev. 57:1386–1389
van Gestel CAM (2008)Physico-chemical and biological parameters determine metal bioavailability in soils. Sci Total Environ 406:385–395. https://doi.org/10.1016/J.SCITOTENV.2008.05.050
van Ginneken L, Meers E, Guisson R et al (2007) Phytoremediation for heavy metal-contaminated soils combined with bioenergy production. J Environ Eng Landsc Manag 15:227–236. https://doi.org/10.1080/16486897.2007.9636935
Vera CL, Malhi SS, Phelps SM, May WE, Johnson EN (2010) N, P, and S fertilization effects on industrial hemp in Saskatchewan. Can J Plant Sci 90:179–184. https://doi.org/10.4141/CJPS09101
Vetterlein D, Wesenberg D, Nathan P, Bräutigam A, Schierhorn A, Mattusch J, Jahn R (2009)Pteris vittata– revisited: uptake of As and its speciation, impact of P, role of phytochelatins and S. Environ Pollut 157:3016–3024. https://doi.org/10.1016/J.ENVPOL.2009.05.057
Wang J, Zhan X, Zhou L, Lin Y (2010) Biological indicators capable of assessing thermal treatment efficiency of hydrocarbon mixture-contaminated soil. Chemosphere 80:837–844. https://doi.org/10.1016/J.CHEMOSPHERE.2010.06.009
Willis RJ(RJ) (2007) The history of allelopathy. Springer, Dordrecht
Wu L, Yi H, Yi M (2010) Assessment of arsenic toxicity using Allium/Vicia root tip micronucleus assays. J Hazard Mater 176:952–956. https://doi.org/10.1016/j.jhazmat.2009.11.132
Wuana RA, Okieimen FE (2011) Heavy metals in contaminated soils: a review sources, chemistry, risks and best available strategies for remediation. ISRN Ecol 2011:1–20. https://doi.org/10.5402/2011/402647
Yang W, Luo L, Bostick BC, Wiita E, Cheng Y, Shen Y (2021) Effect of combined arsenic and lead exposure on their uptake and translocation in Indian mustard. Environ Pollut 274:116549. https://doi.org/10.1016/j.envpol.2021.116549
Yi H, Wu L, Jiang L (2007) Genotoxicity of arsenic evaluated by Allium-root micronucleus assay. Sci Total Environ 383:232–236. https://doi.org/10.1016/j.scitotenv.2007.05.015
Zhao FJ, Ma JF, Meharg AA, McGrath SP (2009) Arsenic uptake and metabolism in plants. New Phytol 181:777–794. https://doi.org/10.1111/j.1469-8137.2008.02716.x
Acknowledgements
The authors would like to thank Manuele Scatena for his valuable technical assistance.
Funding
This research was carried out with the support of the Research & Technological Innovation Department of Eni S.p.A, San Donato Milanese (Italy), and funded by Syndial S.p.A. All activities were conducted at the Italian National Research Council, CNR Area in Pisa, Italy.
Author information
Authors and Affiliations
Contributions
M. B., L. G., and G. P. designed the research and contributed to the conception of the study; C. P., L. G., E. M., M. L., M. G., and I. R. performed the research and analyzed the data; M. B., L. G., E. M., M. L., C. P., and M. G. contributed to the first draft of the manuscript; M. B., M. G., G. P., and E. F. commented on the manuscript draft critically. All authors reviewed the drafts and gave final approval for publication.
Corresponding author
Ethics declarations
Ethics approval
Not applicable
Consent to participate
Not applicable
Consent for publication
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Elena Maestri
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Picchi, C., Giorgetti, L., Morelli, E. et al. Cannabis sativa L. and Brassica juncea L. grown on arsenic-contaminated industrial soil: potentiality and limitation for phytoremediation. Environ Sci Pollut Res 29, 15983–15998 (2022). https://doi.org/10.1007/s11356-021-16673-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-16673-6