Abstract
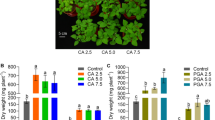
Phytoextraction is currently investigated to effectively remediate soil contaminated by metals and provide highly competitive biomass for energy production. This research aimed to increase arsenic (As) removal from contaminated soil using industrial Cannabis sativa L., a suitable energy crop for biofuel production. Assisted phytoextraction experiments were conducted on a microcosm scale to explore the ability of two friendly treatments, sodium sulphate (SO4) and exogenous cytokinin (CK), in increasing As phytoextraction efficiency. The results showed that the treatments significantly increased As phytoextraction. Cytokinin was the most effective agent for effectively increasing translocation and the amount of As in aerial parts of C. sativa. In fact, the concentration of As in the shoots of CK-treated plants increased by 172% and 44% compared to untreated and SO4-treated plants, respectively. However, the increased As amount accumulated in C. sativa tissues due to the two treatments negatively affected plant growth. Arsenic toxicity caused a significant decrease in aerial C. sativa biomass treated with CK and SO4 of about 32.7% and 29.8% compared to untreated plants, respectively. However, for our research purposes, biomass reduction has been counterbalanced by an increase in As phytoextraction, such as to consider C. sativa and CK an effective combination for the remediation of As-contaminated soils. Considering that C. sativa has the suitable characteristics to provide valuable resources for bioenergy production, our work can help improve the implementation of a sustainable management model for As contaminated areas, such as phytoremediation coupled with bioenergy generation.


Similar content being viewed by others
Data availability
All data generated and analyzed during this study are included in the manuscript.
References
Abbas G, Murtaza B, Bibi I, Shahid M, Niazi N, Khan M, Amjad M, Hussain M, Natasha (2018) Arsenic uptake, toxicity, detoxification, and speciation in plants: physiological, biochemical, and molecular aspects. Int J Environ Res Public Health 15:59. https://doi.org/10.3390/ijerph15010059
Austruy A, Wanat N, Moussard C, Vernay P, Joussein E, Ledoigt G, Hitmi A (2013) Physiological impacts of soil pollution and arsenic uptake in three plant species: Agrostis capillaris, Solanum nigrum and Vicia faba. Ecotoxicol Environ Saf 90:28–34. https://doi.org/10.1016/J.ECOENV.2012.12.008
Barbafieri M (2016) Use of phytohormones for strengthening metal (loid) phytoextraction: limitations and a case study. In: Ahammed GJ, Yu J (eds) Plant hormones under challenging environmental factors. Springer, Dordrecht, pp 157–179
Barbafieri M, Japenga J, Romkens P, Petruzzelli G, Pedron F (2013) Protocols for applying phytotechnologies in metal-contaminated soils. In: Dharmendra Kumar Gupta (ed) Plant-Based Remediation Processes. Springer, Berlin Heidelberg, pp 19–37
Barbafieri M, Morelli E, Tassi E, Pedron F, Remorini D, Petruzzelli G (2018) Overcoming limitation of “recalcitrant areas” to phytoextraction process: the synergistic effects of exogenous cytokinins and nitrogen treatments. Sci Total Environ 639:1520–1529. https://doi.org/10.1016/J.SCITOTENV.2018.05.175
Beesley L, Marmiroli M, Pagano L et al (2013) Biochar addition to an arsenic contaminated soil increases arsenic concentrations in the pore water but reduces uptake to tomato plants (Solanum lycopersicum L.). Sci Total Environ 454–455:598–603. https://doi.org/10.1016/j.scitotenv.2013.02.047
Bulak P, Walkiewicz A, Brzezińska M (2014) Plant growth regulators-assisted phytoextraction. Biol Plant 58:1–8. https://doi.org/10.1007/s10535-013-0382-5
Cappelletto P, Brizzi M, Mongardini F, Barberi B, Sannibale M, Nenci G, Poli M, Corsi G, Grassi G, Pasini P (2001) Italy-grown hemp: Yield, composition and cannabinoid content. Ind Crop Prod 13:101–113. https://doi.org/10.1016/S0926-6690(00)00057-1
Cassina L, Tassi E, Morelli E et al (2011) Exogenous cytokinin treatments of an Ni hyper-accumulator, Alyssum murale, grown in a serpentine soil: implications for phytoextraction. Int J Phytoremediation 13:30–101
Cassina L, Tassi E, Pedron F et al (2012) Using a plant hormone and a thioligand to improve phytoremediation of Hg-contaminated soil from a petrochemical plant. J Hazard Mater 231:36–42
Chen G, Feng T, Li Z, Chen Z, Chen Y, Wang H, Xiang Y (2017) Influence of sulfur on the arsenic phytoremediation using Vallisneria natans (Lour.) Hara. Bull Environ Contam Toxicol 99:411–414. https://doi.org/10.1007/s00128-017-2135-1
Citterio S, Santagostino A, Fumagalli P, Prato N, Ranalli P, Sgorbati S (2003) Heavy metal tolerance and accumulation of Cd, Cr and Ni by Cannabis sativa L. Plant Soil 256:243–252. https://doi.org/10.1023/A:1026113905129
Das HK, Mitra AK, Sengupta PK, Hossain A, Islam F, Rabbani GH (2004) Arsenic concentrations in rice, vegetables, and fish in Bangladesh: a preliminary study. Environ Int 30:383–387. https://doi.org/10.1016/j.envint.2003.09.005
Das L, Liu E, Saeed A, Williams DW, Hu H, Li C, Ray AE, Shi J (2017) Industrial hemp as a potential bioenergy crop in comparison with kenaf, switchgrass and biomass sorghum. Bioresour Technol 244:641–649. https://doi.org/10.1016/j.biortech.2017.08.008
Dastyar W, Raheem A, He J, Zhao M (2019) Biofuel production using thermochemical conversion of heavy metal-contaminated biomass (HMCB) harvested from phytoextraction process. Chem Eng J 358:759–785. https://doi.org/10.1016/j.cej.2018.08.111
de Oliveira LM, Ma LQ, Santos JAG, Guilherme LRG, Lessl JT (2014) Effects of arsenate, chromate, and sulfate on arsenic and chromium uptake and translocation by arsenic hyperaccumulator Pteris vittata L. Environ Pollut 184:187–192. https://doi.org/10.1016/J.ENVPOL.2013.08.025
Dixit G, Singh AP, Kumar A, Mishra S, Dwivedi S, Kumar S, Trivedi PK, Pandey V, Tripathi RD (2016) Reduced arsenic accumulation in rice (Oryza sativa L.) shoot involves sulfur mediated improved thiol metabolism, antioxidant system and altered arsenic transporters. Plant Physiol Biochem 99:86–96. https://doi.org/10.1016/j.plaphy.2015.11.005
Dradrach A, Karczewska A, Szopka K, Lewińska K (2020) Accumulation of arsenic by plants growing in the sites strongly contaminated by historical mining in the sudetes region of poland. Int J Environ Res Public Health 17. https://doi.org/10.3390/ijerph17093342
EPA - United State Environmental Protection Agency (1995a) Method 3051A Microwave assisted acid digestion of sediments, sludges, soils, and oils. In: U.S. Environmental Protection Agency (ed) Test Methods for Evaluating Solid Waste: Physical/Chemical Methods (SW-846), 3rd edn. Washington pp 3051A-1- 3051A-30
EPA - United State Environmental Protection Agency (1995b) Method 3052 Microwave assisted acid digestion of siliceous and organically based matrices. In: U.S. Environmental Protection Agency (ed) Test Methods for Evaluating Soild Waste: Physical/Chemical Methods (SW-846), 3rd edn. Washington D.C., pp 3052–1 3052–21
Fernández MD, Pro J, Alonso C, Aragonese P, Tarazona JV (2011) Terrestrial microcosms in a feasibility study on the remediation of diesel-contaminated soils. Ecotoxicol Environ Saf 74:2133–2140. https://doi.org/10.1016/j.ecoenv.2011.08.009
Fike J (2016) Industrial hemp: renewed opportunities for an ancient crop. CRC Crit Rev Plant Sci 35:406–424. https://doi.org/10.1080/07352689.2016.1257842
Finnan J, Styles D (2013) Hemp: a more sustainable annual energy crop for climate and energy policy. Energy Policy 58:152–162. https://doi.org/10.1016/J.ENPOL.2013.02.046
Fitz WJ, Wenzel WW (2002) Arsenic transformations in the soil–rhizosphere–plant system: fundamentals and potential application to phytoremediation. J Biotechnol 99:259–278. https://doi.org/10.1016/S0168-1656(02)00218-3
Franchi E, Cosmina P, Pedron F, Rosellini I, Barbafieri M, Petruzzelli G, Vocciante M (2019) Improved arsenic phytoextraction by combined use of mobilizing chemicals and autochthonous soil bacteria. Sci Total Environ 655:328–336. https://doi.org/10.1016/j.scitotenv.2018.11.242
Geng H, Wang F, Yan C, Tian Z, Chen H, Zhou B, Yuan R, Yao J (2020) Leaching behavior of metals from iron tailings under varying pH and low-molecular-weight organic acids. J Hazard Mater 383:121136
Girdhar M, Sharma NR, Rehman H, Kumar A, Mohan A (2014) Comparative assessment for hyperaccumulatory and phytoremediation capability of three wild weeds. 3. Biotech 4:579–589. https://doi.org/10.1007/s13205-014-0194-0
Grifoni M, Pedron F, Barbafieri M, et al (2020) Sustainable valorization of biomass: from assisted phytoremediation to green energy production. In: Majeti Narasimha Vara Prasad (ed) Handbook on Assisted and Amendments Enhanced Sustainable Remediation Technology. John Wiley & Sons
Grifoni M, Pedron F, Petruzzelli G, Rosellini I, Barbafieri M, Franchi E, Bagatin R, 1 Institute of Ecosystem Studies, National Council of Research, Pisa, Italy (2017) Assessment of repeated harvests on mercury and arsenic phytoextraction in a multi-contaminated industrial soil. AIMS Environ Sci 4:187–205. https://doi.org/10.3934/environsci.2017.2.187
Grifoni M, Schiavon M, Pezzarossa B, Petruzzelli G, Malagoli M (2015) Effects of phosphate and thiosulphate on arsenic accumulation in the species Brassica juncea. Environ Sci Pollut Res 22:2423–2433. https://doi.org/10.1007/s11356-014-2811-1
Gulz PA, Gupta SK, Schulin R (2005) Arsenic accumulation of common plants from contaminated soils. Plant Soil 272:337–347. https://doi.org/10.1007/s11104-004-5960-z
Ha NTH, Ha NT, Nga TTH, Minh NN, Anh BTK, Hang NTA, Duc NA, Nhuan MT, Kim KW (2019) Uptake of arsenic and heavy metals by native plants growing near Nui Phao multi-metal mine, northern Vietnam. Appl Geochem 108:104368. https://doi.org/10.1016/j.apgeochem.2019.104368
He J, Strezov V, Kumar R, Weldekidan H, Jahan S, Dastjerdi BH, Zhou X, Kan T (2019) Pyrolysis of heavy metal contaminated Avicennia marina biomass from phytoremediation: characterisation of biomass and pyrolysis products. J Clean Prod 234:1235–1245. https://doi.org/10.1016/j.jclepro.2019.06.285
Hu ZY, Zhu YG, Li M, Zhang LG, Cao ZH, Smith FA (2007) Sulfur (S)-induced enhancement of iron plaque formation in the rhizosphere reduces arsenic accumulation in rice (Oryza sativa L.) seedlings. Environ Pollut 147:387–393. https://doi.org/10.1016/j.envpol.2006.06.014
Kashyap L, Garg N (2018) Arsenic toxicity in crop plants: responses and remediation strategies. In: Hasanuzzaman M, Nahar K, Fujita M (eds) Mechanisms of Arsenic Toxicity and Tolerance in Plants. Springer Nature, Singapore, pp 129–169
Kertulis-Tartar GM, Ma LQ, Tu C, Chirenje T (2006) Phytoremediation of an arsenic-contaminated site using Pteris vittata L.: A two-year study. Int J Phytoremediation 8:311–322. https://doi.org/10.1080/15226510600992873
Khalid S, Shahid M, Niazi NK, Rafiq M, Bakhat HF, Imran M, Abbas T, Bibi I, Dumat C (2017) Arsenic behaviour in soil-plant system: biogeochemical reactions and chemical speciation influences. In: Anjum N, Gill S, Tuteja N (eds) Enhancing Cleanup of Environmental Pollutants. Springer, Cham, pp 97–140
Khare R, Kumar S, Shukla T, Ranjan A, Trivedi PK (2017) Differential sulphur assimilation mechanism regulates response of Arabidopsis thaliana natural variation towards arsenic stress under limiting sulphur condition. J Hazard Mater 337:198–207
Kreuger E, Prade T, Escobar F, Svensson SE, Englund JE, Björnsson L (2011) Anaerobic digestion of industrial hemp-Effect of harvest time on methane energy yield per hectare. Biomass Bioenergy 35:893–900. https://doi.org/10.1016/j.biombioe.2010.11.005
Kuglarz M, Alvarado-Morales M, Karakashev D, Angelidaki I (2016) Integrated production of cellulosic bioethanol and succinic acid from industrial hemp in a biorefinery concept. Bioresour Technol 200:639–647. https://doi.org/10.1016/J.BIORTECH.2015.10.081
Kumar S, Asif MH, Chakrabarty D, Tripathi RD, Trivedi PK (2011) Differential expression and alternative splicing of rice sulphate transporter family members regulate sulphur status during plant growth, development and stress conditions. Funct Integr Genomics 11:259–273. https://doi.org/10.1007/s10142-010-0207-y
Kumar S, Singh R, Kumar V, Rani A, Jain R (2017) Cannabis sativa: a plant suitable for phytoremediation and bioenergy production. In: Bauddh K, Singh B, Korstad J (eds) Phytoremediation Potential of Bioenergy Plants. Springer, Cham, pp 269–285
Legislative Decree 152/2006 (2006) Rules in environmental field. Italian Off J
Luo J, Cai L, Qi S, Wu J, Gu XWS (2017) Improvement effects of cytokinin on EDTA assisted phytoremediation and the associated environmental risks. Chemosphere 185:386–393. https://doi.org/10.1016/j.chemosphere.2017.07.036
Maļceva M, Vikmane M, Stramkale V (2011) Changes of photosynthesis-related parameters and productivity of Cannabis sativa under different nitrogen supply. Environ Exp Biol 9:61–69
Mandal A, Purakayastha TJ, Patra AK, Sarkar B (2018) Arsenic phytoextraction by Pteris vittata improves microbial properties in contaminated soil under various phosphate fertilizations. Appl Geochem 88:258–266. https://doi.org/10.1016/J.APGEOCHEM.2017.04.008
Manzoor M, Gul I, Ahmed I, Zeeshan M, Hashmi I, Amin BAZ, Kallerhoff J, Arshad M (2019) Metal tolerant bacteria enhanced phytoextraction of lead by two accumulator ornamental species. Chemosphere 227:561–569. https://doi.org/10.1016/j.chemosphere.2019.04.093
Marchiol L, Assolari S, Sacco P, Zerbi G (2004) Phytoextraction of heavy metals by canola (Brassica napus) and radish (Raphanus sativus) grown on multicontaminated soil. Environ Pollut 132:21–27. https://doi.org/10.1016/J.ENVPOL.2004.04.001
Marwa N, Mishra N, Singh N, Mishra A, Saxena G, Pandey V, Singh N (2020) Effect of rhizospheric inoculation of isolated arsenic (As) tolerant strains on growth, As-uptake and bacterial communities in association with Adiantum capillus-veneris. Ecotoxicol Environ Saf 196:110498. https://doi.org/10.1016/j.ecoenv.2020.110498
Meers E, Ruttens A, Hopgood M, Lesage E, Tack FMG (2005) Potential of Brassica rapa, Cannabis sativa, Helianthus annuus and Zea mays for phytoextraction of heavy metals from calcareous dredged sediment derived soils. Chemosphere 61:561–572. https://doi.org/10.1016/J.CHEMOSPHERE.2005.02.026
Mishra S, Srivastava S, Tripathi RD, Trivedi PK (2008) Thiol metabolism and antioxidant systems complement each other during arsenate detoxification in Ceratophyllum demersum L. Aquat Toxicol 86:205–215. https://doi.org/10.1016/j.aquatox.2007.11.001
Mohan TC, Castrillo G, Navarro C, Zarco-Fernandez S, Ramireddy E, Mateo C, Zamarreño AM, Paz-Ares J, Muñoz R, Garcia-Mina JM, Hernandez LE, Schmülling T, Leyva A (2016) Cytokinin determines thiol-mediated arsenic tolerance and accumulation. Plant Physiol 171:1418–1426. https://doi.org/10.1104/pp.16.00372
Moreno-Jiménez E, Esteban E, Peñalosa JM (2012) The fate of arsenic in soil-plant systems. In: Reviews of environmental contamination and toxicology. Springer, New York, NY, pp 1–37
Moreno-Jiménez E, Manzano R, Esteban E, Peñalosa J (2010) The fate of arsenic in soils adjacent to an old mine site (Bustarviejo, Spain): Mobility and transfer to native flora. J Soils Sediments 10:301–312. https://doi.org/10.1007/s11368-009-0099-4
Moreno-Jiménez E, Peñalosa JM, Carpena-Ruiz RO, Esteban E (2008) Comparison of arsenic resistance in Mediterranean woody shrubs used in restoration activities. Chemosphere 71:466–473. https://doi.org/10.1016/j.chemosphere.2007.10.030
O’Connor D, Zheng X, Hou D et al (2019) Phytoremediation: Climate change resilience and sustainability assessment at a coastal brownfield redevelopment. Environ Int 130:1–9. https://doi.org/10.1016/j.envint.2019.104945
Parvez AM, Lewis JD, Afzal MT (2021) Potential of industrial hemp (Cannabis sativa L.) for bioenergy production in Canada: status, challenges and outlook. Renew Sust Energ Rev 141:110784
Paulelli ACC, Martins AC, Batista BL, Barbosa F (2019) Evaluation of uptake, translocation, and accumulation of arsenic species by six different Brazilian rice (Oryza sativa L.) cultivars. Ecotoxicol Environ Saf 169:376–382. https://doi.org/10.1016/j.ecoenv.2018.11.025
Pedron F, Grifoni M, Barbafieri M, Petruzzelli G, Rosellini I, Franchi E, Bagatin R, Vocciante M (2017) Applicability of a Freundlich-like model for plant uptake at an industrial contaminated site with a high variable arsenic concentration. Environments 4:67. https://doi.org/10.3390/environments4040067
Petrová Š, Benešová D, Soudek P, Vanĕk T (2012) Enhancement of metal(loid)s phytoextraction by Cannabis sativa L. J Food, Agric Environ 10:631–641
Petruzzelli G, Pedron F, Tassi E et al (2014) The effect of thiosulphate on arsenic bioavailability in a multi contaminated soil. A novel contribution to phytoextraction. Res J Environ Earth Sci 6:38–43
Prade T, Svensson S-E, Andersson A, Mattsson JE (2011) Biomass and energy yield of industrial hemp grown for biogas and solid fuel. Biomass Bioenergy 35:3040–3049. https://doi.org/10.1016/J.BIOMBIOE.2011.04.006
Raab A, Williams PN, Meharg A, Feldmann J (2007) Uptake and translocation of inorganic and methylated arsenic species by plants. Environ Chem 4:197. https://doi.org/10.1071/EN06079
Rostami S, Azhdarpoor A (2019) The application of plant growth regulators to improve phytoremediation of contaminated soils: A review. Chemosphere 220:818–827. https://doi.org/10.1016/J.CHEMOSPHERE.2018.12.203
Singh S, Singh A, Srivastava PK, Prasad SM (2018) Cadmium toxicity and its amelioration by kinetin in tomato seedlings vis-à-vis ascorbate-glutathione cycle. J Photochem Photobiol B Biol 178:76–84. https://doi.org/10.1016/j.jphotobiol.2017.10.025
Song Y, Kirkwood N, Maksimović Č, Zheng X, O'Connor D, Jin Y, Hou D (2019) Nature based solutions for contaminated land remediation and brownfield redevelopment in cities: a review. Sci Total Environ 663:568–579. https://doi.org/10.1016/J.SCITOTENV.2019.01.347
Sparks DL, Page AL, Helmke PA, Loeppert RH (1996) Methods of soil analysis Part 3—chemical methods. Soil Science Ssociety of American, Madison
Srivastava S, D’Souza SF (2009) Increasing sulfur supply enhances tolerance to arsenic and its accumulation in Hydrilla verticillata (L.f.) Royle. Environ Sci Technol 43:6308–6313
Sytar O, Kumari P, Yadav S, Brestic M, Rastogi A (2019) Phytohormone priming: Regulator for heavy metal stress in plants. J Plant Growth Regul 38:739–752. https://doi.org/10.1007/s00344-018-9886-8
Tassi E, Pouget J, Petruzzelli G, Barbafieri M (2008) The effects of exogenous plant growth regulators in the phytoextraction of heavy metals. Chemosphere. 71:66–73. https://doi.org/10.1016/j.chemosphere.2007.10.027
Wang HB, Ye ZH, Shu WS, Li WC, Wong MH, Lan CY (2006) Arsenic uptake and accumulation in fern species growing at arsenic-contaminated sites of Southern China: Field surveys. Int J Phytoremediation 8:1–11. https://doi.org/10.1080/16226510500214517
Wei S, Ma LQ, Saha U, Mathews S, Sundaram S, Rathinasabapathi B, Zhou Q (2010) Sulfate and glutathione enhanced arsenic accumulation by arsenic hyperaccumulator Pteris vittata L. Environ Pollut 158:1530–1535. https://doi.org/10.1016/J.ENVPOL.2009.12.024
Wenzel WW, Kirchbaumer N, Prohaska T, Stingeder G, Lombi E, Adriano DC (2001) Arsenic fractionation in soils using an improved sequential extraction procedure. Anal Chim Acta 436:309–323. https://doi.org/10.1016/S0003-2670(01)00924-2
Werner T, Motyka V, Strnad M, Schmülling T (2001) Regulation of plant growth by cytokinin. Proc Natl Acad Sci 98:10487–10492
Yanai J, Omoto T, Nakao A, Koyama K, Hartono A, Anwar S (2014) Evaluation of nitrogen status of agricultural soils in Java, Indonesia. Soil Sci Plant Nutr 60:188–195. https://doi.org/10.1080/00380768.2014.891925
Yildirim D, Sasmaz A (2017) Phytoremediation of As, Ag, and Pb in contaminated soils using terrestrial plants grown on Gumuskoy mining area (Kutahya Turkey). J Geochem Explor 182:228–234. https://doi.org/10.1016/j.gexplo.2016.11.005
Zabłudowska E, Kowalska J, Jedynak et al (2009) Search for a plant for phytoremediation - What can we learn from field and hydroponic studies? Chemosphere 77:301–307. https://doi.org/10.1016/j.chemosphere.2009.07.064
Zhang W, Cai Y, Tu C, Ma LQ (2002) Arsenic speciation and distribution in an arsenic hyperaccumulating plant. Sci Total Environ 300:167–177. https://doi.org/10.1016/S0048-9697(02)00165-1
Zhou M, Ghnaya T, Dailly H, Cui G, Vanpee B, Han R, Lutts S (2019) The cytokinin trans-zeatine riboside increased resistance to heavy metals in the halophyte plant species Kosteletzkya pentacarpos in the absence but not in the presence of NaCl. Chemosphere 233:954–965. https://doi.org/10.1016/j.chemosphere.2019.06.023
Acknowledgements
The authors would like to thank Manuele Scatena for his valuable technical assistance.
Funding
This research was carried out with the support of the Research & Technological Innovation Department of Eni S.p.A, San Donato Milanese (Italy), and funded by Syndial S.p.A. All activities were conducted at the Italian National Research Council, CNR Area in Pisa, Italy.
Author information
Authors and Affiliations
Contributions
Conceptualization: Meri Barbafieri and Gianniantonio Petruzzelli; Methodology: Meri Barbafieri, Gianniantonio Petruzzelli, and Francesca Pedron; Data curation: Martina Grifoni, Irene Rosellini, and Francesca Pedron; Formal analysis and investigation: Martina Grifoni and Irene Rosellini; Writing—original draft preparation: Martina Grifoni and Meri Barbafieri; Writing—review and editing: Martina Grifoni, Meri Barbafieri, Gianniantonio Petruzzelli, Elisabetta Franchi, and Francesca Pedron; Funding acquisition: Meri Barbafieri, Gianniantonio Petruzzelli, and Elisabetta Franchi; Supervision: Meri Barbafieri, Martina Grifoni, Gianniantonio Petruzzelli, and Elisabetta Franchi.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Not applicable.Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Elena Maestri
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Grifoni, M., Rosellini, I., Petruzzelli, G. et al. Application of sulphate and cytokinin in assisted arsenic phytoextraction by industrial Cannabis sativa L.. Environ Sci Pollut Res 28, 47294–47305 (2021). https://doi.org/10.1007/s11356-021-14074-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-14074-3