Abstract
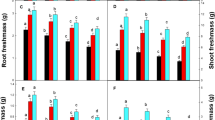
The mechanism of Cu tolerance in plants and its control measures are of considerable significance for the remediation of Cu-contaminated soils. Gibberellic acid (GA3) is involved in plant growth and development and in the response to heavy metal stress. In the present study, changes in the biomass, oxidative stress response responses, and photosynthesis of spinach seedlings were examined under Cu stress with exogenous GA3 applied at concentrations of 0, 3, 5, 10, 20, 40, 60, or 80 mg L−1. Under Cu stress, the plant Cu concentration and oxidative damage were greater, photosynthetic parameters and biomass declined, and antioxidant enzyme activities and the proline concentration increased. However, spinach growth did not terminate, indicating that spinach seedlings had strong Cu tolerance. When low concentrations of GA3 (3–5 mg L−1) were added to Cu-stressed spinach seedlings, the damage caused by Cu stress to spinach seedlings was reduced, and the Cu tolerance of spinach seedlings was enhanced, which mainly manifested as reduced oxidation damage, an increased proline concentration, elevated antioxidant enzyme activities, decreased Cu concentration in leaves, and increased Cu concentration in roots, increased photosynthetic parameters, and an increased in the total biomass. In contrast, additions of GA3 at concentrations higher than 40 mg L−1 intensified oxidative damage and decreased the activities of antioxidant enzymes, photosynthetic parameters, and biomass. Additionally, the Cu concentration increased in leaves and decreased Cu concentration in roots, indicating that high concentrations of GA3 aggravated stress damage and severely influenced physiological functions in spinach seedlings. In summary, the application of 3–5 mg L−1 GA3 to spinach seedlings in Cu-contaminated soil can be used to reduce Cu toxicity to plants and increase Cu tolerance.




Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
Abbreviations
- FW:
-
Fresh weight
- DW:
-
Dry weight
- MDA:
-
Malondialdehyde
- TCA:
-
Trichloroacetic acid
- SOD:
-
Superoxide dismutase
- POD:
-
Peroxidase
- CAT:
-
Catalase
- APX:
-
Ascorbate peroxidase
- ROS:
-
Reactive oxygen species
- Chl a/b:
-
Chlorophyll a/b
- PSII:
-
Photosystem II
- P n :
-
Net photosynthetic rate
- g s :
-
Stomatal conductance
- C i :
-
Internal CO2 concentration
- T r :
-
Transpiration rate
- Fv/Fm:
-
Maximum quantum yield of PSII photochemistry
- qP:
-
Photochemical quenching
- NPQ:
-
Nonphotochemical quenching
- ETR:
-
Electron transport rate
- NBT:
-
Nitroblue tetrazolium
References
Abd EI-Monem MS, Ibrahim IF, Mahmoud RS (2009) Role of gibberellic acid in abolishing the detrimental effects of Cd and Pb on broad bean and lupin plants. Res J Agric Biol Sci 5:668–673
Abdelkader AF (2015) Tolerance strategies of some GA3-pretreated edible plants during growing under drought stress. Egyptian Soc ExpBiolo 11(2):217–225
Acosta JA, Faz A, Martínez-Martínez S, Zornoza R, Carmona DM, Kabas S (2011) Multivariate statistical and GIS-based approach to evaluate heavy metals behavior in mine sites for future reclamation. J Geochem Explor 109:8–17. https://doi.org/10.1016/j.gexplo.2011.01.004
Aehle E, Grandic S, Reynaudle R, Ralainirina S, Baltorarosset F, Mesnard C, Prouillet C, Maziere JC, Fliniaux MA (2004) Development and evaluation of an enriched natural antioxidant preparation obtained from aqueous spinach (Spinacia oleracea) extracts by an adsorption procedure. Food Chem 86:579–585. https://doi.org/10.1016/j.foodchem.2003.10.006
Agami RA (2016) Pre-soaking in indole-3-acetic acid or spermidine enhances copper tolerance in wheat seedlings. S Afr J Bot 104:167–174. https://doi.org/10.1016/j.sajb.2015.10.003
Akman Z (2012) Effects of GA and kinetin pre-sowing treatments on seeding emergence and seeding growth in wheat under saline conditions. J anim Vet Adv 8(2):362–367. https://doi.org/10.1016/j.fsi.2008.03.014
Alia N, Sardar K, Said M, Salma K, Sadia A, Sadaf S, Toqeer A, Miklas S (2015) Toxicity and bioaccumulation of heavy metals in spinach (Spinacia oleracea) grown in a controlled environment. Int J Environ Res Public Health 12:7400–7416. https://doi.org/10.3390/ijerph120707400
Alonso-Ramirez A, Rodriguez D, Reyes JA, Jimenez G, Nicolas M, Lopez-Climent A, Gomez-Cadenas, Nicolas C (2009) Evidence for a role of gibberellins in salicylic acid-modulated early plant responses to abiotic stress in Arabidopsis seeds. Plant Physiol 150:1335–1344. https://doi.org/10.1104/pp.109.139352
Arnon DI (1949) Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol 24:1–15
Batish DR, Singh HP, Setia N, Kaur S, Kohli RK (2006) Effect of 2-benzoxazolinone (BOA) on seedling growth and associated biochemical changes in mung bean (Phaseolus aureus). Zeitschrift Fur Naturforschung C A J Biosci 61:709–714. https://doi.org/10.1515/znc-2006-9-1017
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Becker C, Klaering HP, Kroh LW, Krumbein A (2014) Cool-cultivated red leaf lettuce accumulates cyanidin-3-O-(6″-O-malonyl)-glucoside and caffeoylmalic acid. Food Chem 146:404–411. https://doi.org/10.1016/j.foodchem.2013.09.061
Ben Massoud M, Inès K, Ferjani EE, Chaoui A (2017) Alleviation of copper toxicity in germinating pea seeds by IAA, GA3, Ca and citric acid. J Plant Interact 13:21–29. https://doi.org/10.1080/17429145.2017.1410733
Cai L, Wang Q, Luo J, Chen L, Zhu R, Wang S, Tang C (2019) Heavy metal contamination and health risk assessment for children near a large Cu-smelter in central China. Sci Total Environ 650:725–733. https://doi.org/10.1016/j.scitotenv.2018.09.081
Chandrasekhar, C., Ray, J.G. 2017. Copper accumulation, localization and antioxidant response in Eclipta alba L. in relation to quantitative variation of the metal in soil. Acta Physiol Plant, 39. https://doi.org/10.1007/s11738-017-2508-4.
Chen J, Shafi M, Li S, Wang Y, Wu J, Ye Z, Peng D, Yan W, Liu D (2015) Copper induced oxidative stresses, antioxidant responses and phytoremediation potential of Moso bamboo (Phyllostachys pubescens). Sci Rep 5:13554. https://doi.org/10.1038/srep13554
Cheng H, Hu Y, Luo J, Xu B, Zhao J (2009) Geochemical processes controlling fate and transport of arsenic in acid mine drainage (AMD) and natural systems. J Hazard Mater 165:13–26. https://doi.org/10.1016/j.jhazmat.2008.10.070
Dai H, Xu Y, Zhao L, Shan C (2016) Alleviation of copper toxicity on chloroplast antioxidant capacity and photosystem II photochemistry of wheat by hydrogen sulfide. Braz J Botany 39:787–793. https://doi.org/10.1007/s40415-016-0250-6
Diao M, Ma L, Wang J, Cui J, Fu A, Liu H (2014) Selenium promotes the growth and photosynthesis of tomato seedlings under salt stress by enhancing chloroplast antioxidant defense system. Plant Growth Regul 33:71–682. https://doi.org/10.1007/s00344-014-9416-2
Dresler S, Hanaka A, Bednarek W, Maksymiec W (2014) Accumulation of low-molecular- weight organic acids in roots and leaf segments of Zea mays plants treated with cadmium and copper. Acta Physiol Plant 36:1565–1575. https://doi.org/10.1007/s11738-014-1532-x
Falkowska M, Pietryczuk A, Piotrowaka A, Bajguz A, Grygoruk A, Czerpak R (2011) The effect of gibberellic acid (GA3) on growth, metal biosorption and metabolism of the green algae Chlorella vulgaris (Chlorophyceae) Beijerinck exposed to cadmium and lead stress. Pol J Environ Stud 20:53–59. https://doi.org/10.1002/ldr.1042
Fujita M, Fujita Y, Noutoshi Y, Takahashi F, Narusaka Y, Yamaguchi-Shinozaki K, Shinozaki K (2006) Crosstalk between abiotic and biotic stress responses: a current view from the points of convergence in the stress signaling networks. Curr Opin Plant Biol 9:436–442. https://doi.org/10.1016/j.pbi.2006.05.014
Gong Q, Wang L, Dai T, Zhou J, Kang Q, Chen H, Li K, Li Z (2019) Effects of copper on the growth, antioxidant enzymes and photosynthesis of spinach seedlings. Ecotoxicol Environ Saf 171:771–780. https://doi.org/10.1016/j.ecoenv.2019.01.016
Gou J, Strauss SH, Tsai CJ, Fang K, Chen Y, Jiang X (2010) Gibberellins regulate lateral root formation in Populus through interactions with auxin and other hormones. Plant Cell 22:623–639. https://doi.org/10.1105/tpc.109.073239
Hadi F, Bano A, Fuller MP (2010) The improved phytoextraction of lead (Pb) and the growth of maize (Zea mays L.): the role of plant growth regulators (GA3 and IAA) and EDTA alone and in combinations. Chemosphere. 80:457–462
Hiscox JD, Israeltem GF (1979) A method for extraction of chlorophyll from leaf tissue without maceration using dimethyl sulfoxide. Rev Can Bot 57:1332–1334. https://doi.org/10.1139/b79-163
Ji P, Tang X, Jiang Y, Tong Y, Gao P, Han W (2015) Potential of gibberellic acid 3 (GA3) for enhancing the phytoremediation efficiency of Solanum nigrum L. Bull Environ Contam Toxicol 95:810–814. https://doi.org/10.1007/s00128-015-1670-x
Kaur Kohli S, Handa N, Bali S, Arora S, Sharma A, Kaur R, Bhardwaj R (2018) Modulation of antioxidative defense expression and osmolyte content by co-application of 24-epibrassinolide and salicylic acid in Pb exposed Indian mustard plants. Ecotoxicol Environ Saf 147:382–393. https://doi.org/10.1016/j.ecoenv.2017.08.051
Khan MI, Ahmad N, Anis M (2011) The role of cytokinins on in vitro shoot production in Salix tetrasperma Roxb.: a tree of ecological importance. Trees. 25:577–584. https://doi.org/10.1007/s00468-010-0534-6
Krishnamurthy P, Ranathunge K, Franke R, Prakash HS, Schreiber L, Mathew MK (2009) The role of root apoplastic transport barriers in salt tolerance of rice (Oryza sativa L.). Planata 230(1):119–134. https://doi.org/10.1007/s00425-009-0930-6
Leul M, Zhou WJ (1999) Alleviation of Waterlogging Damage in Winter Rape by Uniconazole Application: Effects on Enzyme Activity, Lipid Peroxidation, and Membrane Integrity. J Plant Growth Regul 18(1):9–14. https://doi.org/10.1007/PL00007046
Li G, Sun G, Williams P, Nunes L, Zhu Y (2011) Inorganic arsenic in Chinese food and its cancer risk. Environ Int 37:1219–1225. https://doi.org/10.1016/j.envint.2011.05.007
Lisiewska Z, Kmiecik W, Gębczyński P, Sobczyńska L (2011) Amino acid profile of raw and as-eaten products of spinach (Spinacia oleracea L.). Food Chem 126:460–465. https://doi.org/10.1016/j.foodchem.2010.11.015
Lu Y, Jiang P, Liu S, Gan Q, Cui H, Qin S (2010) Methyl jasmonate or gibberellins A3-induced astaxanthin accumulation is associated with up-regulation of transcription of beta-carotene ketolase genes (bkts) in microalga Haematococcus pluvialis. Bioresour Technol 101:6468–6474. https://doi.org/10.1016/j.biortech.2010.03.072
Lu Q, Zhang T, Zhang W, Su C, Yang Y, Hu D, Xu Q (2018) Alleviation of cadmium toxicity in Lemna minor by exogenous salicylic acid. Ecotoxicol Environ Saf 147:500–508. https://doi.org/10.1016/j.ecoenv.2017.09.015
Lu H, Waichin L, Nora Fungyee T, Zhihong Y (2019) Effects of root morphology and anatomy on cadmium uptake and translocation in rice (Oryza sativa L.). J Environ Sci 075(001):296–306. https://doi.org/10.1016/j.jes.2018.04.005
Maggio A, Barbieri G, Raimondi G, Pascale S (2010) Contrasting effects of GA treatments to tomato plants exposed to increasing salinity RID A-5646-2011. J plant growth regul 29(1): 63–72. https://doi.org/10.1007/s00344-009-9114-7
Ministry of Environmental Protection, 2014. A national survey of soil pollution bulletin.
Możdżeń K, Wanic T, Rut G, Łaciak T, Rzepka A (2016) Toxic effects of high copper content on physiological processes in Pinus sylvestris L. Photosynthetica 55:193–200. https://doi.org/10.1007/s11099-016-0229-3
Muniandi, Sures Kumar M, Hossain MA, Abdullah MP, Ab Shukor NA (2018) Gibberellic acid (GA3) affects growth and development of some selected kenaf (Hibiscus cannabinus L.) cultivars. Ind Crop Prod 118:180–187. https://doi.org/10.1016/j.indcrop.2018.03.036
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Naz S, Anjum MA, Akhtar S (2016) Monitoring of growth, yield, biomass and heavy metals accumulation in spinach grown under different irrigation sources. Int J Agric Biol 18:689–697. https://doi.org/10.17957/ijab/15.0129
Ouzounidou G, Ilias I (2005) Hormone-induced protection of sunflower photosynthetic apparatus against copper toxicity. Biol Plant 49(2):223–228. https://doi.org/10.1016/S0079-6123(03)46004-4
Piotrowska-Niczyporuk A, Bajguz A, Zambrzycka E, Godlewska-Zylkiewicz B (2012) Phytohormones as regulators of heavy metal biosorption and toxicity in green alga Chlorella vulgaris (Chlorophyceae). Plant Physiol Biochem 52:52–65. https://doi.org/10.1016/j.plaphy.2011.11.009
Poschenrieder C, Bech J, Llugany M, Pace A, Fenés E, Barceló J (2001) Copper in plant species in a copper gradient in Catalonia (North East Spain) and their potential for phytoremediation. Plant Soil 230:247–256
Rombel-Bryzek A, Rajfur M, Zhuk O (2017) The impact of copper ions on oxidative stress in garden cress Lepidium sativum. Ecol Chem Eng S 24. https://doi.org/10.1515/eces-2017-0041
Saeid R, Abooalfazl A (2019) The application of plant growth regulators to improve phytoremediation of contaminated soils. Chemosphere. 220:818–827. https://doi.org/10.1016/j.chemosphere.2018.12.203
Sharma SS, Dietz KJ (2006) The significance of amino acids and amino acid-derived molecules in plant responses and adaptation to heavy metal stress. J Exp Bot 57:711–726. https://doi.org/10.1093/jxb/erj073
Song Y, Zhang H, Chen C, Wang G, Zhuang K, Cui J, Shen Z (2014) Proteomic analysis of copper-binding proteins in excess copper-stressed rice roots by immobilized metal affinity chromatography and two-dimensional electrophoresis. Bio Metals 27:265–276. https://doi.org/10.1007/s10534-014-9707-x
Sun Z, Xie X, Wang P, Hu Y, Cheng H (2018) Heavy metal pollution caused by small-scale metal ore mining activities: a case study from a polymetallic mine in South China. Sci Total Environ 639:217–227. https://doi.org/10.1016/j.scitotenv.2018.05.176
Tamas I, Schwartz J, Hagin J, Simmonds R (1974) Mech Regul Plant Growth
Tewari RK, Kumar P, Sharma PN, Bisht SS (2002) Modulation of oxidative stress responsive enzymes by excess cobalt. Plant Sci 162:381–388
Tuna AL, Kaya C, Dikilitas M, Higgs D (2008) The combined effects of gibberellic acid and salinity on some antioxidant enzyme activities, plant growth parameters and nutritional status in maize plants. Environ Exp Bot 62:1–9. https://doi.org/10.1016/j.envexpbot.2007.06.007
Vinit-Dunand F, Epron D, Alaoui-Sossé B, Badot PM (2002) Effects of copper on growth and on photosynthesis of mature and expanding leaves in cucumber plants. Plant Sci 163:53–58. https://doi.org/10.1016/S0168-9452(02)00060-2
Wang GL, Que F, Xu Z, Wang F, Xiong A (2015) Exogenous gibberellin altered morphology, anatomic and transcriptional regulatory networks of hormones in carrot root and shoot. BMC Plant Biol 15:290–295. https://doi.org/10.1186/s12870-015-0679-y
Yruela I (2009) Copper in plants: acquisition, transport and interactions. Funct Plant Biol 36:409–430
Yuankun L, Qi T, Xinyu G, Jipeng L, Jinxing L, Yongchao L, Tingqiang L (2020) Low calcium-induced delay in development of root apoplastic barriers enhances Cd uptake and accumulation in Sedum alfredii. Sci Total Environ 25:723–734. https://doi.org/10.1016/j.scitotenv.2020.137810
Zhang Y, Xu S, Yang S, Chen Y (2015) Salicylic acid alleviates cadmium-induced inhibition of growth and photosynthesis through upregulating antioxidant defense system in two melon cultivars (Cucumis melo L.). Protoplasma. 252:911–924. https://doi.org/10.1007/s00709-014-0732-y
Zhang JH, Li Z, Sun HL, Wu H, Chen SY (2017) Adversity stress-related responses at physiological attributes, transcriptional and enzymatic levels after exposure to Cu in Lycopersicum esculentm seedlings. Sci Hortic 222:213–220. https://doi.org/10.1016/j.scienta.2017.05.027
Zhang X, Zha T, Guo X, Meng G, Zhou J (2018) Spatial distribution of metal pollution of soils of Chinese provincial capital cities. Sci Total Environ 643:1502–1513. https://doi.org/10.1016/j.scitotenv.2018.06.177
Zhu X, Jiang T, Wang Z, Lei G, Shi Y, Li G, Zheng S (2012) Gibberellic acid alleviates cadmium toxicity by reducing nitric oxide accumulation and expression of IRT1 in Arabidopsis thaliana. J Hazard Mater 239-240:302–307. https://doi.org/10.1016/j.jhazmat.2012.08.077
Funding
This work was funded by grants from key projects of the Hubei Natural Fund (innovation group) of China (2016CFA016) and the research project of Xinjiang Agricultural Vocational and Technical College (XJNZYKJ201901).
Author information
Authors and Affiliations
Contributions
Qong Gin, Ling Wang, and Jing-yi Zhou conducted the experiments, collected and analyzed the samples, and drafted the manuscript. Qun Kang and Duan-dan Niu analyzed the data and revised the manuscript. Zhao-hua Li and Qong Gin conceived and designed this work. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interest
The authors declare no competing interests.
Additional information
Responsible Editor: Philipp Gariguess
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 36 kb)
Rights and permissions
About this article
Cite this article
Gong, Q., Li, Zh., Wang, L. et al. Gibberellic acid application on biomass, oxidative stress response, and photosynthesis in spinach (Spinacia oleracea L.) seedlings under copper stress. Environ Sci Pollut Res 28, 53594–53604 (2021). https://doi.org/10.1007/s11356-021-13745-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-13745-5