Abstract
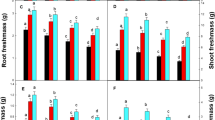
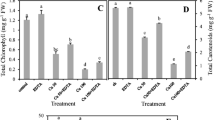
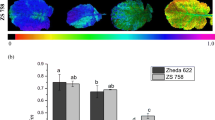
Hydroponic experiments were conducted to compare the effects of excess copper (Cu) on growth and photosynthesis in young Indian mustard (Brassica juncea) and oilseed rape (Brassica napus). We compared the effects of excess Cu on the two Brassica species at different physiological levels from antioxidant levels to photosynthetic activity. Nine-day-old plants were treated with Cu (10, 25 and 50 μM CuSO4) for 7 and 14 days. Both species took up Cu from the external solution to a similar degree but showed slight root-to-shoot translocation. Furthermore, after seven days of treatment, excess Cu significantly decreased other microelement content, such as iron (Fe) and manganese (Mn), especially in the shoots of B. napus. As a consequence, the leaves of young Brassica napus plants showed decreased concentrations of photosynthetic pigments and more intense growth inhibition; however, accumulation of highly reactive oxygen species (hROS) were not detected. After 14 days of Cu exposure the reduction of Fe and Mn contents and shoot growth proved to be comparable in the two species. Moreover, a significant Cu-induced hROS accumulation was observed in both Brassica species. The diminution in pigment contents and photosynthetic efficiency were more pronounced in B. napus during prolonged Cu exposure. Based on all the parameters, B. juncea appears to be more resistant to excess Cu than B. napus, rendering it a species with higher potential for phytoremediation.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Ahmad, M. S. A., Ashraf, M., Tabassam, Q., Hussain, M., Firdous H. (2011) Lead (Pb)- induced regulation of growth, photosynthesis and mineral nutrition in maize (Zea mays L.) plants at early growth stages. Biol. Trace Elem. Res. 144, 1229–1239.
Arshad, M., Murtaza, G., Asada, K. (2006) Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiol. 141, 391–396.
Babu, T. S., Marder, J. B., Tripuranthakam, S., Dixon, D. G., Greenberg, B. M. (2001) Synergistic effects of a photooxidized polycyclic aromatic hydrocarbon and copper on photosynthesis and plant growth: evidence that in vivo formation of reactive oxygen species is a mechanism of copper toxicity. Environ. Toxicol. Chem. 20, 1351–1358.
Boswell, C., Sharma, N. C., Sahi, S. V. (2002) Copper tolerance and accumulation potential of Chlamidomonas reinhardtii. Bull. Environ. Contam. Toxicol. 69, 546–553.
Böddi, B., Oravecz, A. R., Lehoczki, É. (1995) Effect of cadmium on organization and photoreduction of protochlorophyllide in dark-grown leaves and etioplast inner membrane preparations of wheat. Photosynth 31, 411–420.
Burkhead, J. L., Reynolds, K. A. G., Abdel-Ghany, S. E. (2009) Copper homeostasis. New Phytol. 182, 799–816.
Dhindsa, R. S., Plumb-Dhindsa, P., Thorpe, T. A. (1981) Leaf senescence: correlated with increased levels of membrane permeability and lipid peroxidation, and decreased levels of superoxide dismutase and catalase. J. Exp. Bot. 32, 93–101.
Dixon, G. R. (2007) Vegetables Brassicas and related Crucifers. Crop production science in horticulture series:14. Bibbles Ltd, King’s Lynn.
Drążkiewicz, M., Skórzyńska-Polit, E., Krupa, Z. (2004) Copper-induced oxidative stress and antioxidant defence in Arabidopsis thaliana. Biometals 17, 379–387.
Dushenkov, V., Nanda Kumar, P. B. A., Motto, H., Raskin, I. (1995) Rhizofiltration: The use of plants to remove heavy metals from aqueous streams. Environ. Sci. Technol. 29, 1239–1245.
Ebbs, S. D., Kochian, L. V. (1997) Toxicity of zinc and copper to Brassica species: implications for phytoremediation. J. Environ. Qual. 26, 776–781.
Feigl, G., Kumar, D., Lehotai, N., Tugyi, N., Molnár, A., Ördög, A., Szepesi, A., Gémes, K., Laskay, G., Erdei, L., Kolbert, Z. (2013) Physiological and morphological responses of the root system of Indian mustard (Brassica juncea L. Czern.) and rapeseed (Brassica napus L.) to copper stress. Ecotox. Environ. Safety 94, 179–189.
Fellet, G., Marchiol, L., Zerbi, G. (2013) Potential for metal phytoextraction of Brassica oilseed species. In: Naser A. Anjum et al. (eds) Phytotechologies: Remediation of Environmental Contaminants. CRC Press, Taylor & Francis Group, Boca Raton, pp. 180–201.
Halliwell, B., Gutteridge, J. M. C. (1984) Oxygen toxicity, oxygen radical, transition metals and disease. Biochem. J. 219, 1–14.
Jegerschöld, C., Arellano, J. B., Schröder, W. P., van Kan, P. J., Barón, M., Styring, S. (1995) Copper(II) inhibition of electron transfer through photosystem II studied by EPR spectroscopy. Biochem. 34, 12747–12754.
Jegerschöld, C., MacMillan, F., Lubitz, W., Rutherford, A. W. (1999) Effects of copper and zinc ions on photosystem II studied by EPR spectroscopy. Biochem. 38, 12439–12445.
Kolbert, Zs., Pető, A., Lehotai, N., Feigl, G., Ördög, A., Erdei, L. (2012) In vivo and in vitro studies on fluorophore-specificity. Acta Biol. Szeged, 56, 37–41.
Kumar, P. B. A. N., Dushenkov, V., Motto, H., Raskin, I. (1995) Phytoextraction: the use of plants to remove heavy metals from soils. Environ. Sci. Tech. 29, 1232–1238.
Kumar, S., Andy, A. (2012) Health promoting bioactive phytochemicals from Brassica. Int. Food Res. J. 19, 59–66.
Kurepa, J., Hérouart, D., van Montagu, M., Inzé, D. (1997) Differential expression of CuZn- and Fe-superoxide dismutase genes of tobacco during development, oxidative stress, and hormonal treatments. Plant Cell Physiol. 38, 463–470.
Küpper, H., Setlik, I., Spiller, M., Küpper, F., Prasil, O. (2002) Heavy metal-induced inhibition of photosynthesis: targets of in vivo heavy metal chlorophyll formation. J. Phycol. 38, 429–441.
Lahive, E., O’Halloran, J., Jansen, M. A. K. (2012) Frond development gradients are a determinant of the impact of zinc on photosynthesis in three species of Lemnaceae. Aquat. Bot. 101, 55–63.
Law, M. Y., Charles, S. A., Halliwell, B. (1983) Glutathione and ascorbic acid in spinach (Spinacia oleracea) chloroplasts. Biochem. J. 210, 899–903.
Lequeux, H., Hermans, C., Lutts, S., Verbruggen, N. (2010) Response to copper excess in Arabidopsis thaliana: Impact on the root system architecture, hormone distribution, lignin accumulation and mineral profile. Plant Physiol. Biochem. 48, 673–682.
Lichtenthaler, H. K. (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Meth. Enzymol. 148, 350–382.
Lidon, F. C., Henriques, F. S. (1993) Effects of copper toxicity on growth and the uptake and translocation of metals in rice plants. J. Plant Nutr. 16, 1449–1464.
Liu, J., Xiong, Z. T., Li, T. Y., Huang H. (2004) Bioaccumulation and ecophysiological responses to copper stress in two populations of Rumex dentatus L. from Cu contaminated and non-contaminated sites. Environ Exp. Bot. 52, 43–51.
Lombardi, L., Sebastiani, L. (2005) Copper toxicity in Prunus cerasifera: growth and antioxidant enzymes responses of in vitro-grown plants. Plant Sci. 168, 797–802.
Luna, C. M., González, C. A., Trippi, V. S. (1994) Oxidative damage caused by excess of copper in oat leaves. Plant Cell Physiol. 35, 11–15.
Maksymiec, W. (1997) Effect of copper on cellular processes in higher plants. Photosynth 34, 321–342.
Marchiol, L., Sacco, P., Assolari, S., Zerbi, G. (2004) Reclamation of polluted soil: Phytoremediation potential of crop-related Brassica species. Water, Air, and Soil Pollution 158, 345–356.
Mattina, M. J. I., Lannucci-Berger, W., Musante, C., White, J. C. (2003) Concurrent plant uptake of heavy metals and persistent organic pollutants from soil. Environ. Pollut. 124, 375–378.
Møller, I. M., Jensen, P. E., Hansson, A. (2007) Oxidative modifications to cellular components in plants. Annu. Rev. Plant Biol. 58, 459–481.
Nakano, Y., Asada, K. (1981) Hydrogen peroxide is scavenged by ascorbate specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 22, 867–880.
Ouzounidou, G., Lannoye, R., Karataglis, S. (1993) Photoacoustic measurements of photosynthetic activities in intact leaves under copper stress. Plant Sci. 89, 221–226.
Pätsikkä, E., Kairavuo, M., Šeršen, F., Tyystjärvi, E. A. E. (2002) Excess copper predisposes photosystem II to photoinhibition in vivo by outcompeting iron and causing decrease in leaf chlorophyll. Plant Physiol. 129, 1359–1367.
Roháček, K., Soukupová, J., Barták, M. (2008) Chlorophyll fluorescence: A wonderful tool to study plant physiology and plant stress. In: Benoît Schoefs (ed.) Plant Cell Compartments–Selected Topics. Research Signpost, Fort P.O., Trivandrum-695 023, Kerala, pp. 41–104.
Romero-Puertas, M. C., Palma, J. M., Gómez, M., Del Rió, L. A., Sandalio, L. M. (2002) Cadmium causes the oxidative modification of proteins in pea plants. Plant Cell Environ. 25, 677–686.
Russo, M., Sgherri, C., Izzo, R., Navari-Izzo, F. (2008) Brassica napus subjected to copper excess: Phospholipases C and D and glutathione system in signalling. Environ. Exp. Bot. 62, 238–246.
Salt, D. E., Prince, R. C., Pickering, I. J., Raskin, I. (1995) Mechanisms of cadmium mobility and accumulation in Indian mustard. Plant Physiol. 109, 1427–1433.
Sharma, S. S., Dietz, K. J. (2008) The relationship between metal toxicity and cellular redox imbalance. Trends Plant Sci. 14, 43–50.
Shioi, Y., Tamai, H., Sasa, T. (1978) Effects of copper on photosynthetic electron transport systems in spinach chloroplasts. Plant Cell Physiol. 19, 203–209.
Singh, S., Singh, S., Ramachandran, V., Eapen, S. (2010) Copper tolerance and response of antioxidative enzymes in axenically grown Brassica juncea (L.) plants. Ecotoxicol. Environ. Safety 73, 1975–1981.
Taylor, G. J., Foy, C. D. (1985) Differential uptake and toxicity of ionic and chelated copper in Triticum aestivum. Can. J. Bot. 63, 1271–1275.
Upadhyaya, A., Sankhla, D., Davis, T. D., Sankhla, N., Smith, B. N. (1985) Effect of paclobutrazol on the activities of some enzymes of activated oxygen metabolism and lipid peroxidation in senescing soybean leaves. J. Plant Physiol. 121, 453–461.
Zhao, F. J., Lombi, E., McGrath, S. P. (2003) Assessing the potential for zinc and cadmium phytoremediation with the hyperaccumulator Thlaspi caerulescens. Plant Soil 249, 37–43.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Feigl, G., Kumar, D., Lehotai, N. et al. Comparing the Effects of Excess Copper in the Leaves of Brassica Juncea (L. Czern) And Brassica Napus (L.) Seedlings: Growth Inhibition, Oxidative Stress and Photosynthetic Damage. BIOLOGIA FUTURA 66, 205–221 (2015). https://doi.org/10.1556/018.66.2015.2.7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1556/018.66.2015.2.7