Abstract
Background
Stable severe chronic obstructive pulmonary disease (COPD) patients with chronic hypercapnic respiratory failure treated by nocturnal bi-level positive pressure non-invasive ventilation (NIV) may experience severe morning deventilation dyspnea. We hypothesised that in these patients, progressive hyperinflation, resulting from inappropriate ventilator settings, leads to patient–ventilator asynchrony (PVA) with a high rate of unrewarded inspiratory efforts and morning discomfort.
Methods
Polysomnography (PSG), diaphragm electromyogram and transcutaneous capnography (PtcCO2) under NIV during two consecutive nights using baseline ventilator settings on the first night, then, during the second night, adjustment of ventilator parameters under PSG with assessment of impact of settings changes on sleep, patient–ventilator synchronisation, morning arterial blood gases and morning dyspnea.
Results
Eight patients (61 ± 8 years, FEV1 30 ± 8% predicted, residual volume 210 ± 30% predicted) were included. In all patients, pressure support was decreased during setting adjustments, as well as tidal volume, while respiratory rate increased without any deleterious effect on nocturnal PtcCO2 or morning PaCO2. PVA index, initially high (40 ± 30%) during the baseline night, decreased significantly after adjusting ventilator settings (p = 0.0009), as well as subjective perception of PVA leaks, and morning dyspnea while quality of sleep improved.
Conclusion
The subgroup of COPD patients treated by home NIV, who present marked deventilation dyspnea and unrewarded efforts may benefit from adjustment of ventilator settings under PSG or polygraphy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Despite the lack of evidence to support its use in chronic obstructive pulmonary disease (COPD), non-invasive ventilation (NIV) is widely prescribed in this indication in Europe [1]. Although impact on survival remains controversial [2–4], most authors will agree that there appears to be a group of COPD patients who clearly benefit from NIV. Indeed, several studies have described improvements in health-related quality of life [3, 5, 6], hospital admissions [7, 8], dyspnea [6] and quality of sleep [5].
Recent reviews have stressed the importance of identifying abnormal events occurring under NIV, and adjusting ventilator settings accordingly [9]. For instance, it is now established that patient–ventilator asynchrony (PVA) is frequent in patients undergoing chronic NIV who are considered effectively treated [10, 11]. PVA has a negative impact on quality of sleep and potentially on treatment efficacy [10]. Monitoring pulse oximetry (SpO2) and transcutaneous capnography (PtcCO2) alone do not explore this aspect of therapy adequately. Thus, in selected cases, polygraphy or polysomnography (PSG) might be ideal monitoring tools in addition to oxy-capnography for patients with home ventilation [12].
Among severe COPD under home NIV, patients are occasionally burdened by a severe dyspnea when interrupting their treatment after a night of NIV. We hypothesised that in these patients, inappropriate ventilator settings may lead to dynamic hyperinflation, increased intrinsic PEEP, resulting in unrewarded inspiratory efforts [13, 14] and morning discomfort when interrupting NIV [15]. We surmised that adjusting ventilator settings accordingly, during PSG under NIV, might improve quality of sleep, patient–ventilator synchronisation and morning dyspnea (‘deventilation dyspnea’).
Materials and methods
Patients
Eligible patients fulfilled the following inclusion criteria: (1) Severe COPD according to GOLD guidelines [16], (2) home NIV initiated according to prevailing recommendations [17], (3) stable clinical condition defined as absence of exacerbation and of modifications of ventilator settings in the month preceding the study, (4) compliance to NIV of more than 4 h/day assessed by ventilator software, (5) presence of deventilation dyspnea defined as severe dyspnea occurring immediately after interruption of non-invasive ventilation and preventing the patient from getting out of bed and carrying out usual daily activities for more than 30 min after one night of NIV. Severe morning dyspnea was assessed by a chest therapist using a modified Borg scale. All patients with morning dyspnea score >4 were proposed to enter the study. Exclusion criteria were: (1) use of NIV in an acute exacerbation of COPD for hypercapnic ventilatory failure, (2) association of COPD with restrictive thoracic disorders (i.e. morbid obesity, cyphoscoliosis, neuromuscular disease, sequelae of tuberculosis), and (3) patients under 18 years old or not able to give informed consent.
Measurements
Polysomnography
Conventional polysomnography with patient under NIV included: standard electroencephalography (seven electrodes, F4–F3, O1–O2, C3–C4, Cz), left and right electrooculography, submental electromyography (EMG), measurement of airflow using a pneumotachograph, body position, thoracic and abdominal movements (Remlogic 1.1, Embla, Netherlands) and video recording. SpO2 was measured continuously with a pulse oximeter and a finger probe. PtcCO2 measurements were performed using a transcutaneous capnograph with an ear probe heated at 43°C (Tosca 500, Radiometer, Switzerland). Sleep was scored according to standard criteria using 30-s epochs [18]. The investigator scoring the sleep study was blinded to the ventilator settings and subjective evaluation. The following sleep parameters were quantified: total sleep time (TST), sleep efficiency (SE—TST/total recording time × 100), percentage of each sleep stage, wake after sleep onset (WASO) and sleep latency. As indices of sleep fragmentation, we considered the index of micro-arousals (MAI). MAI were defined as a return to alpha or fast frequency, well-differentiated from the background EEG activity lasting >1.5 s.
Diaphragm EMG
Surface diaphragm EMG was recorded using silver electrodes in all subjects only to monitor inspiratory effort [19] and facilitate detection of ineffective inspiratory efforts. The electrodes were placed in the right and left anterior axillary line in the seventh intercostal space with a horizontal distance of 2 cm. Diaphragmatic EMG was sampled at 0.2 kHz, bandpass-filtered between 10 and 70 Hz [20] and recorded with all other parameters on the Embla recording system.
Patient–ventilator asynchrony
Patterns of patient–ventilator asynchrony included ineffective triggering (unrewarded inspiratory efforts), double-triggering and auto-triggering, as defined by Thille et al. [13]. Unrewarded inspiratory efforts were identified as the occurrence of a definite diaphragmatic EMG signal, and/or presence of inspiratory thoracoabdominal movements on strain gauges without subsequent pressurisation by the ventilator. Auto-triggering was defined as a cycle delivered by the ventilator and not triggered by the patient. An unrewarded effort index was calculated as follows: [number of desynchronized cycles/total respiratory rate (i.e. cycles supported by the ventilator plus unrewarded efforts)] × 100 (expressed as a percentage) [21]. Only epochs (3 min.) with an unrewarded effort index above 10% were scored as periods with asynchrony based on a study by Vittaca and al. [22]. PVA index was calculated as the ratio of total sleep time spent with unrewarded efforts on TST [10].
Ventilator built-in software
Compliance, estimated tidal volume (VT), minute ventilation (VE), respiratory rate (RR), leaks and percent (%) of spontaneous inspirations were downloaded after both nights from built-in software of bi-level ventilators (ResScan software, version 1.3, ResMed, Sydney, Australia). Estimation of leaks and VT by ventilator software of VPAP III and VPAP IV ventilators (ResMed, Sydney, Australia) has been shown to be reliable in two recent independent studies [23, 24].
Questionnaires
After both nights, to evaluate patient comfort and patients’ perception of patient–ventilator synchronisation, airflow, leaks and noise of ventilator, an eight-item visual analogue scale (VAS) was administered the morning following each sleep study [25]. Higher values indicate better comfort. Patients were also asked to quantify their dyspnea on a visual analogue scale while withdrawing from pressure support in the morning (10—worst dyspnea ever experienced in the morning, 0—no dyspnea at all).
Study protocol
Patients were admitted to the sleep laboratory for two consecutive nights. On the first night, ventilator settings were those normally used by the patient, thus eliminating the need of an accommodation night. This first night aimed to detect and quantify PVA in highly symptomatic COPD patients on their usual NIV settings. In our institution, ventilation settings are adjusted by chest therapists, under the supervision of the attending pulmonary physician, aiming at patient comfort and optimal control of daytime and nocturnal SpO2 and PtcCO2. On the second night, the therapeutic target was to reduce sleep-disordered breathing and patient–ventilator asynchrony. The investigators adjusted ventilator settings using polysomnography and monitoring of SpO2/PtcCO2.
Our approach was as follows: in case of unrewarded inspiratory efforts, we first reduced pressure support, then, if necessary, we increased expiratory positive airway pressure (EPAP) [15] and set expiratory trigger at a higher percentage of peak inspiratory flow to avoid delayed cycling [26, 27] under close monitoring of PtcCO2. When time to peak pressure (pressurisation time) was set at 100 msec, we increased it by 50–100 msec. Back-up respiratory rate was increased if unrewarded inspiratory efforts persisted. Maximal inspiratory time (TIMAX) was calculated in order to maintain a physiological I/E ratio for COPD patients between 1:2.5 and 1:3.
If auto-triggering was identified, mask fit was adapted to prevent air leaks and inspiratory trigger sensitivity was reduced [28]. Inspiratory time (TIMIN) was increased if double triggering was frequent [13]. Special attention was given to mask leaks in order to limit their impact on patient–ventilator asynchrony.
Statistical analysis
Statistical testing was performed using Prism4 software (GraphPad, San Diego, CA, USA). When data were normally distributed (Schapiro–Wilk test), results were expressed as mean±SD. All variables between the two nights of the study protocol were compared using a paired t test. Differences were considered significant when probability of a type I error was <0.05.
The study protocol was approved by the Ethics Committee for Medical Research of Geneva University Hospital and written informed consent was obtained from all patients. The trial was registered at www.clinicaltrials.gov (Trial No. NCT01180439)
Results
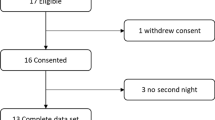
The study flow chart is depicted in Fig. 1. Among severe COPD patients treated by our outpatients clinic, 9/32 (28%) exhibited ‘deventilation dyspnea’ and fulfilled inclusion criteria. We included eight consecutive COPD patients (aged 61 ± 8.3 years, three males, five females, on NIV for 14.3 ± 12.5 months), admitted for a scheduled routine control of their nocturnal ventilation (n = 4 on VPAP ST III, n = 4 on VPAP IV bi-level ventilators; ResMed, Sydney, Australia) between October 2009 and March 2010. One patient declined. Patients’ main characteristics are shown in Table 1. All patients used their ventilator more than 4 h per night (Mean±SD9:06 ± 3:20 hours) and suffered from severe morning dyspnea after interrupting their NIV (referred to as ‘deventilation dyspnea’).
Ventilator settings at baseline and adjustments under PSG
Baseline ventilator settings and settings after adjustment under PSG are shown in Table 2. Seven patients used full-face masks and one patient used a nasal interface. The major change performed in order to decrease unrewarded inspiratory efforts detected on PSG, was a decrease in pressure support in all cases ([IPAP–EPAP] baseline, 13.6 ± 1.8 cm H2O; adjusted, 10.3 ± 1.7 cm H2O; p = .0005). In seven cases, IPAP was reduced by 1–5 cm H2O. In four cases, EPAP was also increased by 1–4 cm H2O. Back-up rate was increased in four cases. Time to peak was slightly increased in all cases by 50–100 msec. TIMAX was adjusted to obtain a I/E ratio between 1:2.5 and 1:3.
Sleep structure and respiratory events under NIV
Unrewarded inspiratory efforts were the most prevalent patient–ventilator asynchrony. PVA index was reduced after ventilator adjustment from 40.5 ± 31.0 to 6.7 ± 7.3%, p = 0.009 (Fig. 2). Neither double triggering nor auto-triggering were identified during both nights. Figure 3 shows a representative correction of unrewarded inspiratory efforts by NIV adjustment under sleep polygraphy.
a Example of unrewarded inspiratory efforts with initial ventilator settings. From top to bottom—flow, pressure, diaphragm EMG, thoracic and abdominal belts. This 60-s epoch shows major patient–ventilator asynchrony. Unrewarded efforts can easily be identified by comparing the EMG trace or that of the abdominal and thoracic belts with the flow or pressure traces. The patients’ inspiratory efforts do not trigger the ventilator which is on its back-up rate. b Correction of patient–ventilator asynchrony by adjustment of ventilator settings with polysomnography (60-s epoch). Flow, pressure, diaphragm EMG and abdominal and thoracic belts are perfectly synchronised. All inspiratory efforts trigger the ventilator resulting in an increased respiratory rate
Neither total sleep time (TST 445 ± 126 vs. 448 ± 96 min), wake after sleep onset (WASO 41 ± 21 vs. 41 ± 24 min), nor micro arousal index (MAI 18 ± 9 vs. 20 ± 12 h) were altered by changes in ventilator settings.
Subjective assessment
The major impact of ventilator adjustment was a marked decrease in morning dyspnea measured by an adapted Borg Scale (values from 0 to 10, increasing values mean increasing dyspnea), for all subjects as shown in Fig. 3 (5.8 ± 2.5 vs. 2.3 ± 1.6, p = 0.006).
The eigh-point VAS also showed significant improvements for perception of patient–ventilator synchronisation (p = 0.04), perception of leaks (p = 0.04) and overall quality of sleep (p = 0.01) (Fig. 4). None of the VAS scales decreased (i.e. worsened).
Correction of deventilation dyspnea by adjusting ventilator settings with polysomnography is illustrated for each individual patient on a modified Borg scale (a, upper left). On a 0–10 visual analogue scale, subjective assessment of ventilator synchronisation (b), leaks (c) and overall quality of sleep (d) all improved
Data provided by ventilator built-in software
Changes in estimated tidal volume (VT), total ventilation (VE), spontaneous respiratory rate (RR) and percentage of inspiratory cycles triggered by the patient are reported in Table 3. Ventilation pattern changed after adjusting ventilator parameters, with a significant decrease in VT, and increase in spontaneous RR. The increase in percentage of inspiratory cycles triggered by the patient most probably reflects a better patient–ventilator synchronisation and improved detection of inspiratory efforts by the ventilator [12]. Average leaks were low during both recordings for all patients with a nonsignificant trend for lower leaks (95% percentile) after adjusting ventilator settings (28 ± 25 vs. 17 ± 19 L/min, p = 0.057).
Nocturnal SpO2 and nocturnal/daytime CO2 values
Mean nocturnal SpO2 was 92 ± 3% on the first night and 91 ± 2% after adjustment of settings under PSG (p value NS). Oxygen desaturation index was low and did not differ between both nights (3.8 ± 6.0 vs. 3.5 ± 2.9, p value NS). Despite a significant reduction in pressure support after adjusting ventilator settings, mean PtcCO2 decreased slightly although not significantly (7.5 ± 1.2 vs. 7.3 ± 1.0 kPa). Average morning PaCO2 under NIV also decreased after adjustment of ventilator settings (7.6 ± 1.3 vs. 7.3 ± 1.5 kPa, p = 0.057).
Discussion
Among severe COPD patients treated at home by NIV, nearly one third complained of ‘deventilation dyspnea’. Our study shows in this population that a very high rate of patient–ventilator asynchrony may occur (mainly unrewarded inspiratory efforts) and remain undetected without PSG. Adjusting ventilator settings after monitoring of nocturnal NIV by PSG can: (1) improve markedly patient–ventilator synchronisation, particularly triggering of ventilator; (2) improve patient comfort; (3) lead to a marked decrease in morning ‘deventilation dyspnea’; (4) change ventilation pattern to a more physiological mode for severe COPD, i.e. increase spontaneous respiratory rate with a slight decrease in tidal volume, without any deterioration of PaCO2 or PtcCO2.
The observation that a high rate of unrewarded efforts occurred and was undetected in these patients confirms previous observations showing a high rate of patient–ventilator asynchrony in stable patients under NIV, most often not reported by the patient [10, 11, 14]. Although systematic polysomnography is clearly not warranted in stable patients under NIV, in these eight cases, PSG was indicated by the importance of morning deventilation dyspnea and the modest impact of NIV on PtcCO2 [12]. Adjusting ventilator settings had a nonsignificant impact on arterial blood gases, but a marked effect on patient comfort, patient–ventilator synchrony and morning dyspnea. Whether these changes are relevant as major endpoints of NIV is debatable, but it seems reasonable to assume that these improvements may have an impact on quality of life and acceptance of treatment. Indeed, morning deventilation dyspnea is a source of important discomfort and distress in these patients.
It may be objected that correction of alveolar hypoventilation in our patients was insufficient. Indeed, both morning arterial blood gases and nocturnal capnography show a residual hypercapnia. Recent publications by Windisch et al. suggest that normalising PaCO2 under NIV may be an important goal, through the use of high levels of pressure support and high back-up respiratory rates: this approach was shown to be feasible, to improve daytime PaCO2, daytime dyspnea, HRQL and FEV1 [6, 29, 30]. The use of high-intensity ventilation and its application in clinical practice still remains a subject of debate. Indeed, several publications on long-term NIV in COPD have used much lower insufflation pressures [2–5]. In our centre, our aim is to obtain the best compromise between reduction of PaCO2/PtcCO2, induced leaks and patient tolerance. Thus, for all patients, initial settings of pressure support were as high as tolerated. Residual hypercapnia in very severe COPD treated by NIV is frequently reported. All patients included in our study had severe COPD according to GOLD criteria, had had several exacerbations with acute hypercapnic respiratory failure, and had a baseline PaCO2 >7.3 kPa.
The mechanisms suggested for the relationship between nocturnal PSG findings and ‘deventilation dyspnea’ are hypothetical and probably differ according to the underlying pathology. In selected subjects with severe COPD and static hyperinflation, excessive pressure support might lead to dynamic hyperinflation and increased PEEPi [31] which are major determinants of unrewarded efforts [21]. Dynamic hyperinflation has been shown to correlate strongly with exercise-induced dyspnea in COPD [32] and may explain the importance of ‘deventilation dyspnea’. Therefore, patient–ventilator asynchrony may, in these subjects, be a marker of inappropriate ventilator settings as demonstrated in the acute setting [26]. Arguments in favour of such a mechanism are—in the patients studied—severity of bronchial obstruction and the presence of static air trapping which may curtail the patients’ ability to take in large tidal volumes on NIV without developing dynamic hyperinflation (Table 1). However, the setting of this study did not allow performing serial measurements of inspiratory capacity after cessation of NIV; furthermore, PEEPI could not be estimated non-invasively.
NIV can thus be considered as a two-edged sword in severe COPD. On one hand, Diaz et al. have shown that efficient NIV may decrease residual volume, functional residual capacity and total lung capacity when compared to sham NIV [33]. On the other hand, inappropriately high pressure support may have in certain patients a negative impact on lung mechanics and increase unrewarded efforts and patient–ventilator asynchrony [26]. Finding the best compromise between too high pressure support (with patient–ventilator asynchrony), efficacy of ventilation and patient comfort under monitoring by polygraphy or PSG might be a relevant clinical target.
Limitations of this study are its non-blinded and non-randomised design. The sequence of night sleep studies were dictated by necessity of a pragmatic step-by-step assessment: the first night to identify patient–ventilator asynchrony during sleep, the second to correct them under appropriate monitoring. As patients were long-time NIV users, the first night effect on sleep quality can be considered as negligible. Moreover, ineffective efforts are objective measurements not influenced by randomization. Thus, we believe that the initial PSG reliably reflected patient–ventilator interactions at home. The advantage of our study design was to validate an effective and feasible procedure for NIV adjustment in clinical practice for COPD complaining from ‘deventilation dyspnea’. The sample size is another limitation of our study. This may have precluded the demonstration of significant improvements in objective sleep quality which is suggested by nearly significant increases in sleep efficiency (69 ± 18% vs. 76 ± 13%, n.s.) and slow wave and REM sleep (% N3 + REM 29 ± 12% vs. 38 ± 14%, n.s.).
In summary, this study shows that in a selected group of stable severe COPD with hyperinflation treated by NIV, patient–ventilator asynchrony and ineffective inspiratory efforts are frequent, most probably increasing work of breathing and decreasing the efficacy of ventilation; this observation may explain the occurrence of morning deventilation dyspnea. After a diagnostic PSG or polygraphy, adjusting ventilator settings may lead to a significant decrease of ineffective inspiratory efforts and a significant improvement of patient comfort during and after NIV.
Abbreviations
- COPD:
-
Chronic obstructive pulmonary disease
- EEG:
-
Electroencephalogram
- EMG:
-
Electromyogram
- EPAP:
-
Expiratory positive airway pressure
- I/E ratio:
-
Inspiratory/expiratory ratio
- IPAP:
-
Inspiratory positive airway pressure
- kHz:
-
Kilohertz
- kPa:
-
Kilopascals
- MAI:
-
Microarousal index
- NIV:
-
Non-invasive ventilation
- PaCO2 :
-
Arterial partial pressure of carbon dioxide
- PEEP:
-
Positive end-expiratory pressure
- PEEPI :
-
Intrinsic positive end-expiratory pressure
- PSG:
-
Polysomnography
- PtcCO2 :
-
Transcutaneous measurement of arterial partial pressure of carbon dioxide
- PVA:
-
Patient–ventilator asynchrony
- RR:
-
Respiratory rate
- SE:
-
Sleep efficiency
- SpO2 :
-
Oxygen saturation of Haemoglobin measured by pulsoximetry
- TIMAX :
-
Maximal inspiratory time
- TST:
-
Total sleep time
- VE:
-
Minute ventilation
- VT :
-
Tidal volume
- WASO:
-
Wake after sleep onset
References
Lloyd-Owen SJ, Donaldson GC, Ambrosino N, Escarabill J, Farre R, Fauroux B, Robert D, Schoenhofer B, Simonds AK, Wedzicha JA (2005) Patterns of home mechanical ventilation use in Europe: results from the Eurovent survey. Eur Respir J 25(6):1025–1031
Casanova C, Celli BR, Tost L, Soriano E, Abreu J, Velasco V, Santolaria F (2000) Long-term controlled trial of nocturnal nasal positive pressure ventilation in patients with severe COPD. Chest 118(6):1582–1590
Clini E, Sturani C, Rossi A, Viaggi S, Corrado A, Donner CF, Ambrosino N (2002) The Italian multicentre study on noninvasive ventilation in chronic obstructive pulmonary disease patients. Eur Respir J 20(3):529–538
McEvoy RD, Pierce RJ, Hillman D, Esterman A, Ellis EE, Catcheside PG, O’Donoghue FJ, Barnes DJ, Grunstein RR (2009) Nocturnal non-invasive nasal ventilation in stable hypercapnic COPD: a randomised controlled trial. Thorax 64(7):561–566
Meecham Jones DJ, Paul EA, Jones PW, Wedzicha JA (1995) Nasal pressure support ventilation plus oxygen compared with oxygen therapy alone in hypercapnic COPD. Am J Respir Crit Care Med 152(2):538–544
Dreher M, Storre JH, Schmoor C, Windisch W High-intensity versus low-intensity non-invasive ventilation in patients with stable hypercapnic COPD: a randomised crossover trial. Thorax 65 (4):303–308
Leger P, Bedicam JM, Cornette A, Reybet-Degat O, Langevin B, Polu JM, Jeannin L, Robert D (1994) Nasal intermittent positive pressure ventilation. Long-term follow-up in patients with severe chronic respiratory insufficiency. Chest 105(1):100–105
Janssens JP, Derivaz S, Breitenstein E, De Muralt B, Fitting JW, Chevrolet JC, Rochat T (2003) Changing patterns in long-term noninvasive ventilation: a 7-year prospective study in the Geneva Lake area. Chest 123(1):67–79
Gonzalez-Bermejo J, Perrin C, Janssens JP, Pepin JL, Mroue G, Leger P, Langevin B, Rouault S, Rabec C, Rodenstein D (2010) Proposal for a systematic analysis of polygraphy or polysomnography for identifying and scoring abnormal events occurring during non-invasive ventilation. Thorax. doi:10.1136/thx.2010.142653
Guo YF, Sforza E, Janssens JP (2007) Respiratory patterns during sleep in obesity-hypoventilation patients treated with nocturnal pressure support: a preliminary report. Chest 131(4):1090–1099
Fanfulla F, Taurino AE, Lupo ND, Trentin R, D’Ambrosio C, Nava S (2007) Effect of sleep on patient/ventilator asynchrony in patients undergoing chronic non-invasive mechanical ventilation. Respir Med 101(8):1702–1707
Janssens JP, Borel JC, Pepin JL (2011) Nocturnal monitoring of home non-invasive ventilation: the contribution of simple tools such as pulse oximetry, capnography, built-in ventilator software and autonomic markers of sleep fragmentation. Thorax 66(5):438–445
Thille AW, Rodriguez P, Cabello B, Lellouche F, Brochard L (2006) Patient-ventilator asynchrony during assisted mechanical ventilation. Intensive Care Med 32(10):1515–1522
Vitacca M, Nava S, Confalonieri M, Bianchi L, Porta R, Clini E, Ambrosino N (2000) The appropriate setting of noninvasive pressure support ventilation in stable COPD patients. Chest 118(5):1286–1293
Jolliet P, Tassaux D (2006) Clinical review: patient-ventilator interaction in chronic obstructive pulmonary disease. Crit Care 10(6):236
Rabe KF, Hurd S, Anzueto A, Barnes PJ, Buist SA, Calverley P, Fukuchi Y, Jenkins C, Rodriguez-Roisin R, van Weel C, Zielinski J (2007) Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease: GOLD executive summary. Am J Respir Crit Care Med 176(6):532–555
(1999) Clinical indications for noninvasive positive pressure ventilation in chronic respiratory failure due to restrictive lung disease, COPD, and nocturnal hypoventilation—a consensus conference report. Chest 116(2):521–534
(1999) Sleep-related breathing disorders in adults: recommendations for syndrome definition and measurement techniques in clinical research. The Report of an American Academy of Sleep Medicine Task Force. Sleep 22(5):667–689
Stoohs RA, Blum HC, Knaack L, Butsch-von-der-Heydt B, Guilleminault C (2005) Comparison of pleural pressure and transcutaneous diaphragmatic electromyogram in obstructive sleep apnea syndrome. Sleep 28(3):321–329
Delhez L (1968) Electromyography of the diaphragm in man. Electromyogr 8(3):253–255
Chao DC, Scheinhorn DJ, Stearn-Hassenpflug M (1997) Patient-ventilator trigger asynchrony in prolonged mechanical ventilation. Chest 112(6):1592–1599
Vitacca M, Bianchi L, Zanotti E, Vianello A, Barbano L, Porta R, Clini E (2004) Assessment of physiologic variables and subjective comfort under different levels of pressure support ventilation. Chest 126(3):851–859
Rabec C, Georges M, Kabeya NK, Baudouin N, Massin F, Reybet-Degat O, Camus P (2009) Evaluating noninvasive ventilation using a monitoring system coupled to a ventilator: a bench-to-bedside study. Eur Respir J 34(4):902–913
Contal O, Vignaux L, Combescure C, Pépin J-L, Jolliet P, Janssens J-P (2011) Performance of built-in software in home ventilators for assessment of leaks: a bench model study. Chest. doi:10.1378/chest.11-0485
Janssens JP, Metzger M, Sforza E (2009) Impact of volume targeting on efficacy of bi-level non-invasive ventilation and sleep in obesity-hypoventilation. Respir Med 103(2):165–172
Thille AW, Cabello B, Galia F, Lyazidi A, Brochard L (2008) Reduction of patient-ventilator asynchrony by reducing tidal volume during pressure-support ventilation. Intensive Care Med 34(8):1477–1486
Tassaux D, Gainnier M, Battisti A, Jolliet P (2005) Impact of expiratory trigger setting on delayed cycling and inspiratory muscle workload. Am J Respir Crit Care Med 172(10):1283–1289
Mehta S, McCool FD, Hill NS (2001) Leak compensation in positive pressure ventilators: a lung model study. Eur Respir J 17(2):259–267
Windisch W, Kostic S, Dreher M, Virchow JC Jr, Sorichter S (2005) Outcome of patients with stable COPD receiving controlled noninvasive positive pressure ventilation aimed at a maximal reduction of PaCO2. Chest 128(2):657–662
Windisch W, Haenel M, Storre JH, Dreher M (2009) High-intensity non-invasive positive pressure ventilation for stable hypercapnic COPD. Int J Med Sci 6(2):72–76
Leung P, Jubran A, Tobin MJ (1997) Comparison of assisted ventilator modes on triggering, patient effort, and dyspnea. Am J Respir Crit Care Med 155(6):1940–1948
O’Donnell DE, Webb KA (1993) Exertional breathlessness in patients with chronic airflow limitation. The role of lung hyperinflation. Am Rev Respir Dis 148(5):1351–1357
Diaz O, Begin P, Torrealba B, Jover E, Lisboa C (2002) Effects of noninvasive ventilation on lung hyperinflation in stable hypercapnic COPD. Eur Respir J 20(6):1490–1498
Acknowledgements
The authors wish to thank the Pulmonary League of Geneva (Ligue Pulmonaire Genevoise: www.lpge.ch) for its financial support.
Conflict of interest
None.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Work performed at: Sleep laboratory, Department of Psychiatry, Geneva University Hospitals
The trial was registered at www.clinicaltrials.gov (Trial no. NCT01180439)
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Adler, D., Perrig, S., Takahashi, H. et al. Polysomnography in stable COPD under non-invasive ventilation to reduce patient–ventilator asynchrony and morning breathlessness. Sleep Breath 16, 1081–1090 (2012). https://doi.org/10.1007/s11325-011-0605-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11325-011-0605-y