Abstract
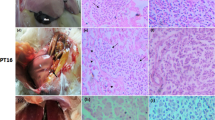
Sudden death of ducklings was reported in a duck farm located at Tiruvallur district in Tamil Nadu, India. Disease investigation began with post mortem findings of dead birds revealing enlarged pale-pink / pale-yellow liver with multifocal petechiae and ecchymosis. A positive amplification with duck hepatitis A virus specific primers by reverse transcription-polymerase chain reaction (RT-PCR) on the tissue samples collected from dead birds indicated infection by duck hepatitis A virus (DHAV), an avian picornavirus, known to cause acute and high-mortality in ducklings. The virus isolation was successful in 9-days old embryonated chicken eggs, in primary chicken embryo fibroblast (CEF) cells and from experimentally infected ducklings. The embryonic death on day 5 to 7 post inoculation in chicken embryos with signs of cutaneous hemorrhage, edema and greenish yellow liver together with histopathology of embryonic liver and kidney further confirmed DHAV infection. TEM analysis of the infected allantoic fluid and infected CEF cell culture supernatant showed the presence of spherical shaped, non-enveloped virion particles of ~ 20–38 nm diameter, typical for DHAV. Experimental infection of ducklings with RT-PCR positive tissue supernatant caused 40% to 50% mortality with typical petechial hemorrhages on the surface of liver. Further, histopathological analysis and RT-PCR of the inoculated duckling’s tissues confirmed the presence of DHAV. Nucleotide sequencing of the 5′UTR region and VP1 region confirmed duck hepatitis A virus genotype 2 (DHAV-2). To the best of our knowledge, this is the first report of laboratory confirmation of DHAV-2 in India. This study warrants the need for the extensive epidemiological surveillance to understand the prevalence of DHAV-2 in India and to take appropriate control measures to curtail the disease spread.






Similar content being viewed by others
Availability of data and materials
The data that supports the findings of this study are available from the corresponding author upon reasonable request.
References
Bancroft JD, Stevens A (1996) Theory and practice of histological techniques, 4th edn. Churchill Livingstone, Nottingham, pp 1–766
Chen LL, Xu Q, Zhang RH, Yang L, Li JX, Xie ZJ, Zhu YL, Jiang SJ, Si XK (2013) Improved duplex RT-PCR assay for differential diagnosis of mixed infection of duck hepatitis A virus type 1 and type 3 in ducklings. J Virol Methods 192:12–17. https://doi.org/10.1016/j.jviromet.2013.04.012
Fabricant J, Rickard CG, Levine PP (1957) The pathology of Duck virus hepatitis. Avian Dis 1:256–275
Felsenstein J (1985) Confidence limits on phylogenies: An approach using the bootstrap. Evolution 39:783–791. https://doi.org/10.1111/j.1558-5646.1985.tb00420.x
Fu Y, Pan M, Wang X, Xu Y, Yang H, Zhang D (2008) Molecular detection and typing of duck hepatitis A virus directly from clinical specimens. Vet Microbiol 131:247–257. https://doi.org/10.1016/j.vetmic.2008.03.011
Hansen WR, Nashold SW, Docherty DE, Brown SE, Knudson DL (2000) Diagnosis of Duck plague in waterfowl by polymerase chain reaction. Avian Dis 44:266–274
Haryanto A, Irianingsih SH, Yudianingtyas DH, Wijayanti N, Budipitojo T (2013) Single step multiplex RT-PCR for detection and differential diagnosis of avian influenza, Newcastle disease and infectious bursal disease viruses in chicken. Int Res J Biotechnol 4:34–39
Kim MC, Kwon YK, Joh SJ, Lindberg AM, Kwon JH, Kim JH, Kim SJ (2006) Molecular analysis of duck hepatitis virus type 1 reveals a novel lineage close to the genus Parechovirus in the family Picornaviridae. J Gen Virol 87:3307–3316. https://doi.org/10.1099/vir.0.81804-0
Kim MC, Kwon YK, Joh SJ, Kim SJ, Tolf C, Kim JH, Sung HW, Lindberg AM, Kwon JH (2007) Recent Korean isolates of duck hepatitis virus reveal the presence of a new geno- and serotype when compared to duck hepatitis virus type 1 type strains. Arch Virol 152:2059–2072. https://doi.org/10.1007/s00705-007-1023-0
Kim MC, Kwon YK, Joh SJ, Kwon JH, Lindberg AM (2008) Differential diagnosis between type-specific duck hepatitis virus type 1 (DHV-1) and recent Korean DHV-1-like isolates using a multiplex polymerase chain reaction. Avian Pathol 37:171–177. https://doi.org/10.1080/03079450801918670
Kimura M (1980) A simple method for estimating evolutionary rate of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16:111–120. https://doi.org/10.1007/bf01731581
Mansour SMG, Mohamed FF, ElBakrey RM, Eid AAM, Mor SK, Goyal SM (2019) Outbreaks of Duck Hepatitis A Virus in Egyptian Duckling flocks. Avian Dis 63:68–74. https://doi.org/10.1637/11975-092118-reg.1
Mason RA, Tauraso NM, Ginn RK (1972) Growth of duck hepatitis virus in chicken embryos and in cell cultures derived from infected embryos. Avian Dis 16:973–979
Mbuthia PG, Njagi LW, Nyaga PN, Bebora LC, Minga U, Kamundia J, Olsen JE (2008) Pasteurella multocida in scavenging family chickens and ducks: carrier status, age susceptibility and transmission between species. Avian Pathol 37:51–57. https://doi.org/10.1080/03079450701784891
Ou X, Mao S, Cao J, Cheng A, Wang M, Zhu D, Chen S, Jia R, Liu M, Sun K, Yang Q, Wu Y, Chen X (2017) Comparative analysis of virus host interactions caused by a virulent and an attenuated duck hepatitis A virus genotype 1. PLoS ONE 12:e0178993. https://doi.org/10.1371/journal.pone.0178993
Phelps NBD, Mor SK, Armien AG, Batts W, Goodwin AE, Hopper L, McCann R, Fan Ng TF, Puzach C, Waltzek TB, Delwart E, Winton J, Goyal SM (2014) Isolation and molecular characterization of a novel picornavirus from baitfish in the USA. PLoS ONE 9:e87593. https://doi.org/10.1371/journal.pone.0178993
Priz NN (1973) Comparative study of the characteristics of the manifestation of viral hepatitis in animals infected by different routes. Vopr Virusol 18:696–700. [Article in Russian]
Rao SBV, Gupta BR (1967) Studies on a filterable agent causing hepatitis in duckling, and biliary cirrhosis and blood dyscrasia in adults. Indian J Poult Sci 2:18–30
Saitou N, Nei M (1987) The neighbor-joining method: A new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Soliman M, Alfajaro MM, Lee MH, Jeong YJ, Kim DS, Son KY, Kwon J, Choi JS, Lim JS, Choi JS, Lee TU, Cho KO, Kang MI (2015) The prevalence of duck hepatitis A virus types 1 and 3 on Korean duck farms. Arch Virol 160:493–498. https://doi.org/10.1007/s00705-014-2264-3
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38:3022–3027. https://doi.org/10.1093/molbev/msab120
Thompson JD, Higgins DG, Gibson TJ (1994) Clustal W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting positions-specific gap penalties and weight matrix choice. Nucl Acids Res 22:4673–4680. https://doi.org/10.1093/nar/22.22.4673
Tseng CH, Tsai HJ (2007) Molecular characterization of a new serotype of duck hepatitis virus. Virus Res 126:19–31. https://doi.org/10.1016/j.virusres.2007.01.012
Wang L, Pan M, Fu Y, Zhang D (2008) Classification of duck hepatitis virus into three genotypes based on molecular evolutionary analysis. Virus Genes 37:52–59. https://doi.org/10.1007/s11262-008-0233-1
WOAH (2018) Manual of diagnostic tests and vaccines for terrestrial animals. Duck Virus Hepatitis;https://www.oie.int/fileadmin/Home/eng/Health_standards/tahm/3.03.08_DVH.pdf. Accessed online October 2021, WOAH Publication, Paris
Woolcock PR (2008) Duck hepatitis. In: Dufour-Zavala L, Swayne DE, Glisson JR, Pearson JE, Reed WM, Jackwood MW, Woolcock PR (eds) A laboratory manual for the isolation, identification and characterization of Avian Pathogens, 5th Edition, 94 eds. American Association of Avian Pathologists, Jacksonville, Florida, USA, pp 175–178
Woolcock PR, Tsai HJ (2013) Duck hepatitis. In: Swayne DE, Glisson JR, McDougald LR, Nolan LK, Suarez DL, Venugopal Nair (eds) Diseases of poultry, Thirteenth Edition., 13th eds. John Wiley & Sons, Inc, Iowa, USA, pp 422–431
Acknowledgements
We are thankful to the Tamil Nadu Veterinary and Animal Sciences University, Chennai and the Director, Centre for Animal Health studies, TANUVAS, Chennai for providing necessary facilities to carry out this work.
Author information
Authors and Affiliations
Contributions
Conceptualization: R. Ramya; Methodology, Data treatment and Formal analysis: R. Ramya, S. Jaisree, N. Jayanthi, G. Kalaiselvi, K. Karthik, M. Madhanmohan, S. Manoharan, Madhuri Subbiah, S. Revathi, Lakshmana Rao Pachinella; Investigation: R. Ramya, N. Jayanthi, S. Manoharan; Resources: R. Sridhar, S. Manoharan; Writing – Original Draft Preparation: R. Ramya, M. Madhanmohan; Writing – Review & Editing: S. Jaisree, N. Jayanthi, G. Kalaiselvi, K. Karthik, S. Revathi, Lakshmana Rao Pachineella, Madhuri Subbiah, R. Sridhar. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical approval
All experimental procedures were reviewed and approved by the Institutional Animal Ethical Committee (IAEC) of Tamil Nadu Veterinary and Animal Sciences University (Approval number—27/SA/IAEC/2022). Experiments were conducted in accordance with approved guidelines.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Competing interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rajendran, R., Srinivasan, J., Natarajan, J. et al. First report of Duck Hepatitis A virus genotype 2 in India. Vet Res Commun 47, 1231–1241 (2023). https://doi.org/10.1007/s11259-022-10063-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11259-022-10063-0