Abstract
Cities are expanding at fast rates across the world, representing one of the main drivers of biodiversity loss due to habitat replacement. Nonetheless, urban and peri-urban areas often feature green spaces that may offer opportunities to wildlife and even represent safe havens for endangered species. Nonetheless, the key drivers that shape wildlife responses to urban landscapes, and in turn their ability to persist within cities, are far from being fully understood. Here we focus on an ecologically specialized butterfly, the endemic Italian festoon Zerynthia cassandra, as a model to assess how endangered species may survive in highly modified urban landscapes. The relatively low mobility and high host plant specialization make Z. cassandra an excellent target for studies in urban ecology, as they make the species able to exploit small suitable patches while at the same time potentially sensitive to habitat fragmentation and loss due to urbanization and land reclamation. We thus first document the relatively widespread occurrence of potentially suitable sites within two highly modified landscapes of central and southern Italy, with 25 and 35% of sites actually occupied by Z. cassandra. By modeling the probability of butterfly occurrence as a function of environmental characteristics, we found that Z. cassandra is strongly influenced by functional connectivity among suitable sites in urban landscapes, as well as by the abundance of Aristolochia host plants, and by the availability of profitable land cover classes in the immediate surroundings of potential oviposition sites. Our results indicate not only that networks of urban and peri-urban green spaces may host populations of protected and endangered species, but that management should also focus on the urban matrix in order to provide connecting corridors, as key assets to guarantee species persistence in cities.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anthropogenic landscapes are extremely complex environments that, despite the intensity of human modifications that occur therein, can still host high levels of biodiversity (MacGregor-Fors et al. 2016; Hall et al. 2017; Labadessa and Ancillotto 2023b). Wildlife may persist in cities and agricultural areas, i.e. the most typical environments that replace natural ones due to human activities, usually in association with remnant green spaces. The latter can be areas with higher levels of naturalness (Sorace 2001; Robinson and Lundholm 2012), and great attention is increasingly being focused on the inclusion of such “wild” spaces—and species—particularly within cities (Aronson et al. 2017; Threlfall et al. 2017). Many species are in fact good indicators of healthy ecosystems, besides having effective roles in providing regulating, provisioning and cultural services, including fostering people's wellbeing. Urban development has been increasing in recent decades, particularly in the Mediterranean Basin, where many cities have experienced intense urbanization of the human population at the expense of natural and agricultural areas (García-Nieto et al. 2018). Nonetheless, several cities still harbor patches—remnants—of the habitats that were originally in the area e.g., by encompassing stretches of more natural territories. The role of these remnants as refugia for wildlife is well recognized, and yet still also hampered by the expansion of urban development, leading to both habitat loss and fragmentation; this has negative effects particularly for poorly mobile species and on those depending upon specific resources and narrow environmental conditions (Grimm et al. 2008; New 2015; Ancillotto and Labadessa 2023). Consequently, functional connectivity among green spaces is usually key to maintaining vital populations of urban wildlife e.g., by fostering and sustaining meta-population dynamics, particularly with the surrounding natural areas that may play as a source of individuals (Lynch 2019; Kirk et al. 2023).
Butterflies have been widely studied in diverse ecological contexts, including urban and agricultural areas, as key elements of ecosystem functioning—by playing major roles in pollination and trophic chains—as well as being effective proxies of biodiversity as a whole, and of environmental health (Van Swaay and van Strien 2005; Feest et al. 2008). Besides, several Lepidoptera are currently protected by the EU international legislation, being listed in Annexes II and IV of the Habitats Directive (HD), thus being recognized as important conservation targets at community scale. Many butterfly species are currently threatened by land use changes, and have reduced in numbers or went locally extinct, albeit several of these may still occur in anthropogenic contexts, when their key resources—e.g., host plants and foraging opportunities, persist (Koh and Sodhi 2004; Ramírez-Restrepo and MacGregor-Fors 2017).
Understanding the drivers of occurrence and/or persistence of threatened species in highly modified environments is key to fostering habitat management practices targeting their conservation i.e., to enhance the role of refuge played by well-conserved remnant green spaces (Jalkanen et al. 2020; Tarabon et al. 2020; Farooq et al. 2023). Potential habitat management practices may though greatly differ not only as a reflection of species’ preferences and ecological needs, but also in terms of the considered spatial scale. As an example, butterflies are known to respond to conditions at very different scales, e.g. from local availability of larval host plant and adult foraging (nectar) opportunities, to landscape metrics such as dominant land cover types and functional connectivity, which in turn is influenced by species’ traits such as mobility and habitat preferences (Dover and Settele 2009; Han et al. 2022). Such complex interplay among site, landscape and species’ traits in shaping the occurrence and survival of butterflies—as of many other taxa—make their conservation particularly challenging—and yet urgent—in highly modified environments such as cities (e.g. Piccini et al. 2022).
Here we focus on the Italian festoon (Zerynthia cassandra), an EU-wide protected butterfly characterized by relatively low adult mobility and a high host-plant specialization; as such, even small patches of suitable habitat may actually represent a profitable resource to this species, at least potentially. Conversely, the same traits may hinder the species’ ability to persist in highly fragmented landscapes due to the difficulties of maintaining the functional connectivity among isolated populations or suitable habitat patches (Öckinger et al. 2009). We thus used Z. cassandra as a model to assess suitability of urban networks of green spaces, and identify environmental drivers of occurrence of endangered butterflies in anthropogenic landscapes, integrating a set of environmental characteristics measured at the habitat patch scale. More specifically, we aim to 1) assess whether urban and peri-urban green areas may actually prove profitable to Z. cassandra, and 2) disentangle the effects of environmental factors at different scales in driving the occurrence of Z. cassandra.
Materials and methods
Study areas and species
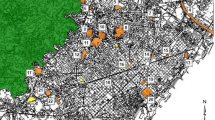
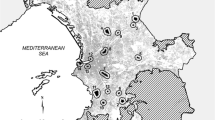
We conducted our study in two anthropogenic landscapes in central-southern Italy, the urbanized areas of the cities of Rome and Bari (Fig. 1), hereafter defined as study “locations”. Rome is the largest Italian city and one of the most ancient urban areas in Europe, hosting ca. 6 million inhabitants, while the city of Bari and its surroundings is among the largest and most populous urbanized territories in southern Italy, containing over 500,000 inhabitants. The locations considered in the present study (see below), each covering approximately 40,000 ha, are dominated by built-up surfaces comprising sparse to high-density districts, yet are also densely interspersed by a network of green spaces, including natural, semi-natural, recreational, and agricultural areas. Climate is typically Mediterranean, with mean temperatures ranging from 18.9–31.7 °C in summer to 2.8–12.7 °C in winter, and precipitation concentrated in autumn–winter season.
For both locations, as for many other Italian cities, most of their urban development occurred during the 40 years after the end of World War II, so that major land-use changes occurred between 1940 and 1980, and mainly consisted in the replacement of semi-natural and agricultural areas with built-up surfaces. Natural and semi-natural vegetation is mostly limited to landscape portions that have been spared from deep land modifications since historical times, as in the case of large protected private properties (e.g., the villas in Rome), or as a consequence of locally rough geomorphology (e.g., the lame of Bari). For this study, we considered the territory encompassed by the great ring road (“Grande Raccordo Anulare”) as the focal location in Rome, and the semi-circular basin of the erosional streams encompassing the city of Bari, thus considering—in both cases – a surface of approximately 350 square kilometers in size (Fig. 1).
The study species, Zerynthia cassandra, is a single-brooded species associated with host plants of the genus Aristolochia (Camerini et al. 2018; Cini et al. 2019, 2021; Cagnetta et al. 2020). Despite being not strictly associated to specific habitat types, Z. cassandra favors open and semi-open areas such as dry grasslands, meadows and forest clearings, that provide wide foraging opportunities to adults; nonetheless, the species needs in terms of level of insolation of the oviposition sites and larval host plants are highly specific (Camerini et al. 2018; Ghesini et al. 2018). In particular, the species is strictly dependent upon Aristolochia rotunda, in Rome, and A. clusii, in Bari, for oviposition and larval development. Recent studies provided important insights on the very limited dispersal abilities of Z. cassandra (Vovlas et al. 2014; Cini et al. 2019), also underlining its marked susceptibility to extinction due to micro-habitat changes (e.g., disappearance of the host plant, and grassland encroachment: Bonelli et al. 2011; Camerini et al. 2018; Ghesini et al. 2018). Zerynthia cassandra is listed—together with the congeneric Z. polyxena—in Annex IV of the EU Habitats Directive (43/92/EEC) and in the Appendix II of the Bern Convention; it is considered as a declining species (Ghesini et al. 2018), with local extinctions being documented (Descimon 1996; Bonelli et al. 2011).
Given its ecological needs, Z. cassandra is potentially – and directly – affected by urbanization, by means of the disappearance of host plant populations and the replacement of the dry open areas needed for foraging with built-up surfaces or intensively managed vegetation.
Data collection
We retrieved all occurrences available for the focal species from both locations, namely Z. cassandra and its host Aristolochia plants. Occurrences were collected from i) published literature (atlases, papers, and reports); ii) iNaturalist.org, and iii) authors’ own data recorded in the field, while systematically screening both study areas during February-June 2023. Namely, we inspected for adults, eggs and larvae all sites with recent and historical records of either the host plant and/or Z. cassandra 2–4 times in 2023, in order to confirm the occurrence of the target species. Sites with no recent confirmation of the plant were excluded from the analyses, while those with no confirmation for Z. cassandra were considered as currently non-occupied by the species (e.g., due to local extinction; N = 2). We only kept observations that had coordinates and did not have geospatial issues (as defined by the Global Biodiversity Information Facility (https://data-blog.gbif.org/post/issues-and-flags/). We removed possible duplicates from analyses by removing any observations that had the same date, latitude, and longitude.
The approach we adopted for locating and mapping occurrence sites of Aristolochia patches mainly relies upon publications exhaustively mapping the flora of both locations, thus representing a comprehensive picture of the actual distribution of the species in our study area (Labadessa and Ancillotto 2023b; Celesti 1995).
Site attributes
To quantify habitat quality at sites suitable to Z. cassandra, we first built 200 m buffers around each occurrence record of Aristolochia, as corresponding to the known maximum distance moved within oviposition sites by this butterfly (Vovlas et al. 2014). Whenever two or more buffers of adjacent occurrences overlapped, we merged the buffers and considered the resulting surface as a single suitable “site”. Based on field surveys and interpretation of most recent available orthophotos, we classified site land covers using seven classes, according to their naturality and a combination of vegetation structure (open or woody) and abiotic features (dry or wet), i.e. anthropogenic, open agricultural, woody agricultural, dry open semi-natural, wet open semi-natural, dry woody semi-natural, wet woody semi-natural. For each suitable site, we also considered host plant abundance i.e., the size (in m2) of Aristolochia patches, as a cumulative measure of the maximum spreading surfaces of plant clusters (see also Cini et al. 2019). Therefore, we characterized sites according to 1) land cover composition (see below) within the considered buffer, 2) land cover diversity, as Shannon’s index (H’) of land cover classes, and 3) host plant abundance.
To classify landscape connectivity, we built a land cover map by combining the most recent high-resolution layers provided by the European Copernicus Programme (https://land.copernicus.eu/pan-european/high-resolution-layers), featuring 10 m resolution raster mapping of forests, grasslands, water and impervious surfaces. We then reclassified land cover into four classes ecologically meaningful to butterflies, namely:
-
Grasslands (including meadows, natural and semi-natural grasslands, discontinuous scrubland);
-
Broadleaved (areas covered in broadleaved trees and shrub);
-
Agricultural surfaces (also including other intensively managed surfaces, e.g. conifer plantations, lawns and recreational parks);
-
Impervious surfaces and water (buildings, roads and permanent water surfaces).
Based on expert opinion and following the approach of Rochat et al. (2017), we built a resistance map by assigning a relative resistance value to each land cover class across both locations, as a function of the capacity of Z. cassandra to cross each type of land cover. Zerynthia cassandra is a butterfly associated with mosaic vegetation and forest edges, especially open and sunny vegetation-covered areas, so we attributed the lowest resistance values to grasslands (resistance = 1) and broadleaved forests (resistance = 2). Agricultural areas are not a barrier per se, but do not offer foraging possibilities to dispersing adults, and are therefore poorly attractive for butterflies (resistance = 5). Water surfaces, buildings, roads and other impervious surfaces can still be potentially crossed but are not vegetated and not necessarily open, and will therefore probably not be adopted by adult Z. cassandra as dispersal corridors (resistance = 10). Based on these resistance values, we produced a resistance map by rasterizing the land cover vectors and assigning to each pixel of the land cover map the corresponding resistance value. We then quantified the functional connectivity among sites by computing a cost-distance matrix, indicating the potential cost of moving from a site to another. The dispersal cost was quantified as the cumulative resistance value of the least-cost path between each pair of sites. We thus used the Least-Cost Path plugin for QGIS 3.30.3 to identify the least cost path (LCP) between a site and all the others, separately for each study area. We then selected the path with lowest cumulative cost value for each site, as a proxy of its functional connectivity for Z. cassandra. As a last connectivity index, for each site we also measured the linear distance (in km) to the closest available site.
Statistical analyses
We first summarized information on land cover composition within the considered buffers by running a Principal Component Analysis (PCA) based on a Bray–Curtis dissimilarity matrix upon the percent amounts of each of the seven land cover classes considered. This resulted in the first component (PC1) explaining > 70% of the total variance, so we kept PC1 scores as a proxy of land cover composition. More specifically, and according to loading values, the variables mainly contributing to PC1 were dry woody natural vegetation (-0.619), woody agricultural (0.583), and dry open semi-natural (0.453) vegetation, with lower – all negative – values featured by the remaining classes (loading values range between -0.001 and -0.218). Differences between sites occupied or not by Z. cassandra were then furtherly explored by running a one-way PERMANOVA test, followed by a Similarity Percentage test (SIMPER) to assess the role of each land cover class in differentiating occupied and non-occupied sites. All these analyses were run with Past ver. 4.15, with default settings (Hammer et al. 2001).
We then modeled the probability of occurrence of Z. cassandra at site level as a function of site attributes, by running a Generalized Linear Model (GLM) with binomial error structure and logit link function. Namely, we used the occurrence of Z. cassandra as binary response variable (present = 1, absent = 0), and site attributes as explaining variables. All variables were first checked for covariation by performing Pearson correlation tests, yet no combination of variables featured a correlation coefficient >|0.5|, so that all variables were retained.
We built a full model including all variables i.e., land cover composition, site size, LCP cost, linear distance to closest site (LCP length), host plant abundance, location (categorical, N = 2), and land cover diversity (H); we did not consider interactions among variables. We then performed a backward stepwise model selection based on AICc values, by running the dredge function from the MuMIn package for R (Barton and Barton 2015). We considered all models valid with ΔAICc < 2 from the best performing model; we then assessed variable importance by following a model averaging approach and calculating estimate average values (using the model.avg function upon the subset of valid models).
Results
We identified 54 sites potentially suitable to Z. cassandra (Fig. 1) as featuring the presence of host plants of the genus Aristolochia, namely A. rotunda from Rome (N = 23), and A. clusii from Bari (N = 31). Of these sites, 34.8 and 25.8% were actually occupied by Z. cassandra, respectively (N = 8 in each of the areas).
Occupied and non-occupied sites significantly differed in their land use composition (PERMANOVA p = 0.024, F = 3.309), with dry woody semi-natural vegetation and dry open vegetation explaining 22.6 and 22.1% of the total difference respectively and being both more represented in occupied sites, followed by anthropic vegetation (20.7% contribution), being more represented in non-occupied sites. Each of all other land cover classes explained < 15% of observed differences.
Model selection identified 8 valid models out of 128 tested variable combinations, cumulatively retaining all of the seven variables considered. Namely, site area, location, LCP cost and land cover composition were featured in all valid models, landcover diversity, LCP length and host plant abundance in 2 models. (Table 1). Model averaging upon all valid models though reported as significant (p < 0.05 and CI not encompassing 0) only LCP cost, site size, host plant abundance, and land cover composition (Table 2), while LCP length and Location were non-significant.
Zerynthia cassandra was significantly more likely to occur at Aristolochia sites with low LCP cost values (Fig. 2), i.e. functionally connected to other sites, as well as at sites showing higher host plant abundance (Fig. 2). A significant drop in the probability of occurrence of Z. cassandra was consistently evident for both locations at LCP costs >100, corresponding to approximately 1,000-1,200 meters of linear LCP length between closest adjacent sites (Fig. 3). Land cover composition also played a major role, mainly driven by differences in the amounts of dry semi-natural vegetation (both woody and open) showing a positive effect on Z. cassandra, and by anthropic vegetation exerting a negative effect (Fig. 4).
Modelled probability of occurrence of the Italian festoon (Zerynthia cassandra) at 54 urban sites in two locations of peninsular Italy (Bari, N = 31; Rome, N = 23) as a function of a) least-cost path cost values among sites, b) least-cost path linear distance (in meters) between closest sites, c) site size (in ha), d) land cover diversity (H), e) host plant (Aristolochia spp.) abundance, and f) land cover composition. Black dots indicate observed data; shaded areas indicate 95% confidence intervals
Least-cost paths (a and c; dashed yellow lines) among sites (yellow circles) potentially suitable to Zerynthia cassandra in the urban landscapes of Rome (a-b) and Bari (c-d), in Italy. Actual habitats from Rome and Bari are also depicted (arrows indicate an oviposition site with Aristolochia rotunda (b) and an adult Z. cassandra in an urban site of Bari (d)
Probability of occurrence of the Italian festoon (Zerynthia cassandra) at 54 urban sites in two locations of peninsular Italy (Bari, N = 31; Rome, N = 23) as a function of land cover composition in terms of percent amounts of a) anthropic vegetation cover, b) dry woody vegetation cover, and c) dry open vegetation cover in a 200 m buffer surrounding potentially suitable sites. Black dots indicate observed data; shaded areas indicate 95% confidence intervals
Discussion
Can urban habitats host Z. cassandra?
Our results indicate that an endangered, highly specialized butterfly of conservation concern such as Z. cassandra may persist in heavily anthropized areas, such as the big cities where we conducted our study, provided that patches of suitable habitat - comprising key resources such as host plants and foraging opportunities to adults - are present, and that such sites are functionally connected to each other. The large urban areas where our study was set differ in some ecological and urbanistic characteristics, and yet the lack of significance of the location variable suggests that the environmental factors we considered were successful in capturing their ecological differences. In fact, biogeographical and urbanistic factors of Rome and Bari make their urban and peri-urban green spaces rather different, yet both capable of hosting populations of the endangered Z. cassandra: Rome’s longest history and intermittent development has led the city to encompass many natural and semi-natural areas within the urban matrix, besides historical recreational parks such as Roman villas. Bari has instead developed from the Adriatic coast towards inner territories, resulting in the preservation of small natural remnants along riverbeds (lame) that stretch from the nearby Alta Murgia plateau down to the city center (Labadessa and Ancillotto 2023b). In both cases, such development patterns have produced a network of green spaces characterized by high ‘naturalness’, and provide vital habitats to wildlife (Planchuelo et al. 2019; Soanes and Lentini 2019; Ives et al. 2016). Such network of green spaces clearly offers profitable opportunities to those species that are not capable of directly exploiting urban or highly modified habitats (e.g., agroecosystems) (Ancillotto and Labadessa 2023), including Z. cassandra, as also evident by the spatial configuration of the network of suitable sites we found. Despite the rapidly changing conditions that usually characterize urban and peri-urban landscapes, the establishment of protected, natural or semi-natural areas and consequent urban planning restrictions provide relatively stable conditions within some of these green spaces, as also suggested by historical records of Z. cassandra at the same sites reported by our study, dating back to early 1900’s (reviewed by Zapparoli 1997). Several Zerynthia species have declined steeply across Europe (van Swaay et al. 2011), also in at least one of our study locations (Fattorini 2011), namely due to the dramatic land use changes associated with agricultural intensification and urbanization that replace the ecotonal habitats favored by Aristolochia plants, and the dry meadows that provide nectar resources in early spring. Both these factors are key to the survival of these species, and yet, surprisingly, Zerynthia butterflies seem to be able to persist in fragmented and deeply modified landscapes, including urban and suburban green areas, such as those from our case study.
Drivers of Z. cassandra occurrence
Functional connectivity to closest alternative sites was among the most important predictors for a patch of suitable habitat to be actually occupied by our target species Z. cassandra. Functional connectivity, as approximated by LCP cost, is known to have a major role in shaping meta-population dynamics in fragmented landscapes, and this may be particularly relevant in urban and intensive agricultural landscapes, where the matrix is particularly hostile to sensitive or specialized species (Öckinger et al. 2009; Soga and Koike 2012). Butterflies are mobile species able to disperse over longer distances than many similarly sized non-flying arthropods, yet Z. cassandra is known as a relatively weak flier, with individuals rarely traveling >200 meters (Vovlas et al. 2014). Nonetheless, it is also likely that even this species is able to occasionally travel longer distances and colonize novel suitable patches of host plants, if low-resistance paths between such sites exist. As an example, newly created habitat patches in an intensive agricultural matrix from northern Italy were spontaneously colonized by Z. cassandra in ca. 30 years after creation (Ghesini et al. 2019), with potential colonization distances being up to 8-15 km. Our results also suggest that, at least in the studied locations, Z. cassandra may perceive suitable sites as functionally connected at linear distances up to 1-1.2 km, at least if low-resistance corridors exist, furtherly suggesting that the species is able to move longer distances than currently known, at least occasionally.
Within suitable sites, the occurrence of Z. cassandra was strongly driven also by the abundance of its host plants, a result in line with those from more natural areas (Labadessa and Ancillotto 2023a), as well as from other studies on isolated populations of specialized butterflies, including other Zerynthia species (Ochoa-Hueso et al. 2014; Örvössy et al. 2014; Kajzer-Bonk et al. 2016; Nowicki et al. 2019). Aristolochia plants, namely A. clusii and A. rotunda, share some common ecological preferences, such as the frequent occurrence at ecotonal margins e.g., between broadleaved woody vegetation and grasslands, and relatively disturbed soils such as gravelous riverbeds and wild boar rooted sites (Labadessa and Ancillotto 2023a; Ghesini et al. 2018; Cini et al. 2021). Among the latter cases, wild boar rooting activity may prove an important driver of the preservation of disturbed soil patches and the consequent expansion of Aristolochia plants (Sims 2005; Labadessa and Ancillotto 2023a). Wild boar is expanding in urban areas worldwide (Melis et al. 2006; Stillfried et al. 2017), including within our study areas (Gaudiano et al. 2018; Pierucci et al. 2015) in recent years. As such, the ecosystem engineering activity provided by urban boars, and its potential effects on both Aristolochia and Z. cassandra habitat suitability in the near future, may deserve further attention. Moreover, little is known on the dispersal abilities of Aristolochia plants, yet - at least in our study locations - disturbance factors such as rooting (both areas) and seasonal flooding (along the lame in Bari) may play a relevant role in shaping patterns of local distributions by fostering plant dispersal.
Moreover, land use composition was also a key driver of site occupancy by Z. cassandra: occupied sites actually featured larger amounts of dry semi-natural vegetation cover, including both open and woody vegetation, and lower presence of synanthropic vegetation, both results in line with the ecological needs of this species (Ghesini et al. 2018). Zerynthia butterflies are in fact associated with early-flowering herbaceous plants - usually characterizing semi-natural dry grasslands (Hicks et al. 2016), as a foraging resource (see Labadessa and Ancillotto 2023a; Cini et al. 2019), in contrast to synanthropic vegetation, known to represent relatively poor nectar resource to pollinators in urban environments (Hicks et al. 2016; Lososová et al. 2006).
Conclusions
The potential role of urban and peri-urban areas as wildlife refuges has been long debated: while urban development clearly depletes natural habitats, thus threatening the wildlife populations occurring therein, a careful governance of green spaces inside and in the immediate surroundings of cities may still prove invaluable to preserve urban and peri-urban populations (Soanes and Lentini 2019). Urban green spaces in fact may suffer from very different threats and pressures than their natural counterparts, and may conversely be subjected to management practices that actually foster species’ persistence, including rare or endangered taxa. In the case of butterflies, most endangered species are those dependent upon semi-natural grassland habitats and ecotones (Maes et al. 2019), so that usual practices undertaken in urban green spaces, such as partial mowing and low-intensity agricultural activities (e.g., grazing and traditional wood crops), may represent essential factors fostering the maintenance of these key habitats (Moranz et al. 2012). Moreover, the involvement of citizens in the creation of suitable habitat patches (e.g., by planting host plants) may furtherly foster the conservation of endangered butterflies, at the same time decreasing the potential risk of illegal collection (Descimon 1996; Sands 2008).
The ability of butterflies to colonize (or persist inside) suitable sites - even though on rare occasions (Nowicki et al. 2014) - should not though be interpreted as a reason to not properly foster habitat connectivity outside sites (Graffigna et al. 2023). In our case, the ecological needs of Aristolochia plants, preferring ecotones and relatively disturbed soils, as well as the need of adult Zerynthia for early-flowering plant species in the immediate surroundings, make habitat conservation particularly challenging. The current lack of specific protection of the species’ favored habitats (despite that the European Habitats Directive indirectly protects the locations where listed species actually occur), and the obvious difficulties in mapping and quantifying ecotonal habitats and their peculiar conditions (Nowicki et al. 2013), make in fact ecotonal-specialized species highly challenging to conserve.
Moreover, further studies are needed to clarify whether, for low-mobility species such as Z. cassandra, urban populations represent demographic sinks or sources within meta-population dynamics, a key factor to be considered to properly manage and conserve populations in highly dynamic landscapes such as cities.
Data availability
No datasets were generated or analysed during the current study.
References
Ancillotto L, Labadessa R (2023) Functional traits drive the fate of Orthoptera in urban areas. Insect Conserv Divers. https://doi.org/10.1111/icad.12683
Aronson MF, Lepczyk CA, Evans KL, Goddard MA, Lerman SB, MacIvor JS, Nilon C, Vargo T (2017) Biodiversity in the city: key challenges for urban green space management. Front Ecol Environ 15:189–196. https://doi.org/10.1002/fee.1480
Barton K, Barton MK (2015) Package ‘mumin.’ Version 1(18):439
Bonelli S, Cerrato C, Loglisci N, Balletto E (2011) Population extinctions in the Italian diurnal Lepidoptera: an analysis of possible causes. J Insect Conserv 15:879–890
Cagnetta G, Labadessa R, Altini E, Clemente D, Vovlas A (2020) New records and an updated checklist of the butterfly fauna (Lepidoptera: Papilionoidea) of Puglia, south-eastern Italy. Phegea 48:113–121
Camerini G, Groppali R, Minerbi T (2018) Observations on the ecology of the endangered butterfly Zerynthia cassandra in a protected area of Northern Italy. J Insect Conserv 22:41–49
Celesti L (1995) Atlante della flora di Roma. Argos Edizioni. Roma, IT
Cini A, Benetello F, Platania L, Bordoni A, Boschi S, Franci E, Ghisolfi G, Pasquali L, Negroni R, Dapporto L (2021) A sunny spot: habitat management through vegetation cuts increases oviposition in abandoned fields in an endemic Mediterranean butterfly. Insect Conserv Diver 14:582–596
Cini A, Bordoni A, Ghisolfi G, Lazzaro L, Platania L, Pasquali L, Negroni R, Benetello F, Coppi A, Zanichelli F, Dapporto L (2019) Host plant selection and differential survival on two Aristolochia L. species in an insular population of Zerynthia cassandra. J Insect Conserv 23:239–246
Descimon H (1996) Zerynthia polyxena Schiffermüller, 1775. In: Van Helsdingen PJ, Willemse L, Speight MCD (eds) Background information on invertebrates of the Habitats directive and the Bern convention, Part I. Nature and environment, vol 79. Council of Europe Publishing, Strasbourg, France, pp 213–217
Dover J, Settele J (2009) The influences of landscape structure on butterfly distribution and movement: a review. J Insect Conserv 13:3–27
Farooq H, Antonelli A, Faurby S (2023) A call for improving the key biodiversity areas framework. Perspect Ecol Conserv 21:85–91
Fattorini S (2011) Insect extinction by urbanization: a long term study in Rome. Biol Cons 144(1):370–375
Feest A, van Swaay C, van Hinsberg A (2008) The linkage between butterfly biodiversity quality and nitrogen deposition. Environment Agency, Department of Civil Engineerng, Bristol, UK
García-Nieto AP, Geijzendorffer IR, Baró F, Roche PK, Bondeau A, Cramer W (2018) Impacts of urbanization around Mediterranean cities: Changes in ecosystem service supply. Ecol Ind 91:589–606
Gaudiano L, Silvestri F, Pucciarelli L (2018) Piano di gestione del cinghiale Sus scrofa nel parco naturale regionale lama balice – 2018/2021. Technical report (in Italian), Available at: https://www.parcolamabalice.it/wp-content/uploads/2018/06/PdG_cinghiale_lama_balice-compresso.pdf
Ghesini S, De Faveri A, Marini M (2019) A successful habitat patch creation for Zerynthia cassandra. Bull Insectol 72(2):261–266
Ghesini S, Magagnoli S, Marini M (2018) Biology and conservation of Zerynthia cassandra (Lepidoptera, Papilionidae) in semi-natural environments and agricultural landscapes. J Insect Conserv 22(1):151–161
Graffigna S, González-Vaquero RA, Torretta JP, Marrero HJ (2023) Importance of urban green areas’ connectivity for the conservation of pollinators. Urban Ecosyst 1–10
Grimm NB, Faeth SH, Golubiewski NE, Redman CL, Wu J, Bai X, Briggs JM (2008) Global change and the ecology of cities. Science 319:756–760
Hall D, Camilo G, Tonietto R, Ollerton J, Ahrné K, Arduser M, Ascher J, Baldock K, Fowler R, Frankie G, Goulson D, Gunnarsson B, Hanley M, Jackson J, Langellotto G, Lowenstein D, Minor E, Philpott S, Potts S, Threlfall C (2017) The city as a refuge for insect pollinators. Conserv Biol 31:24–29. https://doi.org/10.1111/cobi.12840
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: Paleontological statistics software package for education and data analysis. Palaeontologia Electronica 4(1):9. http://palaeo-electronica.org/2001_1/past/issue1_01.htm
Han D, Wang C, Sun Z, She J, Yin L, Bian Q, Han W (2022) Microhabitat preferences of butterflies in urban parks: Both vegetation structure and resources are decisive. Urban Forest Urban Greening 71:127552. https://doi.org/10.1016/j.ufug.2022.127552
Hicks DM, Ouvrard P, Baldock KC, Baude M, Goddard MA, Kunin WE, Stone GN (2016) Food for pollinators: quantifying the nectar and pollen resources of urban flower meadows. PloS one 11(6):e0158117
Ives CD, Lentini PE, Threlfall CG, Ikin K, Shanahan DF, Garrard GE, Kendal D (2016) Cities are hotspots for threatened species. Glob Ecol Biogeogr 25(1):117–126
Jalkanen J, Toivonen T, Moilanen A (2020) Identification of ecological networks for land-use planning with spatial conservation prioritization. Landsc Ecol 35:353–371
Kajzer-Bonk J, Skórka P, Nowicki P, Bonk M, Król W, Szpiłyk D, Woyciechowski M (2016) Relative contribution of matrix structure, patch resources and management to the local densities of two large blue butterfly species. PLoS ONE 11(12):e0168679
Kirk H, Soanes K, Amati M, Bekessy S, Harrison L, Parris K, Threlfall C (2023) Ecological connectivity as a planning tool for the conservation of wildlife in cities. MethodsX 10:101989
Koh LP, Sodhi NS (2004) Importance of reserves, fragments, and parks for butterfly conservation in a tropical urban landscape. Ecol Appl 14(6):1695–1708
Labadessa R, Ancillotto L (2023a) Beauty and the beast: multiple effects of wild boar rooting on butterfly microhabitat. Biodivers Conserv 32(4):1189–1204
Labadessa R, Ancillotto L (2023b) Small but irreplaceable: The conservation value of landscape remnants for urban plant diversity. J Environ Manage 339:117907
Lososová Z, Chytrý M, Kühn I, Hájek O, Horáková V, Pyšek P, Tichý L (2006) Patterns of plant traits in annual vegetation of man-made habitats in central Europe. Perspectives in Plant Ecology, Evolution and Systematics 8(2):69–81
Lynch AJ (2019) Creating effective urban greenways and stepping-stones: four critical gaps in habitat connectivity planning research. J Plan Lit 34(2):131–155
MacGregor-Fors I, Escobar F, Rueda-Hernández R, Avendaño-Reyes S, Baena ML, Bandala VM, Chacón-Zapata S, Guillén-Servent A, González-García F, Lorea-Hernández F, Montes de Oca E, Montoya L, Pineda E, Ramírez-Restrepo L, Rivera-García E, Utrera-Barrillas E (2016) City “green” contributions: The role of urban greenspaces as reservoirs for biodiversity. Forests 7:1–14. https://doi.org/10.3390/f7070146
Maes D, Verovnik R, Wiemers M, Brosens D, Beshkov S, Bonelli S, Warren MS (2019) Integrating national Red Lists for prioritising conservation actions for European butterflies. J Insect Conserv 23(2):301–330
Melis C, Szafrańska PA, Jędrzejewska B, Bartoń K (2006) Biogeographical variation in the population density of wild boar (Sus scrofa) in western Eurasia. J Biogeogr 33:803–811
Moranz RA, Debinski DM, McGranahan DA, Engle DM, Miller JR (2012) Untangling the effects of fire, grazing, and land-use legacies on grassland butterfly communities. Biodivers Conserv 21:2719–2746
New TR (2015) Insect conservation and urban environments. Springer
Nowicki P, Deoniziak K, Dziekańska I, Kostro-Ambroziak A, Plazio E, Rutkowski R, Sielezniew M (2019) What keeps ‘living dead’alive: demography of a small and isolated population of Maculinea (= Phengaris) alcon. J Insect Conserv 23:201–210
Nowicki P, Halecki W, Kalarus K (2013) All natural habitat edges matter equally for endangered Maculinea butterflies. J Insect Conserv 17:139–146
Nowicki P, Vrabec V, Binzenhöfer B, Feil J, Zakšek B, Hovestadt T, Settele J (2014) Butterfly dispersal in inhospitable matrix: rare, risky, but long-distance. Landscape Ecol 29:401–412
Ochoa-Hueso R, Ranea DDLP, Viejo JL (2014) Comparison of trends in habitat and resource selection by the Spanish Festoon, Zerynthia rumina, and the whole butterfly community in a semi-arid Mediterranean ecosystem. J Insect Sci 14:51
Öckinger E, Dannestam Å, Smith HG (2009) The importance of fragmentation and habitat quality of urban grasslands for butterfly diversity. Landsc Urban Plan 93(1):31–37
Örvössy N, Kőrösi A, Batary P, Vozar A, Peregovits L (2014) Habitat requirements of the protected southern festoon (Zerynthia polyxena); adult, egg and larval distribution in a highly degraded habitat complex. Acta Zoologica Academiae Scientiarum Hungaricae 60(4):371–387
Pierucci P, Semenzato P, Mariucci A, Todini A (2015) Piano di gestione del cinghiale (Sus scrofa) nella Riserva Naturale dell’Insugherata. Technical report (in Italian)
Piccini I, Pittarello M, Di Pietro V, Lonati M, Bonelli S (2022) New approach for butterfly conservation through local field-based vegetational and entomological data. Ecosphere 13(4):e4026
Planchuelo G, von Der Lippe M, Kowarik I (2019) Untangling the role of urban ecosystems as habitats for endangered plant species. Landsc Urban Plan 189:320–334
Ramírez-Restrepo L, MacGregor-Fors I (2017) Butterflies in the city: a review of urban diurnal Lepidoptera. Urban Ecosystems 20:171–182
Robinson SL, Lundholm JT (2012) Ecosystem services provided by urban spontaneous vegetation. Urban Ecosyst 15:545–557
Rochat E, Manel S, Deschamps-Cottin M, Widmer I, Joost S (2017) Persistence of butterfly populations in fragmented habitats along urban density gradients: Motility helps. Heredity 119(5):328–338
Sands D (2008) Conserving the Richmond birdwing butterfly over two decades: where to next? Ecol Manag Restor 9:4–16
Sims NKE (2005) The ecological impacts of wild boar rooting in East Sussex. Ph.D. dissertation, School of Biological Sciences, University of Sussex, Brighton, UK.
Soanes K, Lentini PE (2019) When cities are the last chance for saving species. Front Ecol Environ 17(4):225–231
Soga M, Koike S (2012) Relative importance of quantity, quality and isolation of patches for butterfly diversity in fragmented urban forests. Ecol Res 27:265–271
Sorace A (2001) Value to wildlife of urban-agricultural parks: a case study from Rome urban area. Environ Manage 28(4):547–560
Stillfried M, Gras P, Börner K, Göritz F, Painer J, Röllig K, Kramer-Schadt S (2017) Secrets of success in a landscape of fear: urban wild boar adjust risk perception and tolerate disturbance. Front Ecol Evol 5:157. https://doi.org/10.3389/fevo.2017.00157
Tarabon S, Calvet C, Delbar V, Dutoit T, Isselin-Nondedeu F (2020) Integrating a landscape connectivity approach into mitigation hierarchy planning by anticipating urban dynamics. Landsc Urban Plan 202:103871
Threlfall CG, Mata L, Mackie JA, Hahs AK, Stork NE, Williams NSG, Livesley SJ (2017) Increasing biodiversity in urban green spaces through simple vegetation interventions. J Appl Ecol 54:1874–1883. https://doi.org/10.1111/1365-2664.12876
van Swaay C, Maes D, Collins S, Munguira ML, Šašić M, Settele J, Cuttelod A (2011) Applying IUCN criteria to invertebrates: How red is the Red List of European butterflies? Biol Conserv 144(1):470–478
Van Swaay CAM, van Strien A (2005) Using butterfly monitoring data to develop a European grassland butterfly indicator. Studies on the ecology and conservation of Butterflies in Europe, vol 1. General concepts and case studies. pp 106–108
Vovlas A, Balletto E, Altini E, Clemente D, Bonelli S (2014) Mobility and oviposition site-selection in Zerynthia cassandra (Lepidoptera, Papilionidae): implications for its conservation. J Insect Conserv 18:587–597. https://doi.org/10.1007/s10841-014-9662-4
Zapparoli M (1997) Urban development and insect biodiversity of the Rome area. Italy Landscape and Urban Planning 38(1–2):77–86
Acknowledgements
LA was funded by the National Recovery and Resilience Plan (NRRP), Mission 4 Component 2 Investment 1.4—Call for tender, No. 3138 of 16 December 2021, rectified by Decree n.3175 of 18 December 2021 of Italian Ministry of University and Research funded by the European Union – NextGenerationEU; Project code CN_00000033, Concession Decree No. 1034 of 17 June 2022 adopted by the Italian Ministry of University and Research, CUP B83C22002930006, Project title “National Biodiversity Future Center—NBFC”.
Funding
Open access funding provided by Consiglio Nazionale Delle Ricerche (CNR) within the CRUI-CARE Agreement.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection was performed by all authors, analyses were performed by RL and LA. The first draft of the manuscript was written by LA, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ancillotto, L., Mosconi, F. & Labadessa, R. A matter of connection: the importance of habitat networks for endangered butterflies in anthropogenic landscapes. Urban Ecosyst (2024). https://doi.org/10.1007/s11252-024-01542-0
Accepted:
Published:
DOI: https://doi.org/10.1007/s11252-024-01542-0