Abstract
The frictional properties of ZDDP tribofilms at low entrainment speeds in boundary lubrication conditions have been studied in both rolling/sliding and pure sliding contacts. It has been found that the boundary friction coefficients of these tribofilms depend on the alkyl structure of the ZDDPs. For primary ZDDPs, those with linear alkyl chains give lower friction those with branched alkyl chain ZDDPs, and a cyclohexylmethyl-based ZDDP gives markedly higher friction than non-cyclic ones. Depending on alkyl structure, boundary friction coefficient in rolling-sliding conditions can range from 0.09 to 0.14. These differences persist over long duration tests lasting up to 120 h. For secondary ZDDPs, boundary friction appears to depend less strongly on alkyl structure and in rolling-sliding conditions stabilises at ca 0.115 for the three ZDDPs studied. Experiments in which the ZDDP-containing lubricant is changed after tribofilm formation by a different ZDDP solution or a base oil indicate that the characteristic friction of the initial ZDDP tribofilm is lost almost as soon as rubbing commences in the new lubricant. The boundary friction rapidly stabilises at the characteristic boundary friction of the replacement ZDDP, or in the case of base oil, a value of ca 0.115 which is believed to represent the shear strength of the bare polyphosphate surface. The single exception is when a solution containing a cyclohexylethyl-based ZDDP is replaced by base oil, where the boundary friction coefficient remains at the high value characteristic of this ZDDP despite the fact that rubbing in base oil removes about 20 nm of the tribofilm. XPS analysis of the residual tribofilm reveals that this originates from presence of a considerable proportion of C-O bonds at the exposed tribofilm surface, indicating that not all of the alkoxy groups are lost from the polyphosphate during tribofilm formation. Very slow speed rubbing tests at low temperature show that the ZDDP solutions give boundary friction values that vary with alkyl group structure in a similar fashion to rolling-sliding MTM tests. These variations in friction occur immediately on rubbing, before any measurable tribofilm can develop. This study suggest that ZDDPs control boundary friction by adsorbing on rubbing steel or tribofilm surfaces in a fashion similar to organic friction modifiers. However it is believed that, for primary ZDDPs, residual alkoxy groups still chemically bonded to the phosphorus atoms of newly-formed polyphosphate/phosphate tribofilm may also contribute to boundary friction. This understanding will contribute to the design of low friction, fuel efficient crankcase engine oils.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
The lubricant additive zinc dialkyldithiophosphate (ZDDP) has been used as an antiwear agent in engine oils for more than six decades. It limits wear by forming a protective, phosphate-based tribofilm on rubbing surfaces. In recent years its role as an antiwear additive in engine oils has become increasingly important because of the use of very low viscosity oils to improve fuel economy. Such low viscosities mean that engine components operate for longer periods in thin film boundary and mixed lubrication conditions where wear may occur. Unfortunately, as well as controlling wear, ZDDP is also believed to increase friction in both boundary and mixed lubrication conditions and consequently can be detrimental to fuel economy. It is therefore important to determine ways to reduce the friction of ZDDP tribofilms. This paper explores the boundary friction of ZDDP tribofilms and the influence of ZDDP molecular structure on this friction. The aim is to identify ZDDPs that are less harmful to fuel economy than those currently in use.
2 Background
The molecular structure of ZDDP is shown in Fig. 1.
ZDDP is an organometallic compound based on zinc, phosphorus and sulphur. When dissolved in a lubricant it reacts in rubbing contacts to form eventually a quite thick tribofilm (typically 50 to 150 nm). This film forms only on areas of surface that experience rubbing and it has been shown that the rate of film growth increases exponentially with both temperature and with applied shear stress, indicating that the reaction is mechanochemical in nature [1, 2]. The tribofilm has a quite complex structure, with a thin sublayer rich in iron sulphide mixed with and overlain by a much thicker film of zinc and iron phosphate/polyphosphate [3]. When formed initially, the film appears to be mainly polyphosphate with a predominantly amorphous structure, but during prolonged rubbing this depolymerises into nanocrystalline ortho- and pyrophosphate, possibly driven by diffusion of iron from the substrate [4, 5]. XPS analysis suggests that the immediate surface layer contains a high proportion of polyphosphate even after prolonged rubbing.
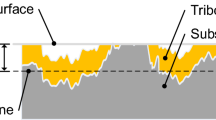
ZDDP tribofilms are relatively rough, with a pad structure consisting of flat-topped islands of tribofilm separated by deep valleys that often contain no detectable film [6]. If formed in reciprocating conditions the pads are quite round but in unidirectional sliding they can be elongated in the sliding direction. Figure 2 shows an AFM topography image taken from [7] of a typical tribofilm formed from a secondary ZDDP solution in a two-hour rolling-sliding test. The rubbed track and thus the tribofilm is ca 125 μm wide.
AFM of typical ZDDP tribofilm from a unidirectional MTM rolling-sliding test [from Ref. 7]
The precise way that ZDDP tribofilms limit wear has not yet been determined but the most widely held view is that the films act to prevent direct contact and thus adhesive or abrasive wear of metal against metal [8]. Instead, opposing tribofilms, that are themselves quite resistant to wear, very slowly abrade. Alternative or additional suggestions are that the tribofilms digest hard and potentially abrasive wear particles [9] or the ZDDP or its tribofilm removes peroxide or peroxy-radical species that would otherwise corrode ferrous surfaces [10, 11].
In the 1980s, potential fuel shortages led to a growing interest in engine fuel economy and it was found that the ZDDP present in lubricants could be detrimental in this respect [12, 13]. In 1996 it was shown that ZDDP could produce a large increase in friction in mixed lubrication conditions compared to the base oil alone [14]. This effect was greater for secondary than primary ZDDPs and developed with rubbing time. In 1998 this friction increase was correlated with the thickness of the ZDDP tribofilm and it was proposed that it originated from the roughness of the film [15], a correlation confirmed in 2003 [16]. Very recently, Dawcyzk et al. have proved that ZDDP’s increase in friction in mixed lubrication conditions results directly from the increase in effective roughness of the rubbing surfaces due to the formation of ZDDP tribofilms [7]. As the rough tribofilm develops, a thicker elastohydrodynamic (EHD) lubricating film must be generated to separate the surfaces and this requires a faster entrainment speed. Figure 3a shows a typical set of friction versus entrainment speed curves (Stribeck curves) obtained by Dawcyzk et al. during a two-hour slow speed rubbing test using a primary ZDDP solution. Prior to rubbing (0 min.) the shape of the curve indicates that the contact operates in mixed boundary/EHD lubrication at the lowest speed and reaches full film EHD lubrication, indicated by a constant and low value of friction, at an entrainment speed of greater than 500 mm/s. However, after 30 min rubbing when a ZDDP tribofilm has formed, the contact is now in boundary lubrication at slow speeds and only reaches full EHD film lubrication at an entrainment speed of 3 m/s. After 2-h rubbing when the ZDDP film has reached its maximum thickness, the contact remains in boundary lubrication up to an entrainment speed of 100 mm/s and never reaches the full EHD lubrication regime even at the highest entrainment speed of 3 m/s. The potential impact of this on friction in a reciprocating piston/liner contact should be evident.
a Stribeck curves measured during a low speed rolling/sliding test using a primary ZDDP solution, b Collapse of friction curves when plotted against lambda ratio [From ref. 7])
Dawcyzk et al. measured the roughness of the tribofilms at each time step and from this and measured EHD film-forming behaviour, determined the prevailing lambda ratio (ratio of film thickness to composite surface roughness). As shown in Fig. 3b all the friction curves collapsed onto a single friction coefficient versus lambda ratio curve, indicating that the change in surface roughness was solely responsible for the evolution of friction [7].
From the above figures it is clear that as a ZDDP tribofilm develops, the value of its boundary friction coefficient becomes increasingly important. This value will determine not just the friction at very low speeds but also contribute strongly to friction up to quite high speeds when the contact operated in mixed film lubrication conditions. Figure 3 also indicates that the ZDDP studied gave the relatively high value of boundary friction coefficient of 0.14.
There has been a limited amount of research on the boundary friction properties of ZDDP tribofilms. In 1987 Studt studied the low speed friction of a range of ZDDP solutions in hexadecane using a ball on disc sliding test rig and found that for linear alkyl ZDDPs, friction coefficient fell with increasing chain length from ca 0.15 for n-propyl, n-butyl and n-pentyl- to about 0.10 for n-octadecyl-, while all branched chain ZDDPs tested gave friction coefficients of ca 0.165 [17, 18]. Studt proposed that the ZDDP molecules were adsorbing to form close-packed monolayers in a similar fashion to organic friction modifiers so that boundary friction depended on how closely the alkyl groups were able to pack together. Much more recently, using a very low speed cylinder on ring sliding test rig, Aoki et al. showed that a secondary ZDDP gave higher boundary friction than primary ZDDPs based on linear alkyl groups and that ZDDPs with longer, linear alkyl groups gave lower boundary friction than those based on shorter chains [19]. Hoshino et al. used a rolling-sliding ball on disc contact to compare the friction properties of three ZDDPs, two primaries, linear n-octyl and branched 2-ethylhexyl, and a secondary ZDDP based on 4-methylpent-2-yl [20]. They showed that boundary friction varied in the order octyl < ethylhexyl < methylpentyl. Also the ZDDP with linear alkyl groups showed friction that increased linearly with log(sliding speed), a phenomenon generally seen with effective organic friction modifiers [21]. The above three studies were made with simple solutions of ZDDP in base oil, but Elvidge and Andrews showed that ZDDP alkyl structure can also influence boundary friction in formulated engine oils [22]. They found that formulations with ZDDPs synthesised in part from long chain alcohols such as oleyl alcohol gave lower HFRR friction than those containing only conventional ZDDPs based on C3 to C8 alcohols.
It is clear from the above that the boundary friction of ZDDPs can depend on the structure of their alkyl groups and that this implies that alkyl groups must be present in some form at the outermost surface of a ZDDP tribofilm. The current study examines the friction behaviour of a range of ZDDP molecular structures to explore further the influence of the nature of their alkyl groups on boundary friction.
3 Test Lubricants
Seven ZDDPs with different alkyl groups (R groups in Fig. 1) were studied as listed in Table 1. All were synthesised from single alcohols and should thus contain just one type of alkyl group. NMR analysis showed that this was the case for six of the ZDDPs but that the oct-2-yl-based additive contained about 2% of oct-1-yl-, suggesting that the alcohol used in its synthesis contained this proportion of octan-1-ol. Negligible alkyl rearrangement is believed to occur during ZDDP synthesis [23].
Four of the ZDDPs were primary, i.e. the C atom directly bonded to the O atom in the alkoxy group was also bonded to one carbon atom and two hydrogen atoms. Three were secondary in that this initial C atom was bonded to two C atoms and one hydrogen. The structures of the ZDDPs are shown in Fig. 4.
The base oil used throughout this study was a Group II mineral oil having viscometric properties listed in Table 2. This is a typical base oil used in modern engine oil formulation. All solutions tested contained ZDDP blended to give a total concentration of 800 ppm phosphorus.
4 Test Methods
4.1 Rolling-Sliding Tests—MTM-SLIM
A Mini Traction Machine with Spacer Layer Imaging (MTM-SLIM, PCS Instruments, UK) was used to study both friction and tribofilm formation of ZDDPs in rolling-sliding contact conditions. In this a 19.05 mm diameter ball is loaded and rotated against the flat surface of a rotating disc immersed in lubricant at controlled temperature. Ball and disc are driven by separate motors to enable any combination of rolling and sliding to be obtained. The ball shaft is angled to minimise spin in the contact and friction is monitored using a load cell attached to the ball shaft housing.
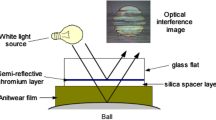
The Spacer Layer Imaging Method (SLIM) allows in situ measurement of tribofilm formed on the rubbed balls in the MTM, as shown schematically in Fig. 5. To make a SLIM measurement, the ball rotation is halted and it is then uploaded against a coated glass mapper disc. White light is shone into the resulting glass-ball contact where some is reflected by a semi-reflective coating on the underside of the glass while the remainder passes through a silica spacer layer and any tribofilm on the ball before being reflected back from the steel substrate. The two beams undergo optical interference depending on their optical path difference and thus on the antiwear film thickness. The result is an interference image as indicated in Fig. 5 which is captured as a RGB pixel map by a high-resolution camera. A calibration chart of RGB colour versus optical path difference obtained using ultrathin film interferometry is then used to convert this RGB pixel map to a ZDDP tribofilm thickness map [24]. A refractive index of 1.6 was assumed for all the ZDDP tribofilms.
The test conditions used in this study are listed in Table 3. For most of a test, the ball and disc are rubbed together in the ZDDP solution at a low entrainment speed of 0.05 m/s to ensure that minimal lubricant is drawn into the contact. This means that the contact operates in the mixed boundary-EHD lubrication regime in which asperity contact occurs and the resulting stresses promote ZDDP tribofilm formation. Periodically throughout a test, a tribofilm thickness map is obtained and a friction curve is measured. For these the ball is first unloaded and its rotation halted before it is uploaded against the coated glass disc to capture a SLIM interference image. To generate the friction curve, the entrainment speed is raised to a maximum value of 3.5 m/s and then reduced in stages while measuring friction at each stage. At each entrainment speed, friction is measured with the ball surface moving faster than the disc and vice versa and the mean determined to eliminate any offset in the load cell. This produces a friction curve or Stribeck curve showing how friction coefficient varies with entrainment speed, as illustrated in Fig. 3a. Since EHD film thickness increases with entrainment speed, this shows how friction varies during the transition from the boundary to EHD lubrication thickness.
In the current study, AISI 52100 steel balls and discs were used with hardness ca 850 Hv and 825 Hv, respectively, and composite surface roughness 15 nm. These were soaked in toluene for at least 24 h and then ultrasonically cleaned successively in toluene and Analar acetone for 10 min prior to use. At the low entrainment speed used throughout a test, the central EHD film thickness based on measurements made in an optical EHD test apparatus was 11.5 nm. This means that the ratio of theoretical EHD film thickness to composite surface roughness was initially 0.8, implying mixed boundary/EHD lubrication conditions.
4.2 Reciprocating Sliding Tests—HFRR
The MTM is normally used to measure friction in rolling-sliding conditions as present in gears, rolling bearings and cam-followers. To determine friction in pure sliding contact, as is present in piston-liner contacts, a high frequency reciprocating rig (HFRR, PCS Instruments, UK) was used. In this a 6 mm diameter ball is loaded and driven in sinusoidal reciprocating motion against the stationary flat surface of a disc immersed in lubricant at controlled temperature. Friction is monitored throughout a test and the wear on the ball is measured at the end, either as the wear scar width or as the wear volume. For this study AISI 52100 steel balls and discs were used, with hardness 880 Hv and 790 Hv, respectively, and composite root mean square roughness 7 nm. Test conditions are listed in Table 4.
4.3 Ball on Flat Unidirectional Sliding Tests—UMT
To study the effect of any initial adsorption of ZDDPs on boundary friction, a few tests were carried out in which a 6 mm diameter stationary steel ball was loaded against the flat surface of a very slowly rotating steel disc (CETR UMT-2 tribometer). The ball specimens were the same as used in the HFRR, while the discs were also AISI 52100 specimens, with hardness 800 Hv and root mean square roughness 12 nm. Test conditions were sliding speed 10–4 m/s, applied load 4 N, corresponding to a maximum Hertz pressure of 1.04 GPa, and test temperature 40 °C.
5 Results
5.1 MTM Friction Curves
All ZDDP solutions were tested at least twice and for repeat tests differences in boundary friction coefficient after 2-h rubbing were always less than 5%. Figure 6a–d shows how MTM friction coefficient versus entrainment speed evolves with rubbing time for the four primary ZDDPs, while Fig. 7 compares their friction behaviour after 120 min rubbing. It should be noted that for clarity only some of the friction curves obtained during the two-hour tests are shown.
The two different features of friction behaviour of ZDDPs as slow speed rubbing progresses can be seen clearly in Fig. 6 i.e. (i) a shift of the mixed lubrication region of the friction curves to higher entrainment speed and also (ii) an increase in slow speed boundary friction for some ZDDPs (EHX and CYC). Figure 7 shows that the four ZDDPs give quite different boundary friction values after two hours rubbing, in the order;
For DOD, OCT and EHX this order is similar to that seen in previous studies [19, 20].
Figure 8a–c shows how MTM friction coefficient versus entrainment speed evolves with rubbing time for the three secondary ZDDPs, while Fig. 9 compares their friction behaviour after 2-h rubbing.
Comparing Figs. 8 and 6 it is evident that mixed friction increases much more rapidly for the secondary than the primary ZDDPs, so that a steady state value is reached after only 30 min. of slow speed rubbing. For BUT-2° and MPE-2° (and to a smaller extent OCT-2°) it can also be seen that boundary friction increases initially but then falls as rubbing progresses beyond 15 min. From Fig. 9 it appears that, compared to the primary ZDDPs there is less difference between the boundary friction of the three secondary ZDDPs, with MPE-2°and OCT-2° being very similar and BUT-2° being slightly lower.
5.2 MTM Tribofilm Thickness
Figure 10 shows how tribofilm thickness varied with rubbing time for the seven ZDDP solutions. Repeat tests were carried out and for film thickness greater than 20 nm the variation was ± 8 nm. Three of the primary ZDDPs, EHX-1°, DOD-1° and CYC-1° show similar film formation rates, while OCT-1° appears to form a film slightly faster. The three secondary ZDDPs all show faster initial film formation than the primaries, in the order BUT-2° > MPE-2° > OCT-2°. MPE-2° and OCT-2° appear to show a levelling-out of film formation at ca 60 nm thickness, but extended testing described in the next section showed that for MPE-2° this is only a temporary slow-down of film growth. BUT-2° forms a film very rapidly and this levels out at ca 150 nm thickness after one hour of rubbing.
5.3 Prolonged MTM Tests
All the above tests were of 2-h duration. Two much longer tests lasting 120 h (5 days) were carried out on DOD-1° and MPE-2° solutions to see whether their difference in boundary friction persisted for this period of rubbing. Figure 11 compares the friction of the ZDDP solutions and it is evident that the lower friction of DOD-1° continues throughout the whole test period. Figure 12a shows how measured tribofilm thicknesses vary over the 120-h tests. MPE-2° tribofilm grows over 30 h rubbing to reach a stable thickness of ca 140 nm thickness, while DOD-1° levels out at ca 50 nm, decreasing slightly over the second half of the test. At first sight there appears to be a discrepancy between the film growth by MPE-2° in Fig. 12a, where the film stabilises at 140 nm, and that in Fig. 10, where it appears to stabilise at ca 60 nm. However, Fig. 12b highlights the first four hours of the 120-h test on MPE-2° and indicates that film growth pauses after 30 min rubbing before resuming growth at a slower rate after about 2 h, from which it eventually reaches 140 nm thickness. This behaviour appears to be characteristic of this ZDDP under these test conditions since it was seen in three repeat tests. It is believed to reflect the interplay between tribofilm formation, its removal by rubbing, and its gradual conversion during rubbing to a more wear-resistant, nanocrystalline structure [5].
5.4 Replacement by Base Oil
Tests were carried out in which, after rubbing in ZDDP solution for two hours, the test was halted and the solution replaced with ZDDP-free base oil before resuming slow speed rubbing for a further hour. To ensure that all free ZDDP was removed, the ZDDP solution was drained and replaced by toluene before rotating ball and disc in the toluene for five minutes at 80 °C without any applied load. This procedure was repeated twice more with fresh toluene and then once with base oil before finally replacing this base oil with a fresh base oil sample.
Figure 13a shows a series of Stribeck curves for a test where ZDDP DOD-1° solution was replaced by base oil (BO). It is evident that boundary friction coefficient rises to 0.13 after just one minute of slow speed rubbing in base oil before falling slightly during further rubbing. Figure 13b shows how the friction coefficient, averaged from the four slow sliding speed measurements made between U = 10 to 15 mm/s, varies with rubbing time throughout the whole test. It can be seen that the effect on friction of rubbing in base oil occurs within one minute of rubbing and that friction coefficient eventually stabilises at ca 0.12. The continuous measurements made from the MTM load cell indicated that the friction increase was, in fact, practically immediate, with high friction present as soon as rolling-sliding in base oil began. Figure 14 shows a similar set of results from a test where ZDDP CYC-1° solution was replaced by base oil (BO). In this case the friction remained at a high value when the ZDDP tribofilm was cleaned in toluene and rubbed in base oil.
Figure 15 shows how tribofilm thickness varied with rubbing time both before and after the DOD-1° and CYC-1° solutions were replaced by base oil. Film build-up is quite linear with rubbing time for the ZDDP solutions, but when this solution is replaced by base oil, about 15 to 20 nm of film is lost quite rapidly.
To test whether the toluene cleaning procedure was too severe, a repeat test was carried out in which the ball and disc were rubbed in DOD-1° solution and then hexane substituted for toluene in the cleaning process outline above, before testing in base oil. This gave the same friction and film thickness behaviour to that seen after cleaning in toluene.
This loss of some ZDDP tribofilm when rubbed in base oil has been previously reported and shown to occur when the film has formed over a relatively short rubbing time under mild conditions [5, 25]. In this case the outer part of the tribofilm has an amorphous structure and is relatively weak. During prolonged rubbing the tribofilm becomes nanocrystalline and much stronger and thus not significantly removed during rubbing in base oil [5].
5.5 Replacement by Another ZDDP Solution
Figure 16a and b shows the effect on boundary friction of replacing DOD-1° solution by MPE-2° solution after two hours rubbing and vice versa. The same toluene rinse method was used as for replacement by base oil and again the boundary friction is an average of measurements made at four entrainments speeds between 10 and 15 mm/s. There is a clear and apparently immediate transition from the friction properties of the initial ZDDP to that characteristic of its replacement.
Figure 17 shows the change in tribofilm thickness when DOD-1° solution is replaced by MPE-2° solution and vice versa. When DOD-1° is replaced by MPE-2°, the film thickness rises very rapidly by about 20 nm before film growth slows down to about the same rate as for the initial DOD-1°. When MPE-2° is replaced by DOD-1°, about 10 nm of film is lost very quickly and then the film stabilises at about 50 nm.
5.6 HFRR Results
Figure 18 shows how the HFRR friction coefficient varies with rubbing time for two of the ZDDP solutions. For OCT-1°, measurements from two tests are shown, while for MPE-2° four tests are shown. For the primary OCT-1° the traces are smooth and very repeatable and this was the case for all four primary ZDDPs. By contrast those for MPE-2° are less regular and less repeatable and this was also seen for the other two secondary ZDDPs.
Figure 19 compares the HFRR friction coefficients for all seven ZDDP solutions, while Fig. 20 compares these HFRR friction values with those from MTM tests after two hours rubbing. The HFRR friction coefficient values are averaged over the last 1.5 h of the two-hour tests, while MTM values are mean values from the four measurements made between U = 10 mm/s and U = 15 mm/s. The HFRR friction coefficient values are higher than those measured in the rolling-sliding MTM contact but, with the exception of BUT-2°, there is close correlation between them. BUT-2° shows low MTM friction but very high HFRR friction.
Figure 21 compares the HFRR wear behaviour of the seven ZDDP solutions. All values shown are the mean of at least two tests and the variation between repeats was less than ± 3% for all solutions except for BUT-2° which had a range of ± 8%. The wear is quite similar for all the ZDDPs except for BUT-2°, which shows very high wear.
5.7 Ultralow Sliding Speed Results
As noted earlier in this paper, Studt measured the boundary friction coefficients of a range of ZDDPs and proposed that their response mirrored that of organic friction modifiers and resulted from ZDDP molecular adsorption on steel. To test this further, the ZDDP solutions of the current study were tested in a very slow speed sliding ball on disc rig. To limit the possibility of both tribochemical and thermal reactions, tests were carried out at 40 °C and involved just ten rotations of the disc. Figures 22 and 23 show measured friction coefficients of the seven ZDDP solutions and also the base oil. Figure 22 shows the variation of friction over just the initial half rotation of the disc, while Fig. 23 shows how friction varied over ten disc rotations.
The friction coefficient of the base oil is quite low since it is a Group 2 oil rather than a PAO or pure hydrocarbon as used in many studies. However there is a clear dependence on friction of ZDDP structure in essentially the same order as seen in MTM and HFRR tests, i.e. DOD < OCT < EHX < CYC. There is also an immediate reduction in friction, strongly suggesting an initial adsorbed film on steel. This is followed by a further fall during ten rubbing rotations, perhaps indicating either a packing density increase or the initial formation of a reaction layer with superior friction or ZDDP adsorption properties.
6 General Discussion
It is evident from these results that for primary ZDDPs, alkyl structure plays a strong role in determining the boundary friction properties of their tribofilms, with friction varying according to;
linear chains < branched chains < cyclic rings
&
long chains < short chains.
Furthermore, in the MTM the linear alkyl ZDDPs have boundary friction that increases linearly with log(SRR) and thus, at fixed entrainment speed, with log(sliding speed). This is commonly seen with linear, saturated organic friction modifiers (OFMs) [21] and was also noted by Hoshino et al. for a linear ZDDP [20]. Boundary friction coefficient is higher in the more severe, pure sliding HFRR tests than in the rolling-sliding MTM tests but the overall dependence on alkyl structure is the same, while the very slow speed linear sliding tests also show this order. With the exception of BUT-2°, ZDDPs with secondary alkyl chains show intermediate friction in MTM and HFRR tests, lower than the branched and ring primaries but higher than the linear ones. However in the very slow speed linear sliding tests, EHX-1° gives lower friction than two of the secondaries. BUT-2° is anomalous, giving quite low friction in the MTM and very slow speed linear sliding but very high friction in the HFRR.
The question that arises is what is the nature of the boundary film that gives rise to this boundary friction dependence on alkyl structure? There are four broad possibilities;
-
(i)
An adsorbed ZDDP monolayer
-
(ii)
An adsorbed monolayer of alkyl species derived from ZDDP
-
(iii)
A viscous surface layer of alkyl-containing, ZDDP-derived material
-
(iv)
A monolayer of residual alkyl chains chemically bonded to polyphosphate
The simplest of these is that ZDDP molecules are adsorbed on the polyphosphate tribofilm surface, with the linear alkyl groups in DOD-1° and OCT-1° arranged vertically to form a close-packed monolayer and the branched and cyclic ones, EHX-1°, cyclic CYC-1° and also the secondaries not forming such close-packing. In consequence they give similar friction coefficients to OFMs with the same alkyl group structures. The increase in boundary friction with log(sliding speed) is also characteristic of OFMs that form close-packed alkyl monolayers [21].
The very slow sliding speed measurements suggest that ZDDP adsorbs on steel surfaces to influence boundary friction in a similar fashion to OFMs and this is the mechanism proposed by Studt [17]. Dacre and Bovington showed that ZDDP adsorbed on steel discs to form a monolayer and that most of this adsorption was reversible except at high temperatures and after lengthy immersion. [26]. Yamaguchi et al. applied inelastic electron tunnelling spectroscopy to show that ZDDP adsorbs on aluminium at room temperature via the P = S bond and that the alkyl groups do not interact directly with the surface [27]. Ueda et al. have recently used quartz crystal microbalance (QCM) to show the rapid adsorption of ZDDP from PAO on to Fe2O3 to form a film of order one monolayer thick [28]. It is not known whether similar adsorption takes place on phosphate/polyphosphate tribofilms. It is evident from the MTM tests in which one ZDDP solution is replaced by another or by base oil that the resulting friction change occurs immediately upon resumption of rubbing. This may be because an initial layer of adsorbed ZDDP molecules has been removed but may equally well represent the removal of another type of thin surface film by rubbing.
The second possibility is that free, alkyl-based polar species eliminated during ZDDP tribofilm formation, such as alcohols or thiols, adsorb on the tribofilm surfaces and act as friction modifiers. The detailed molecular mechanism by which ZDDP polymerises to form polyphosphate is not known but must involve loss of alkyl-containing species. Two suggested products are alkenes and dialkyl sulphides [29]. However these are generally quite volatile and would not be expected to adsorb on solid surfaces at the temperature employed.
The third possible mechanism listed above to explain the noted dependence of friction on alkyl structure of ZDDPs may be the accumulation on the surfaces of a thin viscous layer of alkylphosphate on top of the solid. Such viscous layers were suggested by Bec et al. based on a surface forces apparatus study [30]. If present they would experience high pressures between opposing pads and it is well known that the elastohydrodynamic friction present in thin film, high pressure contacts depends on the ability of small molecules to slip past one another, which is determined by their molecular linearity and flexibility [31]. From friction measurements alone we cannot distinguish between the two possibilities of a close-packed array of vertically-oriented alkyl groups on the tribofilm surface or a very thin micro-elastohydrodynamic film. It should, however, be noted that other studies have failed to find a viscous layer on top of ZDDP tribofilms [32]. The possible origins of this discrepancy have been discussed in [3].
The fourth possibility listed above is that there are residual alkoxy groups bonded to polyphosphate at the surface that control boundary friction in a similar fashion to a chemically-adsorbed organic friction modifier. Only one alkyl group needs to be lost to form a polyphosphate from dialkyldithiophosphate or its isomer dithioalkylphosphate, although both alkyl groups must be lost to form a phosphate. It is therefore possible that initial tribofilm formation results in a film that contains alkoxy or thioalkyl groups bonded to the polyphosphate, especially at the surface where there is newly-formed material. The possibility that not all alkoxy groups are removed from the polyphosphate chains formed during tribofilm formation may be responsible for the persistence of high friction seen when CYC-1° solution is replaced by base oil in Fig. 14. If such alkoxy groups can be detected within the tribofilm, then of course they should also be present at the surface of the initially formed film.
To test for the presence of alkoxy groups within the tribofilms formed by ZDDPs, X-ray photoelectron spectroscopy (XPS) was used to analyse disc tracks from 3 h tests in which the lubricant was changed from ZDDP solution to base oil after 2-h rubbing, as illustrated in Figs. 13, 14, 15. Figure 15 shows that rubbing in base oil removed ca 20 nm of the tribofilm, presumably exposing initially buried film, and since XPS probes only the first few nanometers of a surface this might reveal whether carbon was present in the film and in what form. After one hour rubbing in base oil, the discs were rinsed in analytical grade toluene, dried with compressed air and, to limit contamination, gently wrapped in aluminium foil for transport to the XPS instrument. XPS analysis was carried out without any sputtering. The XPS instrument and conditions employed are described in [5], with charging of the specimen corrected by referring aliphatic carbon binding energy to 284.8 eV (C 1 s signal).
Figure 24a and b shows carbon 1 s XPS spectra from inside and outside the rubbed track of a CYC-1° tribofilm on a disc after rubbed in base oil. In both cases, spectra were obtained from three different regions and were all very similar. The spectra in the track indicate the presence of C–C/C-H, C–O bonds and C=O bonds. The C–C might originate from the toluene used to remove base oil, while all three bonds might, in principle, represent adventitious organic contamination from the atmosphere. However the fact that much more C-O is present within the track than outside it is strong evidence of the presence of alkoxy groups within the original film. Figure 24c and d shows the corresponding spectra for an MPE-2° tribofilm rubbed in base oil. Within the track there is much less C-O than for CYC but still more than outside the track. The level of C-O detected in DOD-1° tribofilm was similar to that of MPE-2°. The O 1 s spectra of the CYC-1° and MPE-2° tribofilms rubbed in base oil in Fig. 24e and f show a oxide (probably zinc and/or iron oxide), together with non-bonded O linked to phosphorus (NBO) and lesser amounts of oxygen linking two phosphorus atoms (BO) [4]. Phosphorus 2p and Zinc 2p spectra showed the presence of these elements, while sulphur 2p spectra showed sulphide but negligible sulphate. Tests were also carried out in which a tribofilm was formed from CYC-1° in polyalphaolefin (PAO4) base oil, followed by rubbing in PAO. These gave very similar results to Fig. 24, confirming that the C-O bonds detected in the CYC-1° tribofilm did not originate from very low levels of oxygenates that might be present the Group II base oil, but not in a PAO.
These XPS results from surfaces from which some tribofilm was removed by rubbing in base oil demonstrate the presence of significant quantities of C-O within the CYC-1° tribofilm, and smaller, though still detectable amounts in DOD-1° and MPE-2° tribofilms. This lends some support, at least for CYC-1°, to the fourth mechanism by which ZDDP might influence boundary friction outlined above, i.e. the presence of residual alkoxy groups chemically bonded to polyphosphate at the tribofilm surface.
HFRR friction values are higher than the MTM ones but this is as expected since the HFRR contact conditions at more severe than those in the MTM, with one surface always in contact and negligible entrainment at reversal points. The way that HFRR friction results depend on ZDDP alkyl structure agree with the MTM ones for all fluids except BUT-2° solution. BUT-2° ZDDP is clearly anomalous in several ways; it forms a tribofilm very quickly in rolling-sliding conditions and it gives high friction in the HFRR; it gives very high wear in the HFRR. The reason for the very different behaviour of this secondary butyl ZDDP from other secondary ZDDPs is not known; there appear to have been very few published comparisons of the behaviour of ZDDPs with different secondary alkyl structures.
7 Conclusions
The frictional properties of ZDDP tribofilms at low entrainment speeds in boundary lubrication conditions have been studied in both rolling/sliding and pure sliding contacts. The boundary friction coefficients of these tribofilms depend on the alkyl structure of the ZDDPs. For primary ZDDPs, those with linear alkyl chains give lower friction than those with branched alkyl chain ZDDPs, and a cyclohexylmethyl-based ZDDP gives markedly higher friction than non-cyclic ones. Depending on alkyl structure, boundary friction coefficient in rolling-sliding conditions can range from 0.09 to 0.14. These differences persist for long duration tests lasting up to 120 h. For secondary ZDDPs, boundary friction appears to depend less strongly on alkyl structure and in rolling-sliding conditions stabilises at ca 0.115 for the three ZDDPs studied.
Experiments in which the ZDDP-containing lubricant is changed after tribofilm formation by a different ZDDP solution or a base oil indicate that the characteristic friction of the initial ZDDP tribofilm is lost almost as soon as rubbing commences in the new lubricant. The boundary friction rapidly stabilises at the characteristic boundary friction of the replacement ZDDP, or in the case of base oil, a value of ca 0.115. One exception is when a solution containing a cyclohexylethyl-based ZDDP is replaced by base oil. In this case, the boundary friction coefficient remains at the high value characteristic of this ZDDP, despite the fact that rubbing in base oil removes about 20 nm of tribofilm. XPS analysis of the residual tribofilm indicates the presence of a considerable proportion of C-O bonds, which indicate that some alkoxy groups remains within the tribofilm to influence boundary friction properties.
Very slow speed rubbing tests at low temperature show that the ZDDP solutions give boundary friction values that vary with alkyl group structure in a similar fashion to rolling-sliding MTM tests. These variations in friction occur immediately on rubbing, before any measurable tribofilm can develop.
These results suggest that ZDDPs influence boundary friction by adsorbing on rubbing steel and tribofilm surfaces, with opposing, outwardly-oriented alkyl groups providing a shear plane in a fashion similar to organic friction modifiers. However, it is also believed that, for primary ZDDPs, residual alkoxy groups still chemically bonded to the phosphorus atoms of newly-formed polyphosphate/phosphate tribofilm may contribute to friction. When adsorbed ZDDP or residual alkoxy groups are removed by rubbing in base oil, the boundary friction coefficient reverts to a value of ca 0.115 to 0.12, characteristic of inorganic polyphosphate/phosphate.
This study shows that friction in mixed and boundary lubrication conditions can be controlled by the structure of a ZDDP’s alkyl groups. It provides further understanding of the frictional properties of engine lubricants and how these can be optimised by ZDDP molecular design.
Change history
24 February 2021
A Correction to this paper has been published: https://doi.org/10.1007/s11249-021-01401-5
References
Gosvami, N.N., Bares, J.A., Mangolini, F., Konicek, A.R., Yablon, D.G., Carpick, R.W.: Mechanisms of antiwear tribofilm growth revealed in situ by single-asperity sliding contacts. Science 348(6230), 102–106 (2015)
Zhang, J., Spikes, H.A.: On the mechanism of ZDDP antiwear film formation. Tribol. Lett. 63(24), 1–15 (2016)
Spikes, H.A.: The history and mechanisms of ZDDP. Trib. Lett. 17, 469–489 (2004)
Heuberger, R., Rossi, A., Spencer, N.D.: XPS study of the influence of temperature on ZnDTP tribofilm composition. Trib. Lett. 25, 185–196 (2007)
Ueda, M., Kadiric, A., Spikes, H.A.: On the crystallinity and durability of ZDDP tribofilm. Trib. Lett. 67, 123 (2019)
Topolovec-Miklozic, K., Forbus, T.R., Spikes, H.A.: Film thickness and roughness of ZDDP antiwear films. Trib Lett. 26, 161–171 (2007)
Dawczyk, J., Morgan, N., Russo, J., Spikes, H.A.: ZDDP tribofilm in rolling-sliding contact. Trib. Lett. 67, 34 (2019)
Hutchins, I.M.: Tribology - Friction & Wear of Engineering Materials. Publ. E, Arnold (1992)
Martin, J.M., Mansot, J.L., Berbezier, I., Belin, M., Balossier, G.: Microstructural aspects of lubricated mild wear with zinc dialkyldithiophosphate. Wear 107, 355–366 (1986)
Habeeb, J.J., Stover, W.H.: The role of hydroperoxides in engine wear and the effect of zinc dialkyldithiophosphates. ASLE Trans. 30, 419–426 (1987)
Rounds, F.: Effects of hydroperoxides on wear as measured in four-ball wear tests. Trib. Trans. 36, 297–303 (1993)
Kennedy, S., Moore, L.D.: Additive effects on lubricant fuel economy. SAE Trans. 96, 681–691 (1987)
Bovington, C. Anghel, V., Spikes, H.A. (1996) Predicting sequence VI and VIA fuel economy from laboratory bench tests. SAE Techn. Paper 961142,
Tripaldi, G., Vettor, A., Spikes, H.A.: Friction behaviour of ZDDP films in the mixed boundary/EHD regime. SAE Tech. Paper 962036, (1996)
Smeeth M. The behaviour of viscosity index improvers in lubricated contacts. Chapter 8. PhD Thesis University of London, May 1998.
Taylor, L., Spikes, H.A.: Friction-enhancing properties of ZDDP antiwear additive Part I Friction and morphology of ZDDP reaction films. Trib. Trans. 46, 303–309 (1996)
Studt, P.: Influence of lubricating oil additives on friction of ceramics under conditions of boundary lubrication. Wear 115, 185–191 (1987)
Studt, P.: Boundary lubrication: adsorption of oil additives on steel and ceramic surfaces and its influence on friction and wear. Trib. Intern. 22, 111–119 (1989)
Aoki, S., Suzuki, A., Masuko, M.: Comparison of sliding speed dependency of friction between steel surfaces lubricated with several ZnDTPs with different hydrocarbon moieties. Proc. Inst. Mech. Eng. J220, 343–351 (2006)
Hoshino, K., Yagashita, K., Tagawa, K., Spikes, H.A.: Tribological properties of sulphur-free antiwear additives zinc dialkylphosphates (ZDPs). SAE Intern. J. Fuels and Lubricants 5, 504–510 (2012)
Campen, S., Green, J.H., Lamb, G.D., Atkinson, D., Spikes, H.A.: On the increase in boundary friction with sliding speed. Trib. Lett. 48, 237–248 (2012)
Elvidge, B.R, Andrews, M.D: A lubricating composition. US Patent 2021/0103299 A1, (2012)
Hanneman, W.W., Porter, R.S.: The thermal decomposition of dialkyl phosphates and O. O-dialkyl dithiophosphates. Journal Org. Chem. 29, 2996–2998 (1964)
Choo, J.W., Olver, A.V., Spikes, H.A.: Influence of surface roughness features on mixed-film lubrication. Lubr. Sci. 15, 219–232 (2003)
Parsaeian, P., Ghanbarzadeh, A., Van Eijk, M.C., Nedelcu, I., Neville, A., Morina, A.: A new insight into the interfacial mechanisms of the tribofilm formed by zinc dialkyl dithiophosphate. Appl. Surf. Sci. 403, 472–486 (2017)
Dacre, B., Bovington, C.H.: The adsorption and desorption of zinc di-isopropyldithiophosphate on steel. ASLE Trans. 25, 546–554 (1982)
Yamaguchi, E.S., Ryason, P.R., Labrador, E.Q.: Inelastic electron tunneling spectra of neutral and basic zinc dithiophosphates on native aluminum oxide surfaces. Trib. Trans. 38, 243–252 (1995)
Ueda, M., Spikes, H.A., Kadiric, A.: ZDDP tribofilm formation on non-ferrous surfaces. Tribology Online 15, 318–331 (2020)
Zhang, J., Ewen, J.P., Ueda, M., Wong, J.S.S., Spikes, H.A.: Mechanochemistry of zinc dialkyldithiophosphate on steel surfaces under full-film elastohydrodynamic lubrication conditions. ACS Appl. Mater. Interfaces 12, 6662–6676 (2020)
Bec, S., Tonck, A., Georges, J.M., Coy, R.C., Bell, J.C., Roper, G.W.: Relationship between mechanical properties and structures of zinc dithiophosphate anti–wear films. Proc. Roy. Soc. Lond. Ser. A 455, 4181–4203 (1992)
Zhang, J., Tan, A., Spikes, H.A.: Effect of base oil structure on elastohydrodynamic friction. Trib. Lett. 65, 13 (2017)
Taylor, L., Spikes, H.A.: Friction-enhancing properties of ZDDP antiwear additive Part II Influence of ZDDP reaction films on EHD lubrication. Trib. Trans. 46, 310–314 (2003)
Acknowledgement
We thank the Engineering and Physical Sciences Research Council (EPSRC) for funding via EP/P030211/1 and AftonChemical and Shell for supply of additives and base oils.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version was revised due to incorrect figures and tables placements. It has been corrected.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, J., Ueda, M., Campen, S. et al. Boundary Friction of ZDDP Tribofilms. Tribol Lett 69, 8 (2021). https://doi.org/10.1007/s11249-020-01389-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11249-020-01389-4