Abstract
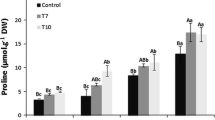
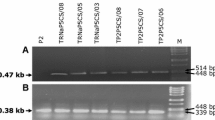
Δ1-pyrroline-5-carboxylate synthetase (P5CS) is one of the key regulatory enzymes involved in the proline biosynthetic pathway. Proline acts as an osmoprotectant, molecular chaperone, antioxidant, and regulator of redox homeostasis. The accumulation of proline during stress is believed to confer tolerance in plants. In this study, we cloned the complete CDS of the P5CS from pearl millet (Pennisetum glaucum (L.) R.Br. and transformed into tobacco. Three transgenic tobacco plants with single-copy insertion were analyzed for drought and heat stress tolerance. No difference was observed between transgenic and wild-type (WT) plants when both were grown in normal conditions. However, under heat and drought, transgenic plants have been found to have higher chlorophyll, relative water, and proline content, and lower malondialdehyde (MDA) levels than WT plants. The photosynthetic parameters (stomatal conductance, intracellular CO2 concentration, and transpiration rate) were also observed to be high in transgenic plants under abiotic stress conditions. qRT-PCR analysis revealed that the expression of the transgene in drought and heat conditions was 2–10 and 2–7.5 fold higher than in normal conditions, respectively. Surprisingly, only P5CS was increased under heat stress conditions, indicating the possibility of feedback inhibition. Our results demonstrate the positive role of PgP5CS in enhancing abiotic stress tolerance in tobacco, suggesting its possible use to increase abiotic stress-tolerance in crops for sustained yield under adverse climatic conditions.






Similar content being viewed by others
References
Amini S, Ghobadi C, Yamchi A (2015) Proline accumulation and osmotic stress: an overview of P5CS gene in plants. J Plant Mol Breed 3(2):44–55. https://doi.org/10.22058/JPMB.2015.17022
Babitha KC, Vemanna RS, Nataraja KN, Udaykumar M (2015) Overexpression of EcbHLH57 transcription factor from Eleusine coracana L. in tobacco confers tolerance to salt, oxidative and drought stress. PLos One 10(9):e0137098. https://doi.org/10.1371/journal.pone.0137098
Bandurska H, Niedziela J, Pietrowska-Borek M, Nuc K, Chadzinikolau T, Radzikowska D (2017) Regulation of proline biosynthesis and resistance to drought stress in two barley (Hordeum vulgare L.) genotypes of different origin. Plant Physiol Biochem 118:427–437. https://doi.org/10.1016/j.plaphy.2017.07.006
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water-stress studies. Plant Soil 39:205–207
Bhatnagar-Mathur P, Vadez V, Jyostna DM, Lavanya M, Vani G, Sharma KK (2009) Genetic engineering of chickpea (Cicer arietinum L.) with the P5CSF129A gene for osmoregulation with implications on drought tolerance. Mol Breeding 23:591–606. https://doi.org/10.1007/s11032-009-9258-y
Chakraborty K, Sairam RK, Bhattacharya RC (2012) Salinity-induced expression of pyrrolline-5-carboxylate synthetase determine salinity tolerance in Brassica spp. Acta Physiol Plant 34:1935–1941. https://doi.org/10.1007/s11738-012-0994-y
Chen JB, Wang SM, Jing RL, Mao XG (2009) Cloning the PvP5CS gene from common bean (Phaseolus vulgaris) and its expression patterns under abiotic stresses. J Plant Physiol 166(1):12–9. https://doi.org/10.1016/j.jplph.2008.02.010
Chen JB, Yang JW, Zhang ZY, Feng XF, Wang SM (2013) Two P5CS genes from common bean exhibiting different tolerance to salt stress in transgenic Arabidopsis. J Genet 92:461–469. https://doi.org/10.1007/s12041-013-0292-5
Chen H, Nelson RS, Sherwood JL (1994) Enhanced recovery of transformants of Agrobacterium tumefaciens after freeze-thaw transformation and drug selection. Biotechniques 16:664–670
Dhandapani G, Kanakachari M, Padmalatha KV, Phanindra MLV, Singh VK, Raghavendrarao S, Jayabalan N, Lakshmi Prabha A, Kumar PA (2014) A gene encoding cold-circadian rhythm-RNA binding-like protein (CCR-Like) from upland cotton (Gossypium hirsutum L) confers tolerance to abiotic stresses in transgenic tobacco. Plant Mol Biol Rep. https://doi.org/10.1007/s11105-014-0729-x
Gang C, Wang Z, Chen Y, Yang Y, Li J, Cheng J, Qi J, Odeh I (2016) Drought-induced dynamics of carbon and water use efficiency of global grasslands from 2000 to 2011. Ecol Indicat 67:788–797. https://doi.org/10.1016/j.ecolind.2016.03.049
Hall TA (1995) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser 41:95–98
Hassan N, El-bastawisy Z, Ebeed H, Nemat Alla MN (2019) Role of defense enzymes, proteins, solutes and Δ1-pyrroline-5-carboxylate synthase in wheat tolerance to drought. Rend Fis Acc Lincei 26:281–291. https://doi.org/10.1007/s12210-015-0429-y
Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A (2012) Role of proline under changing environments: a review. Plant Signal Behav 7(11):1456–1466. https://doi.org/10.4161/psb.21949
Hazariak P, Rajam MV (2011) Biotic and abiotic stress tolerance in transgenic tomatoes by constitutive overexpression of S-adenosylmethionine decarboxylase gene. Physiol Mol Biol Plants 17(2):115–128. https://doi.org/10.1007/s12298-011-0053-y
He M, He C-Q, Ding N-Z (2018) Abiotic stresses: general defenses of land plants and chances for engineering multistress tolerance. Front Plant Sci 9:1771. https://doi.org/10.3389/fpls.2018.01771
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplast. I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Hendrawan VSA, Komori D, Kim W (2023) Possible factors determining global-scale patterns of crop yield sensitivity to drought. PLoS One 18(2):e0281287. https://doi.org/10.1371/journal.pone.0281287
Hiscox JD, Israelstam GF (1979) A method for the extraction of chlorophyll from leaf tissue without maceration. Can J Bot 57(12):1332–1334
Hmida-Sayari A, Gargouri-Bouzid R, Bidani A, Jaoua L, Savoure A, Jaoua S (2005) Overexpression of delta1-pyrroline-5-carboxylate synthetase increases proline production and confers salt tolerance in transgenic potato plants. Plant Sci 169:746–752
Hong Z, Lakkineni K, Zhang Z, Verma DPS (2000) Removal of feedback inhibition of pyrroline-5-carboxylate synthetase results in increased proline accumulation and protection of plants from osmotic stress. Plant Physiol 122:1129–1136
Horsch RB, Fry JE, Hoffmann N, Eicholz D, Rogers SG, Fraley RT (1985) A simple and general method for transferring genes into plants. Science 227:1229–1231. https://doi.org/10.1126/science.227.4691.1229
Hu CA, Delauney AJ, Verma DP (1992) A bifunctional enzyme (delta 1-pyrroline-5-carboxylate synthetase) catalyzes the first two steps in proline biosynthesis in plants. Proc Natl Acad Sci USA 89(19):9354–8. https://doi.org/10.1073/pnas.89.19.9354
Ibragimova SM, Trifonova EA, Filipenko EA, Shymny VK (2015) Evaluation of salt tolerance of transgenic tobacco plants bearing with P5CS1 gene of Arabidosis thaliana. Genetika 51(12):1368–1375
Igarashi Y, Yoshiba Y, Sanada Y, Yamaguchi-Shinozaki K, Wada K, Shinozaki K (1997) Characterization of the gene for Δ1-pyrroline-5-carboxylate synthetase and correlation between the expression of the gene and salt tolerance in Oryza sativa. Plant Mol Biol 33:857–865. https://doi.org/10.1023/a:1005702408601
James D, Tarafdar A, Biswas K, Sathyavathi TC, Padaria JC, Kumar PA (2015) Development and characterization of a high temperature stress responsive subtractive cDNA library in Pearl Millet Pennisetum glaucum (L.) R.Br. Indian J Exp Biol 53:543–550
Kido EA, Ferreira Neto JR, Silva RL, Belarmino LC, Bezerra Neto JP, Soares-Cavalcanti NM et al (2013) Expression dynamics and genome distribution of osmoprotectants in soybean: identifying important components to face abiotic stress. BMC Bioinf. https://doi.org/10.1186/1471-2105-14-S1-S7
Kishor PBK, Hong J, Miao G, Hu C, Verma DPS (1995) Overexpression of ∆1-Pyrroline -5-carboxylate synthetase increases proline production and confers osmotolerance in transgenic plants. Plant Physiol 108:1387–1394
Kishor PBK, Sangam S, Amrutha RN, Lakshmi PS, Naidu KR, Rao KRSS et al (2005) Regulation of proline biosynthesis, degradation, uptake and transport in higher plants: its implications in plant growth and abiotic stress tolerance. Curr Sci 88(3):424–438
Kumar V, Shriram V, Kavi Kishor PB, Jawali N, Shitole MG (2010) Enhanced proline accumulation and salt stress tolerance of transgenic indica rice by over-expressing P5CSF129A gene. Plant Biotechnol Rep 4:37–48. https://doi.org/10.1007/s11816-009-0118-3
Liang X, Zhang L, Natarajan SK, Becker DF (2013) Proline mechanisms of stress survival. Antioxid Redox Signal 19(9):998–1011. https://doi.org/10.1089/ars.2012.5074
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-∆∆CT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Lv WT, Lin B, Zhang M, Hua XJ (2011) Proline accumulation is inhibitory to Arabidopsis seedlings during heat stress. Plant Physiol 156:1921–2193. https://doi.org/10.1104/pp.111.175810
Ma C, Wang M, Zhao M, Yu M, Zheng X, Tian Y et al (2022) The Δ1-pyrroline-5-carboxylate synthetase family performs diverse physiological functions in stress responses in pear (Pyrus betulifolia). Front Plant Sci 13:1066765. https://doi.org/10.3389/fpls.2022.1066765
Maibam A, Lone SA, Ningombam S, Gaikwad K, Amitha Mithra SV, Singh MP et al (2022) Transcriptome analysis of Pennisetum glaucum (L.) R. Br. provides insight into heat stress responses. Front Genet 13:884106. https://doi.org/10.3389/fgene.2022.884106
Mirzaei M, Pascovici D, Atwell BJ, Haynes PA (2012) Differential regulation of aquaporins, small GTPases and V-ATPases proteins in rice leaves subjected to drought stress and recovery. Proteomics 12:864–877. https://doi.org/10.1002/pmic.201100389
Padaria JC, Thuy NT, Tarafdar A, Yadav R (2015) Development of a heat-responsive cDNA library from Prosopis cineraria and molecular characterization of the Pchsp17.9 gene. J Hort Sci Biotechnol 90:318–324. https://doi.org/10.1080/14620316.2015.11513189
Padaria JC, Yadav R, Tarafdar A, Lone SA, Kumar K, Sivalingam PN (2016) Molecular cloning and in-silico characterization of drought stress-responsive abscisic acid-stress ripening (Asr1) gene from wild jujube Ziziphus nummularia (Burm. F.) Wight & Arn. Mol Biol Rep 43(8):849–859. https://doi.org/10.1007/s11033-016-4013-z
Panzade PK, Vishwakarma H, Padaria JC (2020) Heat stress-inducible cytoplasmic isoform of ClpB1 from Z. nummularia exhibits enhanced thermotolerance in transgenic tobacco. Mol Biol Rep 47(5):3821–3831. https://doi.org/10.1007/s11033-020-05472-w
Parida AK, Dagaonkar VS, Phalak MS, Aurangabadkar LP (2008) Differential responses of the enzymes involved in proline biosynthesis and degradation in drought tolerant and sensitive cotton genotypes during drought stress and recovery. Acta Physiol Plant 30:619–627. https://doi.org/10.1007/s11738-008-0157-3
Pavei D, Gonçalves-Vidigal MC, Schuelter AR, Schuster I, Vieira ESN, Vendruscolo ECG et al (2016) Response to water stress in transgenic (p5cs gene) wheat plants (Triticum aestivum L.). Ausn J Crop Sci 10(6):776–783
Ramachandra RA, Chaitanya KV, Jutur PP, Sumithra K (2004) Differential antioxidative responses to water stress among five mulberry (Morus alba L.) cultivars. Environ Exp Bot 52:33–42. https://doi.org/10.1016/j.envexpbot.2004.01.002
Riahi M, Ehsanpour AA (2013) Responses of transgenic tobacco (Nicotiana plambaginifolia) over-expressing P5CS gene under salt stress. Prog Biol Sci 8(2):76–84. https://doi.org/10.22059/PBS.2013.2708
Rogers Scott O, Bendich Arnold J (1994) Extraction of total cellular DNA from plants, algae and fungi. In: Gelvin Stanton B, Schilperoort Robbert A (eds) Plant molecular biology manual. Kluwer Academic Press, Dordrecht. https://doi.org/10.1007/978-94-011-0511-8_12
Rombel IT, Sykes KF, Rayner S, Johnston SA (2002) ORF-FINDER: a vector for high-throughput gene identification. Gene 282(1–2):33–41. https://doi.org/10.1016/s0378-1119(01)00819-8
Sambrook J, Russell DW (2001) Molecular cloning: A laboratory manual, 3rd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Senthil-Kumar M, Govind G, Kang L, Mysore KS, Udayakumar M (2007) Functional characterization of Nicotiana benthamiana homologs of peanut water deficit-induced genes by virus-induced gene silencing. Planta 225(3):523–539. https://doi.org/10.1007/s00425-006-0367-0
Signorelli S, Monza J (2017) Identification of ∆1-pyrroline 5-caboxylate synthase (P5CS) genes involved in the synthesis of proline in Lotus japonicus. Plant Signal Behav 12:11. https://doi.org/10.1080/15592324.2017.1367464
Simon-Sarkadi L, Kocsy G, Várhegyi A, Galiba G, De-Ronde GA (2006) Stress-induced changes in the free amino acid composition in transgenic soybean plants having increased proline content. Biol Plantarum 50(4):793–796. https://doi.org/10.1007/s10535-006-0134-x
Smart RE, Bingham GE (1974) Rapid Estimates of Relative Water Content. Plant Physiol 53(2):258–260. https://doi.org/10.1104/pp.53.2.258.s
Sung DY, Kaplan F, Lee KJ, Guy CL (2003) Acquired tolerance to temperature extremes. Trends Plant Sci 8:179–187. https://doi.org/10.1016/S1360-1385(03)00047-5
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38(7):3022–3027. https://doi.org/10.1093/molbev/msab120
Todaka D, Shinozaki K, Yamaguchi-Shinozaki K (2015) Recent advances in the dissection of drought-stress regulatory networks and strategies for development of drought-tolerant transgenic rice plants. Front Plant Sci 18(6):84. https://doi.org/10.3389/fpls.2015.00084
Vendruscolo EC, Schuster I, Pileggi M, Scapim CA, Molinari HB, Marur CJ, Vieira LG (2007) Stress-induced synthesis of proline confers tolerance to water deficit in transgenic wheat. J Plant Physiol 164(10):1367–1376. https://doi.org/10.1016/j.jplph.2007.05.001
Verdoy D, Coba De La Peña T, Redondo FJ, Lucas MM, Pueyo JJ (2006) Transgenic Medicago truncatula plants that accumulate proline display nitrogen-fixing activity with enhanced tolerance to osmotic stress. Plant Cell Environ 29(10):1913–1923. https://doi.org/10.1111/j.1365-3040.2006.01567.x
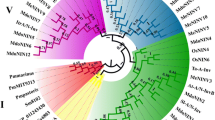
Rai AN, Penna S (2013) Molecular evolution of plant P5CS gene involved in proline biosynthesis. Mol Biol Rep 40:6429–6435. https://doi.org/10.1007/s11033-013-2757-2
Verslues PE, Bray EA (2006) Role of abscisic acid (ABA) and Arabidopsis thaliana ABA-insensitive loci in low water potential-induced ABA and proline accumulation. J Exp Bot 57:201–212. https://doi.org/10.1093/jxb/erj026
Webb B, Sali A (2014) Protein structure modeling with modeller. Methods Mol Biol 1137:1–15. https://doi.org/10.1007/978-1-4939-0366-5_1
Yamada M, Morishita H, Urano K, Shinozaki N, Yamaguchi-Shinozaki K, Shinozaki K et al (2005) Effects of free proline accumulation in petunias under drought stress. J Exp Bot 56:1975–1981. https://doi.org/10.1093/jxb/eri195
Yang L, Tang R, Zhu J, Liu H, Mueller-Roeber B, Xia H et al (2008) Enhancement of stress tolerance transgenic tobacco plants constitutively expressing AtIpk2β, an inositol polyphosphate 6-/3-kinase from Arabidopsis thaliana. Plant Mol Biol 66:329–343. https://doi.org/10.1007/s11103-007-9267-3
Yoshiba Y, Kiyosue T, Katagiri T, Ueda H, Mizoguchi T, Yamaguchi-Shinozaki K et al (1995) Correlation between the induction of a gene for delta 1-pyrroline-5-carboxylate synthetase and the accumulation of proline in Arabidopsis thaliana under osmotic stress. Plant J 5:751–760. https://doi.org/10.1046/j.1365-313x.1995.07050751.x
Zarei S, Ehsanpour AA, Abbaspour J (2012) The role of overexpression of P5CS gene on proline, catalase, ascorbate peroxidase activity and lipid peroxidation of transgenic tobacco (Nicotiana tabacum L.) plant under in vitro drought stress. J Cell Mol Res 4(1):43–49. https://doi.org/10.22067/jcmr.v4i1.18249
Zhang C, Lu Q, Verma DPS (1995) Removal of feedback inhibition of ∆1-Pyrroline-5-carboxylate synthetase, a bifunctional enzyme catalyzing the first two steps of proline biosynthesis in plants. J Biol Chem 270(35):20491–20496. https://doi.org/10.1074/jbc.270.35.20491
Zhang L, Becke DF (2015) Connecting proline metabolism and signaling pathways in plant senescence. Front Plant Sci 6:552. https://doi.org/10.3389/fpls.2015.00552
Zhang M, Huang H, Dai S (2014) Isolation and expression analysis of proline metabolism-related genes in Chrysanthemum lavandulifolium. Gene 537(2):203–13. https://doi.org/10.1016/j.gene.2014.01.002
Acknowledgements
The authors are thankful to the Director NIPB, New Delhi, India for providing facilities to carry out this research. Director IARI (Indian Agricultural Research Institute), New Delhi, India is duly thanked for providing National Phytotron facilities. The support by ICAR (Indian Council of Agricultural Research) under the National Innovations in Climate Resilient Agriculture project till March 2020 is duly acknowledged. We thank RNDr. Cepl Jaroslav, FLD, CZU, for statistical support.
Author information
Authors and Affiliations
Contributions
GS carried out tobacco transformation, physiological and biochemical assays, molecular analysis, and manuscript writing. MC, AT isolated PgP5CS gene. RSJ carried out the bioinformatic analysis. AT, RSJ involved in manuscript editing. HV was involved in the physiological and 2D-SDS PAGE. JCP conceptualized the research plan, designed experiments, involved in manuscript preparation and editing. All the authors have read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors reported no potential conflict of interest.
Research involving human participants and/or animals
Authors declare the research did not involve Human Participants and/or Animals.
Informed consent
Authors consent to the publishing of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sellamuthu, G., Tarafdar, A., Jasrotia, R.S. et al. Introgression of Δ1-pyrroline-5-carboxylate synthetase (PgP5CS) confers enhanced resistance to abiotic stresses in transgenic tobacco. Transgenic Res (2024). https://doi.org/10.1007/s11248-024-00385-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11248-024-00385-x