Abstract
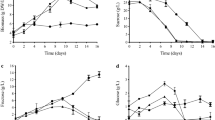
Bioreactors provide suitable conditions for the growth of cells and production of secondary metabolites by regulating physical and chemical factors. In this study, saffron cell growth and the production of crocin metabolite were evaluated in Erlenmeyer flask and in a stirred bioreactor. To optimize the conditions, the effect of different concentrations of 2-(N-morpholino) ethanesulfonic acid (MES) (0, 2.5 and 5 mM) as a buffering agent and sucrose (3%, 6% and gradually 6%) were addressed on saffron cell growth and crocin production in the Erlenmeyer flask. This aim was then pursued in a stirred bioreactor through investigation on the effect of aeration and pH medium adjustment. The results of the first step experiment showed that Schenk and Hildebrandt basal medium with naphthalene acetic acid (2 mg l−1) and 6-benzylaminopurine (1 mg l−1) supplemented with 2.5 mM of MES as well as gradual increment of sucrose from 3 to 6% caused the highest cell biomass and crocin production. The spectrophotometry measurement showed that the highest crocin content of the cells was 0.8 mg g−1 after 6 weeks. The results of the second part revealed that in the stirred bioreactor, constant pH (5.8) during the growth period is a limiting factor for the cell growth and crocin production. Aeration was also found to be an inhibiting factor for the production of crocin. The results showed that, if the evaporated volume of water caused by aeration is constituted, it can be an effective factor to increase cell growth around 2 folds. In addition, total crocin content of the proliferated cells in stirred bioreactor system under the non-constant pH and without aeration conditions, could be raised up to 2 mg g−1 of cell dry weight, based on the HPLC determination. According to the results of this study, it can be concluded that MES and gradual increment of sucrose could increase the cell growth and crocin production. In the tested stirred bioreactor, it was found that the natural pH fluctuation is a suitable condition for saffron biomass cell growth and crocin production. The proper aeration could increase the cell biomass, if the evaporated water is replaced. However based on the obtained results it can be concluded that, aeration at tested value is not capable of increasing the crocin production.
Key message
Saffron, the most expensive spice contains valuable compounds like crocin. Corm derived-cell containing crocin can be produced in higher scales and cheaper price by using cell culture in stirred bioreactor.














Similar content being viewed by others
Data availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
Notes
Volume per Volume Medium.
Abbreviations
- BA:
-
6-Benzylaminopurine
- MES:
-
2-(N-morpholino) ethanesulfonic acid
- NAA:
-
Naphthalene acetic acid
- SH:
-
Schenk and Hildebrandt (1972)
References
Ahmed S, Hahn EJ, Paek KY (2008) Aeration volume and photosynthetic photon flux affect cell growth and secondary metabolite contents in bioreactor cultures of Morinda citrifolia. J Plant Biol 51:209–212. https://doi.org/10.1007/BF03030700
Alam P, Elkholy SF, Hosokawa M, Mahfouz SA, Sharaf-eldin, M.A (2016) Simultaneous extraction and rapid HPLC based quantification of crocin and safranal in saffron (Crocus sativus L.). Int J Pharm Phytopharmac Res 8(10):224–227. https://doi.org/10.22159/ijpps.2016v8i10.12172
Andersone U, Ievinsh G (2008) Medium pH aff ects regeneration capacity and oxidative enzyme activity of Pinus sylvestris in tissue culture. Acta Univ Latv 745:25–35
Baker CJ, Mock NM, Roberts DP et al (2007) Interference by MES [2-(4-morpholino)ethanesulfonic acid] and related buffers with phenolic oxidation by peroxidase. Free Radic Biol Med 43:1322–1327. https://doi.org/10.1016/j.freeradbiomed.2007.07.020
Banthorpe DV, Brown GD (1990) Growth and secondary metabolism in cell cultures of Tanacetum, Mentha and Anethum species in buffered media. Plant Sci 67:107–113
Chen C-C, Bates R, Carlson J (2014) Effect of environmental and cultural conditions on medium pH and explant growth performance of Douglas-fir (Pseudotsuga menziesii) shoot cultures. F1000Research 3:298. https://doi.org/10.12688/f1000research.5919.2
De Klerk GJ, Hanecakova J, Jásik J (2008) Effect of medium-pH and MES on adventitious root formation from stem disks of apple. Plant Cell Tissue Organ Cult 95:285–292. https://doi.org/10.1007/s11240-008-9442-5
Dong Y, Gao W-Y, Man S et al (2013) Effect of bioreactor angle and aeration rate on growth and hydromechanics parameters in bioreactor culture of ginseng suspension cells. Acta Physiol Plant 35:1497–1501. https://doi.org/10.1007/s11738-012-1190-9
Doran PM (2010) Bioreactors, Stirred tank for culture of plant cells. In: Flickinger MC (ed) Encyclopedia of Industrial Biotechnology: Bioprocess, Bioseparation, and Cell Technology. Wiley, Hoboken. https://doi.org/10.1002/9780470054581.eib150
Gorelick J, Bernstein N (2014) Elicitation: an underutilized tool in the development of medicinal plants as a source of therapeutic secondary metabolites. In: Sparks DL (ed) Advances in agronomy. Academic Press, Amsterdam, pp 201–230
Guieysse B, Quijano G, Muñoz R (2011) Airlift bioreactors. In: Moo-Young M (ed) Comprehensive biotechnology, 2nd edn. Academic Press, Burlington, pp 199–212
Hilae A, Te-chato S (2005) Effects of carbon sources and strength of MS medium on germination of somatic embryos of oil palm (Elaeis quineensis Jacq.). Songklanakarin J Sci Technol 7:629–635
Jeong GT, Park DH (2005) Comparative evaluation of modified bioreactors for enhancement of growth and secondary metabolite biosynthesis using Panax ginseng hairy roots. Biotechnol Bioprocess Eng 10:528–534
Kagenishi T, Yokawa K, Baluška F (2016) MES buffer affects arabidopsis root apex zonation and root growth by suppressing superoxide generation in root apex. Front Plant Sci 7:79. https://doi.org/10.3389/fpls.2016.00079
Karuppusamy S (2009) A review on trends in production of secondary metabolites from higher plants by in vitro tissue, organ and cell cultures. J Med Plants Res 3:1222–1239
Kevács G, László M, Rajkai G, Barnabás B (1995) Monitoring of haploid maize cell suspension culture conditions in bioreactors. Plant Cell Tissue Organ Cult 43:123–126. https://doi.org/10.1007/BF00052166
Koch KE (1996) Carbohydrate-modulated gene expression in plants. Annu Rev Plant Physiol Plant Mol Biol 47:509–540. https://doi.org/10.1146/annurev.arplant.47.1.509
Kolewe M (2011) Development of plant cell culture processes to produce natural product pharmaceuticals : Characterization , Analysis , and Modeling of plant cell aggregation. Open Access Dissertations. 467
Leifert C, Pryce S, Lumsden PJ, Waites WM (1992) Effect of medium acidity on growth and rooting of different plant species growing in vitro. Plant Cell Tissue Organ Cult 30:171–179. https://doi.org/10.1007/BF00040019
Leifert C, Murphy KP, Lumsden PJ (1995) Mineral and carbohydrate nutrition of plant cell and tissue cultures. CRC Crit Rev Plant Sci 14:83–109. https://doi.org/10.1080/07352689509701923
Liu C, Guo C, Wang Y, Ouyang F (2002) Effect of light irradiation on hairy root growth and artemisinin biosynthesis of Artemisia annua L. Process Biochem 38:581–585. https://doi.org/10.1016/S0032-9592(02)00165-6
Loyola-Vargas VM, Ochoa-Alejo N (2018) An introduction to plant tissue culture: advances and perspectives. In: Loyola-Vargas VM, Ochoa-Alejo N (eds) Plant cell culture protocols. Springer, New York, pp 3–13
Malik S, Bhushan S, Verma SC, Sharma N, Sinha AK, Sharma M, Ahuja PS (2008) Production of naphthoquinone pigments in cell suspension cultures of Arnebia euchroma (Royale) Johnston: Influence of pH on growth kinetics and acetylshikonin. Med Aromat Plant Sci Biotechnol 2(1):43–49
Meijer JJ, ten Hoopen HJG, Luyben KCAM, Libbenga KR (1993) Effects of hydrodynamic stress on cultured plant cells: a literature survey. Enzyme Microb Technol 15:234–238. https://doi.org/10.1016/0141-0229(93)90143-P
Min JY, Jung HY, Kang SM et al (2007) Production of tropane alkaloids by small-scale bubble column bioreactor cultures of Scopolia parviflora adventitious roots. Bioresour Technol 98:1748–1753. https://doi.org/10.1016/j.biortech.2006.07.033
Minocha SC (1987) pH of the medium and the growth and metabolism of cells in culture. In: Bonga JM, Durzan DJ (eds) Cell and tissue culture in forestry: general principles and biotechnology. Springer, Dordrecht, pp 125–141
Mukundan U, Bhide V, Singh G, Curtis WR (1998) pH-mediated release of betalains from transformed root cultures of Beta vulgaris L. Appl Microbiol Biotechnol 50:241–245. https://doi.org/10.1007/s002530051283
Murthy HN, Lee EJ, Paek KY (2014) Production of secondary metabolites from cell and organ cultures: Strategies and approaches for biomass improvement and metabolite accumulation. Plant Cell Tissue Organ Cult 118:1–16. https://doi.org/10.1007/s11240-014-0467-7
Nagella P, Murthy HN (2010) Establishment of cell suspension cultures of Withania somnifera for the production of withanolide A. Bioresour Technol 101(17):6735–6739 https://doi.org/10.1016/j.biortech.2010.03.078
Nalawade SM, Tsay HS (2004) In vitro propagation of some important Chinese medicinal plants and their sustainable usage. In Vitro Cell Dev Biol Plant 40:143–154. https://doi.org/10.1079/IVP2003504
Nicholas JC, Harper JE (1993) Effect of MES [2(N-morpholino)ethanesulfonic acid] and amberlite IRC-50 resin on nutrient pH control and soybean growth. J Plant Nutr 16:895–909. https://doi.org/10.1080/01904169309364582
Paek KY, Murthy HN, Zhong JJ (2014) Production of biomass and bioactive compounds using bioreactor technology. Springer, Dordrecht
Parfitt DE, Almehdi AA, Bloksberg LN (1988) Use of organic buffers in plant tissue-culture systems. Sci Hortic (amsterdam) 36:157–163. https://doi.org/10.1016/0304-4238(88)90049-0
Pasqua G, Manes F, Monacelli B et al (2002) Effects of the culture medium pH and ion uptake in in vitro vegetative organogenesis in thin cell layers of tobacco. Plant Sci 162:947–955. https://doi.org/10.1016/S0168-9452(02)00048-1
Piao X-C, Zhang W-B, Jiang J et al (2017) Cell suspension culture of Orostachys cartilaginous in bioreactor systems for bioactive compound production and evaluation of their antioxidant properties. Acta Physiol Plant 39:70. https://doi.org/10.1007/s11738-017-2374-0
Pitzschke A, Hirt H (2010) New insights into an old story: Agrobacterium-induced tumour formation in plants by plant transformation. EMBO J 29:1021–1032. https://doi.org/10.1038/emboj.2010.8
Pujol AC (2016) Plant cells and suitable bioreactors. Dissertation, Universitat de Barcelona
Rahaiee S, Shojaosadati SA, Hashemi M et al (2015) Improvement of crocin stability by biodegradable nanoparticles of chitosan-alginate. Int J Biol Macromol 79:423–432. https://doi.org/10.1016/j.ijbiomac.2015.04.041
Ramulifho E, Goche T, Van As J et al (2019) Establishment and characterization of callus and cell suspension cultures of selected sorghum bicolor (L.) moench varieties: a resource for gene discovery in plant stress biology. Agronomy 9:218. https://doi.org/10.3390/agronomy9050218
Rao SR, Ravishankar GA (2002) Plant cell cultures: Chemical factories of secondary metabolites. Biotechnol Adv 20:101–153. https://doi.org/10.1016/s0734-9750(02)00007-1
Ryu DDY, Lee SO, Romani RJ (1990) Determination of growth rate for plant cell cultures: comparative studies. Biotechnol Bioeng 35:305–311
Schenk RU, Hildebrandt AC (1972) Medium and techniques for induction and growth of monocotyledonous and dicotyledonous plant cell cultures. Can J Bot 50:199–204. https://doi.org/10.1139/b72-026
Schmidt BM, Ribnicky DM, Lipsky PE, Raskin I (2007) Revisiting the ancient concept of botanical therapeutics. Nat Chem Biol 3:360–366. https://doi.org/10.1038/nchembio0707-360
Shi X, Yang L, Yan G, Du G (2017) Medium pH between 5.5 and 7.5 has minimal effects on tissue culture of apple. HortScience horts 52:475–478. https://doi.org/10.21273/HORTSCI11443-16
Singh Y, Bramhe P (2017) Cell viability testing with trypan blue exclusion method. Natl Inst Environ Health Sci 8–9. http://brd.nci.nih.gov/brd/sop/show/941
Soeda S, Ochiai T, Shimeno H et al (2007) Pharmacological activities of crocin in saffron. J Nat Med 61:102–111. https://doi.org/10.1007/s11418-006-0120-9
Sotiropoulos T, Molassiotis A, Mouhtaridou G et al (2006) Sucrose and sorbitol effects on shoot growth and proliferation in vitro, nutritional status and peroxidase and catalase isoenzymes of M 9 and MM 106 apple (Malus domestica Borkh.) rootstocks. Eur J Hortic Sci 71:114–119
Steingroewer J, Haas C, Winkler K et al (2017) Monitoring of plant cells and tissues in bioprocesses. In: Pavlov A, Bley T (eds) Bioprocessing of plant in vitro systems. Springer, Cham, pp 1–49
Thompson MR, Thorpe TA (1987) Metabolic and Non-Metabolic Roles of Carbohydrates. In: Bonga JM, Durzan DJ (eds) Cell and tissue culture in forestry: general principles and biotechnology. Springer, Dordrecht, pp 89–112
Thorat AS, Sonone NA, Choudhari VV et al (2017) Plant regeneration from cell suspension culture in Saccharum officinarum L. and ascertaining of genetic fidelity through RAPD and ISSR markers. 3 Biotech 7:16. https://doi.org/10.1007/s13205-016-0579-3
Tu S, Tétu T, Sangwan RS, Sangwan-Norreel BS (1996) Effect of 2-(n-morpholino)ethanesulfonic acid and myo-inositol on somatic embryogenesis and plant are generation from zygotic embryos of Hyoscyamus niger L. Plant Sci 114:193–203. https://doi.org/10.1016/0168-9452(95)04315-2
Wang GR, Qi NM (2010) Influence of mist intervals and aeration rate on growth and second metabolite production of Pseudostellaria heterophylla adventitious roots in a siphon-mist bioreactor. Biotechnol Bioprocess Eng 15:1059–1064. https://doi.org/10.1007/s12257-010-0091-2
Yaseen M, Ahmed T, Abbasi N et al (2009) In vitro shoot proliferation competence of apple rootstocks M. 9 and M. 26 on different carbon sources. Pak J Bot 41(4):1781–1795
Yildiz M, Onde S, Ozgen M (2007) Sucrose effects on phenolic concentration and plant regeneration from sugarbeet leaf and petiole explants. J Sugarbeet Res 44:1–16. https://doi.org/10.5274/jsbr.44.1.1
Zhao J, Davis LC, Verpoorte R (2005) Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol Adv 23:283–333. https://doi.org/10.1016/j.biotechadv.2005.01.003
Acknowledgements
The authors would like to acknowledge from the Research Institute of Food Science and Technology (RIFST) for providing all lab facilities and Iran National Science Foundation (INSF) for financial supports of this project.
Funding
The funding was provided by Research Institute of Food Science and Technology (RIFST), Iran.
Author information
Authors and Affiliations
Contributions
In this experiment, Ziaratnia and Hemmati designed the experiments. Amini carried out all the experiments, analyzed data and wrote the paper. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors hereby declare that there is no conflict of interest.
Additional information
Communicated by Christophe Hano.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Amini, S., Ziaratnia, S.M. & Hemmati, K. Optimization of conditions for increasing of saffron cell biomass and crocin production in stirred bioreactor. Plant Cell Tiss Organ Cult 149, 243–255 (2022). https://doi.org/10.1007/s11240-022-02233-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-022-02233-4