Abstract
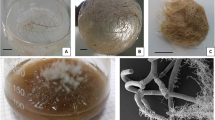
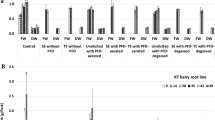
A vincamine positive hairy root clone of Vinca minor with integration of all the three- rolA, rolB and rolC genes and its subsequently raised cell suspensions were treated with culture filtrate of four endophytic fungi namely Chaetomium globosum; Aspergillus niveoglaucus; Paecilomyces lilacinus and Trichoderma harzianum. Addition of 10 % v/v T. harzianum culture filtrate boosted maximum biomass accumulation and highest total alkaloid content (TAC) both in the hairy roots and in the cell suspensions. Treated (10 % v/v T. harzianum) hairy roots registered growth index (GI) of 640 ± 28.3 and TAC of 3.6 ± 0.1 dry wt% in comparison to non-treated roots (GI = 420 ± 14.2; TAC = 3.1 ± 0.3 dry wt%). Treated cell suspensions showed GI of 600 ± 19.1 and TAC of 1.67 ± 0.03 dry wt% in comparison to non-treated cell suspensions (GI = 250 ± 12.6; TAC = 1.15 ± 0.02 dry wt%). The hairy roots and the cell suspensions were successfully up-scaled in the 5 l stirred tank bioreactor with respective GI of 850.0 and 654.0 under optimized conditions. On Real time (qPCR) analysis, treated hairy roots showed fourfold to sixfold enhanced tryptophan decarboxylase (TDC) transcript level [relative quantity value (RQ) = 4.64 ± 0.30 (shake flask); RQ = 5.95 ± 0.31 (bioreactor)] while treated cell suspensions showed only two fold increase in TDC transcript [RQ = 2.1 ± 0.26 (shake flask); RQ = 2.5 ± 0.21 (bioreactor)]. Similarly, fivefold to sixfold [RQ = 5.6 ± 0.20 (shake flask); RQ = 6.7 ± 0.49 (bioreactor)] and threefold to fourfold [RQ = 3.5 ± 0.18 (shake flask); RQ = 3.8 ± 0.68 (bioreactor)] increased transcript of strictosidine synthase (STR) in treated hairy roots and cell suspensions, respectively was observed. Treated shake flask roots showed 0.002 dry wt% vincamine that was further enhanced in the bioreactor grown treated roots (0.015 dry wt%). No vincamine was detected in the shake flask culture or in the bioreactor grown cell suspensions.





Similar content being viewed by others
References
Batra J, Dutta A, Singh D, Kumar S, Sen J (2004) Growth and terpenoid indole alkaloid production in Catharanthus roseus hairy root clones in relation to left and right termini linked Ri T-DNA gene integration. Plant Cell Rep 23:148–154
Belal TS, Barary MH, Ibrahim MEAL, Sabry SM (2009) Kinetic spectrophotometric analysis of naftidrofuryl oxalate and vincamine in pharmaceutical preparations using alkaline potassium permanganate. J Food Drug Anal 17:415–423
Bhagwath SG, Hjortsø MA (2000) Statistical analysis of elicitation strategies for thiarubrine A production in hairy root cultures of Ambrosia artemisiifolia. J Biotech 80:159–167
Bourgaud F, Gravat A, Milesi S, Gontier E (2001) Production of secondary metabolites: a historical perspective. Plant Sci 16:839–851
Bruneton J (1995) Pharmacognosy, phytochemistry, medicinal plants. Lavoisier Publ., Paris, p 915
Bulgakov VP (2008) Functions of rol genes in plant secondary metabolism. Biotechnol Adv 26:318–324
Christey MC (2001) Use of Ri-mediated transformation for production of transgenic plants. In Vitro Cell Dev Biol Plant 37:687–700
Contreras-Cornejo HA, Macias-Rodriguez L, Cortes-Penagos C, Lopez-Bucio J (2009) Trichoderma virens, a plant beneficial fungus, enhances biomass production and promotes lateral root growth through an auxin-dependent mechanism in Arabidopsis. Plant Physiol 149:579–1592
Facchini PJ, De Luca V (2008) Opium poppy and Madagascar periwinkle: model non-model systems to investigate alkaloid biosynthesis in plants. Plant J 54:763–784
Fulzele DP, Heble MR (1994) Large-scale cultivation of Catharanthus roseus cells production of ajmalicine in a 20-1-airlift bioreactor. J Biotechnol 35:1–7
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soyabean root cells. Exp Cell Res 50:151–158
Georgiev MI, Pavlov AI, Bley T (2007) Hairy root type plant in vitro systems as sources of bioactive substances. Appl Microbiol Biotechnol 74:1175–1185
Harman GE, Howell CR, Viterbo A, Chet I, Lorito M (2004) Trichoderma species-opportunities, avirulent plant symbionts. Nat Rev Micro 2:43–56
Huang TK, Mc Donald KA (2009) Bioreactor engineering for recombinant protein production in plant cell suspension cultures. Biochem Eng J 45:168–184
Jung H-Y, Kang S-M, Kang Y-M, Kang M-J, Yun D-J, Bahk J-D, Yang J-K, Choi M-S (2003) Enhanced production of scopolamine by bacterial elicitors in adventitious hairy root cultures of Scopolia parviflora. Enzyme Microb Technol 33:987–990
Khan SA, Hamayun M, Yoon H et al (2008) Plant growth promotion and Penicillium citrinum. BMC Microbiol 8:231–239
Kusari S, Hertweck C, Spiteller M (2012) Chemical ecology of endophytic fungi: origins of secondary metabolites. Chem Biol 19:792–798
Kutchan TM (1995) Alkaloid biosynthesis: the basis for metabolic engineering of medicinal plants. Plant Cell 7:1059–1070
Ming Q, Chunyan S, Zheng C, Jia M, Zhang Q, Zhang H, Huang F, Rahman K, Han T, Qin L (2012) Elicitors from the endophytic fungus Trichoderma atroviride promote Salvia miltiorrhiza hairy root growth and tanshinone biosynthesis. J Exp Bot. doi:10.1093/jxb/ert342
Molchan O, Romashko S, Yurin V (2012) L-tryptophan decarboxylase activity and tryptamine accumulation in callus cultures of Vinca minor L. PCTOC 108:535–539
Moreno-Valenzuela OA, Galaz-Avalos RM, Minero-GarcõÂa Y, Loyola- Vargas VM (1998) Effect of differentiation on regulation of indole alkaloid production in Catharanthus roseus hairy root. Plant Cell Rep 18:99–104
Morgan JA, Shanks JV (2000) Determination of metabolic rate-limitations by precursor feeding in Catharanthus roseus hairy root cultures. J Biotechnol 79:137–145
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Pietrosiuk A, Furmanova M, Lata B (2007) Catharanthus roseus: micropropagation and in vitro techniques. Phytochem Rev 6:459–473
Rao SR, Ravishankar GA (2002) Plant cell culture: chemical factories of secondary metabolites. Biotechnol Adv 20:101–105
Sabry SM, Belal TS, Barary MH, Ibrahim MEAL (2010) A validated HPLC method for the simultaneous determination of vincamine and its potential degradant (metabolite), vincaminic acid: applications to pharmaceutical formulations and pharmacokinetic studies. J Food Drug Anal 18:447–457
Satdive RK, Fulzele DP, Eapen S (2007) Enhanced production of azadirachtin by hairy root cultures of Azadirachta indica A. Juss by elicitation and media optimization. J Biotechnol 128:281–289
Savitha BC, Thimmaraju R, Bhagyalakshmi N, Ravishankar GA (2006) Different biotic and abiotic elicitors influence betalain production in hairy root cultures of Beta vulgaris in shake-flask and bioreactor. Process Biochem 41:50–60
Sevon N, Oksman-Caldentey K (2002) Agrobacterium rhizogenes-mediated transformation: root culture as a source of alkaloids. Planta Med 68:859–868
Taneja J, Jaggi M, Wankhede DP, Sinha AK (2010) Effect of loss of T-DNA genes on MIA biosynthetic pathway gene regulation and alkaloid accumulation in Catharanthus roseus hairy roots. Plant Cell Rep 29:1119–1129
Vasconsuelo A, Boland R (2007) Molecular aspects of the early stages of elicitation of secondary metabolites in plants. Plant Sci 172:861–875
Verma P, Mathur AK, Shankar K (2012a) Enhanced vincamine production in selected tryptophan-overproducing shoots of Vinca minor. PCTOC 111:239–245
Verma P, Mathur AK, Shankar K (2012b) Growth, alkaloid production, rol genes integration, bioreactor up-scaling and plant regeneration studies in hairy root lines of Catharanthus roseus. Plant Biosys 146:27–40
Verma P, Mathur AK, Srivastava A, Mathur A (2012c) Emerging trends in research on spatial and temporal organization of terpenoid indole alkaloids pathway in Catharanthus roseus: a literature up-date. Protoplasma 249:255–268
Verma P, Mathur AK, Masood N, Luqman S, Shankar K (2013) Tryptophan over producing cell suspensions of Catharanthus roseus (L) G. Don and their up-scaling in stirred tank bioreactor: detection of phenolic compound with antioxidant potential. Protoplasma 250:371–380
Verpoorte R, Alfermann AW (2000) Metabolic engineering of plant secondary metabolism. Kluwer, Dordrecht
Yin H, Sun Y-H (2011) Vincamine-producing endophytic fungus isolated from Vinca minor. Phytomedicine 18:802–805
Zhao J, Verpoorte R (2007) Manipulating indole alkaloid production by Catharanthus roseus cell cultures in bioreactors: from biochemical processing to metabolic engineering. Phytochem Rev 6:435–457
Acknowledgments
The work presented here has been supported by DST-FAST TRACK SERC/LS-261/2012. Help rendered by Jyotsana Priya during HPLC analysis has been highly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Verma, P., Khan, S.A., Mathur, A.K. et al. Fungal endophytes enhanced the growth and production kinetics of Vinca minor hairy roots and cell suspensions grown in bioreactor. Plant Cell Tiss Organ Cult 118, 257–268 (2014). https://doi.org/10.1007/s11240-014-0478-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-014-0478-4