Abstract
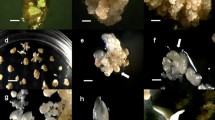
Using mature cotyledonary explants of Fraxinus mandshurica, an efficient plant regeneration system was developed via somatic embryogenesis. More than 67 % of mature cotyledons of zygotic embryos yielded 23–159 somatic embryos (SEs) per explant when incubated on medium consisting of half-strength Murashige and Skoog (MS) salts and vitamins (MS1/2) supplemented with 8.88 μM 6-benzyladenine (BA), 26.84 μM naphthaleneacetic acid (NAA), 75 g L−1 sucrose, and 400 mg L−1 casein hydrolysate (CH). Approximately, 82 % of induced SEs were observed on browning cotyledonary explants. Histological studies of cotyledon explants at various stages of somatic embryogenesis revealed that the SEs originated from single epidermal cells and developed to the globular, heart, torpedo, and cotyledonary stage embryos. Secondary somatic embryos (SSEs) formed on the surface of radicle tips of the SEs. Addition of low concentrations of NAA and 200–400 mg L−1 CH to MS1/2 medium increased SSE induction. Cotyledonary SSEs were cultured on MS1/2 medium with 10 mM abscisic acid in the presence of light to promote maturation, and >92 % of mature SSEs were able to germinate with normal shoots. After 8 weeks in culture in the presence of light on medium with one-third of the MS macroelements as well as 0.06 μM NAA, >94 % of the germinated SSEs converted into plantlets. Plantlets acclimatized successfully to ex vitro conditions and developed normal phenotypes under field conditions.




Similar content being viewed by others
Abbreviations
- 1/3MS:
-
Medium with one-third strength of the macroelements of MS
- 2,4-D:
-
2,4-Dichlorophenoxyacetic acid
- ANOVA:
-
Analysis of variance
- BA:
-
6-Benzyladenine
- CH:
-
Casein hydrolysate
- IBA:
-
Indole-3-butyric acid
- MANOVA:
-
Multivariate analysis of variance
- MS:
-
Medium of Murashige and Skoog (1962)
- MS1/2:
-
Medium with one-half-strength of all elements of MS
- NAA:
-
Naphthaleneacetic acid
- PGR:
-
Plant growth regulator
- SAM:
-
Shoot apical meristem
- SE:
-
Somatic embryo
- SSE:
-
Secondary somatic embryo
- ZE:
-
Zygotic embryo
References
Bates S, Preece JE, Navarrete NE, Van Sambeek JW, Gaffney GR (1992) Thidiazuron stimulates shoot organogenesis and somatic embryogenesis in white ash (Fraxinus americana L.). Plant Cell Tiss Organ Cult 31(1):21–29
Capuana M, Petrini G, Di Marco A, Giannini R (2007) Plant regeneration of common ash (Fraxinus excelsior L.) by somatic embryogenesis. In Vitro Cell Dev Biol 43(2):101–110. doi:10.1007/s11627-007-9030-0
Chen JT, Hong PI (2012) Cellular origin and development of secondary somatic embryos in Oncidium leaf cultures. Biol Plant 56(2):215–220
Chen AH, Yang JL, Niu YD, Yang CP, Liu GF, Yu CY, Li CH (2010) High-frequency somatic embryogenesis from germinated zygotic embryos of Schisandra chinensis and evaluation of the effects of medium strength, sucrose, GA3, and BA on somatic embryo development. Plant Cell Tiss Organ Cult 102(3):357–364. doi:10.1007/s11240-010-9740-6
Correia S, Cunha AE, Salgueiro L, Canhoto JM (2012) Somatic embryogenesis in tamarillo (Cyphomandra betacea): approaches to increase efficiency of embryo formation and plant development. Plant Cell Tiss Organ Cult 109(1):143–152. doi:10.1007/s11240-011-0082-9
Gu DZ, Li YM, Jiang YT (2010) Selection of proper hormone combinations for direct in vitro plant regeneration from bud tips and their in vitro preservation for Fraxinus mandshurica. Sci Silv Sin 46(10):178–182 (in Chinese with English abstract)
Hammatt N (1994) Shoot initiation in the leaflet axils of compound leaves from micropropagated shoots of juvenile and mature common ash (Fraxinus excelsior L.). J Exp Bot 45(6):871–875. doi:10.1093/jxb/45.6.871v
Hammatt N, Ridout MS (1992) Micropropagat ion of common ash (Fraxinus escelsior). Plant Cell Tiss Organ Cult 31(1):67–74
Hu LJ, Uchiyama K, Shen HL, Ide Y (2010) Multiple-scaled spatial genetic structures of Fraxinus mandshurica over a riparian–mountain landscape in Northeast China. Conserv Genet 11(1):77–87. doi:10.1007/s10592-009-0004-0
Kikuchi A, Sanuki N, Higashi K, Koshiba T, Kamada H (2006) Abscisic acid and stress treatment are essential for the acquisition of embryogenic competence by carrot somatic cells. Planta 223(4):637–645. doi:10.1007/s00425-005-0114-y
Kim MS, Schumann CM, Klopfenstein NB (1997) Effects of thidiazuron and benzyladenine on axillary shoot proliferation of three green ash (Fraxinus pennsylvanica Marsh.) clones. Plant Cell Tiss Organ Cult 48(1):45–52
Kong DM, Preece JE, Shen HL (2012a) Somatic embryogenesis in immature cotyledons of Manchurian ash (Fraxinus mandshurica Rupr.). Plant Cell Tiss Organ Cult 108(3):485–492. doi:10.1007/s11240-011-0062-0
Kong DM, Shen HL, Li N (2012b) Influence of AgNO3 on somatic embryo induction and development in Manchurian ash (Fraxinus mandshurica Rupr.). Afr J Biotechnol 11(1):120–125. doi:10.5897/AJB11.3061
Konieczny R, Sliwinska E, Pilarska M, Tuleja M (2012) Morphohistological and flow cytometric analyses of somatic embryogenesis in Trifolium nigrescens Viv. Plant Cell Tiss Organ Cult 109(1):131–141. doi:10.1007/s11240-011-0081-x
Kumar GK, Thomas TD (2012) High frequency somatic embryogenesis and synthetic seed production in Clitoria ternatea Linn. Plant Cell Tiss Organ Cult 110(1):141–151. doi:10.1007/s11240-012-0138-5
Kurczyńska EU, Gaj MD, Ujczak A, Mazur E (2007) Histological analysis of direct somatic embryogenesis in Arabidopsis thaliana (L.) Heynh. Planta 226(3):619–628. doi:10.1007/s00425-007-0510-6
Li WF, Zhang SG, Han SY, Wu T, Zhang JH, Qi LW (2012) Regulation of LaMYB33 by miR159 during maintenance of embryogenic potential and somatic embryo maturation in Larix kaempferi (Lamb.) Carr. Plant Cell Tiss Organ Cult. doi:10.1007/s11240-012-0233-7
Maruyama TE, Hosoi Y (2012) Post-maturation treatment improves and synchronizes somatic embryo germination of three species of Japanese pines. Plant Cell Tiss Organ Cult 110(1):45–52. doi:10.1007/s11240-012-0128-7
Mei L, Gu JC, Zhang ZW, Wang ZQ (2010) Responses of fine root mass, length, production and turnover to soil nitrogen fertilization in Larix gmelinii and Fraxinus mandshurica forests in Northeastern China. J For Res 15(3):194–201. doi:10.1007/s10310-009-0176-y
Naik SK, Chand PK (2011) Tissue culture-mediated biotechnological intervention in pomegranate: a review. Plant Cell Rep 30(5):707–721. doi:10.1007/s00299-010-0969-7
Perez-Parron MA, Gonzalez-Benito ME, Perez C (1994) Micropropagation of Frazinus angustipflia from mature and juvenile plant material. Plant Cell Tiss Organ Cult 37(3):297–302
Pinto G, Silva S, Park YS, Neves L, Araújo C, Santos C (2008) Factors influencing somatic embryogenesis induction in Eucalyptus globulus Labill.: basal medium and anti-browning agents. Plant Cell Tiss Organ Cult 95(1):79–88. doi:10.1007/s11240-008-9418-5
Pinto DLP, Almeida AMR, Rêgo MM, Silva ML, Oliveira EJ, Otoni WC (2011) Somatic embryogenesis from mature zygotic embryos of commercial passionfruit (Passiflora edulis Sims) genotypes. Plant Cell Tiss Organ Cult 107(3):521–530. doi:10.1007/s11240-011-0003-y
Preece JE, Bates SA (1995) Somatic embryogenesis in white ash (Fraxinus Americana L.). In: Jain S, Gupta P, Newton R (eds) Somatic embryogenesis in woody plants, vol 2. Kluwer Acad Publishing, Netherlands, pp 311–325
Preece JE, Christ PH, Ensenberger L, Zhao J (1987) Micropropagation of ash (Fraxinus). Comb Proc Int Plant Prop Soc 37:366–372
Preece JE, Navarrete N, Van Sambeek JW, Gaffney GR (1991) An in vitro microplant bioassay using clonal white ash to test for tall fescue allelopathy. Plant Cell Tiss Organ Cult 27(2):203–210
Reidiboym-Talleux L, Diemer F, Sourdioux M, Chapelain K, March GGD (1999) Improvement of somatic embryogenesis in wild cherry (Prunus avium). Effect of maltose and ABA supplements. Plant Cell Tiss Organ Cult 55(3):199–209
Shen HL, Zhao X, Xing ZB, Huang J, Hu LJ, Liu CL (2005) Influencing factors of vegetative propagation of Fraxinus mandshurica by rooted cuttings. J Northeast For Univ 33(3):5–6 (in Chinese with English abstract)
Silveira CE, Cottignies A (1994) Period of harvest, sprouting ability of cuttings, and in vitro plant regeneration in Fraxinus excelsior. Can J Bot 72(2):261–267. doi:10.1139/b94-035
Stasolla C, Yeung EC (2003) Recent advances in conifer somatic embryogenesis: improving somatic embryo quality. Plant Cell Tiss Organ Cult 74(1):15–35. doi:10.1023/A:1023345803336
Tabrett AM, Hammatt N (1992) Regeneration of shoots from embryo hypocotyls of common ash (Fraxinus excelsior). Plant Cell Rep 11(10):514–518
Tang QY, Feng GM (2002) Practical statistical analysis and DPS data handling system. Science Publishing, Beijing
Tonon G, Capuana M, Di Marco A (2001a) Plant regeneration of Fraxinus angustifolia by in vitro shoot organogenesis. Sci Hortic 87(4):291–301
Tonon G, Capuana M, Rossi C (2001b) Somatic embryogenesis and embryo encapsulation in Fraxinus angustifolia Vhal. J Hortic Sci Biotechnol 76(6):753–757
Troch V, Werbrouck S, Geelen D, Labeke MCV (2009) Optimization of horse chestnut (Aesculus hippocastanum L.) somatic embryo conversion. Plant Cell Tiss Organ Cult 98(1):115–123. doi:10.1007/s11240-009-9544-8
Vooková B, Machava J, Šalgovičová A, Kormuťák A (2010) Optimization of Algerian fir somatic embryos maturation. Biol Plant 54(1):177–180
Yang L, Li YH, Shen HL (2012) Somatic embryogenesis and plant regeneration from immature zygotic embryo cultures of mountain ash (Sorbus pohuashanensis). Plant Cell Tiss Organ Cult 109(3):547–556. doi:10.1007/s11240-012-0121-1
Acknowledgments
This research was supported by China Postdoctoral Science Foundation (2012T50320; 20110491015), Fundamental Research Funds for the Central Universities of China (DL10BA04), and Postdoctoral Science Foundation of Heilongjiang Province of China (LBH–10284). We thank Dr. Robert L. Geneve from Department of Horticulture, University of Kentucky, for his proofreading of our revised manuscript. We thank all the colleagues in our lab for constructive discussion and technical support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, L., Bian, L., Shen, Hl. et al. Somatic embryogenesis and plantlet regeneration from mature zygotic embryos of Manchurian ash (Fraxinus mandshurica Rupr.). Plant Cell Tiss Organ Cult 115, 115–125 (2013). https://doi.org/10.1007/s11240-013-0345-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-013-0345-8