Abstract
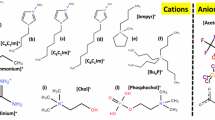
We have investigated the basic mechanism of carbon nanotube (CNT) interactions with various room-temperature ionic liquids (RTILs) using molecular dynamics (MD) simulations. To understand the effects of the cation molecular geometry on the properties of the interface structure in the RTIL systems, we have studied a set of three RTILs with the same [BF4]- (tetrafluoroborate) anion but with different cations, namely, [EMIM]+ (1-ethyl-3-methylimidazolium), [BMIM]+ (1-butyl-3-methylimidazolium), [HMIM]+ (1-hexyl-3-methylimidazolium), and [OMIM]+ (1-octyl-3-methylimidazolium) ions. The simulation results showed that the imidazolium cations exhibit two distinct orientations (perpendicular and parallel to the CNTs surface) at the interface irrespective of the alkyl chain length of the cations. The average number of hydrogen bonds per cations inside the CNT was found to be higher for [OMIM][BF4] (1.01), which suggests that [OMIM]+ imidazolium rings to be concentrated at the center of the CNT, which favors hydrogen bond. The reported results show the diffusion coefficients of ions in confinement are much lower in comparison to the bulk region. The interaction energy between [OMIM][BF4] (-8.75 kcal.mol−1.ion−1) and CNT was found to be higher as compared to other ILs. The cations paralleling the CNT surface are thermodynamically significantly more stable because of the substantial interfacial π-π stacking interactions, as shown by a comparison with the calculated interaction energies between cations and the CNTs. Our simulation results provide a molecular-level understanding of the stabilization and dispersion of CNT bundles in ILs.











Similar content being viewed by others
Availability of data and materials
Not applicable.
References
Salah LS, Ouslimani N, Bousba D, Huynen I, Danlée Y, Aksas H (2021) Carbon nanotubes (CNTs) from synthesis to functionalized (CNTs) using conventional and new chemical approaches. J Nanomater 2021:1–31
Nascimento VV, Neves WQ, Alencar RS, Li G, Fu C, Haddon RC, Bekyarova E, Guo J, Alexandre SS, Nunes RW, Souza Filho AG, Fantini C (2021) Origin of the giant enhanced raman scattering by sulfur chains encapsulated inside single-wall carbon nanotubes. ACS Nano 15(5):8574–8582
Ko J, Joo Y (2021) Review of sorted metallic single-walled carbon nanotubes. Adv Mater Interfaces 8(11):2002106
Wang J, Chen Y, Yu P (2021) The cohesive energy and vibration characteristics of parallel single-walled carbon nanotubes. Molecules 26(24):7470
Singh SK, Savoy AW (2020) Ionic liquids synthesis and applications: an overview. J Mol Liq 297:112038
Pamies R, Espejo C, Carrión FJ, Morina A, Neville A, Bermúdez MD (2017) Rheological behavior of multiwalled carbon nanotube-imidazolium tosylate ionic liquid dispersions. J Rheol 61(2):279–289
Espejo C, Carrión F-J, Martínez D, Bermúdez M-D (2013) Multi-walled carbon nanotube-imidazolium tosylate ionic liquid lubricant. Tribol Lett 50(2):127–136
Chaban VV, Prezhdo OV (2014) Nanoscale carbon greatly enhances mobility of a highly viscous ionic liquid. ACS Nano 8(8):8190–8197
Ohba T, Hata K, Chaban VV (2015) Nanocrystallization of imidazolium ionic liquid in carbon nanotubes. J Phys Chem C 119(51):28424–28429
Hameed N, Church JS, Salim NV, Hanley TL, Amini A, Fox BL (2013) Dispersing single-walled carbon nanotubes in ionic liquids: a quantitative analysis. RSC Adv 3(43):20034–20039
Wang X, Huang Y, Li L, Huang L, Chen X, Yang Z (2021) Molecular-level insights into composition-dependent structure, dynamics, and hydrogen bonds of binary ionic liquid mixture from molecular dynamics simulations. Chem Phys 542:111051
Sappidi P, Bara JE, Turner CH (2021) Molecular-level behavior of imidazolium-based ionic liquid mixtures. Chem Eng Sci 229:116073
Mohammadi M, Foroutan M (2013) Mixture of ionic liquid and carbon nanotubes: comparative studies of the structural characteristics and dispersion of the aggregated non-bundled and bundled carbon nanotubes. Phys Chem Chem Phys 15(7):2482–2494
Peng K, Wang X, Huang Q, Yang Z, Li Y, Chen X (2019) Molecular-level understanding of structures and dynamics of imidazolium-based ionic liquids around single-walled carbon nanotubes: different effects between alkyl chains of cations and nanotube diameters. J Phys Chem C 123(31):18932–18938
Wang X, Fu F, Peng K, Huang Q, Li W, Chen X, Yang Z (2020) Understanding of competitive hydrogen bond behavior of imidazolium-based ionic liquid mixture around single-walled carbon nanotubes. J Phys Chem C 124(12):6634–6645
Huo F, Liu Z (2015) Interfacial layering and orientation ordering of ionic liquid around single-walled carbon nanotube: a molecular dynamics study. Mol Simul 41(4):271–280
García G, Atilhan M, Aparicio S (2015) Theoretical study of renewable ionic liquids in the pure state and with graphene and carbon nanotubes. J Phys Chem B 119(37):12224–12237
Atilhan M, Aparicio S (2018) Theoretical study of low viscous ionic liquids at the graphene interface. J Phys Chem C 122(3):1645–1656
França JMP, Nieto de Castro CA, Pádua AAH (2017) Molecular interactions and thermal transport in ionic liquids with carbon nanomaterials. Phys Chem Chem Phys 19(26):17075–17087
Peng K, Lin J, Yang D, Fu F, Dai Z, Zhou G, Yang Z (2022) Molecular-level insights into interfacial interaction–nanostructure relationships of imidazolium-based ionic liquids around carbon nanotube electrodes. Ind Eng Chem Res 61(37):14051–14065
Jorgensen WL, Maxwell DS, Tirado-Rives J (1996) Development and testing of the OPLS all-atom force field on conformational energetics and properties of organic liquids. J Am Chem Soc 118(45):11225–11236
Price MLP, Ostrovsky D, Jorgensen WL (2001) Gas-phase and liquid-state properties of esters, nitriles, and nitro compounds with the OPLS-AA force field. J Comput Chem 22(13):1340–1352
Canongia Lopes JN, Pádua AAH (2004) Molecular force field for ionic liquids composed of triflate or bistriflylimide anions. J Phys Chem B 108(43):16893–16898
Canongia Lopes JN, Deschamps J, Pádua AAH (2004) Modeling ionic liquids using a systematic all-atom force field. J Phys Chem B 108(6):2038–2047
Huddleston JG, Visser AE, Reichert WM, Willauer HD, Broker GA, Rogers RD (2001) Characterization and comparison of hydrophilic and hydrophobic room temperature ionic liquids incorporating the imidazolium cation. Green Chem 3(4):156–164
Essmann U, Perera L, Berkowitz ML, Darden T, Lee H, Pedersen LG (1995) A smooth particle mesh Ewald method. J Chem Phys 103(19):8577–8593
Phillips JC, Braun R, Wang W, Gumbart J, Tajkhorshid E, Villa E, Chipot C, Skeel RD, Kalé L, Schulten K (2005) Scalable molecular dynamics with NAMD. J Comput Chem 26(16):1781–1802
Humphrey W, Dalke A, Schulten K (1996) VMD: Visual molecular dynamics. J Mol Graph 14(1):33–38
Martínez L, Andrade R, Birgin EG, Martínez JM (2009) PACKMOL: A package for building initial configurations for molecular dynamics simulations. J Comput Chem 30(13):2157–2164
Fan X-H, Chen Y-P, Su C-S (2016) Density and viscosity measurements for binary mixtures of 1-Ethyl-3-methylimidazolium Tetrafluoroborate ([Emim][BF4]) with Dimethylacetamide, Dimethylformamide, and Dimethyl Sulfoxide. J Chem Eng Data 61(2):920–927
Song D, Chen J (2014) Densities and viscosities for ionic liquids mixtures containing [eOHmim][BF4], [bmim][BF4] and [bpy][BF4]. J Chem Thermodyn 77:137–143
Ning H, Hou M, Mei Q, Liu Y, Yang D, Han B (2012) The physicochemical properties of some imidazolium-based ionic liquids and their binary mixtures. Sci China Chem 55(8):1509–1518
Brela MZ, Kubisiak P, Eilmes A (2018) Understanding the structure of the hydrogen bond network and its influence on vibrational spectra in a prototypical aprotic ionic liquid. J Phys Chem B 122(41):9527–9537
Mananghaya MR, Santos GN, Yu D (2019) Solubility of aminotriethylene glycol functionalized single wall carbon nanotubes: a density functional based tight binding molecular dynamics study. J Comput Chem 40(8):952–958
Liu Y, Wang Q, Wu T, Zhang L (2005) Fluid structure and transport properties of water inside carbon nanotubes. J Chem Phys 123(23)
Sahu P, Ali SM, Shenoy K, Mohan S (2019) Nanoscopic insights of saline water in carbon nanotube appended filters using molecular dynamics simulations. Phys Chem Chem Phys 21(16):8529–8542
Harris KR, Kanakubo M (2016) Revised and extended values for self-diffusion coefficients of 1-Alkyl-3-methylimidazolium tetrafluoroborates and hexafluorophosphates: relations between the transport properties. J Phys Chem B 120(50):12937–12949
Bagno A, D’Amico F, Saielli G (2007) Computer simulation of diffusion coefficients of the room-temperature ionic liquid [bmim][BF4]: problems with classical simulation techniques. J Mol Liq 131:17–23
Zhang X, Wang D, Song X, Li Y, Leng X, Wei Y, Liu G, Xia Q (2023) Insight into the stabilization mechanism of imidazole-based ionic liquids at the interface of the carbon nanotubes: a computational study. J Mol Liq 375
Acknowledgements
Vellore Institute of Technology is gratefully acknowledged for providing high-performance computing technology for this work.
Author information
Authors and Affiliations
Contributions
Rima Biswas conceived and designed the study. Prateek Banerjee and Rima Biswas performed simulation calculations. Rima Biswas and Kavathekar Soham Sudesh wrote the paper. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Biswas, R., Banerjee, P. & Sudesh, K.S. Molecular dynamics studies on interfacial interactions between imidazolium-based ionic liquids and carbon nanotubes. Struct Chem (2024). https://doi.org/10.1007/s11224-024-02323-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11224-024-02323-3