Abstract
Purpose
Owing to their topographic location and nutrient rich soils, riparian forests are often converted to pastures for grazing. In recent decades, remnant riparian forests cleared for grazing pastures have been restored with native species. The impacts of such land-use changes on soil fungal communities are unclear, despite the central roles that soil fungi play in key ecosystem processes. We investigated how soil fungal taxonomic and functional composition are affected by land-use change at different depths, and if variation in soil fungal communities is related to edaphic properties and extant vegetation.
Methods
The study was conducted in six waterways in south-eastern Australia, each comprising three land-use types: remnant riparian forest, cleared forest converted to pasture, and pastures restored with native plants. We surveyed three strata of vegetation and sampled top-soil and sub-soil to characterise physicochemical properties and soil fungal communities. ITS1 region sequences were used to assign soil fungal taxonomic and functional composition.
Results
Fungal taxonomic and functional composition infrequently varied with land-use change or soil depth. Overall, environmental properties (soil and vegetation) explained 35–36% of variation in both fungal taxonomic and functional composition. Soil fungal taxonomic composition was related to soil fertility (N, P, K, pH and Ca) and ground cover characteristics, whereas functional composition was related to clay content, sub-canopy cover and tree basal area.
Conclusion
Across the six studied waterways, fungal taxonomic and functional composition were more strongly associated with land-use mediated changes in site-scale soil physicochemical properties and vegetation structure than broad-scale classes of land-use type.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Riparian forests are multifunctional hotspots able to provide a wide range of functions and services, and are host to a high diversity of organisms (Allan 2004; Harner et al. 2011; Naiman et al. 2010). Despite their ecological significance and due to their topographic location and nutrient-rich soils, riparian forests have often been cleared for agricultural purposes, including grazing, resulting in the loss of a large proportion of these ecosystems. In Europe and America, the extent of riparian vegetation has declined by 80 – 95%, with most losses attributed to land clearing for grazing (Naiman et al. 1993; Riis et al. 2020). In Victoria, Australia, 32% of riparian land is in poor or very poor condition (Victoria State Government 2015). Several policy-driven restoration initiatives have aimed to reverse the effects of land-use conversion by restoring grazing pastures with native riparian species (Daigneault et al. 2017). These initiatives, however, are often focused on restoring and assessing vegetation and seldom focus on soil microbial communities, even though knowledge about changes in soil fungal communities with changing land uses could inform restoration practices. Previous studies in grasslands and prairies have shown that the restoration of soil fungal communities through inoculation in degraded ecosystems can influence the pace and trajectory of vegetation recovery and improve restoration outcomes (Barber et al. 2017; Bever et al. 2003; Middleton and Bever 2012; Stürmer et al. 2012; Wubs et al. 2016). Further, active replanting of native vegetation can also return important fungal functional groups (Yan et al. 2018), as well as introducing novel fungal associations (Bermúdez-Contreras et al. 2020).
Soil fungal communities encompass a large phylogenetic diversity and are generally classified into three functional groups—symbiotrophs, saprotrophs, and pathogens (Borrell et al. 2017). Symbiotrophs include mycorrhizal fungi, which form symbiotic associations that expand plant root surface area, giving plants greater access to nutrients and water in exchange for carbon (Kramer et al. 2012; Schmidt et al. 2019; van der Heijden et al. 2008). Approximately 20,000 plant species are dependent on soil microbial symbionts for growth and survival, based on estimates for the northern hemisphere (Hamonts et al. 2017; Jobim et al. 2016; van der Heijden et al. 2008). Saprotrophs decompose organic matter and are broken into trophic guilds based on whether they decompose plants, litter, soil, or wood (Otsing et al. 2018). During the decomposition process, saprotrophs translocate carbon, nitrogen and phosphorus into the soil, which is then available for uptake by plants (Crowther et al. 2012). Soil pathogens can be harmful but can also play an important role in controlling populations of insects, animals, plants or fungal pests (Schmidt et al. 2019). Soil fungal communities are shaped by environmental filters related to soil, vegetation and climate properties (Aponte et al. 2010a; Gómez Gallego et al. 2013; Peay et al. 2013; Rincón et al. 2015; Sun et al. 2019) and can respond rapidly to alterations in local conditions, such as those induced by land-use change (Banning et al. 2011; Guo et al. 2019).
Soil fungal communities respond to soil properties including pH, nutrient availability, and texture (Fierer et al. 2012; Lauber et al. 2008; Semchenko et al. 2018; Siciliano et al. 2014). Clearing native riparian vegetation for grazing has been shown to alter soil properties in various ways (Richardson et al. 2007). Tilling or soil pugging from stock can affect soil structure, which influences soil aeration, decreasing nutrient content, and water-holding capacity and has been related to differences in fungal taxonomic composition (Alvarez-Sanchez et al. 2012; Bouffaud et al. 2017; Xiang et al. 2014). Soil particle size distribution has also been found to affect fungal community composition in tropical riparian forests, with clay soils harbouring a higher relative abundance of arbuscular mycorrhizal fungi compared to sandy soil (Peay et al. 2013). Other farming practices include the addition of fertilisers, particularly nitrogen and phosphate, to increase the growth of grass or crops (Henzell 2007). Soils higher in phosphorus and nitrogen have a lower richness of ectomycorrhizal and arbuscular mycorrhizal fungi compared to soils that are depleted in phosphorus and nitrogen (Henry et al. 2015; Pagano et al. 2007; Sharma et al. 2010). The use of lime is also a common way to improve grazing pastures by increasing nutrient availability by changing soil pH. Many studies indicate that pH is a determinant of fungal community structure in grasslands and forests (e.g., Aponte et al. 2010b; Henry et al. 2015; Liang et al. 2015; Wasof et al. 2019). Additionally, soil fertility and pH have been found to alter the abundance of Glomeromycota, symbionts that form endomycorrhizal associations with the roots of forbs and grasses (Leff et al. 2015; Sepp et al. 2019). To this end, land-use change effects on soil physicochemical properties can play an important role in shaping fungal communities.
Soil fungal communities are also tightly related to vegetation properties through host specificity and the production of diverse organic substrates, including leaf litter and root exudates (Aponte et al. 2010a; Kasel et al. 2008; Peay et al. 2013; Sun et al. 2019). For instance, symbiotrophs exhibit a great degree of specificity, with Glomeromycota being the dominant mycorrhizal symbionts in grasses or forbs, while Basidiomycota and Ascomycota are dominant in woody vegetation (Brundrett 2002; Molina et al. 1992; Sepp et al. 2019). The canopy cover in contrast to canopy gaps and proximity to tree trunks have also been related to differences in soil fungal taxonomic and functional composition and abundance (Bennett et al. 2009; Branco et al. 2013; Perreault et al. 2020). Other features of vegetation have been shown to have an impact on soil fungal community composition such as the presence of exotic plants in ground cover vegetation (Ba et al. 2018; Sielaff et al. 2018; Smith et al. 2018) or density of roots (Peay et al. 2013). The latter, which decreases exponentially with soil depth (Poesen and De Baets 2005), has been positively related to fungal taxonomic and functional abundance (Wang et al. 2013). Furthermore, vegetation can indirectly influence soil fungal communities by modifying soil structure and soil chemical properties (Aponte et al. 2010a; Klopf et al. 2017). Land-use change from native vegetation to grazing pastures results in a loss of diversity, structure and cover of native species that are replaced with exotic pasture and thus could have a profound effect on soil fungal communities (Calvo-Polanco et al. 2016; Wasof et al. 2019). Understanding feedbacks between above- and below-ground biota could inform restoration practices that facilitate the return of soil communities that enhance the persistence of above-ground vegetation (Ettema and Wardle 2002; Wardle et al. 2004).
Our study characterises changes in soil fungal taxonomic and functional community compositions in response to the conversion of riparian vegetation to grazing pasture and subsequent restoration with native plant species of the remnant riparian vegetation. Specifically, we hypothesised: (1) that soil fungal taxonomic and functional composition would change with land use, with communities in remnant forests and pastures being most dissimilar and communities in restored sites showing more similarities with remnant vegetation; (2) that land use related changes would be concentrated in the top-soil relative to sub-soil; and (3) that fungal communities would be related to both vegetation and soil physicochemical properties, which are both expected to vary strongly with land-use change.
Materials and Methods
Study sites
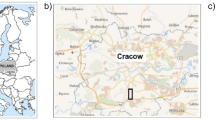
Study sites were located near six different waterways in south-eastern Australia (Fig. 1) and encompassed a range of topographies and soil types. Mean annual maximum temperature ranged from 17–19 °C and mean minimum temperature ranged from 7–11 °C (Bureau of Meteorology 2020). Each site supported three different land-use types; pasture, restored and remnant riparian forests. These three land uses represent the transition from intact riparian forest that was cleared for grazing pasture and then restored with native species that were common to the riparian forest. Remnant riparian forest comprised of species common in well-watered areas of south-eastern Australia such as Eucalyptus viminalis Labill., Eucalyptus ovata Labill., and Acacia dealbata Link (Online Resource 1). In contrast pasture was primarily exotic grasses including Holcus lanatus L. and Anthoxanthum odoratum L. and regularly grazed by sheep or cattle (Online Resource 1). Restoration sites were formerly pasture that had been planted with nursery grown indigenous trees and shrubs typical of the riparian forest three to nine years prior to our study (Online Resource 1).
Vegetation surveys
Vegetation surveys were conducted as described in Waymouth et al. (2021). Briefly, one 25 m × 25 m quadrat (625 m2) was located within each land-use type, at each of the six study sites for sampling of vegetation and soils. Vegetation surveys characterised three strata: canopy (trees with diameter at breast height (DBH) ≥ 15 cm), sub-canopy (trees and tall shrubs with DBH < 15 cm); and ground cover (herbaceous vegetation) within each quadrat. Canopy species were assessed at four random locations within the 625 m2 the quadrat using the point-centred quarter method (total of 16 points assessed; Cottam and Curtis 1956). From the point-centred quarter method we obtained measurements of cover, basal area, and average plant to plant distance (APPD). Basal area was calculated across all plants and for Eucalyptus species, which dominated the canopy. Sub-canopy cover was assessed via the line-intercept method, with three 20 m transects established within each quadrat (Canfield 1941). We used the line intercept data to determine the total number of species and total number of Acacia spp. (dominant species) in the sub-canopy. For ground cover, the cover of litter, bare ground, and each plant species was assessed using a 1 m2 sub-quadrat placed every 5 m along each transect (5 sub-quadrats per transect).
Soil sampling and analyses
Soil sampling and physicochemical analyses
Soils were sampled from within each quadrat according to protocols established by the Biomes of Australian Soil Environments (BASE; Bissett et al. 2016). Soil was sampled as per Waymouth et al. (2021). Briefly, each soil sample was a composite of 27 cores, taken from nine locations (3 cores per location) that were evenly spaced along two diagonal transects that dissected each quadrat. Soil samples were taken from two depths (0–10 cm and 20–30 cm) and kept separate for further analysis. Soils were kept on ice until homogenised and sieved; material > 5 mm was removed e.g. litter, roots and stones. Approximately 20 g of field moist soil was frozen at − 80 °C for later genetic analysis. The remaining soil was weighed, then dried in an oven at 40 °C and weighed again for soil moisture (%). Approximately 500 g of soil was sent to CSBP Soil and Plant Analysis Laboratory (Bibra Lake, Western Australia) for soil physical and chemical analysis. Tests included: pH, electrical conductivity, available nitrogen, available phosphorus, sulphur, organic carbon, total nitrogen, exchangeable cations (Mg, K, Na, Ca), extractable trace elements (Cu, Fe, Mn, Zn, Al, B), and soil particle size (CSBP Lab 2019). Soil bulk density was measured by the volumetric method for 0–10 cm depth, on one core per quadrat. Samples were not adjusted for stone content and were dried at 80 °C.
Soil DNA extraction, sequencing, OTU identification and function
DNA extraction and sequence analyses were conducted using methods described in Bissett et al. (2016) and Waymouth et al. (2020). Sample identifiers of samples used are listed in Online Resource 2. Briefly, soil DNA was extracted and then pooled from 3 × 0.25 g soil samples per replicate at the Australian Genome Research Facility (AGRF, Adelaide, Australia) using MoBio powersoil DNA extraction kits according to manufacturer’s instructions. The fungal internal transcribed spacer (ITS) region was PCR-amplified using primers ITS1F (Gardes and Bruns 1993) and ITS4 (White et al. 1990). Sequences (300 bp PE) were then produced using the Illumina MiSEQ platform. The ITS1 region was extracted from Illumina R1 reads using ITSx (Bengtsson-Palme et al. 2013), and sequences containing ambiguous bases removed. Sequences were denoised using the UNOISE3 algorithm (Edgar and Flyvbjerg 2015) in the program USEARCH v11 (Edgar 2010) (–minsize 4) and abundance profiles built by mapping all data to identified zOTUs (usearch –otutab –maxaccepts 0). Amplicon sequence variants were classified using the RDP classifier (Wang et al. 2007) in MOTHER at a 60% probability cutoff, against UNITE SH v7 (Nilsson et al. 2018).
In total, across all samples, we found 9891 ITS1 zOTUs across 12 phyla. Rare phyla were characterised as < 0.05% relative abundance. The number of sequence reads ranged from 14,157 to 180,164, with a mean of 47,400 per sample. 35% of sequence reads that were assigned to the kingdom fungi could not be assigned further. Preliminary analysis showed no significant difference in the sequence numbers per sample with land use and depth, and observed richness was strongly correlated with rarefied richness, as such samples were not rarefied (see Online Resources 3 and 4; refer to McMurdie and Holmes 2014).
We used FUNGuild v1.0 (Nguyen et al. 2016) to assign fungal zOTUs to different functional guilds based on trophic strategies: ‘symbiotroph’, ‘saprotroph’ and ‘pathotroph’ with a confidence rating of ‘highly probable’, ‘probable’ and ‘possible’. We labelled all zOTUs with a ‘possible’ confidence rating as unassigned in the stacked bar graph due to the low confidence rating. In total we had 2781 different zOTUs assigned to functional guilds across 24 different trophic guilds. The 5% of zOTUs with a mixed trophic mode (e.g., saprotroph-symbiotroph-pathotroph), or guild were categorised as multimode. Rare trophic guilds were characterised as those < 0.05% relative abundance (trophic guilds with trophic modes are listed in Online Resource 5). Samples within the dataset had sequence reads ranging from 773 to 46,790 reads, with a mean sample size of 9649, and 79% of sequence reads were unassigned, either due to low confidence rating or no functional data available.
Datasets for overall fungal taxonomic composition (within-phyla composition and within-class) and functional composition (trophic mode and guild) were used to investigate differences in abundance (sequence abundance of single fungal species), richness and Shannon diversity index (Peet 1974; referred to from here on as diversity) among land uses, and with soil depth.
Statistical analysis
Differences in vegetation, soil physicochemical properties and fungal taxonomic and functional composition between land uses were assessed using GLMs, with soil analyses also including sampling depth as an explanatory variable. Land use and depth were fixed factors, and site (waterway) was a random factor. Dependent variables are listed in Online Resources 6 (for vegetation properties) and 10 (for soil properties). Models were implemented in R package lme4 (Bates et al. 2014), using the gaussian method. Pairwise differences were assessed using post hoc Tukey HSD tests in the R package emmeans (Lenth 2020).
Non-parametric Permutational Multivariate Analysis of Variance (PERMANOVA) was used to test the overall effects of land use, sampling depth (for soil properties only) and their interactions on environmental properties (vegetation and soil properties) and fungal community (taxonomic and functional composition). Euclidean dissimilarity measures of environmental properties were significance tested using post hoc comparisons with 999 permutations in the R package vegan version 2.4–6 (Oksanen et al. 2018). Bray–Curtis distances were used for fungal taxonomic and functional composition using 999 permutations with outputs visualised using non-metric multidimensional scaling (NMDS). Dispersion was assessed by calculating the average distance between data points and the centroid in multivariate space with differences (by land use or depth) analysed using ANOVAs, with post hoc Tukey HSD tests. Analyses were conducted using vegan version 2.4–6 R package (Oksanen 2015).
Principal component analysis (PCA) was used to identify differences in variables between land-use types and depth and to generate independent PCs representative of vegetation and soil properties for use in the subsequent Redundancy Analysis (RDA). Visualisation of PCA used the function prcomp in R core stats package (Kassambara and Mundt 2017), and packages factoextra (Husson et al. 2020) and FactoMineR (Barber et al. 2017). Dimensions for variables with eigenvalues ≥ 1 that cumulatively explained 80% or more of the variation were used in the RDA that was used to assess the relationship between environmental properties and soil fungal taxonomic and functional composition. This approach was used by Waymouth et al. (2021) where variables included in the RDA are dimensions from the PCA and were fitted using the envfit function in vegan package (Oksanen 2015). Vegetation factors were duplicated for both soil depths. Bulk density and soil moisture were removed as they were only sampled in top-soil.
Mantel tests were used to test bivariate relationships between environmental properties and soil fungal community composition (taxonomic and functional; Mantel 1967). Pearson correlation coefficients were used to test for correlations between dissimilarity matrices using 9999 comparative trials. As in the PERMANOVA, Euclidean distance dissimilarities were used for environmental properties and Bray–Curtis dissimilarities were used for soil fungal communities. Variance partitioning was used to examine the relative importance of environmental properties in determining the variability of soil fungal community taxonomic and functional composition and using the function varpart (Borcard et al. 1992; Legendre and Legendre 2012).
Prior to analyses, environmental data (soil physicochemical and vegetation) were either log10 or square-root transformed as necessary to meet assumptions of normality and homogeneity of variance. Hellinger transformations were performed on fungal community data, using the vegan package (Oksanen 2015) and decostand function (Anderson et al. 2006; Legendre and Gallagher 2001; Oksanen 1983). All statistical analyses were conducted in R version 1.0.153 (RStudio Team 2016).
Results
Environmental properties – vegetation and soil physicochemical properties
Vegetation properties were clearly distinct among land uses, with pasture having more exotic ground cover species, lower sub-canopy cover and more distance between plants (APPD) than restored and remnant quadrats (Fig. 2; Online Resource 6). Pasture sites had significantly fewer plant species across all strata compared to restored and remnant sites (Online Resource 6). Pasture had significantly different vegetation properties relative to restored and remnant land-use types (PERMANOVA P < 0.01; Online Resource 6). More details about differences with land use in both vegetation and soil physicochemical properties can be found in Waymouth et al. (2021).
Key vegetation properties in ground cover (a, Litter; b, Richness of Exotic Species), sub-canopy (d, Total Cover), Canopy (e, Average Point to Plant Distance) and PCA of vegetation properties (c). For a, b, d and e values represent means (± SE) with P values from general linear models (GLM) testing for the effect of land use (L). Significant differences (Tukey HSD test) between land uses are indicated by superscript letters. PCA of vegetation properties, points are coloured based on land use (red = pasture (P), green = restored (RS), and blue = remnant (RM)). Abbreviations are as follows; for Ground Cover – Bareground (BG), Litter (L), Richness of exotic species (GC_Exotic), Richness of native species (GC_Native), and Total Cover (GC_TC); For Sub-Canopy – Richness of Acacia spp. (SC_Acacia), Total Cover (SC_TC), Total Richness of Native and Exotic species (SC_TR); For Canopy—Average Basal Area (BA), Average Basal Area of Eucalyptus spp. (BA_Euc), Average Point to Plant Distance (APPD), Relative Density (RD), Abundance of Eucalyptus spp. (C_Euc), Total Density (C_Den), and Total Richness (C_Rich)
Principal component analysis (PCA) of vegetation properties grouped pasture quadrats distinct to other land-use types (Fig. 2c for PC1 and 2; Online Resource 7). 84% of the total variance was explained by the first four components, the remaining components explained less than 10% of the variance (Online Resource 8). PC1 (45% variance explained) reflected the results of the GLM and PERMANOVA (Fig. 2a, b, d, and e; Online Resource 6) and separated pasture from restored and remnant quadrats with separation most strongly correlated with canopy richness, average basal area, abundance of eucalypts, and total richness of sub-canopy (contribution > 10%; Online Resource 9). None of the other PC axes showed a clear separation of quadrats based on land use, instead reflecting the variability in ground cover characteristics (bare ground, litter, total ground cover; Online Resource 8).
There were few differences in soil physicochemical properties among land uses (Fig. 3; Online Resource 10). For example, pasture quadrats had greater available phosphorus and less acidic soil (pH ~ 5) than remnant (pH ~ 4, Fig. 3a, b). Pasture also had greater proportions of silt and clay, with less sand than restored quadrats (Fig. 3c; Online Resource 10). Across all land uses top-soil was characterised as having greater C, NH4+-N, available P, Fe and Mn than sub-soil (Fig. 3a, e, f; Online Resource 10).
Key soil physicochemical properties (a, Phosphorus; b, pH; c, Clay; e, Carbon and; f, Nitrogen), and PCA of soil physicochemical properties (d). For a, c, e and f values represent means (± SE); for b values represent mean pH (95% confidence interval – lower limit, upper limit). P values are from general linear models (GLM) testing for the effects of land use (L) and depth (D). Significant differences (Tukey HSD test) between land uses are indicated by superscript letters. Points are coloured based on land use (red = pasture (P), green = restored (RS), and blue = remnant (RM)) with symbols distinguishing soil depth (circle = surface soil and triangle = subsoil)
The PCA of soil physicochemical properties indicated no clear separation of quadrats according to land use or depth (Fig. 3d; Online Resource 11). The soil fertility gradient separated quadrats in PC1 (33%), whereas differences in soil texture separated the quadrats along PC2 (26%; Online Resource 12). Mn and Na separated quadrats along PC3 (11%) and PC4 (7%) respectively (Online Resource 7).
Land use and depth effects on soil fungal taxonomic composition
Fungal taxonomic composition by phylum and class did not differ with land use (P = 0.34 F = 1.07) or depth (P = 0.52 F = 0.77; Fig. 4). No dispersion effect was found for land use (P = 0.71 F = 0.34) or depth (P = 0.74 F = 0.11). Overall fungal taxonomic abundance, richness, and diversity did not differ with either land use or depth (Online Resource 13). Once separated into different phyla however, remnant soils tended to have lower fungal abundance and diversity than restored and pasture, which had the greatest abundance and diversity (Table 1). GLM results within class showed that only three of the 14 taxonomic classes investigated (Archaeorhizomycetes, Glomeromycetes and Tremellomycetes) differed in abundance, richness, or diversity with land use (Table 1).
Archaeorhizomycetes (Ascomycota) were relatively more abundant in pasture quadrats than in remnant quadrats, with restored showing intermediate values (Table 1). Glomeromycetes were more abundant in restored, where Tremellomycetes (Basidiomycota) also showed the greater diversity than in remnant, with pasture having intermediate values (Table 1). Top-soil tended to have higher richness and diversity of phyla and classes compared to sub-soil (Table 1). Glomeromycota had significantly higher diversity and Mucoromycota had significantly higher richness in top-soil than sub-soil (Table 1). Arthoniomycetes on the other hand had higher abundance in sub-soil than top-soil (Table 1).
Few differences were observed in the distribution of taxa across the vertical profile in relative abundance of taxonomic groups (Fig. 5). Top-soil in pasture quadrats had the greatest proportion of Ascomycota (48%) and lowest of Basidiomycota (13%; Fig. 5b and c), while sub-soil had more equal proportions with 29% and 24% respectively. Sub-soil of restored quadrats had the largest relative abundance of Glomeromycota (4%), driven by increased abundance of Glomeromycetes (Fig. 5d and Table 1).
Relative abundance of fungal community OTUs: (a) overall by phylum, and within the classes of (b) Ascomycota, (c) Basidiomycota, (d) Glomeromycota, (e) Mucoromycota and (f) Rozellomycota by land-use type; Pasture (P), Restored (RS) and Remnant (RM) across both depths (0–10 cm = Top-soil (T) and 20–30 cm = Sub-soil (S). Data are means of n = 6 per land use
Land use and depth effects on soil fungal functional composition
PERMANOVA analyses by trophic guild revealed marginal differences in fungal community function with land use (P = 0.07 F = 1.70), but not with depth (P = 0.99 F = 0.25; Fig. 6). Pairwise comparisons showed that remnant quadrats were significantly different in functional composition to restored quadrats, but pasture quadrats were not different to restored or remnant quadrats (Fig. 6). There were no differences in dispersion of soil fungal functional data by land use (P = 0.80 F = 0.23), or depth (P = 0.66 F = 0.20; Fig. 6).
Abundance, richness and diversity did not differ consistently within trophic modes between land-use types or soil depth (Online Resource 13). In contrast, when examined at the level of trophic guilds, abundance, richness, or diversity of seven of the ten guilds differed with land use (Table 2). Remnant quadrats had lower richness and diversity of all pathogen types and dung saprotrophs than pasture and restored (Table 2). In contrast, both pasture and remnant had similar abundance of wood saprotrophs, which was greater than that found in restored sites. Ectomycorrhizal fungi showed higher diversity in pasture and restored than remnant quadrats (Table 2). Only arbuscular mycorrhizal fungi showed a difference with depth, where top-soil had higher richness and diversity than sub-soil (Table 2).
Comparisons of relative abundances of sequences by trophic mode and guild revealed the largest differences between pasture and other land uses (Fig. 7). Pasture soils were characterised by higher relative abundance of dung saprotrophs (Ascomycota; Fig. 7c) and a lower relative abundance of pathotrophs and symbiotrophs (Fig. 7bb, d; Online Resources 14–16). Restored soils harboured greater relative proportions of plant pathogens particularly in top-soil (primarily comprised of taxonomic class Agaricomycetes and Sordariomycetes from phylum Ascomycota), wood saprotrophs and arbuscular mycorrhizal fungi (Fig. 6; Online Resources 14–16). Remnant soils had the greatest proportions of animal pathogens (in top-soil) and ectomycorrhizal fungi (symbiotrophs; class Agaricomycetes from phylum Basidiomycota, in sub-soil, Fig. 6b, d; Online Resources 14–16).
Relative abundance of fungal community OTUs (a) overall by trophic mode, and showing trophic guilds within (b) Pathotroph, (c) Saprotroph and (d) Symbiotroph, by land use; Pasture (P), Restored (RS) and Remnant (RM) across both depths (0–10 cm = Top-soil (T) and 20–30 cm = Sub-soil (S)). Data are means of n = 6 per land use
Relationships between fungal taxonomic composition and environmental properties
Environmental properties explained 35% of the total variation in fungal taxonomic composition by phyla (P = 0.03, Fig. 8a). RDA1 explained 18.5% and RDA2 explained 7.4% of the constrained variation. No clear groups were observed based on land use or depth (Fig. 8a). One vegetation and two soil principal components were related to fungal taxonomic composition (Fig. 8a; Online Resource 17). These were Veg PC3 (P = 0.04), which represents a gradient of variation in ground cover features (litter, ground cover of exotics, total ground cover, bare ground and average point-plant distance (APPD; Online Resource 9), Soil PC1 (P = 0.01) and Soil PC2 (P < 0.001), which represent gradients in soil fertility (N, P, K and Ca) and soil texture, respectively (Online Resource 12). RDA results indicate that Ascomycota, Basidiomycota and Mucoromycota were more abundant in areas with greater canopy cover, little ground cover, lower proportion of clay and lower soil fertility (N, P, K and Ca) relative to other areas (Fig. 8a).
Redundancy analysis using environmental properties from PCA dimensions (Figs. 2c, 3d) to explain variation in (a) Fungal community taxonomic phyla and (b) Fungal community function shown through trophic guilds. The direction of constraints is derived from principal components and shown as a grey arrow, black lines indicate constraints that were overlapping other text. Fungal community had a P value of 0.03, and Trophic Guild had a P value < 0.01. Land-use type abbreviations; Pasture (P), Restored (RS) and Remnant (RM). Names for phyla have been abbreviated in Fig. 8a, as follows: Ascomycota (Asco), Basidiomycota (Basi), Glomeromycota (Glom), Mucoromycota (Muco), Rozellomycota (Roze), with rare Phyla grouped (Rare) and unclassified fungi (Uncl). Names for trophic guilds have been abbreviated in Fig. 8b, as follows: animal pathogens (Apath), plant pathogens (Ppath), rare pathogens (Rpath), arbuscular mycorrhizal fungi (AMF), ectomycorrhizal fungi (EcM), rare symbiotrophs (Rsym), dung saprotroph (Dsap), wood saprotroph (Wsap), rare saprotrophs (Rsap) and undefined saprotrophs (Usap)
Mantel tests showed that variation in taxonomic composition of soil fungal communities was significantly correlated with soil chemical properties at both depths and with soil physical properties only in top-soil (Table 3). Notably, no significant correlations were observed between variation in sub-canopy vegetation and variation in fungal taxonomic composition (Table 3). Variation in ground cover vegetation correlated with variation in sub-soil fungal taxonomic composition, except for Glomeromycota where no correlation was observed (Table 3). Ascomycota and Mucoromycota at both depths were significantly correlated with variation in canopy vegetation, whereas Basidiomycota and Glomeromycota in top-soil were significantly correlated with variation in canopy vegetation (Table 3).
Variance partitioning analysis indicated that the total variation in fungal taxonomic composition in top-soil explained by environmental properties (vegetation and soil physicochemical properties) ranged from 7–35%, a lower proportion was explained in sub-soil (6–21%; Online Resource 18). Soil chemical properties explained the largest proportion of variation in fungal taxonomic composition (average 8.7%, Online Resource 18). Vegetation properties explained the largest proportion of variation in top-soil communities of Glomeromycota (11%), whereas none of the variation in sub-soil communities of Glomeromycota was explained by vegetation properties (Online Resource 18).
Relationships between fungal functional composition and environmental properties
In total 36% of the variation in fungal community trophic guilds was explained by environmental properties (P < 0.001; Fig. 8b). RDA1 and RDA2 explained 12.5% and 7.3% of the constrained variation respectively. As for the taxonomy, no clear groupings were observed with land use or depth (Fig. 8b). Variation in functional composition was related to Veg PC4 (a gradient of increasing sub-canopy cover and decreasing canopy tree basal area and bareground; P = 0.02) and Soil PC2 (a textural-pH gradient of increasing clay, and decreasing sand content and pH, P < 0.01; Online Resources 7, 9 and 12). Soil PC2 was positively corelated with RDA1 and 2, whereas Veg PC4 was negatively corelated with RDA1 but positively correlated with RDA2 (Fig. 8b). Therefore, sites with higher clay content, lower sub-canopy cover and higher tree basal area tended to harbour more plant pathogens (PPath), dung saprotrophs (DSap) and arbuscular mycorrhizal fungi (AMF; Fig. 8b). In contrast, areas with higher sub-canopy cover and lower tree basal area encompassed a greater abundance of animal pathogens (APath), wood saprotrophs (WSap) and Ectomycorrhizal fungi (EcM; Fig. 8b). Our results from Mantel test and the variance partitioning analysis support these results and show differences with soil depth (Table 3; Online Resource 18). A notable difference with depth was in the composition of saprotrophs, in top-soil, relationships between composition and environmental properties were found, but not in sub-soil (Table 3; Online Resource 18).
Discussion
Our study investigated changes to soil fungal taxonomic and functional composition in response to the conversion of riparian vegetation to grazing pasture and subsequent restoration. In contrast to our hypothesis, we found few differences in fungal taxonomic and functional composition between land uses. Instead, soil fungal community composition responded more strongly to variations in soil physicochemical properties and canopy cover.
Land use effects on soil fungal (taxonomic and functional) composition
Fungal community composition (both taxonomic and functional) changed little with land use, with few specific differences observed within phyla, class or guild. Soils at restored sites were characterised by a higher abundance of class Glomeromycetes within Glomeromycota (which includes arbuscular mycorrhizal fungi), and a greater richness and diversity of Tremellomycetes within Basidiomycota (which includes pathogens) compared to remnant and pasture soils. Investigations of functional guilds showed soils at restored sites had a higher abundance of pathogens, arbuscular mycorrhizal fungi and wood saprotrophs than pasture or remnant soils.
Fungi within the phylum Glomeromycota are arbuscular mycorrhizal fungi that are known to associate with grasses and forbs (Sepp et al. 2019). Previously it has been shown that grazing pasture had lower relative abundance of arbuscular mycorrhizal fungi than grasslands with high plant diversity (Oehl et al. 2003; Vályi et al. 2015). Our results agree with this finding as we found intermediate abundances of class Glomeromycetes and functional group arbuscular mycorrhizal fungi in pasture soils, which also had a lower richness of native ground cover than restored. Other studies also show that a change in land use from pasture to forest reduces the abundance of arbuscular mycorrhizal communities (Balami et al. 2020; Melo et al. 2020). Similarly, we found that remnant soils consistently had the lowest abundance of class Glomeromycetes and trophic guild arbuscular mycorrhizae. Furthermore, the comparatively higher abundance of arbuscular mycorrhizal fungi in restored soils may be related to changes in nutrient acquisition due to the dual mycorrhizal status of some myrtaceous species. Eucalyptus species had been planted in the restored areas and this is a group in which root associations shift from arbuscular mycorrhizal fungi to ectomycorrhizal dominance as plants age (Chen et al. 2000; Teste et al. 2020). Therefore, mature extant vegetation would have a lower colonisation of arbuscular mycorrhizal fungi compared with the young plants in the restored sites as previously shown by Bermúdez-Contreras et al. (2020).
Our results indicated that land use also influenced soil fungal communities that decompose organic matter (saprotrophs). Soil at restored sites had the greatest abundance of wood saprotrophs, pasture had the greatest richness of dung saprotrophs and remnant had similar abundance of wood saprotrophs to pasture, but lowest diversity and richness of dung saprotrophs. Previous studies have shown higher abundance of wood saprotrophs in natural forests than in plantation forests or farmland (Zhang et al. 2021), a difference that was related to plant community composition and soil chemical characteristics (pH and NH4+-N concentration). A similar relationship is shown in our results, with restored sites having the lowest soil pH and the greatest wood saprotroph abundance suggesting an increase in nutrient cycling 3–9 years after restoration. The greater diversity of dung saprotrophs found in our pasture compared to remnant sites is consistent with the history of grazing. We support research by Eldridge and Delgado-Baquerizo (2018) that showed that increasing intensity of cattle grazing and historic grazing by livestock in pasture and woodland communities had a positive effect on the relative abundance of dung saprotrophs. Consistent with that, the restoration and destocking of pastures here was associated with a decrease in richness and diversity of dung saprotrophs.
The main group of potential fungal pathogens (disease causing) found here were plant pathogens, which had twice the relative abundance in restored soils than other land-use types. It has been shown that plant pathogens have higher diversity in grazed pasture than in natural systems (Makiola et al. 2019) which contrasts with the result in our study. The greater relative abundance of plant pathogens in restored soils here may be due to the planting technique introducing new fungal taxonomic groups, bearing in mind that functional guild is only known for ~ 30% of the sequences. Previous research by Bermúdez-Contreras et al. (2020) showed transplanted plants had novel fungi compared to plants sown directly on site. It may be that these novel fungi were pathogens, and account for the observed higher abundance of plant pathogens, however that study reported fungal taxonomy, not functional guilds. Another avenue for the high relative abundance of plant pathogens may be due to a lack of sterilisation procedures in the nursery or during the transplanting process. Consequently, the restoration provides opportunities for the introduction of plant pathogens (Hancock 2015; Rooney-Latham et al. 2019; Simamora et al. 2018). The increase in plant pathogens may also be due to differences in plant species richness particularly between pasture and restored land-use types, the latter having significantly greater species richness than pasture. Previous studies report that plant species richness is a dominant factor affecting plant pathogens as plants provide diverse substrates for pathogens (Chen et al. 2018; Yang et al. 2017). Soil disturbance from farming practices may also play a role in pathogen abundance (Hu et al. 2021) and explain the differences between restored and remnant which both had high plant species richness. A higher plant species richness could have a greater root biomass and diversity of root exudates (Cui et al. 2018; Zhang et al. 2019), thus providing a higher abundance of resources for fungi. Therefore, increasing plant species richness would likely increase the biomass and diversity of plant pathogens. Furthermore, in remnant riparian forests it is likely that some degree of equilibrium had been reached between soil fungal communities and pathogens as the vegetation community is relatively stable. On the other hand, restored sites are in a state of flux, with the vegetation community changing rapidly as the planted species grow, increasing the likelihood of opportunistic pathogens. Consequently, the observed higher relative abundance of plant pathogens in restored soils has implications for plant establishment and restoration outcomes and requires further investigation.
Depth effects on soil fungal (taxonomic and functional) composition
Contrary to our hypotheses we found a lack of differences with depth in both fungal taxonomic and functional composition. The few differences we observed showed a similar pattern of Mucoromycota and Glomeromycota taxa having greater diversity in top-soil. Root exudates, root density, litter, carbon and nutrient availability change along the soil profile (Carnovale et al. 2019; Zhang et al. 2020) inducing a shift in fungal community composition (Aponte et al. 2010a; Gregory 2008; Kasel et al. 2008; Otsing et al. 2018; Peay et al. 2011). Mucoromycota are saprophytic fungi that inhabit litter and coarse woody debris and therefore, and consistent with previous research (Posada et al. 2012), it is expected that they are more diverse in the top-soil than in the sub-soil. Furthermore, greater carbon and nitrogen were observed in the top-soil compared with sub-soil, a difference that could underpin the observed changes. Glomeromycota form arbuscular mycorrhizal associations with non-woody species and their diversity is strongly dependent on root depth (Peay et al. 2011). Forbs and grasses have shallower roots than woody species therefore underpinning the observed greater diversity of Glomeromycota in top-soil than sub-soil (Gregory 2008). In our study, few differences were found in soil chemistry with soil depth, which agrees with the few changes noted in the fungal community.
Arthoniomycetes (phyla Ascomycota) abundances showed a contrasting pattern with soil depth. In pastures they were more abundant on the top-soil, whereas in restored quadrats they were more abundant on the sub-soil. Arthoniomycetes are usually lichen-forming fungi that inhabit exposed surfaces (Nelsen et al. 2011). It is possible that the soil tillage and turn-over that was implemented during the restoration may have altered the distribution of this taxa through the soil profile.
Relationships between soil fungal community composition and environmental properties
Consistent with our hypothesis, fungal taxonomic and functional composition were related to several vegetation properties. Interestingly soil fungal taxonomic composition was related to ground cover vegetation, but functional composition was more strongly related to canopy vegetation. This is in contrast with our previous research where variation in soil fungal communities was strongly related to variation in sub-canopy vegetation at one site (Waymouth et al. 2020). It may be that at different scales (within a site or across multiple sites) relationships with vegetation properties differ. Ascomycota and Basidiomycota, which contain ectomycorrhizal species, were more abundant in areas with greater canopy cover, and little ground cover, a relationship which may be due to shading and associated differences in soil microclimate which vary with canopy cover. Previous investigations by Perreault et al. (2020) showed a relationship between canopy density and both soil fungal taxonomic and functional composition, where fungal abundance was higher in areas with canopy gaps, soil temperature was higher and more phosphorus was available compared to areas without canopy gaps. Another study by Branco et al. (2013) reported differences in fungal community in two canopy defined-zones (inner – close to trunk and outer – further away) under single trees, showing higher diversity of ectomycorrhizal fungi close to tree trunks, where root densities are higher. Other vegetation properties, such as native plant species richness, can also influence fungal diversity (Sielaff et al. 2018), and exotic plant species can reinforce the degraded state of soils by changing nutrient cycles and soil biota composition (Ba et al. 2018; Smith et al. 2018). We found that Ascomycota and Basidiomycota were more abundant in areas with greater canopy cover, and less ground cover possibly related to differences in the richness of native plant species in ground cover between land-use types.
Our results also highlight a strong relationship between soil chemical attributes and soil fungal community composition, where these differences may underpin to a great extent the observed variation in the fungal community composition. Despite the absence of overall differences in soil chemistry with land use, we found significant differences in P, Ca and pH. Calcium (and pH) was higher in pasture quadrats and top-soil compared to remnant quadrats, potentially due to increased inputs from farming practices. Available P was higher in pasture and restored soils than in remnant forests. Soil pH, Ca concentration (as a modulator of soil pH) and the availability of nutrients (P and N) may select for particular taxonomic groups and functions in the soil fungal community (e.g., Aponte et al. 2010b, Newbound et al. 2012, Leff et al. 2015, Cassman et al. 2016, and Ye et al. 2020). The greater abundance of symbiotrophic phyla Ascomycota, and Basidiomycota in areas with lower soil fertility (N, P, K and Ca) in this study is consistent with previous work in which sites with low nutrient content and higher pH were characterised by a higher fungal taxonomic and functional diversity and greater abundance of symbiotrophs (Aponte et al. 2010b; Henry et al. 2015; Lauber et al. 2008).
Soil texture also emerged as a strong predictor of soil fungal composition. Soil texture modulates soil fertility (N, P, K and Ca), influencing soil microbial communities and restoration (Bach et al. 2010). We found soil texture, primarily the proportions of clay and sand, were related to changes in both soil fungal taxonomic and functional composition. Other studies have shown that variations in soil clay content are related to changes in soil fungal communities (Cassman et al. 2016; Schmidt et al. 2019). Our results are also in agreement with Xiang et al. (2014) who found a relationship between fungal taxonomic composition and soil texture (particle size distribution), and suggested it was due to the influence of soil texture on soil aeration, nutrient content, and water-holding capacity. Contrastingly, investigations by Klopf et al. (2017) in grasslands showed no correlation between soil texture and soil fungal community. Our research indicates that in riparian ecosystems of south-eastern Australia soil texture is an important determinant for soil fungal taxonomic and functional composition.
Conclusion
Fungal taxonomic and functional composition varied more with soil physicochemical properties, rather than land use or depth. We found that Ascomycota and Basidiomycota were more abundant in areas with little ground cover and lower soil fertility (N, P, K and Ca), whereas the opposite was true for Glomeromycota. Sites with higher clay content, lower sub-canopy cover and higher tree basal area tended to harbour more plant pathogens, dung saprotrophs and arbuscular mycorrhizae. In contrast, areas with higher sub-canopy cover and lower tree basal area encompassed a greater abundance of animal pathogens, wood saprotrophs and ectomycorrhizal fungi. Contrary to our hypothesis, land-use change had little effect on the taxonomic and functional composition of soil fungal communities, however changes in soil chemistry and vegetation related to the conversion of riparian forest to grazing pasture were associated with a shift in soil fungal community composition.
Data Availability
The datasets generated during the current study are available in the Bioplatforms Australia Data Portal, https://data.bioplatforms.com/organization/about/australian-microbiome. Data from vegetation surveys are available from the corresponding author on reasonable request.
References
Allan JD (2004) Landscapes and riverscapes: the influence of land use on stream ecosystems. Annu Rev Ecol Evol Syst 35:257–284
Alvarez-Sanchez J, Johnson N, Antoninka A, Chaudhary VB, Lau MK, Owen SM, Sanchez-Gallen I, Guadarrama P, Castillo S (2012) Large-scale diversity patterns in spore communities of arbuscular mycorrhizal fungi [Chapter 2]. In: Pagano MC (ed) Mycorrhiza: occurrence in natural and restored environments. Nova Science Publishers Inc, Hauppauge, p 29–47
Anderson MJ, Ellingsen KE, McArdle BH (2006) Multivariate dispersion as a measure of beta diversity. Ecol Lett 9:683–693
Aponte C, Garcia LV, Maranon T, Gardes M (2010a) Indirect host effect on ectomycorrhizal fungi: Leaf fall and litter quality explain changes in fungal communities on the roots of co-occurring Mediterranean oaks. Soil Biol Biochem 42:788–796
Aponte C, Marañón T, García LV (2010b) Microbial C, N and P in soils of Mediterranean oak forests: influence of season, canopy cover and soil depth. Biogeochemistry 101:77–92
Ba L, Facelli E, Facelli JM (2018) Plant-mycorrhizal fungi feedbacks: potential accomplices of Avena barbata’s high invasiveness. Plant Ecol 219:1045–1052
Bach EM, Baer SG, Meyer CK, Six J (2010) Soil texture affects soil microbial and structural recovery during grassland restoration. Soil Biol Biochem 42:2182–2191
Balami S, Vašutová M, Godbold D, Kotas P, Cudlín P (2020) Soil fungal communities across land use types. iForest-Biogeosciences and Forestry 13:548
Banning NC, Gleeson DB, Grigg AH, Grant CD, Andersen GL, Brodie EL, Murphy D (2011) Soil microbial community successional patterns during forest ecosystem restoration. Appl Environ Microbiol 77:6158–6164
Barber NA, Chantos-Davidson KM, Amel PR, Sherwood JP, Swingley WD (2017) Soil microbial community composition in tallgrass prairie restorations converge with remnants across a 27-year chronosequence. Environ Microbiol 19:3118–3131
Bates D, Mächler M, Bolker B, Walker S (2014) Fitting linear mixed-effects models using lme4. arXiv preprint arXiv:1406.5823
Bengtsson-Palme J, Ryberg M, Hartmann M, Branco S, Wang Z, Godhe A, De Wit P, Sánchez-García M, Ebersberger I, de Sousa F (2013) Improved software detection and extraction of ITS1 and ITS 2 from ribosomal ITS sequences of fungi and other eukaryotes for analysis of environmental sequencing data. Methods Ecol Evol 4:914–919
Bennett LT, Kasel S, Tibbits J (2009) Woodland trees modulate soil resources and conserve fungal diversity in fragmented landscapes. Soil Biol Biochem 41:2162–2169
Bermúdez-Contreras AI, Ede F, Waymouth V, Miller RE, Aponte C (2020) Revegetation technique changes root mycorrhizal colonisation and root fungal communities: the advantage of direct seeding over transplanting tube-stock in riparian ecosystems. Plant Ecol 221:813–828
Bever J, Schultz P, Miller R, Gades L, Jastrow J (2003) Prairie mycorrhizal fungi inoculant may increase native plant diversity on restored sites (Illinois). Ecol Restor 21:311–312
Bissett A, Fitzgerald A, Meintjes T, Mele PM, Reith F, Dennis PG, Breed MF, Brown B, Brown MV, Brugger J, Byrne M, Caddy-Retalic S, Carmody B, Coates DJ, Correa C, Ferrari BC, Gupta VVSR, Hamonts K, Haslem A, Hugenholtz P, Karan M, Koval J, Lowe AJ, Macdonald S, McGrath L, Martin D, Morgan M, North KI, Paungfoo-Lonhienne C, Pendall E, Phillips L, Pirzl R, Powell JR, Ragan MA, Schmidt S, Seymour N, Snape I, Stephen JR, Stevens M, Tinning M, Williams K, Yeoh YK, Zammit CM, Young A (2016) Introducing BASE: The biomes of Australian soil environments soil microbial diversity database. GigaScience 5:1–11
Borcard D, Legendre P, Drapeau P (1992) Partialling out the spatial component of ecological variation. Ecology 73:1045–1055
Borrell AN, Shi Y, Gan Y, Bainard LD, Germida JJ, Hamel C (2017) Fungal diversity associated with pulses and its influence on the subsequent wheat crop in the Canadian prairies. Plant Soil 414:13–31
Bouffaud M-L, Bragalini C, Berruti A, Peyret-Guzzon M, Voyron S, Stockinger H, van Tuinen D, Lumini E, Wipf D, Plassart P, Lemanceau P, Bianciotto V, Redecker D, Girlanda M (2017) Arbuscular mycorrhizal fungal community differences among European long-term observatories. Mycorrhiza 27:331–343
Branco S, Bruns TD, Singleton I (2013) Fungi at a small scale: spatial zonation of fungal assemblages around single trees. PLoS ONE 8:e78295
Brundrett MC (2002) Coevolution of roots and mycorrhizas of land plants. New Phytol 154:275–304
Bureau of Meteorology (2020) Climate statistics for Australian locations. Melbourne, Australia
Calvo-Polanco M, Sánchez-Castro I, Cantos M, García JL, Azcón R, Ruiz-Lozano JM, Beuzón CR, Aroca R (2016) Effects of different arbuscular mycorrhizal fungal backgrounds and soils on olive plants growth and water relation properties under well-watered and drought conditions. Plant, Cell Environ 39:2498–2514
Canfield RH (1941) Application of the line interception method in sampling range vegetation. J Forest 39:388–394
Carnovale D, Bissett A, Thrall PH, Baker G (2019) Plant genus (Acacia and Eucalyptus) alters soil microbial community structure and relative abundance within revegetated shelterbelts. Appl Soil Ecol 133:1–11
Cassman NA, Leite MFA, Pan Y, de Hollander M, van Veen JA, Kuramae EE (2016) Plant and soil fungal but not soil bacterial communities are linked in long-term fertilised grassland. Sci Rep 6:23680
Chen W, Xu R, Wu Y, Chen J, Zhang Y, Hu T, Yuan X, Zhou L, Tan T, Fan J (2018) Plant diversity is coupled with beta not alpha diversity of soil fungal communities following N enrichment in a semi-arid grassland. Soil Biol Biochem 116:388–398
Chen Y, Brundrett M, Dell B (2000) Effects of ectomycorrhizas and vesicular–arbuscular mycorrhizas, alone or in competition, on root colonization and growth of Eucalyptus globulus and E. urophylla. New Phytol 146:545–555
Cottam G, Curtis JT (1956) The use of distance measures in phytosociological sampling. Ecology 37:451–460
Crowther TW, Boddy L, Hefin JT (2012) Functional and ecological consequences of saprotrophic fungus-grazer interactions. ISME J 6:1992–2001
CSBP Lab (2019) CSBP Lab Methods. https://www.csbp.com.au/docs/default-source/csbp-lab/csbp-lab-methods-1118.pdf. CSBP Lab, 2 Altona Street, Bibra Lake, WA 6163
Cui Y, Fang L, Guo X, Wang X, Zhang Y, Li P, Zhang X (2018) Ecoenzymatic stoichiometry and microbial nutrient limitation in rhizosphere soil in the arid area of the northern Loess Plateau, China. Soil Biol Biochem 116:11–21
Daigneault AJ, Eppink FV, Lee WG (2017) A national riparian restoration programme in New Zealand: Is it value for money? J Environ Manage 187:166–177
Edgar RC (2010) Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26:2460–2461
Edgar RC, Flyvbjerg H (2015) Error filtering, pair assembly and error correction for next-generation sequencing reads. Bioinformatics 31:3476–3482
Eldridge DJ, Delgado-Baquerizo M (2018) Functional groups of soil fungi decline under grazing. Plant Soil 426:51–60
Ettema CH, Wardle DA (2002) Spatial soil ecology. Trends Ecol Evol 17:177–183
Fierer N, Lauber CL, Ramirez KS, Zaneveld J, Bradford MA, Knight R (2012) Comparative metagenomic, phylogenetic and physiological analyses of soil microbial communities across nitrogen gradients. ISME J 6:1007–1017
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for Basidiomycetes-application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Gómez Gallego T, García-Rabasa S, Flores-Rentería D, Rincón A (2013) Effect of temperature on ectomycorrhizal fungi associated with Pinus sylvestris L. in organic vs. mineral soils. II IBEMPA Conference microorganisms for future agriculture, pp 176–177
Gregory PJ (2008) Plant roots: growth, activity and interactions with the soil. John Wiley & Sons
Guo Y, Hou L, Zhang Z, Zhang J, Cheng J, Wei G, Lin Y (2019) Soil microbial diversity during 30 years of grassland restoration on the Loess Plateau, China: Tight linkages with plant diversity. Land Degrad Dev 30:1172–1182
Hamonts K, Bissett A, Macdonald BCT, Barton PS, Manning AD, Young A (2017) Effects of ecological restoration on soil microbial diversity in a temperate grassy woodland. Appl Soil Ecol 117–118:117–128
Hancock D (2015) Towards new nursery industry protocols for Phytophthora control on supply of stock for restoration and revegetation. Acta Horticulturae 1140:81–84. https://doi.org/10.17660/ActaHortic.2016.1140.15
Harner MJ, Opitz N, Geluso K, Tockner K, Rillig MC (2011) Arbuscular mycorrhizal fungi on developing islands within a dynamic river floodplain: An investigation across successional gradients and soil depth. Aquat Sci 73:35–42
Henry C, Raivoarisoa J-F, Razafimamonjy AR, Heriniaina R, Andrianaivomahefa P, Selosse M-A, Ducousso M (2015) Asteropeia mcphersonii, a potential mycorrhizal facilitator for ecological restoration in Madagascar wet tropical rainforests. For Ecol Manage 358:202–211
Henzell T (2007) Australian agriculture: its history and challenges. CSIRO Publishing, Collingwood
Hu X, Liu J, Liang A, Li L, Yao Q, Yu Z, Li Y, Jin J, Liu X, Wang G (2021) Conventional and conservation tillage practices affect soil microbial co-occurrence patterns and are associated with crop yields. Agriculture, Ecosystems & Environment 319:107534
Husson F, Josse J, Le S, Mazet J (2020) Multivariate exploratory data analysis and data mining. R package
Jobim K, Silva IR, Assis DMA, Vieira LC, Silva DKA, Oehl F, Maia LC, Goto BT (2016) Arbuscular mycorrhizae in coastal areas. In: Pagano MC (ed) Recent Advances on Mycorrhizal Fungi. Springer, Switzerland, pp 101–108
Kasel S, Bennett LT, Tibbits J (2008) Land use influences soil fungal community composition across central Victoria, south-eastern Australia. Soil Biol Biochem 40:1724–1732
Kassambara A, Mundt F (2017) Extract and visualize the results of multivariate data analyses. R Package, https://CRAN.R-project.org/package=factoextra. Accessed 24 Dec 2021
Klopf RP, Baer SG, Bach EM, Six J (2017) Restoration and management for plant diversity enhances the rate of belowground ecosystem recovery. Ecol Appl 27:355–362
Kramer S, Marhan S, Ruess L, Armbruster W, Butenschoen O, Haslwimmer H, Kuzyakov Y, Pausch J, Scheunemann N, Schoene J (2012) Carbon flow into microbial and fungal biomass as a basis for the belowground food web of agroecosystems. Pedobiologia 55:111–119
Lauber CL, Strickland MS, Bradford MA, Fierer N (2008) The influence of soil properties on the structure of bacterial and fungal communities across land-use types. Soil Biol Biochem 40:2407–2415
Leff JW, Jones SE, Prober SM, Barberan A, Borer ET, Firn JL, Harpole WS, Hobbie SE, Hofmockel KS, Knops JMH, McCulley RL, La Pierre K, Risch AC, Seabloom EW, Schutz M, Steenbock C, Stevens CJ, Fierer N (2015) Consistent responses of soil microbial communities to elevated nutrient inputs in grasslands across the globe. Proc Natl Acad Sci USA 112(35):10967–10972
Legendre P, Gallagher ED (2001) Ecologically meaningful transformations for ordination of species data. Oecologia 129:271–280
Legendre P, Legendre L (2012) Numerical ecology. Elsevier
Lenth R (2020) Estimated marginal means, aka least-squares means. R package https://CRAN.R-project.org/package=emmeans. Accessed 24 Dec 2021
Liang Y, He X, Chen C, Feng S, Liu L, Chen X, Zhao Z, Su Y (2015) Influence of plant communities and soil properties during natural vegetation restoration on arbuscular mycorrhizal fungal communities in a Karst region. Ecol Eng 82:57–65
Makiola A, Dickie IA, Holdaway RJ, Wood JR, Orwin KH, Glare TR (2019) Land use is a determinant of plant pathogen alpha-but not beta-diversity. Mol Ecol 28:3786–3798
Mantel N (1967) The detection of disease clustering and a generalised regression approach. Can Res 27:209–220
McMurdie PJ, Holmes S (2014) Waste not, want not: why rarefying microbiome data is inadmissible. PLoS Comput Biol 10(4):1–12
Melo CD, Pimentel R, Walker C, Rodríguez-Echeverría S, Freitas H, Borges PAV (2020) Diversity and distribution of arbuscular mycorrhizal fungi along a land use gradient in Terceira Island (Azores). Mycol Prog 19:643–656
Middleton EL, Bever JD (2012) Inoculation with a native soil community advances succession in a grassland restoration. Restor Ecol 20:218–226
Molina R, Massicotte H, Trappe JM (1992) Specificity phenomena in mycorrhizal symbioses: community-ecological consequences and practical implications. In: Allen MF (ed) Mycorrhizal functioning: an integrative plant-fungal process. Chapman and Hall, New York, p 357–423
Naiman RJ, Decamps H, McClain ME (2010) Riparia: Ecology, conservation, and management of streamside communities. Elsevier
Naiman RJ, Decamps H, Pollock M (1993) The role of riparian corridors in maintaining regional biodiversity. Ecol Appl 3:209–212
Nelsen MP, Plata ER, Andrew CJ, Lücking R, Lumbsch HT (2011) Phylogenetic diversity of Trentepohlialean algae associated with lichen-forming fungi. J Phycol 47:282–290
Newbound M, Bennett LT, Tibbits J, Kasel S (2012) Soil chemical properties, rather than landscape context, influence woodland fungal communities along an urban-rural gradient. Austral Ecol 37:236–247
Nguyen NH, Song Z, Bates ST, Branco S, Tedersoo L, Menke J, Schilling JS, Kennedy PG (2016) FUNGuild: an open annotation tool for parsing fungal community datasets by ecological guild. Fungal Ecology 20:241–248
Nilsson RH, Larsson K-H, Taylor AFS, Bengtsson-Palme J, Jeppesen TS, Schigel D, Kennedy P, Picard K, Glöckner FO, Tedersoo L (2018) The UNITE database for molecular identification of fungi: Handling dark taxa and parallel taxonomic classifications. Nucleic Acids Res 47:D259–D264
Oehl F, Sieverding E, Ineichen K, Mader P, Boller T, Wiemken A (2003) Impact of land use intensity on the species diversity of arbuscular mycorrhizal fungi in agroecosystems of central Europe. Appl Environ Microbiol 69:2816–2824
Oksanen J (1983) Ordination of boreal heath-like vegetation with principal component analysis, correspondence analysis and multidimensional scaling. Vegetatio 52:181–189
Oksanen J (2015) Multivariate analysis of ecological communities in R: vegan tutorial. R package version. R Doc, https://john-quensen.com/wp-content/uploads/2018/10/Oksanen-Jari-vegantutor.pdf. Accessed 24 Dec 2021
Oksanen J, Blanchet FG, Friendly M, Kindt R, Legendre P, McGlinn D, Minchin PR, O'Hara RB, Simpson GL, Solymos P, Stevens MHH, Szoecs E, Wagner H (2018) Vegan: community ecology package. R package version 1.17–4
Otsing E, Barantal S, Anslan S, Koricheva J, Tedersoo L (2018) Litter species richness and composition effects on fungal richness and community structure in decomposing foliar and root litter. Soil Biol Biochem 125:328–339
Pagano M, Cabello M, Scotti M (2007) Phosphorus response of three native Brazilian trees to inoculation with four arbuscular mycorrhizal fungi. Journal of Agriculture Technology 3:231–240
Peay KG, Baraloto C, Fine PV (2013) Strong coupling of plant and fungal community structure across western Amazonian rainforests. ISME J 7:1852–1861
Peay KG, Kennedy PG, Bruns TD (2011) Rethinking ectomycorrhizal succession: are root density and hyphal exploration types drivers of spatial and temporal zonation? Fungal Ecology 4:233–240
Peet RK (1974) The measurement of species diversity. Annu Rev Ecol Syst 5:285–307
Perreault L, Forrester JA, Wurzburger N, Mladenoff DJ (2020) Emergent properties of downed woody debris in canopy gaps: A response of the soil ecosystem to manipulation of forest structure. Soil Biology and Biochemistry 151:108053
Poesen J, De Baets S (2005) The effects of plant root characteristics on soil erosion. RECONDES: conditions for restoration and mitigation of desertified areas in southern Europe using vegetation: 113
Posada RH, Madriñan S, Rivera E-L (2012) Relationships between the litter colonization by saprotrophic and arbuscular mycorrhizal fungi with depth in a tropical forest. Fungal Biol 116:747–755
Richardson DM, Holmes PM, Esler KJ, Galatowitsch SM, Stromberg JC, Kirkman SP, Pyšek P, Hobbs RJ (2007) Riparian vegetation: Degradation, alien plant invasions, and restoration prospects. Divers Distrib 13:126–139
Riis T, Kelly-Quinn M, Aguiar FC, Manolaki P, Bruno D, Bejarano MD, Clerici N, Fernandes MR, Franco JC, Pettit N (2020) Global overview of ecosystem services provided by riparian vegetation. Bioscience 70:501–514
Rincón A, Santamaría-Pérez B, Rabasa SG, Coince A, Marçais B, Buée M (2015) Compartmentalised and contrasted response of ectomycorrhizal and soil fungal communities of Scots pine forests along elevation gradients in France and Spain. Environ Microbiol 17:3009–3024
Rooney-Latham S, Blomquist C, Kosta K, Gou Y, Woods P (2019) Phytophthora species are common on nursery stock grown for restoration and revegetation purposes in California. Plant Dis 103:448–455
RStudio Team (2016) RStudio: integrated development environment for R. RStudio, Inc., Boston, MA, USA, http://www.r-project.org/. Accessed 24 Dec 2021
Schmidt R, Mitchell J, Scow K (2019) Cover cropping and no-till increase diversity and symbiotroph:saprotroph ratios of soil fungal communities. Soil Biol Biochem 129:99–109
Semchenko M, Leff JW, Lozano YM, Saar S, Davison J, Wilkinson A, Jackson BG, Pritchard WJ, Jonathan R, Oakley S (2018) Fungal diversity regulates plant-soil feedbacks in temperate grassland. Science advances 4:eaau4578
Sepp SK, Davison J, Jairus T, Vasar M, Moora M, Zobel M, Öpik M (2019) Non-random association patterns in a plant–mycorrhizal fungal network reveal host–symbiont specificity. Mol Ecol 28:365–378
Sharma SK, Ramesh A, Sharma MP, Joshi OP, Govaerts B, Steenwerth KL, Karlen DL (2010) Microbial community structure and diversity as indicators for evaluating soil quality. Sustainable Agriculture Reviews 5:317–358
Siciliano SD, Palmer AS, Winsley T, Lamb E, Bissett A, Brown MV, van Dorst J, Ji M, Ferrari BC, Grogan P (2014) Soil fertility is associated with fungal and bacterial richness, whereas pH is associated with community composition in polar soil microbial communities. Soil Biol Biochem 78:10–20
Sielaff AC, Upton RN, Hofmockel KS, Xu X, Polley HW, Wilsey BJ (2018) Microbial community structure and functions differ between native and novel (exotic-dominated) grassland ecosystems in an 8-year experiment. Plant Soil 432:359–372
Simamora AV, Paap T, Howard K, Stukely MJ, Hardy GESJ, Burgess TI (2018) Phytophthora contamination in a nursery and its potential dispersal into the natural environment. Plant Dis 102:132–139
Smith ME, Facelli JM, Cavagnaro TR (2018) Interactions between soil properties, soil microbes and plants in remnant-grassland and old-field areas: a reciprocal transplant approach. Plant Soil 433(1):1–19
Stürmer SL, Sgrott AF, Braghirolli FL, Uhlmann A, Pescador R (2012) Importance of arbuscular mycorrhizal fungi for recovery of riparian sites in Southern Brazil [Chapter 13]. In: Pagano MC (ed) Mycorrhiza: occurrence in natural and restored environments. Nova Science Publishers, pp 275–289
Sun Y, Zhang Y, Feng W, Qin S, Liu Z (2019) Revegetated shrub species recruit different soil fungal assemblages in a desert ecosystem. Plant Soil 435:81–93
Teste FP, Jones MD, Dickie IA (2020) Dual-mycorrhizal plants: their ecology and relevance. New Phytol 225:1835–1851
Vályi K, Rillig MC, Hempel S (2015) Land-use intensity and host plant identity interactively shape communities of arbuscular mycorrhizal fungi in roots of grassland plants. New Phytol 205:1577–1586
van der Heijden MGA, Bardgett RD, Van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310
Victoria State Government (2015) Regional riparian action plan. In: The State of Victoria Department of Environment, Land, Water and Planning (eds), pp 1–53
Wang M, Qu L, Ma K, Yuan X (2013) Soil microbial properties under different vegetation types on Mountain Han. Science China Life Sciences 56:561–570
Wang Q, Garrity GM, Tiedje JM, Cole JR (2007) Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl Environ Microbiol 73:5261–5267
Wardle DA, Bardgett RD, Klironomos JN, Setälä H, Van Der Putten WH, Wall DH (2004) Ecological linkages between aboveground and belowground biota. Science 304:1629–1633
Wasof S, De Schrijver A, Schelfhout S, Perring MP, Remy E, Mertens J, de la Peña E, De Sutter N, Viaene N, Verheyen K (2019) Linkages between aboveground and belowground community compositions in grasslands along a historical land-use intensity gradient. Plant Soil 434:289–304
Waymouth V, Miller RE, Ede F, Bissett A, Aponte C (2020) Variation in soil microbial communities: elucidating relationships with vegetation and soil properties, and testing sampling effectiveness. Plant Ecol 221:837–851
Waymouth V, Miller RE, Kasel S, Ede F, Bissett A, Aponte C (2021) Soil bacterial community responds to land-Use change in riparian ecosystems. Forests 12:157
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. PCR Protocols: a Guide to Methods and Applications 18:315–322
Wubs EJ, Van der Putten WH, Bosch M, Bezemer TM (2016) Soil inoculation steers restoration of terrestrial ecosystems. Nature Plants 2:1–5
Xiang D, Verbruggen E, Hu Y, Veresoglou SD, Rillig MC, Zhou W, Xu T, Li H, Hao Z, Chen Y (2014) Land use influences arbuscular mycorrhizal fungal communities in the farming–pastoral ecotone of northern China. New Phytol 204:968–978
Yan D, Mills JG, Gellie NJ, Bissett A, Lowe AJ, Breed MF (2018) High-throughput eDNA monitoring of fungi to track functional recovery in ecological restoration. Biol Cons 217:113–120
Yang T, Adams JM, Shi Y, He J.s., Jing X., Chen L., Tedersoo L. and Chu H. (2017) Soil fungal diversity in natural grasslands of the Tibetan Plateau: associations with plant diversity and productivity. New Phytol 215:756–765
Ye G, Lin Y, Luo J, Di HJ, Lindsey S, Liu D, Fan J, Ding W (2020) Responses of soil fungal diversity and community composition to long-term fertilization: Field experiment in an acidic Ultisol and literature synthesis. Applied Soil Ecology 145:103305
Zhang Q, Deng J, Zhu W, Wu S, Bai X, Zhou Y, Yin Y (2021) Land-use types combined with plant species alter soil fungal community and functional guilds in the Eastern Mountainous Region of Liaoning Province. China Polish Journal of Environmental Studies 30(1):477–495
Zhang X, Chen X, Liu M, Xu Z, Wei H (2020) Coupled changes in soil organic carbon fractions and microbial community composition in urban and suburban forests. Sci Rep 10:15933
Zhang Y, Cao C, Cui Z, Qian W, Liang C, Wang C (2019) Soil bacterial community restoration along a chronosequence of sand-fixing plantations on moving sand dunes in the Horqin sandy land in northeast China. J Arid Environ 165:81–87
Acknowledgements
We thank Lisa Wittick, Sascha Andrusiak, Nicholas Osborne for technical support, and field assistants Cordula Gutekunst, Ana Bermudez Contreras, Sarah Fischer, Tony Lovell, Kathy Russell, Robert Dabal, Anu Singh and Scott McKendrick; and Joanne McGimpsey for reviewing early drafts.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions This study was funded by Holsworth Wildlife Research Endowment from the Ecological Society of Australia, Melbourne Water, and the Madeleine Selwyn Smith Memorial Scholarship. Vicky Waymouth was a recipient of a Research Training Program Scholarship and the Faculty of Science write-up Award supported by the Albert Shimmins Fund. Cybec Foundation supported Rebecca Miller during her lectureship at the University of Melbourne. Sabine Kasel is supported by the Integrated Forest Ecosystem Research Program funded by the Victorian Government Department of Environment, Land, Water and Planning. We would like to acknowledge the contributions of the Biomes of Australian Soil Environments (BASE) and Australian Microbiome consortiums for the generation of genetic data for this study. The Australian Microbiome initiative is supported by funding from Bioplatforms Australia and the Integrated Marine Observing System (IMOS) through the Australian Government’s National Collaborative Research Infrastructure Strategy (NCRIS), Parks Australia through the Bush Blitz program funded by the Australian Government and BHP, and CSIRO. Soil collection and field sampling was conducted under Parks Victoria permit number 10008494.
Author information
Authors and Affiliations
Contributions
Conceptualization, V.W., R.E.M., F.E., and C.A.; methodology, V.W., R.E.M., F.E., and C.A.; software, V.W., A.B., and C.A.; formal analysis, V.W., S.K., A.B., R.E.M., and C.A.; investigation, V.W., R.E.M., S.K., F.E., and C.A.; resources, V.W., F.E., A.B., R.E.M., and C.A.; data curation, V.W., and A.B.; writing—original draft preparation, V.W., R.E.M., S.K., and C.A.; writing, review and editing, V.W., R.E.M., S.K., and C.A.; validation, V.W., R.E.M., S.K., and C.A.; project administration, V.W., R.E.M., S.K., F.E., and C.A.; funding acquisition, V.W., R.E.M., A.B., F.E., and C.A. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest/Competing interests
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Responsible Editor: Matthew A. Bowker.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Waymouth, V., Miller, R.E., Kasel, S. et al. Riparian fungal communities respond to land-use mediated changes in soil properties and vegetation structure. Plant Soil 475, 491–513 (2022). https://doi.org/10.1007/s11104-022-05383-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-022-05383-8