Abstract
Although increasing concentrations of atmospheric CO2 are predicted to have substantial impacts on plant growth and functioning of ecosystems, there is insufficient understanding of the responses of belowground processes to such increases. We investigated the effects of different dark septate root endophytic (DSE) fungi on growth and nutrient acquisition by Pinus sylvestris seedlings under conditions of N limitation and at ambient and elevated CO2 (350 or 700 μ1 CO2 l−1). Each seedling was inoculated with one of the following species: Phialocephala fortinii (two strains), Cadophora finlandica, Chloridium paucisporum, Scytalidium vaccinii, Meliniomyces variabilis and M. vraolstadiae. The trial lasted 125 days. During the final 27 days, the seedlings were labeled with 14CO2 and 15NH +4 . We measured extraradical hyphal length, internal colonization, plant biomass, 14C allocation, and plant N and 15N content. Under elevated CO2, the biomass of seedlings inoculated with DSE fungi was on average 17% higher than in control seedlings. Simultaneously, below-ground respiration doubled or trebled, and as a consequence carbon use efficiency by the DSE fungi significantly decreased. Shoot N concentration decreased on average by 57% under elevated CO2 and was lowest in seedlings inoculated with S. vaccinii. Carbon gain by the seedlings despite reduced shoot N concentration indicates that DSE fungi increase plant nutrient use efficiency and are therefore more beneficial to the plant under elevated CO2.
Similar content being viewed by others
Explore related subjects
Find the latest articles, discoveries, and news in related topics.Avoid common mistakes on your manuscript.
Introduction
The rising level of atmospheric CO2 is predicted to have substantial impacts on plant growth and the functioning of ecosystems. Predicting plant response to increasing CO2 will require more detailed data concerning dynamics of roots and microbial populations (Phillips 2007; Alberton et al. 2005; Heinemeyer et al. 2007; Alberton and Kuyper 2009).
Roots of a very large number of plant species are regularly colonized by a group of ascomycete fungi with usually dark-pigmented (melanized) septate hyphae (Mandyam and Jumpponen 2005; Sieber and Grünig 2006). These fungi are now referred to as dark septate root endophytic (DSE) fungi; in former times they were also known under the collective name Mycelium radicis atrovirens (MRA) and as pseudomycorrhizal fungi (Summerbell 2005a). DSE fungi constitute a polyphyletic assemblage, with most species belonging to the Leotiomycetes (Kernaghan et al. 2003; Hambleton and Sigler 2005; Wang et al. 2006).
DSE fungi exhibit a very broad host range. A review by Jumpponen and Trappe (1998a) reported that close to 600 plant species from more than 110 families across the world are hosts to DSE fungi. DSE fungi occur and co-occur in plants that are also able to form arbuscular, orchid, ericoid and ectomycorrhizal (ECM) associations. Summerbell (2005b) noted that DSE fungi often co-occur with healthy ectomycorrhizas, although they are also common on non-mycorrhizal tree roots (Ahlich and Sieber 1996). Read and Haselwandter (1981) suggested that DSE fungi often replace arbuscular mycorrhizal (AM) and ECM fungi in arctic and alpine ecosystems, however Ruotsalainen (2003) failed to confirm this pattern in northern Finland. Postma et al. (2007) suggested that DSE fungi replace AM fungi at very low pH. Their occurrence on tree seedlings (Horton et al. 1998) and in the earliest stages of primary succession (Jumpponen 2003; Cázares et al. 2005) has been mentioned, but DSE fungi can be equally abundant on mature trees (Ahlich and Sieber 1996).
Despite their almost ubiquitous occurrence, the behavior of DSE fungi on plant roots, and hence their impact on plant performance and fitness, are poorly understood. Wilcox and Wang (1987) indicated pathological (parasitic) behavior on tree seedlings at low pH, as shown by reduced performance of the host plant. The abundance of DSE fungi in healthy-looking roots, however, suggests that they are unlikely to be primary pathogens (Sieber and Grünig 2006). In line with that suggestion, many studies (summarized in Mandyam and Jumpponen 2005) indicated that DSE fungi could improve plant performance (improved nutrient uptake, increased tolerance against root pathogens, improved ability to withstand adverse environmental conditions).
Associations with DSE fungi share with mycorrhizal associations that they span the continuum from mildly antagonistic to mutualistic interactions (Johnson et al. 1997; Jumpponen 2001). In the case of DSE fungi, it is not known to what extent such differences in behavior are context-dependent, being influenced by the availability of plant C on the one hand and external nutrients on the other hand (Jumpponen and Trappe 1998a,1998b; Jumpponen 2001). Different outcomes of experiments with DSE fungi on host plants can also be due to differences among various fungal species.
Sieber and Grünig (2006) suggested that elevated CO2 as a consequence of burning fossil fuels and land use change could affect functioning of DSE fungi, but noted that nothing was known about the direction and magnitude of effects. The objectives of our study were therefore to determine plant biomass (C gain) and nutrient acquisition (N gain) by Scots pine (Pinus sylvestris) seedlings in association with different DSE species under low N availability and at ambient and elevated CO2.
Materials and methods
Plant, fungi and substrate
Around 1200 (10 g) seeds of Scots pine, purchased from PVM (Grubbenvorst, The Netherlands), were soaked overnight in a 200 ml beaker containing demineralized water, and then surface-sterilized during 30 min in a 200 ml beaker containing 30% H2O2-solution and a drop of Tween 20. Around 700 surface-sterilized seeds were germinated in Petri dishes (Ø 14.5 cm), in a horizontal position, on sterile water agar containing 5 g glucose l−1. Germination was around 80 %. After three weeks, 96 visually equal-sized seedlings were transferred to 96 Petri dishes (Ø 14.5 cm), in a vertical position, filled with 50 g dry weight of a wet-sterilized (1 h at 110°C) peat-vermiculite mixture (2:5, w/w). Each seedling was inoculated around the root system with 12 plugs of actively growing mycelium of one of the following DSE fungal species: Phialocephala fortinii (two strains), Cadophora finlandica, Chloridium paucisporum, Scytalidium vaccinii, Meliniomyces variabilis, M. vraolstadiae, or uninoculated (See Table S1 for further details on fungal species). The uninoculated controls also received around the root system 12 plugs with uncolonized agar. The Petri dishes were hermetically sealed with parafilm and tape, covered with aluminum foil, but contained two lateral openings on the top. One opening allowed the shoot to grow out of the dish using lanolin for sealing, while the other opening could be instantaneously opened in the flow hood for addition of nutrient solution and prevention of anoxia in the root compartment, after which it was closed again with parafilm and tape. The peat-vermiculite mixture contained 29.9 mg available mineral N kg−1 and 1.4 mg available P kg−1. The peat-vermiculite mixture was moistened with 50 ml of full-strength modified Melin-Norkrans (MMN; Marx 1969) solution (without agar, glucose and malt extract) at the start of the experiment. MMN solution was used to ensure that both fungal and control treatments were solely limited by N (and not by P), and to prevent a decrease in pH due to ammonium-N uptake during the experiment (Kamminga-van Wijk and Prins 1989). During the first 98 days, we added 28 ml MMN five times. Just before the start of 14C-labeling we added 35 ml MMN with (15NH4)2HPO4 at 5% 15N (corresponding to 1.84 mg of mineral N). This labeling of 15N during the final four weeks allowed us to separate the net effect over the full experimental period (125 days) from the effect over the final 27 days. In total, 11.8 mg of N and 38.8 mg of P were added to each Petri dish. Addition of the MMN-solution to the experimental system and the set up of the below-ground respiration trap were done as described by Alberton et al. (2007).
Plant growth and experimental conditions
The seedlings were placed into two growth chambers (one for each CO2 level) with a steady flow of either 350 or 700 µl CO2 l−1. A previous study, where the seedlings were placed into four growth chambers (two for each CO2 level), showed no differences between both growth chambers for each CO2 level (Alberton et al. 2007), and for that reason we used only two growth chambers. Temperature was maintained at 20°C during the day (16 h, photosynthetic photon fluence rate (PPFR) of 250 µmol m−2 s−1) and at 16°C during the night (8 h), and relative humidity was kept around 70%. The Petri dishes were randomized every two weeks within each growth chamber. After 98 days, the treatments under ambient CO2 were transferred to an Experimental Soil Plant Atmosphere System (ESPAS; Gorissen et al. 1996) and treatments under elevated CO2 to another ESPAS compartment. The ESPAS facility consists of closed and sealed growth chambers with controlled condition of light, humidity, temperature and CO2 availability. The seedlings were exposed to 14CO2 atmosphere (specific radioactivity 0.88 ± 0.1 kBq mg−1 C) supplied from a pressurized cylinder for an additional 27 days. This labeling of 14C during the final four weeks allowed us to separate the net effect over the full experimental period (125 days) from the effect over the final 27 days. Growth conditions (temperature, relative humidity and CO2 levels) in the ESPAS were equal to the growth chambers, except for PPFR, which was 375 µmol m−2 s−1.
Analyses
Seedlings were harvested 27 days after 14C and 15N-labeling started. Plants were removed from Petri dishes; shoots, roots and the peat-vermiculite mixture were separated and weighed; dried at 70°C for 48 h and weighed again. A subsample of fresh roots and the peat-vermiculite mixture from each replicate was used to estimate fractional colonization by DSE fungi and extraradical hyphal length. The same conversion from fresh weight to dry weight was used for the subsamples. The dried fractions were subsequently ground and analyzed for 14C content with the modified wet combustion method (Dalal 1979). Plant material (30 mg) and peat-vermiculite (0.5 g) were digested in duplicate in 5 ml of a 10% (w/v) solution of K2Cr2O7 in a mixture of concentrated H2SO4 and H3PO4 (3:2, v/v) at 160°C for 2 h. The 14CO2 evolved was trapped in 10 ml of 0.5 M NaOH. The 14C was determined in 0.5 ml of NaOH by liquid scintillation counting (Tri-Carb 2100TR; Packard Instrument Company, Meriden, CT, USA) using 3 ml of Ultima Gold (Packard). The soda-lime containing the below-ground respired 14CO2 was transferred to bottles (0.5 l) and dissolved in 50 ml excess 6 M HCl, injected through a septum in the lid. The 14CO2 evolved was captured in 10 ml of 5 M NaOH and analyzed by liquid scintillation counting as described.
Carbon and nitrogen concentrations and atom% 15N of shoot and root samples were determined at the UC Davis Stable Isotope Facility using an automated elemental analyzer-continuous flow isotope ratio mass spectrometer (RoboPrepSample Converter interfaced with a TracerMass Stable Isotope Detector; Europa Scientific, Crewe, England).
Extraradical hyphal length in the peat-vermiculite mixture was determined with the gridline intersection method after fluorescent staining (Bloem and Vos 2004). For each sample, 6 g of the homogenized substrate that contains hyphae, fine roots and 180 ml of dH2O were mixed in a blender for 1 min at maximum speed (20000 rev min−1). A 9 ml sample was collected, put in a plastic tube of 13 ml with screw cap, and fixed by adding 1 ml of 37% formaldehyde. The suspension was shaken for 10 sec and after 2 min of settling to remove coarse particles 12 μl of the suspension was evenly smeared in a hole (12 mm diameter) on a glass microscope slide and left to dry for 2 hrs at 50°C. Afterwards 50 μl of fluorescent brightener 28 (Sigma F3397, Sigma Chemical Co., St Louis, MD, USA) solution (1 mg ml−1) were added to the slides, and stained for 2 hrs in the dark at room temperature. The fluorescent brightener 28, which stains chitin in fungal cell walls, was used to determine hyphal length in the soil (substrate) samples. After staining the slides were rinsed 3 times for 20 min in dH2O, and finally air-dried overnight in the dark at room temperature. After air drying a coverslip was mounted with a small drop of immersion oil. The edges of the coverslip were sealed with nail varnish. Extraradical hyphal length was estimated under an epifluorescence microscope using the gridline intersection method over 100 randomly selected microscope fields at 400x magnification.
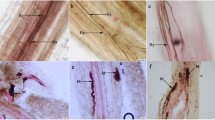
Root samples from each treatment were carefully washed under tap water and kept in 50% ethanol, then cleared in 10% KOH by autoclaving for 20 min at 121°C, rinsed in dH2O, acidified in 2% HCl for 5 min, and then stained in 0.05% Trypan blue by autoclaving for 15 min (Peterson et al. 2004). For each seedling 15 root segments of approx. 2 cm in length were placed on microscope slides (5 root segments per slide) in several drops of lactoglycerol. A coverslip was placed over the root segments and pressed gently. From each slide, approx. 33 (100 intercepts per seedling) randomly selected root intercepts were examined for hyphae and microsclerotia in the cortex under a compound light microscope at 400x magnification. Fractional root colonization was calculated by dividing the number of intersections with septate hyphae in the cortex by the total number of root intersections.
Statistical analyses
The experiment consisted of a fungal treatment (seven species of DSE fungi and uninoculated), and CO2 treatment (with 2 levels of 350 and 700 µl CO2 l−1) and six replicates, resulting in 96 seedlings (8 × 2 × 6). In order to comply with ANOVA assumptions for homogeneity (Levene’s test) some variables were log10 transformed before analysis: extraradical hyphal length, 14C in shoots, 14C in substrate, below-ground 14C respired, and below-ground 14C use efficiency [(14C in roots + 14C in substrate)/ 14C in roots + 14C in substrate + below-ground 14C respired]. We excluded two outliers of below-ground 14C respired using the box plot method from the S. vaccinii and M. vraolstadiae treatments at ambient CO2. ANOVA followed by the least significant difference (LSD) method post hoc test was performed using PROC General Linear Model (GLM) procedure in SAS 9.0 for Windows. Considering the large number of parameters measured and calculated (19), a sequential Bonferroni correction was applied to the ANOVA results to keep P value constant at P < 0.05. Pearson pairwise correlation coefficients were calculated and their significance determined in two-tailed tests as indicators of direction of relationship between plant and fungal parameters. All correlations (12 degrees of freedom) excluded the control.
Results
All seedlings remained alive and healthy, showing no symptoms of fungal disease during the experiment. Almost all parameters were significantly affected by DSE fungi and CO2 level. Five of the parameters (extraradical hyphal length, below-ground 14C respired, below-ground 14C use efficiency, total shoot N concentration and total shoot N content) were affected by the interaction between DSE fungi and CO2 (Table 1). The complete data set is provided as Supplementary material (Table S2).
Root colonization by DSE fungi and length of the extraradical mycelium
All controls remained free of fungi. One strain of P. fortinii showed very high levels of colonization, with values over 50%. Colonization by the other DSE fungal strains was below 12% (Table S2). Microsclerotia were not observed. Extraradical hyphal length varied between 26 and 166 m g−1 substrate. It was highest in Chloridium paucisporum and lowest in Cadophora finlandica and M. vraolstadiae (Fig. 1). The length of the extraradical mycelium increased under elevated CO2, on average by 53%, but the difference was statistically significant only for M. variabilis. The interaction between DSE fungi and CO2 was also significant for extraradical hyphal length (Table 1). Fractional colonization and hyphal length were not correlated.
Main effect of seven DSE fungal strains and uninoculated treatment under 350 or 700 µl CO2 l−1 on plant shoot and root biomass (g), and extraradical hyphal length (m g−1 substrate) values. Letters refer to plant shoot (white bars) and root biomass (shaded bars) and between parenthesis for hyphal length. Means followed by different letters are significantly different according to one-way ANOVA
Plant biomass
Plant biomass was significantly affected by DSE fungi and CO2 levels (Table 1 and Fig. 1). The most beneficial fungi as judged from plant biomass were S. vaccinii and M. vraolstadiae under elevated CO2 (Fig. 1). Under ambient CO2, plants inoculated with DSE fungi were on average 4% heavier than control plants and under elevated CO2, plants inoculated with DSE fungi were 17% heavier than control plants, but the difference was not statistically significant. Across all plants (DSE-inoculated and controls plants) under elevated CO2, shoot and root biomass increased significantly by 21% and 19% respectively, relative to ambient. Incorporation of 14C showed the same trends, with higher values over the final four weeks (increase by 40% and 30% for shoots and roots respectively). The shoot-to-root ratio and 14C in shoot-to-root ratio were not affected by CO2 level (Table 1). Plant biomass was neither correlated with levels of internal colonization nor extraradical hyphal length.
Below-ground respiration of 14C accounted for 4–10% of the 14C budget under ambient CO2 and for 7–27% under elevated CO2. Below-ground respiration increased significantly under elevated CO2 except in the control seedlings and in seedlings inoculated with M. vraolstadiae (Fig. 2 and Table S2). As a consequence, below-ground carbon-use efficiency declined significantly under elevated CO2, except for the control plants and plants inoculated with M. vraolstadiae. Below-ground CUE was significantly positively correlated with shoot N concentration and content (r = 0.70 and 0.67 respectively; P < 0.01).
Plant nitrogen uptake
Shoot N concentration was significantly different among DSE fungal treatments (Table 1). Under ambient CO2, shoot N concentration was highest in plants inoculated with P. fortinii – MRA and S. vaccinii. Shoot N concentration ranged between 8.2 and 11.5 mg g−1 under ambient CO2 and declined to 4.7 to 7.5 mg g−1 under elevated CO2 (Fig. 3 and Table S2). The decline in shoot N concentration was less in control plants than in plants inoculated with DSE fungi. The decline was largest in plants inoculated with S. vaccinii. On average, shoot N concentration was 57% lower under elevated CO2. The interaction between DSE fungi and CO2 levels was also significant. Root N concentration was significantly affected by DSE fungi and CO2 levels (Table 1). Elevated CO2 decreased root N concentration on average by 16%. It was lowest in the treatment with M. vraolstadiae and highest in the control (Fig. 3). The decline in root N concentration under elevated CO2 was much smaller than the decline in shoot N concentration. Shoot and root N concentration were not significantly correlated (r = 0.27; P > 0.05).
Main effect of seven DSE fungal strains and uninoculated treatment under 350 or 700 µl CO2 l−1 on plant shoot and root N concentration (mg g−1). Letters refer to plant shoot N concentration (white bars) and root N concentration (shaded bars). Means followed by different letters are significantly different according to one-way ANOVA
Shoot and root N content, and total plant N uptake were not affected by DSE fungi (Table 1). In total 13.3 mg of mineral N was available for plant growth. Total N uptake ranged between 10.5 and 12.7 mg under ambient CO2, and between 8.3 and 11.8 mg under elevated CO2 (Table S2). Total N uptake declined under elevated CO2 by 12%. The decline was largest in plants inoculated with M. variabilis and M. vraolstadiae. Shoot N content declined by 32% under elevated CO2 and was significant for seedlings inoculated with S. vaccinii, M. variabilis and M. vraolstadiae, whereas N content of roots did not change. Total N content was not significantly correlated with plant biomass, shoot or root dry weight. Likewise, shoot N content was not significantly correlated with plant biomass. Mycelium length was significantly negatively correlated with shoot N content (r = −0.70; P < 0.01). Plant N uptake (assessed by 15N over the final 27 days) was not affected by DSE fungi (Table 1).
Discussion
The relationship between pine seedlings and DSE fungi
Our study showed a neutral to weakly beneficial effect of DSE fungi in terms of plant carbon gain under ambient CO2 with almost no differences between shoot and root biomass. DSE fungi had a larger positive effect on plant biomass under elevated CO2, but the difference was statistically significant only for S. vaccinii and M. vraolstadiae. Sieber and Grünig (2006) had already suggested that enhanced CO2 could affect functioning of DSE fungi, although they also noted that nothing was known about the direction and magnitude of effects.
The results of studies between DSE fungi and their host plants show a large variation and the ecological roles of DSE fungi are still poorly understood. For P. fortinii, Jumpponen (2001) listed examples of negative, neutral and positive effects on plant performance. Jumpponen et al. (1998) noted that the effects of P. fortinii on the growth of Pinus contorta were neutral at low N supply but positive at higher N supply. Kernaghan et al. (2003) observed that P. fortinii was relatively common in nurseries with higher levels of applied N, but that several other DSE fungi including C. finlandica and S. vaccinii were more common in nurseries with lower levels of N application.
In order to compare our results with the literature we performed a meta-analysis (For details see Alberton et al. 2005) based on 11 studies (Fernando and Currah 1996; Cameron 1998; Jumpponen et al. 1998; Jumpponen and Trappe 1998b; Yu 2000; Hashimoto and Hyakumachi 2001; Schulz et al. 2002; Vohník et al. 2003, 2005; Wu and Guo 2008; Grelet et al. 2009). The meta-analysis showed for shoot biomass no significant effect (Response ratio R [inoculated/control] = 1.05 (95% confidence interval of R = 0.92–1.19; n = 34), and a significant positive effect for root mass (R = 1.30; 95% confidence interval of R = 1.05–1.61; n = 16). The difference in response ratio between shoot and root weight was marginally significant (P = 0.06). Our data support the conclusion that DSE fungi have little or no effect on shoot biomass, and suggest that under some conditions (possibly related to N availability) root biomass increases.
Fungal performance
All seven DSE fungal strains in this experiment colonized the roots of P. sylvestris seedlings. Phialocephala fortinii is considered the most important representative of the DSE fungi (Stoyke et al. 1992; Harney et al. 1997; Grünig et al. 2004; Peterson et al. 2008). The species has been described as a co-inhabitant of ECM roots (Summerbell 2005b) or an ECM fungus (Kaldorf et al. 2004). Menkis et al. (2004) observed that the species is very plastic in its ecological behavior. The two strains of P. fortinii were very different in colonization of pine seedlings. It could be possible that differential behavior is due to the experimental conditions, especially as the second strain (P. fortinii – MRA) has already been in culture for almost 60 years. However, we cannot exclude the possibility that our two study strains belong to closely related cryptic species with divergent evolutionary histories.
Strains of M. vraolstadiae have been described as mycorrhizal and non-mycorrhizal (Hambleton and Sigler 2005). Meliniomyces variabilis is also variable in its trophic habit, with some strains obtained from ECM and other strains from ericoid mycorrhizal and AM roots (Hambleton and Sigler 2005). Cadophora finlandica forms both ECM and ericoid mycorrhizal associations (Hambleton and Sigler 2005). Chloridium paucisporum is considered ectendomycorrhizal (Harney et al. 1997). Scytalidium vaccinii, the anamorph of Rhizoscyphus ericae is known as the ericoid mycorrhizal fungus par excellence. However, Hambleton and Sigler (2005) mentioned an ECM isolate, while Piercey et al. (2002) reported that one strain of that fungus formed an intracellular association.
Despite the fact that this study is, to our knowledge, the most comprehensive study of various DSE species under elevated CO2, we cannot exclude the possibility that the life history strategy adopted by these fungi could depend on experimental conditions, or alternatively that there is no relationship between taxonomy and ecology.
Potential mechanisms for mutualistic behavior by DSE fungi
The DSE fungi usually form incomplete mantles that only partially cover the root, and very little of a Hartig net. On the other hand, Jumpponen (2001) claimed that DSE fungi possess intracellular (and possibly intercellular) interfaces. He therefore argued that DSE fungi are capable of forming mutualistic associations with plant roots that are functionally similar to mycorrhiza. Peterson et al. (2008) stated that DSE fungi lack a host-derived perifungal membrane and interfacial matrix material, both hallmarks for a biotrophic nutritional habit.
Addy et al. (2005) and Mandyam and Jumpponen (2005) suggested alternative explanations for enhanced plant growth without a nutritional interface over which C and nutrients are exchanged. These include increased mineralization in the rhizosphere, improved nutrient uptake, enhanced pathogen resistance and the production of phytohormones (especially indole acetic acid (IAA) and auxins).
Ruotsalainen (2003) observed that a strain of DSE fungi did not colonize seedlings of Gnaphalium norvegicum while still resulting in benefits for the seedling, suggesting a saprotrophic role for DSE fungi. Based on our study a mechanism through saprotrophy only is unlikely. Increased hyphal length and, especially, increased below-ground respiration for six DSE fungal species (but not for control seedlings and seedlings inoculated with M. vraolstadiae) under elevated CO2 indicate that the carbon fixed by the plant was rapidly available to the fungus.
Our study did not suggest a role for DSE fungi in increased nutrient uptake. Under elevated CO2 DSE fungi even reduced N content of the pine seedlings. These data suggest increased competition for N between plant and fungus due to the fact that elevated CO2 allowed the fungus to produce more biomass. However, only a small part of the additional C was incorporated in fungal biomass. The larger part of the additional carbon captured by the fungus was rapidly respired (Fig. 2). Because below-ground respiration of 14C doubled or trebled in 6 (of 7) DSE fungi, but not in the control, we ascribe this effect to fungal respiration. Heinemeyer et al. (2007) proposed a mechanism of a mycorrhizal fungal overflow CO2 tap through which additional carbon is directly returned to the atmosphere. Gorissen and Kuyper (2000) had earlier described increased respiration for a N-limited ECM fungus, Laccaria bicolor.
If elevated CO2 increases fungal immobilization of N, we would expect increased competition for N and decreased availability of N for the host plant, which constrains plant response to elevated CO2 (Reich et al. 2006; Hobbie et al. 2007). Surprisingly, even under reduced N availability elevated CO2 led to increases in both above-ground and below-ground plant biomass.
Shoot N concentration ranged between 8.2 and 11.5 mg g−1 under ambient CO2 and declined to 4.7 to 7.5 mg g−1 under elevated CO2. Especially these latter values are clearly below the deficiency limit of 10 mg g−1 as mentioned by Reuter et al. (1997) for pines. Data from northern Finland in Scots pine and Norway spruce (Picea abies) stands on very poor soils indicate that this degree of N limitation also occurs in natural systems (Helmisaari et al. 2007). The mechanisms underlying this higher N use efficiency through which growth increases were sustained, even under decreased N availability, merit further study.
A potential mechanism for the increase of plant biomass even when plant nutrient uptake decreases is the production of phytohormones by DSE fungi. We did not measure the production of phytohormones, but earlier authors noted that DSE fungi enhance plant growth by producing phytohormones or inducing the host hormone production without any apparent facilitation of host nutrient uptake or stimulation of host nutrient metabolism (Addy et al. 2005; Schulz and Boyle 2005). Rudawska et al. (1992) reported that DSE fungi are able to release IAA.
Behavioral differences and similarities between DSE and ECM fungi
In an earlier experiment, Alberton et al. (2007) described the impact of seven different ECM fungi on the growth performance of Scots pine seedlings under ambient and elevated CO2. Because the same experimental conditions were applied in that experiment, a comparison between both experiments provides likely explanations of behavioral differences and similarities between DSE and ECM fungi.
In the study with ECM fungi there was a very significant negative correlation between the amount of the extraradical mycelium and plant performance (Alberton et al. 2007). Direct competition for N between plant and fungus increased when elevated CO2 decreased fungal C limitation, leading to ECM fungal-driven progressive nitrogen limitation (Luo et al. 2004; Hu et al. 2006). With DSE fungi the situation was different. The amount of the extraradical mycelium was much smaller. Even though the fungi were also C-limited (as evidenced from a 53% increase in the length of the extraradical mycelium, an increase in root colonization, and a large increase in carbon that was respired under elevated CO2), hyphal length was not negatively correlated with plant biomass. Because the decline in shoot N concentration resulted in downregulation of photosynthesis in the case of ECM fungi, but not in the case of DSE fungi, it is likely that different mechanisms regulate the interaction. Furthermore, the increased sink strength of the ECM fungi resulted in a significant increase in C and N allocation below-ground. In the case of seedlings inoculated with DSE fungi, elevated CO2 resulted in significant higher N allocation, but not in increased C allocation to roots.
In the present study, the combination of increased fungal respiration and decline of shoot N content and concentration under elevated CO2 suggests that the association is not strongly mutualistic, while the increase in plant biomass and 14C assimilated suggest that mutualistic behavior is maintained. These observations confirm that it is difficult to position DSE fungi in the symbiotic continuum between beneficial and antagonistic (see reviews by Jumpponen and Trappe 1998a; Jumpponen 2001; Addy et al. 2005; Mandyam and Jumpponen 2005; Peterson et al. 2008; Schulz and Boyle 2005). Because the cost – benefit ratio for the pine seedlings in association with ECM fungi was more negatively affected by ECM fungi under elevated CO2 than DSE fungi, we hypothesize that the predicted increases in atmospheric CO2 will benefit these DSE fungi.
References
Addy HD, Piercey MM, Currah RS (2005) Microfungal endophytes in roots. Can J Bot 83:1–13. doi:10.1139/b04-171
Ahlich K, Sieber TN (1996) The profusion of dark septate endophytic fungi in non-ectomycorrhizal fine roots of forest trees and shrubs. New Phytol 132:259–270. doi:10.1111/j.1469-8137.1996.tb01845.x
Alberton O, Kuyper TW (2009) Ectomycorrhizal fungi associated with Pinus sylvestris seedlings respond differently to increased carbon and nitrogen availability: implications for ecosystem responses to global change. Glob Chang Biol 15:166–175. doi:10.1111/j.1365-2486.2008.01714.x
Alberton O, Kuyper TW, Gorissen A (2005) Taking mycocentrism seriously: mycorrhizal fungal and plant responses to elevated CO2. New Phytol 167:859–868. doi:10.1111/j.1469-8137.2005.01458.x
Alberton O, Kuyper TW, Gorissen A (2007) Competition for nitrogen between Pinus sylvestris and ectomycorrhizal fungi generates potential for negative feedback under elevated CO2. Plant Soil 296:159–172. doi:10.1007/s11104-007-9306-5
Bloem J, Vos A (2004) Fluorescent staining of microbes for total direct counts. In: Kowalchuk GA, de Bruijn FJ, Head IM, Akkermans AD, van Elsas JD (eds) Molecular microbial ecology manual, vol 402, 2nd edn. Kluwer Academic, Dordrecht, pp 861–874
Cameron SL (1998) Colonization of Populus tremuloides seedlings by the fungus Phialocephala fortinii in the presence of the ectomycorrhizal fungus Thelephora terrestris. M.Sc. thesis, University of Guelph, Guelph, Ont
Cázares E, Trappe JM, Jumpponen A (2005) Mycorrhiza-plant colonization patterns on a subalpine glacier forefront as a model system of primary succession. Mycorrhiza 15:405–416. doi:10.1007/s00572-004-0342-1
Dalal RC (1979) Simple procedure for the determination of total carbon and its radioactivity in soils and plant materials. Analyst 104:151–154
Fernando AA, Currah RS (1996) A comparative study of the effects of root endophytes Leptodontidium orchidicola and Phialocephala fortinii (Fungi Imperfecti) on the growth of some subalpine plants in culture. Can J Bot 74:1071–1078
Gorissen A, Kuyper TW (2000) Fungal species-specific responses of ectomycorrhizal Scots pine (Pinus sylvestris) to elevated [CO2]. New Phytol 146:163–168. doi:10.1046/j.1469-8137.2000.00610.x
Gorissen A, Kuikman PJ, van Ginkel JH, van de Beek H, Jansen AG (1996) ESPAS – an advanced phytotron for measuring carbon dynamics in a whole plant-soil system. Plant Soil 179:81–87. doi:10.1007/bf00011645
Grelet G-A, Johnson D, Paterson E, Anderson IC, Alexander IJ (2009) Reciprocal carbon and nitrogen transfer between an ericaceous dwarf shrub and fungi isolated from Piceirhiza bicolorata ectomycorrhizas. New Phytol 182:359–366. doi:10.1111/j.1469-8137.2009.02813.x
Grünig CR, McDonald BA, Sieber TN, Rogers SO, Holdenrieder O (2004) Evidence for subdivision of the root-endophyte Phialocephala fortinii into cryptic species and recombination within species. Fungal Genet Biol 41:676–687. doi:10.1016/j.fgb.2004.03.004
Hambleton S, Sigler L (2005) Meliniomyces, a new anamorph genus for root-associated fungi with phylogenetic affinities to Rhizoscyphus ericae (= Hymenoscyphus ericae), Leotiomycetes. Stud Mycol 53:1–27
Harney SK, Rogers SO, Wang CJK (1997) Molecular characterization of dematiaceous root endophytes. Mycol Res 101:1397–1404. doi:10.1017/s095375629700419x
Hashimoto Y, Hyakumachi M (2001) Effects of isolates of ectomycorrhizal fungi and endophytic Mycelium radicis atrovirens that were dominant in soil from disturbed sites on growth of Betula platyphylla var. japonica seedlings. Ecol Res 16:117–125. doi:10.1046/j.1440-1703.2001.00377.x
Heinemeyer A, Hartley IP, Evans SP, de la Fuentes JAC, Ineson P (2007) Forest soil CO2 flux: uncovering the contribution and environmental responses of ectomycorrhizas. Glob Chang Biol 13:1786–1797. doi:10.1111/j.1365-2486.2007.01383.x
Helmisaari HS, Derome J, Nöjd P, Kukkola M (2007) Fine root biomass in relation to site and stand characteristics in Norway spruce and Scots pine stands. Tree Physiol 27:1493–1504
Hobbie EA, Rygiewicz PT, Johnson MG, Moldenke AR (2007) 13C and 15N in microarthropods reveal little response of Douglas-fir ecosystems to climate change. Glob Chang Biol 13:1386–1397. doi:10.1111/j.1365-2486.2007.01379.x
Horton TR, Cázares E, Bruns TD (1998) Ectomycorrhizal, vesicular-arbuscular and dark septate fungal colonization of bishop pine (Pinus muricata) seedlings in the first 5 months of growth after wildfire. Mycorrhiza 8:11–18. doi:10.1007/s005720050205
Hu S, Tu C, Chen X, Gruver JB (2006) Progressive N limitation of plant response to elevated CO2: a microbiological perspective. Plant Soil 289:47–58. doi:10.1007/s11104-006-9093-4
Johnson NC, Graham JH, Smith FA (1997) Functioning of mycorrhizal associations along the mutualism-parasitism continuum. New Phytol 135:575–585. doi:10.1046/j.1469-8137.1997.00729.x
Jumpponen A (2001) Dark septate endophytes - are they mycorrhizal? Mycorrhiza 11:207–211. doi:10.1007/s00570100112
Jumpponen A (2003) Soil fungal community assembly in a primary successional glacier forefront ecosystem as inferred from rDNA sequence analyses. New Phytol 158:569–578. doi:10.1046/j.1469-8137.2003.00767.x
Jumpponen A, Trappe JM (1998a) Dark septate endophytes: a review of facultative biotrophic root-colonizing fungi. New Phytol 140:295–310. doi:10.1046/j.1469-8137.1998.00265.x
Jumpponen A, Trappe JM (1998b) Performance of Pinus contorta inoculated with two strains of root endophytic fungus, Phialocephala fortinii: effects of synthesis system and glucose concentration. Can J Bot 76:1205–1213. doi:10.1139/cjb-76-7-1205
Jumpponen A, Mattson KG, Trappe JM (1998) Mycorrhizal functioning of Phialocephala fortinii with Pinus contorta on glacier forefront soil: interactions with nitrogen and organic matter. Mycorrhiza 7:261–265. doi:10.1007/s005720050190
Kaldorf M, Renker C, Fladung M, Buscot F (2004) Characterization and spatial distribution of ectomycorrhizas colonizing aspen clones released in an experimental field. Mycorrhiza 14:295–306. doi:10.1007/s00572-003-0266-1
Kamminga-van Wijk C, Prins HBA (1989) The influence of pH on ectomycorrhizal development of Pseudotsuga menziesii inoculated with Laccaria bicolor in hydroculture. Agr Ecosys Environ 28:213–217. doi:10.1016/0167-8809(90)90043-d
Kernaghan G, Sigler L, Khasa D (2003) Mycorrhizal and root endophytic fungi of containerized Picea glauca seedlings assessed by rDNA sequence analysis. Microb Ecol 45:128–136. doi:10.1007/s00248-002-1024-1
Luo Y, Su B, Currie WS et al (2004) Progressive nitrogen limitation of ecosystem responses to rising atmospheric carbon dioxide. Bioscience 54:731–739. doi:10.1641/0006-3568(2004)054[0731:PNLOER]2.0.CO;2
Mandyam K, Jumpponen A (2005) Seeking the elusive function of the root-colonising dark septate endophytic fungi. Stud Mycol 53:173–189
Marx DH (1969) The influence of ectotrophic mycorrhizal fungi on the resistance of pine roots to pathogenic infections. I. Antogonism of mycorrhizal fungi to root pathogenic fungi and soil bacteria. Phytopathology 59:153–163
Menkis A, Allmer J, Vasiliauskas R, Lygis V, Stenlid J, Finlay R (2004) Ecology and molecular characterization of dark septate fungi from roots, living stems, coarse and fine woody debris. Mycol Res 108:965–973. doi:10.1017/S0953756204000668
Peterson RL, Massicotte HB, Melville LH (2004) Mycorrhizas: anatomy and cell biology. NRC Research Press, Ottawa
Peterson RL, Wagg C, Pautler M (2008) Associations between microfungal endophytes and roots: do structural features indicate function? Can J Bot 86:445–456. doi:10.1139/B08-016
Phillips RB (2007) Towards a rhizo-centric view of plant-microbial feedbacks under elevated atmospheric CO2. New Phytol 173:664–667. doi:10.1111/j.1469-8137.2007.02006.x
Piercey MM, Thormann MN, Currah RS (2002) Saprobic characteristics of three fungal taxa from ericalean roots and their association with the roots of Rhododendron groenlandicum and Picea mariana in culture. Mycorrhiza 12:175–180. doi:10.1007/s00572-002-0166-9
Postma JWM, Olsson PA, Falkengren-Grerup U (2007) Root colonisation by arbuscular mycorrhizal, fine endophytic and dark septate fungi across a pH gradient in acid beech forests. Soil Biol Biochem 39:400–408. doi:10.1016/j.soilbio.2006.08.007
Read DJ, Haselwandter K (1981) Observations on the mycorrhizal status of some alpine plant communities. New Phytol 88:341–352. doi:10.1111j.1469-8137.1981.tb01729.x
Reich PB, Hobbie SE, Lee T et al (2006) Nitrogen limitation constrains sustainability of ecosystems response to CO2. Nature 440:922–925. doi:10.1038/nature04486
Reuter DJ, Robinson JB, Dutkiewicz C (1997) Plant analysis: an interpretation manual. CSIRO, Collingwood
Rudawska M, Bernillon J, Gay G (1992) Indole compounds released by the ectendomycorrhizal fungal strain MrgX isolated from a pine nursery. Mycorrhiza 2:17–23. doi:10.1007/BF00206279
Ruotsalainen AL (2003) Mycorrhizal colonization and plant performance in arcto-alpine conditions. Ph.D thesis, Department of Biology, University of Oulu, Oulu, Finland
Schulz B, Boyle C (2005) The endophytic continuum. Mycol Res 109:661–686. doi:10.1017/S095375620500273X
Schulz B, Boyle C, Draeger S, Römmert A-K, Krohn K (2002) Endophytic fungi: a source of novel biologically active secondary metabolites. Mycol Res 106:996–1004. doi:10.1017/S0953756202006342
Sieber TN, Grünig CR (2006) Biodiversity of fungal root-endophyte communities and populations, in particular of the dark septate endophyte Phialocephala fortinii s.l. In: Schulz B, Boyle C, Sieber TN (eds) Microbial root endophytes. Soil biology, vol 9. Springer-Verlag, Berlin Heidelberg, pp 107–132
Stoyke G, Egger KN, Currah RS (1992) Characterization of sterile endophytic fungi from the mycorrhizae of subalpine plants. Can J Bot 70:2009–2016. doi:10.1139/b92-250
Summerbell RC (2005a) From Lamarckian fertilizers to fungal castles: recapturing the pre-1985 literature on endophytic and saprotrophic fungi associated with ectomycorrhizal root systems. Stud Mycol 53:191–256
Summerbell RC (2005b) Root endophyte and mycorrhizosphere fungi of black spruce, Picea mariana, in a boreal forest habitat: influence of site factors on fungal distributions. Stud Mycol 53:121–145
Vohník M, Lukančič S, Bahor E, Regvar M, Vosátka M, Vodnik D (2003) Inoculation of Rhododendron cv. Belle-Heller with two strains of Phialocephala fortinii in two differents substrates. Folia Geobot 38:191–200
Vohník M, Albrechtová J, Vosátka M (2005) The inoculation with Oidiodendron maius and Phialocephala fortinii alters phosphorus and nitrogen uptake, foliar C:N ratio and root biomass distribution in Rhododendron cv. Azurro. Symbiosis 40:87–96
Wang Z, Johnston PR, Takamatsu S, Spatafora JW, Hibbett DS (2006) Toward a phylogenetic classification of the Leotiomycetes based on rDNA data. Mycologia 98:1065–1075. doi:10.3852/mycologia.98.6.1065
Wilcox HE, Wang CJK (1987) Mycorrhizal and pathological associations of dematiaceous fungi in roots of 7-month-old tree seedlings. Can J For Res 17:884–899
Wu L, Guo S (2008) Interaction between an isolate of dark-septate fungi and its host plant Saussurea involucrata. Mycorrhiza 18:79–85. doi:10.1007/s00572-007-0159-9
Yu TEJ-C (2000) Characterization of the interaction between Phialocephala fortinii and two plant species, Asparagus officinalis and Lupinus latifolius. M.Sc. thesis, University of Guelph, Guelph, Ont
Acknowledgements
This study was funded by the C.T. de Wit Graduate School for Production Ecology and Resource Conservation (PE&RC) of Wageningen University, the Netherlands. We thank Antonie Gorissen and Glaciela Kaschuk for pre-submission review of the manuscript. Comments on an earlier version by anonymous referees are very gratefully acknowledged.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Erik A. Hobbie.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License (https://creativecommons.org/licenses/by-nc/2.0), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Alberton, O., Kuyper, T.W. & Summerbell, R.C. Dark septate root endophytic fungi increase growth of Scots pine seedlings under elevated CO2 through enhanced nitrogen use efficiency. Plant Soil 328, 459–470 (2010). https://doi.org/10.1007/s11104-009-0125-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-009-0125-8