Abstract
Arbuscular mycorrhizal symbiosis (AM) is a beneficial trait originating with the first land plants, which has subsequently been lost by species scattered throughout the radiation of plant diversity to the present day, including the model Arabidopsis thaliana. To explore if elements of this apparently beneficial trait are still present and could be reactivated we generated Arabidopsis plants expressing a constitutively active form of Interacting Protein of DMI3, a key transcription factor that enables AM within the Common Symbiosis Pathway, which was lost from Arabidopsis along with the AM host trait. We characterize the transcriptomic effect of expressing IPD3 in Arabidopsis with and without exposure to the AM fungus (AMF) Rhizophagus irregularis, and compare these results to the AM model Lotus japonicus and its ipd3 knockout mutant cyclops-4. Despite its long history as a non-AM species, restoring IPD3 in the form of its constitutively active DNA-binding domain to Arabidopsis altered expression of specific gene networks. Surprisingly, the effect of expressing IPD3 in Arabidopsis and knocking it out in Lotus was strongest in plants not exposed to AMF, which is revealed to be due to changes in IPD3 genotype causing a transcriptional state, which partially mimics AMF exposure in non-inoculated plants. Our results indicate that molecular connections to symbiosis machinery remain in place in this nonAM species, with implications for both basic science and the prospect of engineering this trait for agriculture.
Key message
Arabidopsis lost its ability to host mycorrhizal symbiosis over 65 million years ago. Restoring expression of an important mycorrhizae-related gene from a legume into Arabidopsis regulated genes for defense and stress.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Arbuscular mycorrhizae (AM) are formed during symbiosis between host plants and soil fungi. In AM, the plant provides carbon in the form of lipids and sugar to the fungus, and receives water and nutrients in return (Oldroyd 2013; Rich et al. 2017). AM can also confer resistance to pathogens and abiotic stress (Ceballos et al. 2013; Aliyu et al. 2019; Begum et al. 2019; Ramírez-Flores et al. 2020). AM are conserved in the majority of plant species from a single origin through the present day, and are thought to have aided in the first colonization of land (Wang et al. 2010; Delaux et al. 2013; Genre et al. 2020). However, multiple independent losses of the AM trait and its genetic machinery have occurred in diverse plant clades (Cosme et al. 2018). Why a trait considered to be beneficial has been so often lost is a puzzle important for not only basic understanding of AM symbiosis' role in plant resilience, but also for potential crop improvement through re-engineering the trait (Hornstein and Sederoff 2023).
Arabidopsis thaliana is one of the ~ 7% of plant species that lack the ability to form AM and which have not replaced them with an alternative endosymbiosis (Veiga et al. 2013; Brundrett 2017; Brundrett and Tedersoo 2018; Cosme et al. 2018; Radhakrishnan et al. 2020). Other such non-mycorrhizal (nonAM) plants include economically important species in the Brassicaceae and Amaranthaceae (Brundrett 2017). Proposed reasons for AM loss include changes in root morphology and lifestyle, altered insect and pathogen resistance, and the carbon cost of supporting the symbiont (Sikes 2010; Field et al. 2012; Brundrett 2017; Ma et al. 2018; Poveda et al. 2019). Little experimental validation exists for drivers of AM loss in individual species, let alone an overarching explanation for which species lose AM, why, and when. The proximate genetic causes for mycorrhizal loss, however, are quite clear: a shared subset of specific genes is deleted in all independent nonAM evolutions (Delaux et al. 2014; Radhakrishnan et al. 2020).
Gene losses in nonAM plants include the Common Symbiosis Pathway (CSP) that mediates signal perception and transduction (Cope et al. 2019; Radhakrishnan et al. 2020; Genre et al. 2020). Member genes lost in Arabidopsis include the cell-surface receptor SymRK, the ion channel DMI1, the kinase DMI3, and the transcription factor IPD3 (Delaux et al. 2014; Bravo et al. 2016; Radhakrishnan et al. 2020). These genes mediate a signal transduction pathway leading from AMF perception to activation of DMI3 by calcium (Demchenko et al. 2004; Pan et al. 2018; Feng et al. 2019). DMI3 then phosphorylates IPD3, enabling its DNA-binding activity for regulation of downstream targets (Oldroyd 2013; Pimprikar and Gutjahr 2018). Outside the CSP, groups of genes involved in lipid flux from plant to AMF and in vesicle trafficking to the arbuscule are also lost (Radhakrishnan et al. 2020).
IPD3 (Interacting Protein of DMI3) is notable among lost genes in nonAM plants. While most such genes belong to families with non-AM-specific homologs, IPD3 does not, suggesting its function is distinct to symbiosis. Genetic work has also demonstrated the powerful role of this gene in turning CSP signaling into regulation of AM response genes. The ipd3 knockout phenotype is near-complete elimination of mycorrhization (Watts-Williams and Cavagnaro 2015). Ectopic expression of IPD3 constitutively activated via phosphomimicking (IPD3S50D) or truncation to the DNA-binding domain (IPD3Min), however, results in symbiosis-like gene regulation even in the absence of a microbial signal or upstream CSP genes (Singh et al. 2014; Pimprikar et al. 2016).
Here, we express IPD3 in Arabidopsis based on knowledge of its uniquely powerful function in AM. By restoring expression of this gene that was present in the mycorrhizal ancestors of Arabidopsis before loss of AM in the Brassicaceae, we apply a novel means of identifying retained or unrecognized connections between AM and other genetic networks conserved in nonAM plants. Characterization of IPD3-expressing Arabidopsis targets two specific questions. First, does IPD3 have roles outside of its canonically narrow function in AM establishment? Second, does IPD3 retain functional molecular relationships when restored to Arabidopsis, which lost its ancestral copy of this gene along with the trait approximately 65 million years ago (Hohmann et al. 2015)? If so, could reconnecting such relationships alter the response to AMF or even restore symbiosis?
We use transcriptomics to understand the gene-regulatory impact of restoring IPD3 expression to Arabidopsis, and we also compare the inverse case of IPD3 knockout in a mycorrhizal host plant, via the cyclops-4 mutant of Lotus japonicus. While Lotus has the upstream CSP that normally activates IPD3 in the presence of AMF, the cyclops-4 mutant lacks functioning IPD3 and results in Lotus that fails to form mycorrhizae despite having the upstream CSP elements intact (Yano et al. 2008; Singh et al. 2014; Pimprikar et al. 2016). This Lotus mutant phenotypically mirrors wild type Arabidopsis in lacking AM, though Arabidopsis further lacks many other genes as described above (Delaux et al. 2013). We subjected both genotypes of both species to AMF treatment, thus, our experiment generates a factorial set of transcriptome data for every combination of IPD3 genotype, [non]symbiotic species background, and AMF exposure (Fig. 1). Because AMF cannot colonize or survive in long-term monoculture with nonAM plants, we use a pre-germinated spore treatment as described by Fernández et al. to study the early phase of plant-AMF interaction after 48 h of co-culture (Fernández et al. 2019). This corresponds to the pre-symbiotic phase of AM host plant-AMF interaction where significant CSP activity takes place but little colonization is established (Gutjahr and Parniske 2013).
Experimental design comparing different species and genotypes. A The AM-host Lotus japonicus wildtype (ecotype Gifu) and IPD3 knockout (cyclops-4; cyc4) genotypes differ in their ability to establish AM. Arabidopsis thaliana as a non-host species was engineered to express the DNA binding domain of IPD3 (OX-IPD3Min). B Phenotypic and transcriptomic analysis was carried out on the genotypes with or without AM inoculation and C under high and low macronutrient-containing media. Two independent transgenic lines of Arabidopsis were used for analysis in B and 1 transgenic line of Arabidopsis was used for analysis in C
We additionally examined a set of AMF-exposed Arabidopsis plants under low-macronutrient conditions (N, P, K) as a known regulator of symbiotic interactions in host plants (Bonneau et al. 2013; Nouri et al. 2014; Carbonnel and Gutjahr 2014). We perform correlation network and differential expression analyses to identify patterns in transcriptomic data from this experiment, and explore their functional implications using gene ontology (GO).
Materials and methods
Please see supplemental experimental procedures in Online Resource 1 for additional detail on all sections.
Generation of transgenic Arabidopsis
The coding sequences of MtIPD3, S50D-IPD3, and IPD3-Min (Genbank EF569224.1; Yano et al. 2008; Singh et al. 2014) were synthesized (Integrated DNA Technologies, Research Triangle Park, NC) and assembled by Hi-Fi assembly (New England Biolabs, Ipswich, MA) into a modified pCAMBIA0380 expression construct (Genbank AF234290.1) under control of the Arabidopsis Ubiquitin 10 promoter (Ivanov and Harrison 2014). 35S:mCherry amplified from pC-GW-mCherry (Genbank KP826771.1) (Dalal et al. 2015) was the selection marker. Arabidopsis were transformed as described by Davis et al. (2009). Seeds were screened by fluorescence and PCR during segregation and lines were brought to homozygosity.
Protein analysis
Protein was extracted from roots and shoots of 6-week-old seedlings grown on 1/2MS plates. For untargeted shotgun proteomics, 5 samples per line and tissue type were ground and lysed in SDT buffer, then prepared as described in Wiśniewski et al. (2009) before 1D-LC-MS/MS using an UltiMateTM 3000 RSLCnano Liquid Chromatograph and Orbitrap Eclipse Tribrid mass spectrometer (Thermo Fisher) as described in Mordant and Kleiner (2021). Mass spectra were searched against a database of Col-0 A. thaliana proteins (Uniprot:UP000006548) using the SEQUEST HT algorithm in Proteome Discoverer. Protein abundance was quantified as normalized spectral abundance factor (NSAF) in Microsoft Excel (Zybailov et al. 2006).
For analysis of specific protein size ranges corresponding to bands observed on blots or predicted protein length, we used in-gel digestion of excised SDS-PAGE prior to LC-MS/MS analysis (GeLC-MS/MS). Only 1 sample per construct was used for GeLC-MS/MS. 30–40 µg of total protein in lysate generated for untargeted proteomics was denatured by heating with Laemmli buffer, then run on an SDS-PAGE 12% separating gel with 5% stacking gel. Excised bands were processed according to Shevchenko et al. (2006), and 10 μL of peptide mixture was injected to LC as described above, connected to an Orbitrap Exploris 480 mass spectrometer (Thermo-Fisher) for MS/MS with the same settings used for untargeted proteomics.
For Western blotting, frozen tissue was hand-ground with extraction buffer, centrifuged, and the supernatant denatured by heating in LDS sample buffer. Protein was run on a 12% Tris-Glycine acrylamide gel, then transferred to PVDF membranes and blocked in TBS+2% BSA with 4 μL/mL Tween-20, then incubated with 1:1000 dilution of custom rabbit anti-IPD3 polyclonal antibody (Genscript, China) followed by 1:2500 donkey anti-rabbit AlexaFluor 488-conjugated secondary antibodies (Thermo Fisher, Waltham, MA) and imaged on a GelDoc SR (Bio-Rad, Hercules, CA).
Growth phenotyping
Plants were grown in the NCSU phytotron under long day conditions in 8 oz pots filled with SunGro propagation mix (Sungro, Agawam, MA). Pots were hand-watered with deionized water. Seeds were harvested from dried mature plants and hand-cleaned before weighing. The seed yield experiment was repeated 3 times with n = 9–15. The flowering time experiment was performed once with n = 14 for line 312 and n = 15 for all other lines, with plants censused daily for onset of bolting.
Transcriptome experiment
Seeds were sterilized and grown for 6 weeks under long-day conditions (16 h light/8 h dark, 21/18 ℃) on sterile petri dishes containing either 1/2MS or low-nutrient MS media (Phytotech Labs, Lenexa, KS). Following the protocols established by Fernández et al. (2019) and Mukherjee and Ané (2011), sterile spores of R. irregularis (Premier Tech, Canada) were pre-germinated for 1 week at 26 ℃, and a suspension of ~ 200 germinated spores was added to the roots of each AMF-treated plant 48 hours prior to sample collection. Control plants were mock-inoculated with sterile water. Each replicate consisted of the pooled roots of 5 seedlings from the same plate.
RNA was extracted with the Purelink RNA Mini kit and treated with Turbo DNAse (Invitrogen, Waltham, MA), then sequenced by DNBSEQ at BGI Group (China) using strand-specific, poly-A enrichment to obtain 100 bp paired-end reads. Lotus and Arabidopsis alignments were performed using BBSplit, a multi-reference aligner, to separate plant and fungal reads (Bushnell 2014). Reference genomes used were Araport 11 for Arabidopsis (TAIR), the Joint Genomics Institute assembly for R. irregularis (Genbank: GCA_000439145.3), the Gifu V1.2 assembly for Lotus (GCA_012489685.2), the IPD3-Min transformation construct, and the LjCYCLOPS sequence. Reads were counted at the transcript level using featureCounts (Liao et al. 2014).
Gene expression networks were constructed using WGCNA v1.69 (Langfelder and Horvath 2008, 2012) with soft-threshold power of 16 (Arabidopsis) or 24 (Lotus). Correlation coefficients were calculated between the eigengenes of each module and treatment variables to identify significant module-trait relationships, with p < 0.05. Gene Ontology enrichment and hierarchical term clustering was performed with PANTHER (Mi et al. 2013). Terms were filtered by specificity and significance, then subjected to semantic similarity clustering in reviGO (Supek et al. 2011).
Differential expression analysis was performed in R using the edgeR package (Liao et al. 2014; Robinson et al. 2010). The estimateGLMCommonDisp function with FDR-adjusted p-value < 0.05 was used to test for differential expression. Cross-comparison of gene lists was done in Excel.
Results
Generation of transgenic Arabidopsis
The transcription factor IPD3 consists of an activation domain that can be phosphorylated on Serine-50 upon AM inoculation and a DNA-binding domain essential for transcriptional regulation of downstream effectors (Singh et al. 2014; Jin et al. 2018). To ensure that we can identify transcriptional regulation via IPD3 in the non-host that is likely lacking signaling and activation of IPD3, we generated 7 homozygous transgenic Arabidopsis lines expressing Medicago truncatula IPD3 (IPD3Mt), 4 lines of phosphomimic IPD3 (IPD3S50D), and 5 lines expressing IPD3Min (aa 254-513 of IPD3Mt) (Singh et al. 2014). All transgenes were expressed under the Arabidopsis UBIQUITIN10 promoter (Grefen et al. 2010; Ivanov and Harrison 2014). RT-PCR confirmed RNA expression of all versions of the transgene.
Western blot of root and shoot tissue of T3 individuals confirmed presence of IPD3Min protein, but not IPD3Mt and IPD3S50D (Online Resource 2). We noted that Western blot visualization was difficult to obtain, requiring the precise method described in Online Resource 2, and that root and shoot IPD3Min protein bands appear to run at slightly different size, which might represent influence of unknown post-translational modification or cellular localization that interferes with expression and running conditions. We therefore performed targeted protein sequencing using mass spectrometry to confirm that the protein bands observed on the blot were in fact IPD3Min. Excised SDS-PAGE gel bands at the expected size of IPD3Min in shoot and root samples were used for shotgun proteomics (Online Resources 1 and 3). IPD3Mt and IPD3S50D lines were analyzed in the same manner, however, and as with Western blot of these lines, protein expression was again not detected. To further confirm that our ability to detect and identify all forms of IPD3 was not affected by gel running conditions or extraction, we then performed untargeted shotgun proteomics of shoots and roots (n = 5) using independent processing methods (Online Resource 1). In untargeted proteomic analysis, IPD3Mt and IPD3S50D were again not detected, while IPD3Min was highly abundant (top 10% NSAF) in respective lines (Online Resource 3). Consequently, we focused only on characterizing the 5 IPD3Min lines (numbered 303; 308; 310; 312; 357).
In IPD3Min lines used we confirmed that protein and RNA of the gene of interest were highly expressed relative to native sequences (Online Resources 3 and 15), however, these quantitative data do not provide a direct measurement of transcription factor activity of the protein product.
Phenotypic effect of IPD3 Min expression in Arabidopsis
Plants were surveyed for differences in growth under long-day conditions (Fig. 2, Online Resource 4). All 5 transgenic IPD3Min lines were significantly slower to initiate the transition to flowering than wild type Col-0, measured as days to onset of bolting (Fig. 2A, B, Online Resource 5). Four of 5 lines (excluding 303) were also significantly slower to flower than a null segregant control (Fig. 2B). Despite flowering differences, no significant difference in seed yield was detected over 3 repetitions of the experiment (Fig. 2C). No difference in germination timing was observed (Online Resource 5). An unexpected strong phenotype was the pink coloration of four of the five IPD3Min transgenic plant roots, while this color in IPD3Mt and IPD3S50D lines was very weak in 4 out of the 11 plants, and visually absent in the remaining 7 (Online Resource 6). In addition, 7 of the 8 independent T1 empty vector lines showed a range of pink coloration in roots. Because anthocyanins or carotenoids can cause this coloration in plant tissues, we performed extraction protocols for those secondary metabolites, but were not able to confirm any measurable quantities. The most likely conclusion is that the color could be caused by the marker mCherry that was used as a reporter gene. This reporter gene was present in the IPD3Mt and IPD3S50D constructs as well, where little to no pink coloration was observed, which may relate to the lack of expression and potential silencing of those transgenes as assessed through proteomics (Online Resources 2, 3).
Phenotypes of independent homozygous transgenic lines expressing the DNA-binding domain of IPD3 (IPD3Min). A Shoots of most IPD3Min transgenic lines appear shorter, less branched and delayed in development; scale bar 10 cm. B IPD3Min Transgenic lines (blue) had a delayed transition to reproductive development (onset of bolting) relative to nontransgenic controls (orange) (ANOVA, p < 0.05, n = 15), but C showed no significant differences in total seed yield (MCM p < 0.05) (in B and C mean is marked with X and median marked with –, outliers shown as dots) (see Online Resources 4–6)
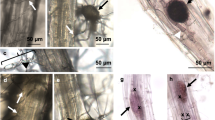
We also monitored Lotus japonicus plants used in cross-species comparison for overt differences between wild type (Gifu ecotype) and the cyclops-4 knockout mutant of IPD3. Consistent with prior literature we noted no obvious growth phenotype in the ipd3 mutant (Yano et al. 2008; Horváth et al. 2011) (Online Resource 7). The genotype of cyclops-4 Lotus was confirmed by Sanger sequencing of the CYCLOPS genomic region.
Transcriptional consequences of manipulating IPD3
Before a functional AM host trait could be engineered into Arabidopsis as a reference plant for Brassicaceae (Hornstein and Sederoff 2023), the potential for evolutionarily conserved interactions and pathways can be explored via comparative transcriptome analysis of non-host and host plants with and without IPD3/IPD3Min. We sequenced total mRNA of 6-week-old roots of transgenic IPD3Min, mutant cyclops-4, and wild type Arabidopsis Col-0 & L. japonicus Gifu plants with or without 48 hours of AMF germinated spore treatment prior to collection (Fig. 1). Two independent transgenic lines of Arabidopsis (308 and 310) with similar phenotype (Fig. 2, Online resources 4, 6) and IPD3Min transcript abundance were included in the experiment to account for possible variation due to genomic insertion site effects and epigenetic consequences in transgenic lines (Schnell et al. 2015).
We used statistical methods like Principle Component Analysis (PCA) and Differential Gene Expression as well as machine learning for network analysis to identify genes, pathways and networks in Arabidopsis and Lotus plants that are responsive to AMF inoculation and dependent or independent of IPD3 regulation. The comparison of the species and genotype specific transcriptome analyses provides an insight into those pathways that are still conserved in Arabidopsis after the evolutionary loss of the AM host trait through IPD3 loss. Correlation network construction used quantitative expression of IPD3Min in each sample of both lines as the primary transgenic trait. In differential expression analysis an IPD3Min category including only genes independently attested as DEGs in both transgenic lines was used. We also compared Col-0 Arabidopsis to a single IPD3Min transgenic line (308) with AMF inoculation on low-nutrient (LN) MS medium containing 0.5% P, 1% N, and 1% K relative to 1/2 MS (Fig. 1).
In principal component analysis (PCA) of Lotus, Gifu transcriptomes are clustered according to AMF treatment along PC1 (Online Resource 8). cyclops-4 transcriptomes were separated from those of Gifu plants along PC2. Interestingly, both AMF-treated and untreated cyclops-4 transcriptomes clustered along PC1 with those of AMF-treated Gifu plants. In Arabidopsis, IPD3Min transgenic lines clustered relative to Col-0 along PC1 (Online Resource 8). Line 308 clustered farther from Col-0 than line 310, most likely due to the higher IPD3Min expression in individuals of this line (Online Resources 8, 9). Col-0 plants clustered by AMF treatment along PC2, but there was no clear separation by treatment among IPD3Min lines (Online Resource 8). PCA of the low-nutrient experiment also shows strong clustering by genotype (Online Resource 10). In conclusion, the variance in gene expression changes in wild type Lotus and Arabidopsis plants corresponds more to the inoculation with AMF, while gene expression variation in response to presence or absence of IPD3/IPD3Min is less affected by presence or absence of AMF and this variance is explained by a different component.
Correlation network and gene ontology analysis
We used weighted gene co-expression network analysis (WGCNA) as a transcriptome analysis tool which captures global expression patterns reflective of the underlying reality of coordinated expression of many genes affected by multiple factors (Langfelder and Horvath 2008, 2012). WGCNA clusters the transcriptome into modules with similar expression profiles which suggest they may regulate each other or are co-regulated. WGCNA also correlates expression of a representative eigengene for each module to traits and treatments, in this case quantitative IPD3Min expression, AMF treatment, and nutrient level. The two-step process of module construction and module-trait correlation makes WGCNA sensitive to links between potentially subtle expression changes at the gene level and experimental variables, and can be used to guide more targeted analysis. WGCNA analysis incorporates transgene expression agnostic of categorical information about the 2 transgenic lines, instead reflecting how well expression of native Arabidopsis genes correlate to IPD3Min across a range of variation in transgene expression levels in each individual plant. WGCNA of Arabidopsis identified 22 co-regulatory modules (Online Resources 11, 12). Figure 3A includes all modules significantly correlated to IPD3Min expression, and selected modules correlated to AMF treatment and nutrient level. We used gene ontology (GO) enrichment to link module membership to biological functions (Fig. 3B). We also performed targeted pathway analysis of specific gene members within modules to better connect these broad patterns to physiological functions (Fig. 4). Statistical significance in Fig. 4 is a hypothesis test of whether mean expression of the given gene differs between specific treatment groups, an independently calculated and complementary measure to the network analysis correlations which establish module membership.
Correlation network modules and gene ontology analysis for transcription of Arabidopsis thaliana IPD3Min and Col-0 genotypes across AMF and low-nutrient treatments. A Selected correlation modules, including all those significantly correlated to IPD3Min expression. Modules are labeled E–T in order of appearance in the full network (Online Resource 7). Color of cells within the heatmap reflects correlation of that module to trait values; p-value of the module-trait correlation is listed in parentheses. B Gene Ontology enrichment of all modules shown in A; terms have been reduced by applying a cutoff for the top 3 most-enriched and most-significant terms in each module followed by overlap analysis to establish representative terms. Color intensity corresponds to FDR-corrected p-value of term enrichment and circle area corresponds to scale of enrichment. (See Online Resource 11, Online Resources 12, 14, S10)
Targeted analysis for pathways of interest within correlation network modules. All genes included in this figure are statistically significant members of the respective network module (having module-trait correlations at p_GS < 0.05 as indicated in Fig. 3) at p_kME < 0.05. Significance reflects 1-tailed t-test of group means for transcript counts of specific genes, a complementary measure which is calculated independently from both the correlation network and untargeted DEG analysis; n = 3–5, *p < 0.05; **p < 0.01; ***p < 0.005. Standard Error of all treatment groups is provided in Online Resource 15 in addition to t-test results. Some interactions in pathway maps have been omitted for clarity. A Expression of a subset of individual genes belonging to module O related to extracellular wax synthesis. B Expression of well-known circadian clock related genes in module T. C Pathway map of the WRKY70-SA-NPR1-mediated defense response with expression of individual genes. Genes belong to module S unless marked otherwise in parentheses. (See Online Resources 12, 15). Gene acronyms are expanded as follows: A LACS2, Long-chain Acyl-CoA Synthetase 2; KCS1, 3-Ketoacyl-CoA Synthase 1; CYP86B1, Cytochrome P450, Family 86, Subfamily B, Polypetide 1; GPAT5, Glycerol-3-phosphate sn-2-Acyltransferase 5; ABCG20, ATP-binding Cassette G20. B FKF1, Flavin-binding, Kelch Repeat, F Box 1; ELF3, Early Flowering 3; APRR1, Pseudo-response Regulator 1; Spatula; Gigantea. C BDA1, Bian Da 1; WRKY70, WRKY DNA-binding Protein 70; WRKY54, WRKY DNA-binding Protein 54; SARD1, SAR Deficient 1; NPR1, Nonexpressor of PR Genes 1; lnc_NPR1, antisense long non-coding RNA of NPR1; NIMIN1, NIM1-interacting 1; PR5, Pathogenesis-related Gene 5; PR2, Pathogenesis-related Gene 2
The WGCNA analysis identified clusters of co-regulated transcripts that were either responsive to the nutrient level, the exposure to AMF, the expression of IPD3Min, or combinations of those treatments. Further GOterm analysis of those specific clusters showed the potential functional connections of genes that were differentially co-regulated in response to either treatment of combinations of those. Only one cluster of transcripts (cluster O) significantly correlated only with AMF inoculation in Arabidopsis, and three clusters (E, F, H) significantly correlated only with IPD3Min expression. One cluster (M) showed positive correlation with AMF inoculation but negative correlation with IPD3Min expression suggesting repression of those genes by IPD3Min.
The Arabidopsis response to AMF includes altered defense and lipid metabolism
Despite its nonAM status, Arabidopsis transcriptome networks showed 10 modules of co-regulated genes with trait correlations to AMF treatment, indicating a clear transcriptional response to the fungus (Online Resource 11). We considered modules O and R that correlate to AMF treatment but not IPD3Min expression to reflect portions of the native Arabidopsis AMF response that are unaffected by IPD3Min expression. Expression of genes in module R positively correlated with AMF treatment but negatively correlated with nutrient level. Top GO terms in this module R related to both nutrient stress (GO:0016036; phosphate starvation) and biotic interactions (GO:0010200; chitin response) (Fig. 4B, Online Resources 13, 14).
Module O expression correlated solely to AMF treatment, independent of IPD3Min genotype and was highly enriched for terms related to biosynthesis of extracellular lipids (GO:0010143; cutin biosynthesis, GO:0010345; suberin biosynthesis, GO:0042761; very long-chain fatty acid biosynthesis) (Fig. 3B). These genes fall along the biosynthetic pathway of 2-monoacylglycerols (2-MAGs), which affect pathogen and stress resistance as components of cutin and suberin (Fig. 4A, Online Resources 12, 14). 2-MAGs are also the primary form of carbon exported to AMF fungus from AM host plants (Luginbuehl et al. 2017; Rich et al. 2017). Mean expression of all tested genes in the pathway was significantly higher in AMF-treated Col-0 plants than in untreated plants (Fig. 4A). Significantly higher expression was also detected in some but not all AMF-treated transgenic lines for the same genes. This indicates that upregulation of lipid biosynthetic genes in AMF-treated Arabidopsis is IPD3Min-independent, consistent with the module’s correlation to AMF treatment but not IPD3Min expression in Fig. 3A. The gene level comparisons of transcript abundance for this module shows that both independent transgenic lines (308 and 310) have similar transcript abundance differences to Col-0 and are comparable in their response prior to AMF treatment (Fig. 4B, C). Other genes in module O also function in lipid synthesis, including GPAT6, a functional homolog of RAM2 which is strictly required for AM in host species (Online Resource 12) (Gobbato et al. 2013; Dai et al. 2022). While it is a member of module O, AMF-mediated increase of GPAT6 gene expression was only significant in the IPD3Min line 308, but not in Col-0 nor in the IPD3Min line 310 (Online Resource 15).
IPD3 Min expression imitates AMF treatment in Arabidopsis Col-0 plants
Transcription in module T correlated negatively to AMF treatment and IPD3Min expression (Fig. 3A). This is consistent with IPD3Min expression enhancing the effect of AMF treatment or replicating the effect of AMF in untreated samples. A similar pattern was present in modules N (both positive) and S (both negative) (Fig. 3A). Module T contains only 56 genes and was enriched solely for terms related to circadian rhythm (GO:0007623; circadian rhythm, GO:0048511; rhythmic process) (Fig. 3B, Online Resource 14). Members of this module include components of the CONSTANS-FT daylength sensing system (Online Resources 12, 15) (Takagi et al. 2023). As shown in Fig. 4B, AMF treatment of Col-0 plants results in significantly reduced expression for genes across the pathway, and IPD3Min expression replicates this effect even in the absence of AMF.
Module S transcription was negatively correlated with both IPD3Min and AMF treatment, and is enriched for GO terms related to pathogen defense and systemic acquired resistance (SAR) (GO:0002229, oomycete defense; GO:0002237, bacterial molecule response; GO:0009627, systemic acquired resistance) (Fig. 3). This module contains the key defense regulator WRKY70, which mediates selection of defense responses by positively regulating salicylic acid (SA) immunity and negatively regulating jasmonic acid (JA) related pathways (Li et al. 2006). As shown in Fig. 4C, genes acting up- and downstream of WRKY70 in SA-mediated defense are also present in this module, including cell-surface ankyrin protein BDA1, and pathogen response gene PR2 (Thomma et al. 2001; Yang et al. 2012a). NPR1, another key positive regulator of SA-mediated defense was detected in the transcriptome but not placed in a network module (Fig. 4C) (Thomma et al. 2001).
Several SA defense genes (BDA1; WRKY70; WRKY54; NIMIN1; PR5) were significantly downregulated in one or both transgenic lines even in the absence of biotic interaction with AMF (Fig. 4C). This suggests that IPD3Min expression in isolation can replicate the suppressive effect of AMF interaction on SA-related defense. Consistent with downregulation of SA-related genes, we also found evidence of antagonistic crosstalk between jasmonic and salicylic acid-mediated defenses (Li et al. 2006; Hou and Tsuda 2022). Module R, which in contrast to Module S was upregulated in response to AMF, is functionally enriched for jasmonic acid (JA) signaling (GO:009753) (Online Resource 14). JA genes upregulated by AMF in module R included upstream (jasmonate methlytransferase JMT) and downstream (chitinases PR3 and PR4) members of the JA defense pathway (Samac et al. 1990; Thomma et al. 1998; Seo et al. 2001). Module R also contained a set of genes enriched for salicylic acid signaling, some of which are negative regulators of SA defense such as At1g08667.1 (lnc_NPR1), a putative antisense transcript of NPR1 whose upregulation would correspond to downregulation of NPR1 and the SA response (Fig. 4C, Online Resource 15). Other SA-related genes were placed into module R due to expression correlation at the individual sample level, but upon comparison of treatment group means matched the pattern of downregulation in module S; this included WRKY54, a co-regulator with WRKY70 of SA synthesis (Chen et al. 2021b) (Fig. 4C, Online Resource 15).
Positive IPD3Min correlation with modules F, G, H, and N corresponded to GO enrichment for background processes, e.g. GO:0000466, rRNA maturation (Fig. 3). These functions are essential to the organism, but limited in GO analysis from being linked to specific effects. These modules contained 4132 genes, indicating that about 10% of all gene models in the Arabidopsis genome were upregulated in connection to IPD3Min (Online Resource 12). Module E had the strongest correlation to IPD3Min expression but resulted in no significant GO term enrichment (Fig. 3A, Online Resources 12, 14). Notably, 8 of 25 genes subsequently identified as IPD3-responsive via differential transcription analysis in Fig. 6B belonged to module E and are discussed in later sections (Online Resource 16).
Network analysis of Lotus transcriptomes
We also constructed a correlation network for Lotus, which is available in Online Resources 17 and 18. A distinctive feature of the Lotus network is that while expression of 8 modules correlated with IPD3 genotypes and 15 modules correlated with the AMF treatment, no modules correlated with both treatments. We confirmed transcription of CSP and AMF marker genes including CCaMK, NSP2, and PT4, however, all detectable CSP genes were placed into network modules not correlated to either treatment.
Cross-species comparison of differential transcription
The major question of our experiment was whether elements of the AMF response network are still present in the non-host Arabidopsis after the evolutionary loss of the transcription factor IPD3. To directly characterize transcript abundance changes related to the presence or absence of the respective IPD3/IPD3Min versions in a manner that enables cross-comparison of species, we conducted differential transcript abundance analysis for genotype and AMF treatment in Lotus and Arabidopsis.
The question to answer in this cross-species comparison was which genes are IPD3-dependent in their response to AMF inoculation in the host plant (Lotus) and then compare it to the non-host Arabidopsis (Fig. 5A, Online Resource #19) with the goal to identify genes that still can be regulated by IPD3Min. Transgenic expression used to create the +IPD3Min genotype in Arabidopsis introduces two additional factors which make this genotype only partially parallel to the Lotus wild type. First, the transgene is constitutively expressed under the Ubiquitin promoter and constitutively active at the protein level due to truncation of the activation domain, as opposed to inducible as in Lotus (Pimprikar et al. 2016; Singh et al. 2014). Second, Agrobacterium mediated transformation randomly inserts the transgene into different genomic locations in different independent lines, resulting in differing expression levels and potential effects on native genes (Edwards et al. 2022). To limit influence of such across-line variation, in the primary analysis we considered only those Arabidopsis DEGs independently attested for a given contrast in both transgenic lines (Fig. 5). Contrasts of individual transgenic lines 308 and 310 vs. Col-0 are provided in Online Resource 16. In the −ipd3 genotype of Lotus (cyclops-4), AMF exposure induced only 15 DEGs (Fig. 5A contrast A), while AMF treatment of +IPD3 Lotus induced 461 DEGs (Fig. 5A, contrast B). This is consistent with prior knowledge that AM symbiosis is strongly IPD3-dependent (Yano et al. 2008). In −ipd3 Col-0 Arabidopsis, however, AMF regulated 497 DEGs (Fig. 5A contrast C), while AMF treatment of +IPD3Min Arabidopsis regulated abundance of only 3 transcripts (contrast D). In -AMF Lotus the presence of IPD3 in +IPD3 Gifu produced 338 DEGs relative to −ipd3 cyclops-4 (Fig. 5A contrast E), while the equivalent contrast in Arabidopsis produced 25 DEGs (contrast F). Contrast of the two genotypes under AMF treatment resulted in 38 DEGs for Lotus and 77 DEGs for Arabidopsis (Fig. 5A, contrasts G and H). This comparison showed that regulation of only 15 transcripts in Lotus were independent of IPD3 when Lotus roots were challenged with AMF in contrast to 497 genes that did not require IPD3Min in Arabidopsis to be regulated in response to AMF inoculation. When we interpret these DEGs for the two Arabidopsis genotypes, it is important to keep in mind that the IPD3Min protein is constitutively active and the transgene was controlled by the constitutive UBI10 promoter and therefore both highly expressed and active prior to the AMF-inoculation, unlike in Lotus. If IPD3-responsive genes were already regulated by constitutive IPD3Min expression in Arabidopsis, further activation upon AMF exposure would not be expected.
Differential transcription contrasts for IPD3[Min] expression with and without AMF treatment in two species. A Overview of all contrasts executed and number of DEGs detected. Contrast labels A–H in B and C and Fig. 6 correspond to those noted in A. DEGs reported for the +IPD3Min genotype in Arabidopsis are those attested in both of the individually conducted contrasts for the 2 transgenic lines 308 and 310. B Comparison of DEGs resulting from AMF treatment in −ipd3 (cyc4) and +IPD3 (Gifu) genotypes of Lotus. L2FC is Log2(fold change) of mean expression. C Comparison of DEGs resulting from AMF treatment in −ipd3 (Col-0) and +IPD3 (OX-IPD3Min) genotypes of Arabidopsis. Differential transcription is expressed as log2(fold change) (L2FC); more-positive numbers indicate higher expression and more-negative numbers indicate lower. (See Online Resource 16)
Cross-species comparison of AMF and IPD3[Min] responsive DEGs. A When the AMF-responsive DEGs within the respective +IPD3 and −ipd3 genotypes of Lotus and Arabidopsis are directly compared using annotated Arabidopsis homologs of Lotus genes, OBP4 is shared by −ipd3 Col-0 Arabidopsis with both Lotus genotypes, but oppositely regulated across species. Thirteen additional genes are shared by −ipd3 Col-0 Arabidopsis and +IPD3 Gifu Lotus, also with opposite regulation across species. B When the IPD3-responsive DEGs in non-AMF-treated Lotus and Arabidopsis are compared, LEA4-5 and His1-3 are shared across species, with LEA4-5 being oppositely regulated. C When IPD3-responsive DEGs in non-AMF-treated Arabidopsis are compared to AMF-responsive DEGs in −ipd3 Col-0 Arabidopsis, there is significant overlap. Eleven of 25 DEGs induced by IPD3Min in both transgenic lines 308 and 310 are also induced by AMF in −ipd3 Col-0. 103 out of 497 AMF-responsive DEGs (21%) in −ipd3 Col-0 are also responsive to IPD3Min in transgenic line 308, which has higher IPD3Min expression and a larger overall DEG set. (See Online Resource 18)
Next, we asked if the genes that remained responsive to AMF in −ipd3 Lotus would be conserved in Arabidopsis. To detect candidate genes that are part of the Lotus AMF host response but independent of IPD3, we looked for overlap between AMF-responsive DEGs for both Lotus genotypes (Fig. 5B). Only 3 DEGs were shared: Lj1g0209300.1 (homolog of OBF Binding Protein 4 (LjOBP4)); Lj2g0329400.1 (homolog of Cinnamyl Alcohol Dehydrogenase 8 (LjCAD8)); and Lj5g0253700.1 (homolog of Binding to TOMV RNA 1 (LjBTR1)). All of these genes were regulated in the same direction and at similar scale in both Lotus genotypes, suggesting they are elements of the native Lotus AMF response that are governed by an IPD3-independent mechanism. Despite IPD3's effect on the scale of AMF response in Lotus, IPD3Min expression in Arabidopsis did not enable any new AMF-responsive DEGs, and the scale of response to AMF was much larger in the −ipd3 Col-0 genotype than in IPD3Min (Fig. 5C).
We then looked for overlaps in DEGs for AMF response between both genotypes of both species (Fig. 6). For direct cross-species comparisons Lotus genes were labeled with their closest Arabidopsis homologs as recorded in genome annotations; total DEG count for AMF response of Gifu was slightly reduced due to exclusion of 33 genes without an annotated Arabidopsis homolog (Online Resource 19). One of the 3 IPD3-independent genes characterized in Fig. 5B as a member of the IPD3-independent Lotus AMF response, OBP4 (Lj1g0209300; At1g68050), was also shared with the AMF response of Col-0 Arabidopsis (Fig. 6A). OBP4 is a transcription factor that negatively regulates lateral root and root hair development in response to nitrate and abscisic acid (Ramirez‐Parra et al. 2017; Rymen et al. 2017; Xu and Cai 2019). Interestingly, OBP4 was upregulated in the AMF response of Col-0 Arabidopsis, but downregulated in Lotus. OBP4 was not differentially regulated in IPD3Min plants.
In addition to OBP4, 13 genes were differentially regulated in response to AMF in Gifu Lotus and Col-0 Arabidopsis, but not cyclops-4 Lotus or IPD3Min Arabidopsis (Fig. 6A, Online Resource 19). In most of these genes, as for OBP4, the direction of regulation was reversed between species, including the next two most strongly regulated, multi-stress-responsive Blue Copper Binding Protein and pathogen response-related WRKY22 (Fig. 6A). Consistent with defense effects of AMF observed in the network analysis, WRKY22, which was downregulated in Arabidopsis, is implicated in systemic acquired resistance and is characterized as a positive regulator of the SA pathway in SA-JA defense crosstalk (Kloth et al. 2016).
We also compared the effect of IPD3 presence and absence across the two species in the absence of AMF treatment. Two genes differentially transcribed in +IPD3 Lotus relative to −ipd3 Lotus were also differentially transcribed in +IPD3Min Arabidopsis relative to −ipd3 (Fig. 6B). Of these, osmotic stress responsive Late Embryogenesis Abundant 4–5 was strongly downregulated by the presence of IPD3 in Lotus but upregulated by the presence of IPD3Min in Arabidopsis, while drought stress responsive Histone H1-3 was downregulated in both species by the presence of respective IPD3 versions.
Beyond specific genes, a shared and unexpected feature of the +IPD3/−ipd3 contrast in both species was a pattern of many differentially expressed genes resulting from manipulation of IPD3 even in the absence of AMF (Figs. 5A, 6B). Many of the affected genes are related to biotic and abiotic stress. These included strong downregulation in Lotus of Lj4g0393400.1 and Lj1g0155200_LC.1, both of which are homologs of AtFRS5, a gene involved in linking light perception to stress and pathogen defense via JA, and upregulation of Lj4g0167200.1, a homolog of the AtJASSY JA biosynthesis enzyme (Ma and Li 2018; Guan et al. 2019). The top most strongly regulated gene that was dependent on the presence of IPD3 in Lotus was Lj1g0431600.1; its homolog Lj1g0070100.1 was also upregulated, with an average L2FC of 7.14. These genes are homologous to an uncharacterized MYB/SANT transcription factor in Arabidopsis, At2g24960.2. In Arabidopsis, IPD3Min expression also resulted in strong upregulation of genes related to JA pathogen response and auxin signaling including Cytochrome P450 76C2 and Tryptophan Aminotransferase Related 1 (Fig. 6B) (Lorenzo et al. 2003; Stepanova et al. 2008). Notably, both of these genes were included in module E of the network analysis, which had the strongest correlation to quantitative IPD3Min expression (Online Resource 16).
While the differential response to AMF was nearly eliminated in −ipd3 Lotus, 337 Differentially Expressed Genes (DEGs) respond to IPD3 in the contrast of +IPD3 and −ipd3 Lotus without AMF inoculation (Fig. 5A). 135 of these DEGs resulting from knockout of IPD3 in the absence of AMF treatment, were also found among DEGs resulting from AMF treatment of Gifu plants, accounting for 29% of the wildtype Lotus AMF response (Online Resource 19). Thus, although AMF-responsive upregulation of genes by IPD3 is known to be essential for normal AM function, its deletion also appears to partially replicate the AM response under the conditions of this experiment. This suggests that IPD3 in Lotus may act as a context-dependent transcriptional repressor of a portion of its own targets.
A similar pattern is present in Arabidopsis, where 11 of 25 genes (44%) differentially expressed in response to IPD3Min in the absence of AMF treatment were also induced by AMF treatment of Col-0 plants (Fig. 6C). These genes included, for example, the FKF1 circadian clock gene identified in Fig. 5C as well as in the IPD3-Min responsive circadian clock module of the correlation network (Figs. 4, 5) (Online Resource 19). In transgenic line 308 where higher IPD3Min expression corresponds to a larger absolute number of DEGs (Fig. 5A, Online Resource 9), this effect is much larger. The no-AMF contrast of IPD3Min-308 vs Col-0 recapitulates differential regulation of 103 of the 497 (21%) AMF-responsive DEGs in Col-0 (Fig. 6C). These results confirm evidence in the correlation network that effects of IPD3Min expression in Arabidopsis overlap partially with the AMF response. They indicate that while the nature of the response may differ, expression of constitutively activated IPD3 in Arabidopsis can activate the transcriptional response to AMF even in the symbiont's absence, much as it does in AM host plants (Singh et al. 2014).
While the detailed comparisons of DEGs provide interesting insight into those genes and pathways that are apparently dependent on IPD3 activation in Lotus for a response to AMF challenge, the conservation of genes regulated by the constitutive expression of the DNA binding domain of IPD3 (IPD3Min) in Arabidopsis shows that there is still a set of genes regulated by this domain. While we used a conservative approach by only considering those DEGs that were regulated in both transgenic lines, the differential gene expression in the respective individual transgenic lines by IPD3Min was much larger and can provide targets for further studies.
Discussion
We successfully expressed IPD3Min, the DNA binding domain of the AM symbiosis-essential transcription factor IPD3, in the nonAM host plant Arabidopsis which lost both the AM trait and IPD3 about 60–70 million years ago (Hohmann et al. 2015; Radhakrishnan et al. 2020). To understand the impact of IPD3Min on remaining genetic pathways and biotic interactions, we compared the response of Arabidopsis genotypes to AMF inoculation, as well as the AM host plant Lotus and its ipd3 mutant cyclops-4. Our results indicate that despite the long intervening period as a nonAM plant and the further loss of related genes, expressing IPD3Min in Arabidopsis resulted in phenotypic and transcriptional effects. The preservation of molecular connections for IPD3 raises the prospect of re-wiring nonAM plants to restore AM symbiosis, with applications in agriculture (French 2017; Lynch 2019). Although they are a phylogenetic minority, nonAM crops are responsible for almost half a billion metric tons of agricultural harvest every year (Hornstein 2022). With reported yield increases from mycorrhizae of 20–100% in various host crops, significant economic advantages could result if these benefits can be conferred on nonAM crops (Eo and Eom 2009; Pellegrino et al. 2015; Berruti et al. 2016). In 2021, such an increase in US canola alone would have resulted in an additional 250,000–1.25 million metric tons of harvest worth 175–870 million dollars (USDA NASS 2022).
We also found evidence that IPD3 may be subject to targeted regulation in Arabidopsis. Two full-length versions of IPD3 transgenically expressed in Arabidopsis could not be detected at the protein level, while IPD3Min was successfully expressed in roots and shoots (Figs. 2, S1). This suggests IPD3 may be subject to silencing or degradation specific to the N-terminal portion excluded from IPD3Min. In prior work, we attempted to express IPD3Mt and IPD3S50D in the oilseed crop Camelina sativa, and observed low expression levels in multiple independent lines, consistent with evidence in the present study for silencing of these constructs in Arabidopsis (Hornstein 2022). We have also observed potentially deleterious effects for IPD3S50D in Camelina. T1 transformants were repeatedly lost to severe fungal infections, and later generations produced unusual dwarfed growth phenotypes at nonmendelian ratios (Hornstein and Sederoff unpublished). It is possible that both the present study and past work in Camelina were biased by survivorship of T1 individuals which avoid a deleterious effect due to silencing.
Mechanisms for functional AM go beyond the CSP to essential plant functions including lipid biosynthesis, membrane and vesicle functions, nutrient transport, and defense (Wang et al. 2012; Behie and Bidochka 2014; Luginbuehl et al. 2017). Although symbiosis-specific genes that affect these functions in AM hosts have been lost in nonAM species, most belong to families with non-symbiosis-specific members that have similar molecular functions. In our experiment we observed IPD3-independent upregulation of lipid and 2-MAG biosynthesis genes in response to AMF in Arabidopsis (Figs. 3, 4). Activation of 2-MAG biosynthesis via RAM1 and RAM2 is well-characterized as an essential feature of AM symbiosis that strictly depends on IPD3 in host species (Wang et al. 2012; Gobbato et al. 2013; Pimprikar and Gutjahr 2018). In our results, 2-MAG-related transcription appeared to occur in response to AMF, but by action of related genes which are not symbiosis-specific (Yang et al. 2010, 2012b). Given that AMF was the only microbe tested, the biotic interaction terms and linkage to phosphate starvation as shown in the WGCNA and GOterm analysis may represent a generalized microbial response consistent with known effects in Arabidopsis (Finkel et al. 2019). In the context of engineering AM as an agriculturally useful trait, this raises the interesting possibility that non-symbiosis-specific genes retained in nonAM plants can be recruited to perform symbiotic functions (Hornstein and Sederoff 2023).
Figure 7 lays out the main findings of this study in relation to the canonical knowledge of IPD3’s role in AM. In Arabidopsis, IPD3Min plants showed a > 99% reduced transcriptional response to AMF compared to wild type (Fig. 5). This is in part because IPD3Min expression places plants in an AMF-exposure-like transcriptional state prior to actual exposure to the fungus (Fig. 6C). This is reminiscent of results in symbiosis models where expression of phosphomimic IPD3 leads to a constitutive symbiotic response in the absence of biotic stimulus (Singh et al. 2014; Gobbato 2015). Our results suggest that although the nature of the transcriptional response may differ, IPD3Min acts analogously in Arabidopsis by activating the native AMF response even in the absence of the fungus. In Lotus, surprisingly, we found that −ipd3 knockout was associated with a similar priming effect. The AMF response in this Lotus genotype is ~ 90% reduced in our experiment, consistent with canonical understanding of IPD3 (Yano et al. 2008; Pimprikar et al. 2016). However, in our experiment this was partly because a set of genes in uninoculated plants is regulated similarly upon ipd3 knockout and AMF exposure (Fig. 5). This suggests IPD3 may have an unrecognized function as a direct or indirect repressor of some of its own targets.
Novel roles for IPD3 and responses to AMF in Arabidopsis and Lotus. In Lotus, we add to the current understanding of IPD3 by showing that its knockout has a large effect even in the absence of AMF, and that the reduction in overall scale of AMF response in the cyclops-4 genotype is due to a surprising partial replication of AMF-exposure-like transcription. In Arabidopsis we find evidence that AM-related lipid metabolism is altered by AMF independently of IPD3 and despite the plant's nonAM status. Expression of IPD3Min vastly reduced the amount of AMF-responsive differential expression in Arabidopsis, but not because IPD3Min has little effect. IPD3Min expression results in many differentially expressed genes in the absence of AMF, and the response to AMF in transgenic plants is small at least in part because many of the DEGs acting in the wild type Arabidopsis AMF response are similarly regulated in IPD3Min transgenics even before AMF treatment is applied. Genes affected by IPD3Min in Arabidopsis relate to functions including abiotic stress, pathogen defense, and circadian rhythm
The striking inverse effects in Lotus and Arabidopsis also highlight two limitations of our results for drawing comparisons between the two species and with prior work. First, the IPD3 genotypes across the two species are not identical. The native LjIPD3/CYCLOPS gene in Lotus encodes a protein product that undergoes extensive protein-protein interactions and requires phosphorylation for activation, while the IPD3Min protein we used in Arabidopsis is constitutively active (Yu et al. 2014; Singh et al. 2014; Pimprikar et al. 2016; Jin et al. 2016, 2018). Second, the effects we observed for AMF interaction may be specific to the early time point sampled in this study (48 h). We selected an early time point for the whole experiment because Arabidopsis cannot sustain AMF in monoxenic culture, but most studies allow interaction with AMF on the scale of weeks to establish mature colonization; earlier stages can involve rapidly shifting responses not seen later (Siciliano et al. 2007; Genre et al. 2008; Gutjahr et al. 2009; Handa et al. 2015; Nanjareddy et al. 2017; Prihatna et al. 2018). An idealized experiment might use cyclops-4 Lotus rescued by expression of IPD3Min, and a nurse-pot colonization system to support Arabidopsis AM colonization (Veiga et al. 2013; Fernández et al. 2019).
Prior studies in AM model plants at this early phase of colonization have found 20–650 DEGs in response to AMF treatment, germinated spore exudates, and various AM signal molecules at timepoints from 6 h to 48 days from contact (Czaja et al. 2012; Hohnjec et al. 2015; Giovannetti et al. 2015; Siciliano et al. 2007). Importantly, CSP gene knockout has previously been found to significantly alter the AMF (and nodulating rhizobacteria) response even at this early time point (Hohnjec et al. 2015; Høgslund et al. 2009). However, early-stage results in this experiment should also be viewed in recognition of the fact that late-stage colonization of AM host plants, as well as nurse-pot supported late-stage colonization of Arabidopsis, implicate much larger plant gene sets in the AM relationship (Fernández et al. 2019).
One area where IPD3Min expression duplicates the effect of AMF treatment is downregulation of circadian clock genes that result in delayed flowering (Fig. 4) (Sawa et al. 2007; Para et al. 2007; Niinuma et al. 2008; Shim et al. 2017). This gene regulation may explain the delayed flowering phenotype in IPD3Min transgenic Arabidopsis seen in our experiment if the root transcriptome effect extends to aboveground tissue (Figs. 1, S1). AM symbiosis affects flowering time in host plants, and the circadian clock is implicated in maintenance of the symbiosis itself as well as AM-mediated abiotic stress resistance (Hernandez and Allen 2013; Lee et al. 2019; Bennett and Meek 2020; Liu et al. 2022). In Arabidopsis, circadian rhythm genes are also known to act in microbiome construction, pathogen defense, development, and abiotic stress (Lee et al. 2005; Nakamichi et al. 2016; Newman et al. 2022; Xu et al. 2022; Singh 2022a, b). Remarkably, in mosses, the sole group of nonAM plants that retain CSP genes, IPD3 specifically has been shown to mediate a stress-responsive reproductive transition (Kleist et al. 2022).
In uninoculated plants, two DEGs result from the respective +IPD3/−ipd3 contrasts in both Arabidopsis and Lotus: LEA4-5 and HIS1-3 (Fig. 6B). Both of these genes are associated with response to abiotic stress including drought, heat, and cold (Dalal et al. 2009; Olvera-Carrillo et al. 2010; Rutowicz et al. 2015). HIS1-3 is downregulated in the +IPD3 genotype of both species, and LEA4-5 is upregulated in Arabidopsis but downregulated in Lotus (Fig. 6).
A molecular function in stress response would subject IPD3 to selection on factors unrelated to its role in AM symbiosis, and might help explain both the reasons for this gene’s loss in nonAM species, and its presence in charophyte ancestors of land plants long before the existence of AM symbiosis (Delaux et al. 2015; Delaux and Schornack 2021).
We also identified AMF and IPD3-Min-responsive downregulation of Arabidopsis genes involved in salicylic acid-mediated defense (Fig. 4C). Reduced transcription of WRKY70, WRKY54, and PR5 by IPD3Min in the absence of AMF suggests that plants expressing IPD3Min have reduced baseline levels of SA-mediated defense to biotrophic pathogens (Glazebrook 2005; Blanco et al. 2005; Li et al. 2006; Yang et al. 2012a). Early AM colonization of host plants can involve a transient increased defense response followed by reduction, while Arabidopsis shows an early symbiosis-like response to AMF, followed by a strong defensive response under forced long-term interaction (Giovannetti et al. 2015) (Fernández et al. 2019; Cosme et al. 2021). While AM can confer resistance to pathogens and insects, genes acting in AM also enable infection by some pathogens (Wang et al. 2012; Siebers et al. 2016; Ried et al. 2019; Chen et al. 2021a; Dey and Ghosh 2022). The full mechanisms for such effects are not known, but comport with our evidence for perturbation of Arabidopsis defenses by a nominally AM-specific gene.
Recently, significant portions of the AMF genetic network were revealed to be under control of the phosphate-stress-responsive PHR-SPX system, which is conserved in Arabidopsis (Shi et al. 2021, 2022). In Arabidopsis, PHR also regulates the microbiome by reducing plant defenses when phosphate is low, resulting in recruitment of beneficial microbes (Finkel et al 2019; Martin-Rivilla et al. 2019; Castrillo et al 2017; Cho et al. 2013; Kotchoni and Gachomo 2006; Dangl 1998). Given knowledge that both AM and non-AM microbial relations are part of the PHR-regulated phosphate response network, it is possible that the IPD3Min effect in our experiment relates to conserved, non-AM-specific points of crosstalk with defense and symbiosis (Fig. 7).
Regardless of whether plants are formally labeled non-hosts in isolation, in natural settings they continue to interact with ubiquitous AM fungi and can display partial and transitional phenotypes (Ma et al. 2018; Cosme et al. 2018). It is not surprising that an ancient, conserved trait is interconnected to other aspects of plant life. Our results show that despite the loss of genes essential for AM symbiosis, connections to this trait remain in place in Arabidopsis, and can be highlighted, even re-activated by expression of IPD3Min. The transcriptional effects we identified suggest specific targets for follow-up studies to directly assess pleiotropic effects, including pathogen sensitivity and abiotic stress resilience.
Data availability
Transcriptome data is publicly available via GEO accession number GSE225213, https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE225213. Proteomics data have been deposited to the ProteomeXchange Consortium via PRIDE identifier PXD040665 (Reviewer access at https://www.ebi.ac.uk/pride/login with Username: reviewer_pxd040665@ebi.ac.uk and Password: CVDhdrfh).
References
Aliyu IA, Yusuf AA, Uyovbisere EO, Masso C, Sanders IR (2019) Effect of co-application of phosphorus fertilizer and in vitro-produced mycorrhizal fungal inoculants on yield and leaf nutrient concentration of cassava. PLoS ONE 14:e0218969
Begum N, Qin C, Ahanger MA, Raza S, Khan MI, Ashraf M, Ahmed N, Zhang L (2019) Role of arbuscular mycorrhizal fungi in plant growth regulation: implications in abiotic stress tolerance. Front Plant Sci 10:1068
Behie SW, Bidochka MJ (2014) Nutrient transfer in plant–fungal symbioses. Trends Plant Sci 19:734–740
Bennett AE, Meek HC (2020) The influence of arbuscular mycorrhizal fungi on plant reproduction. J Chem Ecol 46:707–721
Berruti A, Lumini E, Balestrini R, Bianciotto V (2016) Arbuscular mycorrhizal fungi as natural biofertilizers: let’s benefit from past successes. Front Microbiol. https://doi.org/10.3389/fmicb.2015.01559
Blanco F, Garretón V, Frey N, Dominguez C, Pérez-Acle T, Van der Straeten D, Jordana X, Holuigue L (2005) Identification of NPR1-dependent and independent genes early induced by salicylic acid treatment in arabidopsis. Plant Mol Biol 59:927–944
Bonneau L, Huguet S, Wipf D, Pauly N, Truong H (2013) Combined phosphate and nitrogen limitation generates a nutrient stress transcriptome favorable for arbuscular mycorrhizal symbiosis in Medicago truncatula. New Phytol 199:188–202
Bravo A, York T, Pumplin N, Mueller LA, Harrison MJ (2016) Genes conserved for arbuscular mycorrhizal symbiosis identified through phylogenomics. Nat Plants 2:15208
Brundrett MC (2017) Global diversity and importance of mycorrhizal and nonmycorrhizal plants. In: Tedersoo L (ed) Biogeography of mycorrhizal symbiosis. Springer, Cham, pp 533–556
Brundrett MC, Tedersoo L (2018) Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol 220(4):1108–1115
Bushnell B (2014) “BBMap: a fast, accurate, splice-aware aligner.” BBMap: a fast, accurate, splice-aware (No. LBNL-7065E). Lawrence Berkeley National Lab (LBNL), Berkeley
Carbonnel S, Gutjahr C (2014) Control of arbuscular mycorrhiza development by nutrient signals. Front Plant Sci. https://doi.org/10.3389/fpls.2014.00462
Castrillo G, Teixeira PJPL, Paredes SH, Law TF, de Lorenzo L, Feltcher ME, Finkel OM, Breakfield NW, Mieczkowski P, Jones CD et al (2017) Root microbiota drive direct integration of phosphate stress and immunity. Nature 543:513–518
Ceballos I, Ruiz M, Fernández C, Peña R, Rodríguez A, Sanders IR (2013) The in vitro mass-produced model mycorrhizal fungus, Rhizophagus irregularis, significantly increases yields of the globally important food security crop cassava. PLoS ONE 8:e70633
Chen M, Bruisson S, Bapaume L, Darbon G, Glauser G, Schorderet M, Reinhardt D (2021) VAPYRIN attenuates defence by repressing PR gene induction and localized lignin accumulation during arbuscular mycorrhizal symbiosis of Petunia hybrida. New Phytol 229:3481–3496
Chen S, Ding Y, Tian H, Wang S, Zhang Y (2021) WRKY54 and WRKY70 positively regulate SARD1 and CBP60g expression in plant immunity. Plant Signal Behav 16:1932142
Cho S-M, Kang BR, Kim YC (2013) Transcriptome analysis of induced systemic drought tolerance elicited by Pseudomonas chlororaphis O6 in Arabidopsis thaliana. Plant Pathol J 29:209–220
Cope KR, Bascaules A, Irving TB, Venkateshwaran M, Maeda J, Garcia K, Rush TA, Ma C, Labbé J, Jawdy S et al (2019) The ectomycorrhizal fungus Laccaria bicolor produces Lipochitooligosaccharides and uses the common symbiosis pathway to colonize Populus Roots. Plant Cell 31:2386–2410
Cosme M, Fernández I, Van der Heijden MGA, Pieterse CMJ (2018) Non-mycorrhizal plants: the exceptions that prove the rule. Trends Plant Sci 23:577–587
Cosme M, Fernández I, Declerck S, van der Heijden MGA, Pieterse CMJ (2021) A coumarin exudation pathway mitigates arbuscular mycorrhizal incompatibility in Arabidopsis thaliana. Plant Mol Biol 106:319–334
Czaja LF, Hogekamp C, Lamm P, Maillet F, Martinez EA, Samain E, Dénarié J, Küster H, Hohnjec N (2012) Transcriptional responses toward diffusible signals from symbiotic microbes reveal MtNFP-and MtDMI3-dependent reprogramming of host gene expression by arbuscular mycorrhizal fungal lipochitooligosaccharides. Plant Physiol 159(4):1671–1685
Dai H, Zhang X, Zhao B, Shi J, Zhang C, Wang G, Yu N, Wang E (2022) Colonization of mutualistic mycorrhizal and parasitic blast fungi requires OsRAM2-regulated fatty acid biosynthesis in rice. Mol Plant–Microbe Interactions. https://doi.org/10.1094/MPMI-11-21-0270-R
Dalal M, Tayal D, Chinnusamy V, Bansal KC (2009) Abiotic stress and ABA-inducible Group 4 LEA from Brassica napus plays a key role in salt and drought tolerance. J.Biotechnol 139:137–145
Dalal J, Yalamanchili R, La Hovary C, Ji M, Rodriguez-Welsh M, Aslett D, Ganapathy S, Grunden A, Sederoff H, Qu R (2015) A novel gateway-compatible binary vector series (PC-GW) for flexible cloning of multiple genes for genetic transformation of plants. Plasmid 81:55–62
Dangl J (1998) Plants just say NO to pathogens. Nature 394:525–527
Davis AM, Hall A, Millar AJ, Darrah C, Davis SJ (2009) Protocol: Streamlined sub-protocols for floral-dip transformation and selection of transformants in Arabidopsis thaliana. Plant Methods 5:3
Delaux P-M, Schornack S (2021) Plant evolution driven by interactions with symbiotic and pathogenic microbes. Science 371:eaba6605
Delaux P-M, Séjalon-Delmas N, Bécard G, Ané J-M (2013) Evolution of the plant–microbe symbiotic ‘toolkit.’ Trends Plant Sci 18:298–304
Delaux P-M, Varala K, Edger PP, Coruzzi GM, Pires JC, Ané J-M (2014) Comparative phylogenomics uncovers the impact of symbiotic associations on host genome evolution. PLoS Genet 10:e1004487
Delaux P-M, Radhakrishnan GV, Jayaraman D, Cheema J, Malbreil M, Volkening JD, Sekimoto H, Nishiyama T, Melkonian M, Pokorny L et al (2015) Algal ancestor of land plants was preadapted for symbiosis. Proc Natl Acad Sci 112:13390–13395
Demchenko K, Winzer T, Stougaard J, Parniske M, Pawlowski K (2004) Distinct roles of Lotus japonicus SYMRK and SYM15 in root colonization and arbuscule formation. New Phytol 163:381–392
Dey M, Ghosh S (2022) Arbuscular mycorrhizae in plant immunity and crop pathogen control. Rhizosphere 22:100524
Edwards B, Hornstein ED, Wilson NJ, Sederoff H (2022) High-throughput detection of T-DNA insertion sites for multiple transgenes in complex genomes. BMC Genomics 23(1):1–20
Eo J-K, Eom A-H (2009) Differential growth response of various crop species to arbuscular mycorrhizal inoculation. Mycobiology 37:72–76
Feng F, Sun J, Radhakrishnan GV, Lee T, Bozsóki Z, Fort S, Gavrin A, Gysel K, Thygesen MB, Andersen KR et al (2019) A combination of chitooligosaccharide and lipochitooligosaccharide recognition promotes arbuscular mycorrhizal associations in Medicago truncatula. Nat Commun 10:5047
Fernández I, Cosme M, Stringlis IA, Yu K, Jonge R, Saskia CM, Pozo MJ, Pieterse CMJ (2019) Molecular dialogue between arbuscular mycorrhizal fungi and the nonhost plant Arabidopsis thaliana switches from initial detection to antagonism. New Phytol 223:867–881
Field KJ, Cameron DD, Leake JR, Tille S, Bidartondo MI, Beerling DJ (2012) Contrasting arbuscular mycorrhizal responses of vascular and non-vascular plants to a simulated Palaeozoic CO2 decline. Nat Commun 3:835
Finkel OM, Salas-González I, Castrillo G, Spaepen S, Law TF, Teixeira PJPL, Jones CD, Dangl JL (2019) The effects of soil phosphorus content on plant microbiota are driven by the plant phosphate starvation response. PLoS Biol 17:e3000534
French KE (2017) Engineering mycorrhizal symbioses to alter plant metabolism and improve crop health. Front Microbiol 8:1403
Genre A, Chabaud M, Faccio A, Barker DG, Bonfante P (2008) Prepenetration apparatus assembly precedes and predicts the colonization patterns of arbuscular mycorrhizal fungi within the root cortex of both Medicago truncatula and Daucus carota. Plant Cell 20:1407–1420
Genre A, Lanfranco L, Perotto S, Bonfante P (2020) Unique and common traits in mycorrhizal symbioses. Nat Rev Microbiol 18:649–660
Giovannetti M, Mari A, Novero M, Bonfante P (2015) Early Lotus japonicus root transcriptomic responses to symbiotic and pathogenic fungal exudates. Front Plant Sci. https://doi.org/10.3389/fpls.2015.00480
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43:205–227
Gobbato E (2015) Recent developments in arbuscular mycorrhizal signaling. Curr Opin Plant Biol 26:1–7
Gobbato E, Wang E, Higgins G, Bano SA, Henry C, Schultze M, Oldroyd GE (2013) RAM1 and RAM2 function and expression during arbuscular mycorrhizal symbiosis and Aphanomyces euteiches colonization. Plant Signal Behav 8:e26049
Grefen C, Donald N, Hashimoto K, Kudla J, Schumacher K, Blatt MR (2010) A ubiquitin-10 promoter-based vector set for fluorescent protein tagging facilitates temporal stability and native protein distribution in transient and stable expression studies: fluorescence tagging and expression in Arabidopsis. Plant J 64:355–365
Guan L, Denkert N, Eisa A, Lehmann M, Sjuts I, Weiberg A, Soll J, Meinecke M, Schwenkert S (2019) JASSY, a chloroplast outer membrane protein required for jasmonate biosynthesis. Proc Natl Acad Sci 116:10568–10575
Gutjahr C, Novero M, Guether M, Montanari O, Udvardi M, Bonfante P (2009) Presymbiotic factors released by the arbuscular mycorrhizal fungus Gigaspora margarita induce starch accumulation in Lotus japonicus roots. New Phytol 183:53–61
Handa Y, Nishide H, Takeda N, Suzuki Y, Kawaguchi M, Saito K (2015) RNA-seq transcriptional profiling of an arbuscular mycorrhiza provides insights into regulated and coordinated gene expression in Lotus japonicus and Rhizophagus irregularis. Plant Cell Physiol 56:1490–1511
Hernandez RR, Allen MF (2013) Diurnal patterns of productivity of arbuscular mycorrhizal fungi revealed with the soil ecosystem observatory. New Phytol 200:547–557
Høgslund N, Radutoiu S, Krusell L, Voroshilova V, Hannah MA, Goffard N, Sanchez DH, Lippold F, Ott T, Sato S, Tabata S (2009) Dissection of symbiosis and organ development by integrated transcriptome analysis of Lotus japonicus mutant and wild-type plants. PLoS ONE 4(8):e6556
Hohmann N, Wolf EM, Lysak MA, Koch MA (2015) A time-calibrated road map of Brassicaceae species radiation and evolutionary history. Plant Cell. https://doi.org/10.1105/tpc.15.00482
Hohnjec N, Czaja-Hasse LF, Hogekamp C, Küster H (2015) Pre-announcement of symbiotic guests: transcriptional reprogramming by mycorrhizal lipochitooligosaccharides shows a strict co-dependency on the GRAS transcription factors NSP1 and RAM1. BMC Genomics 16:1–16
Hornstein ED (2022) Genetic engineering to retrace the evolutionary loss of arbuscular mycorrhizae, with implications for sustainable agriculture. PhD Thesis NCSU
Hornstein ED, Sederoff H (2023) Back to the future: re-engineering the evolutionarily lost arbuscular mycorrhiza host trait to improve climate resilience for agriculture. Crit Rev Plant Sci 43:1–33
Horváth B, Yeun LH, Domonkos Á, Halász G, Gobbato E, Ayaydin F, Miró K, Hirsch S, Sun J, Tadege M et al (2011) Medicago truncatula IPD3 Is a member of the common symbiotic signaling pathway required for rhizobial and mycorrhizal symbioses. Mol Plant Microbe Interactions 24:1345–1358
Hou S, Tsuda K (2022) Salicylic acid and jasmonic acid crosstalk in plant immunity. Essays Biochem 66:647–656
Ivanov S, Harrison MJ (2014) A set of fluorescent protein-based markers expressed from constitutive and arbuscular mycorrhiza-inducible promoters to label organelles, membranes and cytoskeletal elements in Medicago truncatula. Plant J 80:1151–1163
Jin Y, Liu H, Luo D, Yu N, Dong W, Wang C, Zhang X, Dai H, Yang J, Wang E (2016) DELLA proteins are common components of symbiotic rhizobial and mycorrhizal signalling pathways. Nat Commun 7:12433
Jin Y, Chen Z, Yang J, Mysore KS, Wen J, Huang J, Yu N, Wang E (2018) IPD3 and IPD3L function redundantly in rhizobial and mycorrhizal symbioses. Front Plant Sci 9:267
Kleist TJ, Bortolazzo A, Keyser ZP, Perera AM, Irving TB, Venkateshwaran M, Atanjaoui F, Tang R-J, Maeda J, Cartwright HN et al (2022) Stress-associated developmental reprogramming in moss protonemata by synthetic activation of the common symbiosis pathway. iScience 25:103754
Kloth KJ, Wiegers GL, Busscher-Lange J, van Haarst JC, Kruijer W, Bouwmeester HJ, Dicke M, Jongsma MA (2016) AtWRKY22 promotes susceptibility to aphids and modulates salicylic acid and jasmonic acid signalling. J Exp Bot 67:3383–3396
Kotchoni SO, Gachomo EW (2006) The reactive oxygen species network pathways: an essential prerequisite for perception of pathogen attack and the acquired disease resistance in plants. J Biosci 31:389–404
Langfelder P, Horvath S (2008) WGCNA: an R package for weighted correlation network analysis. BMC Bioinf 9:559
Langfelder P, Horvath S (2012) Fast R functions for robust correlations and hierarchical clustering. J Stat Soft 46:i11
Lee B, Henderson DA, Zhu J-K (2005) The Arabidopsis cold-responsive transcriptome and its regulation by ICE1. Plant Cell 17:3155–3175
Lee S-J, Morse D, Hijri M (2019) Holobiont chronobiology: mycorrhiza may be a key to linking aboveground and underground rhythms. Mycorrhiza 29:403–412
Li J, Brader G, Kariola T, Tapio Palva E (2006) WRKY70 modulates the selection of signaling pathways in plant defense. Plant J 46:477–491
Liao Y, Smyth GK, Shi W (2014) featureCounts: an efficient general purpose program for assigning sequence reads to genomic features. Bioinformatics 30:923–930
Liu R-C, Ding Y-E, Wu Q-S, Zou Y-N (2022) Mycorrhizae enhance drought tolerance of trifoliate orange by regulating circadian clock response patterns. Sci Hortic 305:111426
Lorenzo O, Piqueras R, Sánchez-Serrano JJ, Solano R (2003) ETHYLENE RESPONSE FACTOR1 integrates signals from ethylene and jasmonate pathways in plant defense. Plant Cell 15:165–178
Luginbuehl LH, Menard GN, Kurup S, Van Erp H, Radhakrishnan GV, Breakspear A, Oldroyd GED, Eastmond PJ (2017) Fatty acids in arbuscular mycorrhizal fungi are synthesized by the host plant. Science 356:1175–1178
Lynch JP (2019) Root phenotypes for improved nutrient capture: an underexploited opportunity for global agriculture. New Phytol 223:548–564
Ma L, Li G (2018) FAR1-RELATED SEQUENCE (FRS) and FRS-RELATED FACTOR (FRF) family proteins in Arabidopsis growth and development. Front Plant Sci 9:692
Ma Z, Guo D, Xu X, Lu M, Bardgett RD, Eissenstat DM, McCormack ML, Hedin LO (2018) Evolutionary history resolves global organization of root functional traits. Nature 555:94–97
Martin-Rivilla H, Garcia-Villaraco A, Ramos-Solano B, Gutierrez-Mañero FJ, Lucas JA (2019) Extracts from cultures of Pseudomonas fluorescens induce defensive patterns of gene expression and enzyme activity while depressing visible injury and reactive oxygen species in Arabidopsis thaliana challenged with pathogenic Pseudomonas syringae. AoB PLANTS 11:49
Mi H, Muruganujan A, Casagrande JT, Thomas PD (2013) Large-scale gene function analysis with the PANTHER classification system. Nat Protoc 8:1551–1566
Mordant A, Kleiner M (2021) Evaluation of sample preservation and storage methods for metaproteomics and analysis of intestinal microbes. Microbiol Spectrum 9(3):01877–21
Nakamichi N, Takao S, Kudo T, Kiba T, Wang Y, Kinoshita T, Sakakibara H (2016) Improvement of Arabidopsis biomass and cold, drought and salinity stress tolerance by modified Circadian clock-associated PSEUDO-RESPONSE REGULATORs. Plant Cell Physiol 57:1085–1097
Nanjareddy K, Arthikala M-K, Gómez B-M, Blanco L, Lara M (2017) Differentially expressed genes in mycorrhized and nodulated roots of common bean are associated with defense, cell wall architecture, N metabolism, and P metabolism. PLoS ONE 12:e0182328
Newman A, Picot E, Davies S, Hilton S, Carré IA, Bending GD (2022) Circadian rhythms in the plant host influence rhythmicity of rhizosphere microbiota. BMC Biol 20:235
Niinuma K, Nakamichi N, Miyata K, Mizuno T, Kamada H, Mizoguchi T (2008) Roles of Arabidopsis PSEUDO-RESPONSE REGULATOR (PRR) genes in the opposite controls of flowering time and organ elongation under long-day and continuous light conditions. Plant Biotechnol 25:165–172
Nouri E, Breuillin-Sessoms F, Feller U, Reinhardt D (2014) Phosphorus and nitrogen regulate arbuscular mycorrhizal symbiosis in Petunia hybrida. PLoS ONE 9:e90841
Oldroyd GED (2013) Speak, friend, and enter: signalling systems that promote beneficial symbiotic associations in plants. Nat Rev Microbiol 11:252–263
Olvera-Carrillo Y, Campos F, Reyes JL, Garciarrubio A, Covarrubias AA (2010) Functional analysis of the group 4 late embryogenesis abundant proteins reveals their relevance in the adaptive response during water deficit in Arabidopsis. Plant Physiol 154:373–390
Pan H, Stonoha-Arther C, Wang D (2018) Medicago plants control nodulation by regulating proteolysis of the receptor-like kinase DMI2. Plant Physiol 177:792–802
Para A, Farré EM, Imaizumi T, Pruneda-Paz JL, Harmon FG, Kay SA (2007) PRR3 is a vascular regulator of TOC1 stability in the Arabidopsis Circadian clock. Plant Cell 19:3462–3473
Pellegrino E, Öpik M, Bonari E, Ercoli L (2015) Responses of wheat to arbuscular mycorrhizal fungi: a meta-analysis of field studies from 1975 to 2013. Soil Biol Biochem 84:210–217
Pimprikar P, Gutjahr C (2018) Transcriptional regulation of arbuscular mycorrhiza development. Plant Cell Physiol 59:678–695
Pimprikar P, Carbonnel S, Paries M, Katzer K, Klingl V, Bohmer MJ, Karl L, Floss DS, Harrison MJ, Parniske M et al (2016) A CCaMK-CYCLOPS-DELLA complex activates transcription of RAM1 to regulate arbuscule branching. Curr Biol 26:987–998
Poveda J, Hermosa R, Monte E, Nicolás C (2019) Trichoderma harzianum favours the access of arbuscular mycorrhizal fungi to non-host Brassicaceae roots and increases plant productivity. Sci Rep 9:11650
Prihatna C, Larkan NJ, Barbetti MJ, Barker SJ (2018) Tomato CYCLOPS/IPD3 is required for mycorrhizal symbiosis but not tolerance to Fusarium wilt in mycorrhiza-deficient tomato mutant rmc. Mycorrhiza 28:495–507
Radhakrishnan GV, Keller J, Rich MK, Vernié T, Mbadinga Mbadinga DL, Vigneron N, Cottret L, Clemente HS, Libourel C, Cheema J et al (2020) An ancestral signalling pathway is conserved in intracellular symbioses-forming plant lineages. Nat Plants 6:280–289
Ramírez-Flores MR, Perez-Limon S, Li M, Barrales-Gamez B, Albinsky D, Paszkowski U, Olalde-Portugal V, Sawers RJ (2020) The genetic architecture of host response reveals the importance of arbuscular mycorrhizae to maize cultivation. eLife 9:e61701
Ramirez-Parra E, Perianez-Rodriguez J, Navarro-Neila S, Gude I, Moreno-Risueno MA, del Pozo JC (2017) The transcription factor OBP 4 controls root growth and promotes callus formation. New Phytol 213:1787–1801
Rich MK, Nouri E, Courty P-E, Reinhardt D (2017) Diet of arbuscular mycorrhizal fungi: bread and butter? Trends Plant Sci 22:652–660
Ried MK, Banhara A, Hwu F-Y, Binder A, Gust AA, Höfle C, Hückelhoven R, Nürnberger T, Parniske M (2019) A set of Arabidopsis genes involved in the accommodation of the downy mildew pathogen Hyaloperonospora arabidopsidis. PLOS Pathog 15:e1007747
Robinson MD, McCarthy DJ, Smyth GK (2010) edgeR: a bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26:139–140
Rutowicz K, Puzio M, Halibart-Puzio J, Lirski M, Kroteń MA, Kotliński M, Kniżewski Ł, Lange B, Muszewska A, Śniegowska-Świerk K et al (2015) A specialized histone H1 variant is required for adaptive responses to complex abiotic stress and related DNA methylation in Arabidopsis. Plant Physiol. https://doi.org/10.1104/pp.15.00493
Rymen B, Kawamura A, Schäfer S, Breuer C, Iwase A, Shibata M, Ikeda M, Mitsuda N, Koncz C, Ohme-Takagi M et al (2017) ABA suppresses root hair growth via the OBP4 transcriptional regulator. Plant Physiol 173:1750–1762
Samac DA, Hironaka CM, Yallaly PE, Shah DM (1990) Isolation and characterization of the genes encoding basic and acidic chitinase in Arabidopsis thaliana. Plant Physiol 93:907–914
Sawa M, Nusinow DA, Kay SA, Imaizumi T (2007) FKF1 and GIGANTEA complex formation is required for day-length measurement in Arabidopsis. Science 318:261–265
Schnell J, Steele M, Bean J, Neuspiel M, Girard C, Dormann N, Pearson C, Savoie A, Bourbonniere L, Macdonald P (2015) A comparative analysis of insertional effects in genetically engineered plants: considerations for pre-market assessments. Transgenic Res 24:1–17
Seo HS, Song JT, Cheong J-J, Lee Y-H, Lee Y-W, Hwang I, Lee JS, Choi YD (2001) Jasmonic acid carboxyl methyltransferase: a key enzyme for jasmonate-regulated plant responses. Proc Natl Acad Sci 98:4788–4793
Shevchenko A, Tomas H, Havli J, Olsen JV, Mann M (2006) In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nat Protoc 1(6):2856–2860
Shi J, Zhao B, Zheng S, Zhang X, Wang X, Dong W, Xie Q, Wang G, Xiao Y, Chen F et al (2021) A phosphate starvation response-centered network regulates mycorrhizal symbiosis. Cell 184:5527-5540.e18
Shi J, Zhao B, Jin R, Hou L, Zhang X, Dai H, Yu N, Wang E (2022) A phosphate starvation response-regulated receptor-like kinase, OsADK1, is required for mycorrhizal symbiosis and phosphate starvation responses. New Phytol 236:2282–2293
Shim JS, Kubota A, Imaizumi T (2017) Circadian clock and photoperiodic flowering in Arabidopsis: CONSTANS is a hub for signal integration. Plant Physiol 173:5–15
Siciliano V, Genre A, Balestrini R, Cappellazzo G, deWit PJGM, Bonfante P (2007) Transcriptome analysis of arbuscular mycorrhizal roots during development of the prepenetration apparatus. Plant Physiol 144:1455–1466
Siebers M, Brands M, Wewer V, Duan Y, Hölzl G, Dörmann P (2016) Lipids in plant–microbe interactions. Biochim Biophys Acta BBA Mol Cell Biol Lipids 1861:1379–1395
Sikes BA (2010) When do arbuscular mycorrhizal fungi protect plant roots from pathogens? Plant Signal Behav 5:763–765
Singh A (2022) GIGANTEA regulates PAD4 transcription to promote pathogen defense against Hyaloperonospora arabidopsidis in Arabidopsis thaliana. Plant Signal Behav 17:2058719
Singh A (2022) GIGANTEA regulates lateral root formation by modulating auxin signaling in Arabidopsis thaliana. Plant Signal Behav 17:2096780
Singh S, Katzer K, Lambert J, Cerri M, Parniske M (2014) CYCLOPS, a DNA-binding transcriptional activator, orchestrates symbiotic root nodule development. Cell Host Microbe 15:139–152
Stepanova AN, Robertson-Hoyt J, Yun J, Benavente LM, Xie D-Y, Doležal K, Schlereth A, Jürgens G, Alonso JM (2008) TAA1-mediated auxin biosynthesis is essential for hormone crosstalk and plant development. Cell 133:177–191
Supek F, Bošnjak M, Škunca N, Šmuc T (2011) REVIGO summarizes and visualizes long lists of gene ontology terms. PLoS ONE 6:e21800
Takagi H, Hempton AK, Imaizumi T (2023) Photoperiodic flowering in Arabidopsis: multilayered regulatory mechanisms of CONSTANS and the florigen FLOWERING LOCUS T. Plant Commun. https://doi.org/10.1016/j.xplc.2023.100552
Thomma BPHJ, Eggermont K, Penninckx IAMA, Mauch-Mani B, Vogelsang R, Cammue BPA, Broekaert WF (1998) Separate jasmonate-dependent and salicylate-dependent defense-response pathways in Arabidopsis are essential for resistance to distinct microbial pathogens. Proc Natl Acad Sci 95:15107–15111
Thomma BP, Penninckx IA, Cammue BP, Broekaert WF (2001) The complexity of disease signaling in Arabidopsis. Curr Opin Immunol 13:63–68
USDA National Agricultural Statistical Service (2022) Quick Stats
Veiga RSL, Faccio A, Genre A, Pieterse CMJ, Bonfante P (2013) Arbuscular mycorrhizal fungi reduce growth and infect roots of the non-host plant Arabidopsis thaliana: effects of AMF on the non-host Arabidopsis. Plant Cell Environ. https://doi.org/10.1111/pce.12102
Wang B, Yeun LH, Xue J, Liu Y, Ané J, Qiu Y (2010) Presence of three mycorrhizal genes in the common ancestor of land plants suggests a key role of mycorrhizas in the colonization of land by plants. New Phytol 186:514–525
Wang E, Schornack S, Marsh JF, Gobbato E, Schwessinger B, Eastmond P, Schultze M, Kamoun S, Oldroyd GED (2012) A common signaling process that promotes mycorrhizal and oomycete colonization of plants. Curr Biol 22:2242–2246
Watts-Williams SJ, Cavagnaro TR (2015) Using mycorrhiza-defective mutant genotypes of non-legume plant species to study the formation and functioning of arbuscular mycorrhiza: a review. Mycorrhiza 25:587–597
Wisniewski JR, Zougman A, Nagaraj N, Mann M (2009) Universal sample preparation method for proteome analysis. Nat Commun 6(5):359–362
Xu P, Cai W (2019) Nitrate-responsive OBP4-XTH9 regulatory module controls lateral root development in Arabidopsis thaliana. PLOS Genet 15:e1008465
Xu X, Yuan L, Xie Q (2022) The circadian clock ticks in plant stress responses. Stress Biol 2:15
Yang W, Pollard M, Li-Beisson Y, Beisson F, Feig M, Ohlrogge J (2010) A distinct type of glycerol-3-phosphate acyltransferase with sn -2 preference and phosphatase activity producing 2-monoacylglycerol. Proc Natl Acad Sci 107:12040–12045
Yang Y, Zhang Y, Ding P, Johnson K, Li X, Zhang Y (2012) The ankyrin-repeat transmembrane protein BDA1 functions downstream of the receptor-like protein SNC2 to regulate plant immunity. Plant Physiol 159:1857–1865
Yang W, Simpson JP, Li-Beisson Y, Beisson F, Pollard M, Ohlrogge JB (2012) A land-plant-specific glycerol-3-phosphate acyltransferase family in Arabidopsis: substrate specificity, sn -2 preference, and evolution. Plant Physiol 160:638–652
Yano K, Yoshida S, Müller J, Singh S, Banba M, Vickers K, Markmann K, White C, Schuller B, Sato S et al (2008) CYCLOPS, a mediator of symbiotic intracellular accommodation. Proc Natl Acad Sci 105:20540–20545
Yu N, Luo D, Zhang X, Liu J, Wang W, Jin Y, Dong W, Liu J, Liu H, Yang W et al (2014) A DELLA protein complex controls the arbuscular mycorrhizal symbiosis in plants. Cell Res 24:130–133
Zybailov B, Mosley AL, Sardiu ME, Coleman MK, Florens L, Washburn MP (2006) Statistical analysis of membrane proteome expression changes in Saccharomyces cerevisiae. J Proteome Res 5:2339–2347
Acknowledgements
We thank Swathi Barampuram, Matthew Occena, and Asa Budnick for assistance with laboratory work in this study. We thank Caroline Gutjahr and Makoto Hayashi for the use of the cyclops-4 Lotus mutant. This work was supported by the Novo Nordisk Foundation InRoot project under Award No. NNF19SA0059362 (H.S. & M.K.) and the Department of Energy, DE-SC0018269 (H.S., M.C., E.D.H.). Graduate student fellowships from the National Science Foundation (NRT-INFEWS #1828820) and NCSU Provost’s Doctoral Fellowship provided support for E.H. and NIH Molecular Biotechnology Training Grant # 1T32GM133366-01 Fellowship to M.C. and a UsDoEd GAANN fellowship (P200A210002) to M.F. The LC-MS/MS measurements of protein samples were made using equipment in the Molecular Education, Technology, and Research Innovation Center (METRIC) at North Carolina State University.
Funding
This work was supported by the Novo Nordisk Foundation InRoot project under award No. NNF19SA0059362 (H.S. & M.K.) and the Department of Energy, DE-SC0018269 (H.S., M.C., E.D.H.). Graduate student fellowships from the National Science Foundation (NRT-INFEWS #1828820) and NCSU Provost’s Doctoral Fellowship provided support for E.H. and NIH Molecular Biotechnology Training Grant # 1T32GM133366-01 Fellowship to M.C. and a UsDoEd GAANN fellowship (P200A210002) to M.F.. The LC-MS/MS measurements of protein samples were made using equipment in the Molecular Education, Technology, and Research Innovation Center (METRIC) at North Carolina State University.
Author information
Authors and Affiliations
Contributions
EDH and HS designed the research; EDH and SV performed the research; BE, SV, and MK contributed analytical tools; MC, MF, SV, and EDH analyzed the data; EDH and HS drafted the manuscript with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors have no financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Supplementary file1 (DOCX 33 KB)
Online Resource 1 Supplemental experimental procedures providing further protocol detail for bioinformatic and laboratory analysis
Supplementary file2 (PNG 3327 KB)
Online Resource 2 Comparison of IPD3 protein versions used in this study and validation of RNA and protein expression in transgenic plants. The +C control sample is a synthetic peptide of the 349 C-terminal residues of MtIPD3 with expected size 39.98 kDa; expected size of IPD3-Min is 29.19 kDa and expected size of full-length IPD3 versions is 57.98 kDa. (A) Protein sequence alignment of Medicago truncatula IPD3, S50D phosphomimic IPD3, constitutively active DNA binding domain IPD3Min, Lotus japonicus IPD3/CYCLOPS, and the truncated C-terminal fragment resulting from the cyclops-4 mutation in Lotus. (B) Schematic of protein domains as affected by variants in this study. (C) Western blot of 3 IPD3 versions from transgenic Arabidopsis roots and shoots. This blot was visualized with fluorescent secondary antibody under 488/530 nm excitation/emission conditions (see Methods and Online Resource 1), causing the target protein bands to appear as light areas of high exposure while protein size ladder bands are visualized by dye and appear dark.(D) rt-PCR to confirm expression of IPD3 versions as RNA in leaves of T3 individuals.
Supplementary file3 (XLSX 1185 KB)
Online Resource 3 Shotgun proteomic quantification data of IPD3 and native proteins in Arabidopsis. Protein abundance is quantified as Normalized Spectral Abundance Factor (NSAF) according to Zybailov et al (2006), which is the number of unique protein fragments mapped to a specific reference sequence (spectral counts, SpC) divided by the total length of that protein (L), divided by the sum of the latter across all proteins present in the dataset (e.g. for protein n, [(SpCn)/(Ln)]/Σ[(SpC)/(L)]. NSAF is therefore a positive metric between 0 and 1, and the sum of NSAF for all proteins in a sample always adds up to 1, with the protein with highest NSAF having the highest estimated abundance. Note that matches of IPD3Mt and IPD3S50D are detected in the IPD3Min transgenic line due to the fact that IPD3Min is identical to the C-terminal formation of those reference sequences. No protein sequence originating from the N-terminal portion of IPD3 was detected in IPD3Min plants, and no IPD3 protein sequence of any kind was detected in plants transformed with IPD3Mt or IPD3S50D.
Supplementary file4 (PNG 3488 KB)
Online Resource 4 Mature phenotypes of IPD3-Min transgenic and control Arabidopsis lines in T3. (A-E) Transgenic lines 303, 308, 310, 312, 357, respectively; (F) wild type Col-0; (G) null segregants of transgenic line 308. Red line in all pictures shows 30 cm from surface
Supplementary file5 (XLSX 32 KB)
Online Resource 5 Raw phenotypic data and statistical output for Arabidopsis phenotyping experiments
Supplementary file6 (PNG 5009 KB)
Online Resource 6 Representative root images of transgenic and control lines. In an original set of experiments, the roots of soil-grown IPD3Min transgenic plants were bright pink while wild type and null segregant controls were white, and IPD3Mt and IPD3S50D were faint pink to white. In a followup experiment, multiple independent empty vector controls containing the mCherry marker also ranged from pink to white. Red stars mark those root images visually assessed to show any degree of pink coloration.
Supplementary file7 (PNG 1262 KB)
Online Resource 7 Images of 5-week-old Lotus japonicus seedlings of Gifu wildtype and cyclops-4 knockout mutant of ipd3 as grown on sterile petri dishes for the transcriptome experiment. Scale bar = 2 cm (scale tiles within image are denoted in cm)
Supplementary file8 (PNG 455 KB)
Online Resource 8 Principal component analysis shows clustering of transcriptomes by IPD3 genotype and AMF treatment in two species. (A) Lotus Gifu samples cluster according to AMF treatment along PC1. Gifu and cyclops-4 samples cluster by genotype along PC2. In contrast to Gifu, cyclops-4 plants do not separate along PC1 according to AMF treatment; both treatment groups instead cluster near AMF-treated Gifu plants along this axis. (B) Arabidopsis samples cluster by genotype along PC1 (IPD3M = IPD3Min transgenic). While wild type Col-0 plants cluster by AMF treatment along PC2, there is no equivalent separation by AMF treatment within either IPD3Min line (308; 310)
Supplementary file9 (PNG 172 KB)
Online Resource 9 IPD3-Min and mCherry transcript level in Arabidopsis transcriptome samples. Average IPD3-Min transcript count in line 308 is 36,591, ~4X higher than line 310 with average IPD3-Min count of 9,250
Supplementary file10 (PNG 181 KB)
Online Resource 10 Principal Component Analysis of Arabidopsis transcriptomes under low-nutrient conditions with AMF treatment. Only IPD3Min transgenic line 308 was used for the low-nutrient experiment. Transcriptomes cluster by IPD3 genotype along PC1, which accounts for 62% of variation
Supplementary file11 (PNG 672 KB)
Online Resource 11 Heatmap of complete gene expression correlation network (WGCNA) with module-trait correlations. Cells included in figure 3 of the main text are highlighted in yellow. The top number in each cell is the signed eigengene correlation of that module to value of trait shown on the X axis. A value of 1 indicates perfect positive correlation, -1 would indicate perfect inverse correlation. Bottom value shown in parentheses is the P-value of that module-trait correlation. Trait/treatment labels are as follows: Nutrient Level: determined by medium composition as described in Methods, positive correlation of a module to Nutrient Level indicates positive correlation of gene expression for module members with combined macronutrient concentration; +/-AMF: determined by presence or absence of germinated AM fungus spore treatment, positive correlation of a module to +/-AMF indicates positive correlation of gene expression for module members with presence of AMF; Transgenic Line: determined by transgenic genotype, discriminating between independent IPD3Min lines 308 and 310. Positive or negative correlation to Transgenic Line indicates that gene expression for that module is affected by transgenic genotype but is non-quantitative (see following); IPD3-Min Expression: determined by quantitative transcript level of IPD3Min, agnostic of line identity. Positive correlation of a module with IPD3-Min Expression indicates positive correlation of gene expression for module members with level of IPD3Min expression across all samples. Correlation of a module to Transgenic Line but not IPD3-Min Expression (i.e. Modules B, I, P), or correlation in opposite directions (not observed), would suggest that effects in that module could be due to insertion site effects rather than IPD3Min expression itself; mCherry Expression: determined by quantitative transcript level of mCherry marker gene across all samples.
Supplementary file12 (XLSX 15519 KB)
Online Resource 12 WGCNA gene module membership and trait correlations in Arabidopsis
Supplementary file13 (PDF 156 KB)
Online Resource 13 Semantic similarity clustering of gene ontology (GO) terms of all modules shown in figure 3 of the main text (including all Arabidopsis modules significantly correlated to IPD3Min expression). Size of the point on the plot for each GO term corresponds to its fold enrichment in that module, and intensity of color corresponds to FDR-corrected p-value of the enrichment. Proximity of the points represents their semantic similarity as determined by ReviGO (Supek et al 2011) from relatedness and frequency within the Arabidopsis gene ontology reference
Supplementary file14 (XLSX 65 KB)
Online Resource 14 Gene Ontology enrichment of Arabidopsis network modules
Supplementary file15 (XLSX 7511 KB)
Online Resource 15 Transcript count data of Arabidopsis
Supplementary file16 (XLSX 463 KB)
Online Resource 16 Gene sets resulting from differential expression contrasts summarized in figure 5
Supplementary file17 (PNG 404 KB)
Online Resource 17 Gene co-regulatory modules resulting from WGCNA analysis of Lotus transcriptomes. AMF corresponds to AM fungus treatment. Genotype reflects the presence of functional IPD3 in the wild type with a value of +1 and its absence in the cyclops-4 mutant with a value of 0
Supplementary file19 (XLSX 27421 KB)
Online Resource 18 WGCNA gene module membership and trait correlations in Lotus.
Supplementary file20 (XLSX 396 KB)
Online Resource 19 Cross-species comparisons of DEGs summarized in figure 6
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hornstein, E.D., Charles, M., Franklin, M. et al. IPD3, a master regulator of arbuscular mycorrhizal symbiosis, affects genes for immunity and metabolism of non-host Arabidopsis when restored long after its evolutionary loss. Plant Mol Biol 114, 21 (2024). https://doi.org/10.1007/s11103-024-01422-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11103-024-01422-3