Abstract
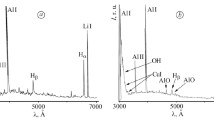
This paper represents emission spectrometric analysis of a hollow-cathode glow discharge plasma with helium–oxygen mixed gas for surface treatment of a cobalt-based alloy, together with surface analysis of the resulting oxide layer. A Co–28Cr–6Mo alloy was employed as a specimen. The objective of this work is to obtain plasma information for the operating conditions to be optimized for producing a stoichiometric oxide layer on the alloy surface. Helium atomic lines, atomic and ionic lines of oxygen atom, and band heads of oxygen molecule ion were observed in the emission spectra. These intensities drastically changed depending on a mixing ratio of helium–oxygen mixed gas; particularly, the emission intensity of the molecular bands was largely enhanced in the mixed gas plasma compared to the pure oxygen plasma. This band spectrum is assigned to an electronic transition from the 4 Σ g to 4 Π u states of oxygen molecule ion, whose excitation energies are 18–19 eV from the ground state of oxygen molecule. It is thus suggested that a Penning-type ionization process with metastables of helium atom (1s2s 1 S 0, 20.6 eV and 3 S 1, 19.8 eV) is an excitation mechanism how the number density of the corresponding excited state can be elevated in the helium–oxygen mixed gas plasma. This effect, which more populated the excited oxygen species in the plasma, also exerted influence on the resultant oxide layer, such as the chemical composition and the layer thickness. Surface analysis by X-ray photoelectron spectroscopy indicated that an oxide layer consisting of iron and chromium oxides was formed by this plasma treatment, and that chromium atom was enriched in it. The thickness of the oxide layer varied with a mixing ratio of the plasma gas.











Similar content being viewed by others
References
Steinemann SG (1980) Evaluation of biomaterials. Wiley, New York
Long M, Rack HJ (1998) Biomaterials 19:1621–1639
Akahori T, Niinomi M (1998) Mater Sci Eng A 243:237–243
Denkhaus E, Salnikow K (2002) Crit Rev Oncol/Hematol 42:35–56
Yamanaka K, Mori M, Chiba A (2016) Acta Biomater 31:435–447
Mori M, Yamanaka K, Kuramoto K, Ohmura K, Ashino T, Chiba A (2015) Mater Sci Eng C 55:145–154
Ng BS, Annergren I, Soutar AM, Khor KA, Jarfors AEW (2005) Biomaterials 26:1087–1095
Zaffe D, Bertoldi C, Consolo U (2004) Biomaterials 25:3837–3844
Satoh K, Ohtsu N, Sato S, Wagatsuma K (2013) Surf Coat Technol 232:298–302
Satoh K, Sato S, Wagatsuma K (2016) Surf Coat Technol 302:82–87
Broekaert JA (2002) Analytical atomic spectrometry with flames and plasmas. Wiley-VCH Verlag, Weinheim
Wagatsuma K, Hirokawa K (1987) Spectrochim Acta 42B:523–531
Wagatsuma K, Hirokawa K (1988) Anal Chem 60:702–705
Wagatsuma K (2001) Spectrochim Acta 56B:465–486
Wagatsuma K, Hirokawa K (1995) Anal Chim Acta 806:193–200
Mushtaq S, Steers EBM, Pickering JC, Gusarova T, Sm P, Weinstein V (2011) J Anal At Spectrom 26:766–775
Mushtaq S, Steers EBM, Pickering JC, Weinstein V (2014) J Anal At Spectrom 29:2027–2041
Zaidel AN, Prokofev VK, Raikill SM (1961) Tables of spectrum lines. Pergamon Press, Berlin
Moore CE (1949, 1953) Atomic energy levels, vols 1 and 2. NBS Circular No. 467, U.S. Government Printing Office, Washington
Pearse RWB, Gaydon AG (1965) The identification of molecular spectra, 3rd edn. Chapman & Hall, London
Seah MP (2001) Surf Interface Anal 31:721–723
Shirley DA (1972) Phys Rev B 5:4709–4714
Proctor A, Sherwood PMA (1982) Anal Chem 54:13–19
Krupenie PH (1972) J Phys Chem Ref Data 1:423–534
von Engel A (1965) Ionized gases. Clarendon Press, Oxford
Zhang L, Kashiwakura S, Wagatsuma K (2011) Spectrochim Acta Part B 66:785–792
Bell KL, Kennedy DJ, Kingston AE (1968) J Phys B 1:218–231
Herzberg G (1950) Molecular spectra and molecular structure. I. Spectra of diatomic molecules. Van Nostrand Reinhold, New York
Briggs D, Seah MP (1983) Practical surface analysis by Auger and X-ray photoelectron spectroscopy. Wiley, Chichester
Hess DW (1984) J Vac Sci Technol A 2:244–252
Szymanowski H, Sobczyk A, Gazicki-Lipman M, Jakubowski W, Klimek L (2005) Surf Coat Technol 200:1036–1040
Acknowledgements
The authors are grateful to Drs. M. Mori and A. Chiba, IMR Tohoku University, Japan, for a gift of the Co–Cr–Mo alloy sample. This work was supported by the Grant-in-Aid for Japan Society for the Promotion of Science Fellowships (No. 267212).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shimazaki, K., Satoh, K. & Wagatsuma, K. Emission Spectrometric Evaluation of a Hollow-Cathode Glow Discharge Plasma with Helium–Oxygen Mixed Gas for Surface Modification of Co–Cr–Mo Alloy. Plasma Chem Plasma Process 37, 1265–1279 (2017). https://doi.org/10.1007/s11090-017-9801-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11090-017-9801-2