Abstract
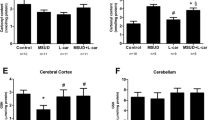
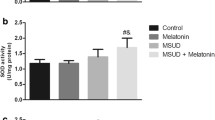
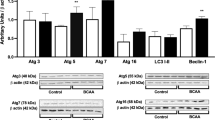
Maple syrup urine disease (MSUD) is an autosomal recessive inborn error of metabolism caused by deficiency of the activity of the mitochondrial enzyme complex branched-chain α-keto acid dehydrogenase (BCKAD) leading to accumulation of the branched-chain amino acids (BCAA) leucine, isoleucine and valine and their corresponding branched-chain α-keto acids. Affected patients present severe brain dysfunction manifested such as ataxia, seizures, coma, psychomotor delay and mental retardation. The mechanisms of brain damage in this disease remain poorly understood. Recent studies have shown that oxidative stress may be involved in neuropathology of MSUD. L-Carnitine (L-Car) is considered a potential antioxidant through its action against peroxidation as a scavenger of reactive oxygen species and by its stabilizing effect of damage to cell membranes. In this study we evaluate the possible neuroprotective in vivo effects of L-Car against pro-oxidative effects of BCAA in cerebral cortex of rats. L-Car prevented lipoperoxidation, measured by thiobarbituric acid-reactive substances, protein damage, measured by sulfhydryl and protein carbonyl content and alteration on catalase and glutathione peroxidase activity in rat cortex from a chemically-induced model of MSUD. Our data clearly show that L-Car may be an efficient antioxidant, protecting against the oxidative stress promoted by BCAA. If the present results are confirmed in MSUD patients, this could represent an additional therapeutic approach to the patients affected by this disease.


Similar content being viewed by others
References
Aebi H (1984) Meth Enzymol 105:121–126
Agarwal A, Said TM (2004) Carnitines and male infertility. Reprod Biomed Online 8:376–384
Aksenov MY, Markesbery WR (2001) Changes in thiol content and expression of glutathione redox system genes in the hippocampus and cerebellum in Alzheimer’s disease. Neurosci Lett 302:141–145
Amaral AU, Leipnitz G, Fernandes CG, Seminotti B, Schuck PF, Wajner M (2010) Alpha-ketoisocaproic acid and leucine provoke mitochondrial bioenergetic dysfunction in rat brain. Brain Res 1324:75–84
Araújo P, Wassermann GF, Tallini K, Furlanetto V, Vargas CR, Wannmacher CMD, Dutra-Filho CS, Wyse ATS, Wajner M (2001) Reduction of large neutral amino acid level in plasma and brain of hyperleucinemic rats. Neurochem Int 38:529–537
Augustyniak A, Skrzydlewska E (2010) L-Carnitine in the lipid and protein protection against ethanol-induced oxidative stress. Alcohol 43:217–223
Barschak AG, Sitta A, Deon M, Oliveira MH, Haeser A, Dutra-Filho CS, Wajner M, Vargas CR (2006) Evidence that oxidative stress is increased in plasma from patients with Maple Syrup Urine Disease. Metab Brain Dis 21:279–286
Barschak AG, Sitta A, Deon M, Barden AT, Schmitt GO, Dutra-Filho CS, Wajner M, Vargas CR (2007) Erythrocyte glutathione peroxidase activity and plasma selenium concentration are reduced in maple syrup urine disease patients during treatment. Int J Dev Neurosci 25:335–338
Barschak AG, Sitta A, Deon M, Barden AT, Dutra-Filho CS, Wajner M, Vargas CR (2008) Oxidative stress in plasma from maple syrup urine disease patients during treatment. Metab Brain Dis 23:71–80
Bridi R, Araldi J, Sgarbi MB, Testa CG, Durigon K, Wajner M, Dutra-Filho CS (2003) Induction of oxidative stress in rat brain by the metabolites accumulating in maple syrup urine disease. Int J Dev Neurosci 21:327–332
Bridi R, Braum CA, Zorzi GK, Wannmacher CM, Wajner M, Lissi EG, Dutra-Filho CS (2005a) Alpha-keto acids accumulating in maple syrup urine disease stimulate lipid peroxidation and reduce antioxidant defences in cerebral cortex from young rats. Metab Brain Dis 20:155–167
Bridi R, Latini A, Braum CA, Zorzi GK, Moacir W, Lissi E, Dutra-Filho CS (2005b) Evaluation of the mechanism involved in leucine-induced oxidative damage in cerebral cortex of young rats. Free Radic Res 39:71–79
Bridi R, Fontella FU, Pulrolnik V, Braun C, Zorzi GK, Coelho D, Wajner M, Vargas CR, Dutra-Filho CS (2006) A chemically-induced acute model of maple syrup urine disease in rats for neurochemical studies. J Neurosci Meth 155:224–230
Chuang DT, Shih VE (2001) Maple syrup urine disease (branched-chain ketoaciduria). In: Scriver CR, Beaudt AL, Sly WL, Valle D (eds) The metabolic and molecular bases of inherited disease. McGraw-Hill, New York, pp 1971–2005
Derin N, Izgut-Uysal VN, Agac A, Aliciguzel Y, Demir N (2004) L-carnitine protects gastric mucosa by decreasing ischemia –reperfusion induced lipid peroxidation. J Physiol Pharmacol 55:595–606
Fontella FU, Gassen E, Pulrolnik V, Wannmacher CMD, Klein AB, Wajner M, Dutra CS (2002) Stimulation of lipid peroxidation in vitro in rat brain by metabolites accumulating in maple syrup urine disease. Metab Brain Dis 17:47–54
Gulcin I (2006) Antioxidant and antiradical activities of L-carnitine. Life Sci 78:803–811
Gutteridge MC, Halliwell B (2010) Antioxidants: molecules, medicines, and myths. Biochem Biophys Res Commun 393:561–564
Halliwell B (2001) Role of free radicals in the neurodegenerative diseases: therapeutic implications for antioxidant treatment. Drugs Aging 18:685–716
Halliwell B (2006) Oxidative stress and neurodegeneration: where are we now? J Neurochem 97:1634–1658
Halliwell B, Gutteridge MC (2007) Free Radicals in Biology and Medicine, 4ªth edn. Oxford University Press Inc., New York
Hoppel C (2003) The role of carnitine in normal and altered fatty acid metabolism. Am J Kidney Dis 41:S4–12
Jouvet P, Rustin P, Taylor DL, Pocock JM, Felderhoff-Mueser U, Mazarakis ND, Sarraf C, Joashi U, Koszma M, Greewood K, Edwards AD, Mehmet H (2000) Branched chain amino acids induce apoptosis in neural cells without mitochondrial membrane despolarization or cytochrome c release: implications for neurological impairment associated with maple syrup urine disease. Mol Biol Cell 11:1919–1932
Kido Y, Tamai I, Ohnari A, Sai Y, Kagami T, Nezu J, Nikaido H, Hashimoto N, Asano M, Tsuji A (2001) Functional relevance of carnitine transporter OCTN2 to brain distribution of L-carnitine and acetyl-L-carnitine across the blood-brain barrier. J Neurochem 79:959–969
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Mancuso C, Bates TE, Butterfield DA, Calafato S, Cornelius C, De Lorenzo A, Dinkova Kostova AT, Calabrese V (2007) Natural antioxidants in Alzheimer’s disease. Expert Opin Investig Drugs 16:1921–1931
Marklund SL (1985) Pyrogallol autoxidation. In: Greenwald RA (ed) Handbook of methods for oxygen radical research. CRC Press, Boca Raton, pp 243–247
Muthuswamy AD, Vedagiri K, Ganesan M, Chinnakannu P (2006) Oxidative stress-mediated macromolecular damage and dwindle in antioxidant status in aged rat brain regions: role of L-carnitine and DL-alpha-lipoic acid. Clin Chim Acta 368:84–92
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358
Pederzolli CD, Mescka CP, Magnusson AS, Deckmann KB, de Souza SE, Sgaravatti AM, Sgarbi MB, Wyse AT, Wannmacher CM, Wajner M, Dutra-Filho CS (2010) N-Acetylaspartic acid impairs enzymatic antioxidant defenses and enhances hydrogen peroxide concentration in rat brain. Metab Brain Dis 25:251–259
Reznick AZ, Packer L (1994) Oxidative damage to proteins: spectrophotometric method for carbonyl assay. Meth Enzymol 233:357–363
Reznick AZ, Kagan VE, Ramsey R, Tsuchiya M, Khwaja S, Serbinova EA, Packer L (1992) Antiradical effects in L-propionyl carnitine protection of the heart against ischemia-reperfusion injury: the he possible role of iron chelation. Arch Biochem Biophys 296:394–401
Ribas GS, Manfredini V, De Mari JF, Wayhs CY, Vanzin CS, Biancini GB, Sitta A, Deon M, Wajner M, Vargas CR (2010a) Reduction of lipid and protein damage in patients with disorders of propionate metabolism under treatment: a possible protective role of l-carnitine supplementation. Int J Dev Neurosci 28:127–132
Ribas GS, Manfredini V, De Marco MG, Vieira RB, Wayhs CY, Vanzin CS, Biancini GB, Wajner M, Vargas CR (2010b) Prevention by L-carnitine of DNA damage induced by propionic and L-methylmalonic acids in human peripheral leukocytes in vitro. Mutat Res 702:123–128
Ribeiro CA, Sgaravatti AM, Rosa RB, Schuck PF, Grando V, Schmidt AL, Ferreira GC, Perry MLS, Dutra-Filho CS, Wajner M (2008) Inhibition of brain energy metabolism by the branched-chain amino acids accumulating in maple syrup urine disease. Neurochem Int 33:114–124
Sgaravatti AM, Rosa RB, Schuck PF (2003) Inhibition of brain energy metabolism by the α-keto acids accumulating in maple syrup urine disease. Biochem Biophys Acta 1639:232–238
Sitta A, Barschak AG, Deon M, Mari J, Barden AT, Vanzin CS, Biancini GB, Schwartz IV, Wajner M, Vargas CR (2009) L-Carnitine blood levels and oxidative stress in treated phenylketonuric patients. Cell Mol Neurobiol 29:211–218
Snyderman SE, Norton PM, Roitman E (1964) Maple syrup urine disease with particular reference to diet therapy. Pediatrics 34:454–472
Stadtman ER, Levine RL (2003) Free-radical mediated oxidation of free amino acids and amino acid residues in proteins. Amino Acids 25:207–218
Tavares RG, Santos CES, Tasca C, Wajner M, Souza DO, Dutra-Filho CS (2000) Inhibition of glutamate uptake into synaptic vesicles of rat brain by the metabolites accumulating in maple syrup urine disease. J Neurol Sci 181:44–49
Treacy E, Clow CL, Reade TR, Chitayat D, Mamer OA, Scriver CR (1992) Maple syrup urine disease: interrelationship between branched chain amino-, oxo- and hydroxylacids implications for treatment association with CNS dysmyelination. J Inherit Metab Dis 15:121–135
Wajner M, Latini A, Wyse AT, Dutra-Filho CS (2004) The role of oxidative damage in the neuropathology of organic acidurias: insights from animal studies. J Inherit Metab Dis 27:427–448
Wendel A (1981) Glutathione peroxidase. Meth Enzymol 77:325–333
Zielke HR, Huang Y, Tildon JT (1996) Elevation of amino acids in the interstitial space of the rat brain following infusion of large neutral amino and keto acids by microdialysis: leucine infusion. Dev Neurosci 18:420–425
Acknowledgements
This work was supported by the research grants from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Programa de Núcleos de Excelência (PRONEX), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS) and FINEP Rede Instituto Brasileiro de Neurociência (IBN-Net #01.06.0842-00).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mescka, C., Moraes, T., Rosa, A. et al. In vivo neuroprotective effect of L-carnitine against oxidative stress in maple syrup urine disease. Metab Brain Dis 26, 21–28 (2011). https://doi.org/10.1007/s11011-011-9238-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-011-9238-x