Abstract
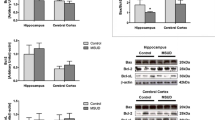
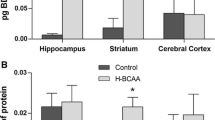
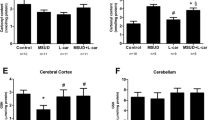
Maple Syrup Urine Disease (MSUD) is an inborn error of metabolism (EIM) biochemically characterized by the tissue accumulation of branched-chain amino acids (BCAA) and their branched-chain alpha-keto acids. The mechanisms by which BCAA and their branched-chain alpha-keto acids lead to the neurological damage observed in MSUD are poorly understood. Mounting evidence has demonstrated that BCAA induce the overproduction of reactive oxygen species, which may modulate several important signaling pathways necessary for cellular homeostasis maintenance, such as autophagy. Taking this into account, we evaluated the effects of BCAA on the autophagic pathway in brain structures of rats submitted to the administration of these amino acids (animal model of MSUD). Our findings showed that BCAA significantly increased the levels of Beclin-1, ATG7, and ATG5 in the cerebral cortex of rats. In addition, BCAA augmented ATG12 levels in the striatum and ATG5 and LC3 I-II in the hippocampus. Therefore, our work demonstrates that the administration of BCAA increases autophagy and autophagic cell death, possibly mediated by the elevated levels of reactive species generated by BCAA.



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and are also available upon request.
References
Baehrecke EH (2003) Autophagic programmed cell death in Drosophila. Cell Death Differ 10:940–945. https://doi.org/10.1038/sj.cdd.4401280
Bar-Yosef T, Damri O, Agam G (2019) Dual role of autophagy in diseases of the central nervous system. Front Cell Neurosci 13:196. https://doi.org/10.3389/fncel.2019.00196
Bridi R, Araldi J, Sgarbi MB et al (2003) Induction of oxidative stress in rat brain by the metabolites accumulating in maple syrup urine disease. Int J Dev Neurosci 21:327–332. https://doi.org/10.1016/S0736-5748(03)00074-1
Bridi R, Braun CA, Zorzi GK et al (2005a) α-keto acids accumulating in maple syrup urine disease stimulate lipid peroxidation and reduce antioxidant defences in cerebral cortex from young rats. Metab Brain Dis 20:155–167. https://doi.org/10.1007/s11011-005-4152-8
Bridi R, Latini A, Braum CA et al (2005b) Evaluation of the mechanisms involved in leucine-induced oxidative damage in cerebral cortex of young rats. Free Radic Res 39:71–79. https://doi.org/10.1080/10715760400022350
Bridi R, Fontella FU, Pulrolnik V et al (2006) A chemically-induced acute model of maple syrup urine disease in rats for neurochemical studies. J Neurosci Methods 155:224–230. https://doi.org/10.1016/J.JNEUMETH.2006.01.005
Chuang DT, Shih VE, Wynn M (2001) Maple syrup urine disease (branched chain ketoaciduria). In: Scriver C, Beaudet A, Sly W, Valle D (eds) The metabolic and molecular bases of inherited disease. McGraw-Hill, New York, pp 1971–2005
Chuang DT, Chuang JL, Wynn RM (2006) Lessons from genetic disorders of branched-chain amino acid metabolism. J Nutr 136:243–249. https://doi.org/10.1093/jn/136.1.243s
Chuang D, Shih V, Wynn R (2020) Maple syrup urine disease (branched chain ketoaciduria). In: Valle D, Antonarakis S, Ballabio A, et al. (eds) The Online Metabolic and Molecular Bases of Inherited Disease. McGraw-Hill, New York
Clarke PGH, Puyal J (2012) Autophagic cell death exists. Autophagy 8:867–869. https://doi.org/10.4161/auto.20380
Farias HR, Gabriel JR, Cecconi ML et al (2021) The metabolic effect of α-ketoisocaproic acid: in vivo and in vitro studies. Metab Brain Dis 36:185–192. https://doi.org/10.1007/S11011-020-00626-Y
Filippone A, Esposito E, Mannino D et al (2022) The contribution of altered neuronal autophagy to neurodegeneration. Pharmacol Ther 238. https://doi.org/10.1016/J.PHARMTHERA.2022.108178
Filomeni G, De Zio D, Cecconi F (2015) Oxidative stress and autophagy: the clash between damage and metabolic needs. Cell Death Differ 22:377. https://doi.org/10.1038/CDD.2014.150
Fontella FU, Gassen E, Pulrolnik V et al (2002) Stimulation of lipid peroxidation in vitro in rat brain by the metabolites accumulating in maple syrup urine disease. Metab Brain Dis 17:47–54. https://doi.org/10.1023/A:1014004414733
Frazier DM, Allgeier C, Homer C et al (2014) Nutrition management guideline for maple syrup urine disease: an evidence- and consensus-based approach. Mol Genet Metab 112:210–217. https://doi.org/10.1016/J.YMGME.2014.05.006
Funderburk SF, Wang QJ, Yue Z (2010) The Beclin 1-VPS34 complex - at the crossroads of autophagy and beyond. Trends Cell Biol 20:355–362. https://doi.org/10.1016/J.TCB.2010.03.002
Gibson GE, Blass JP (1976) Inhibition of acetylcholine synthesis and of carbohydrate utilization by maple-syrup-urine disease metabolites. J Neurochem 26:1073–1078. https://doi.org/10.1111/J.1471-4159.1976.TB06988.X
Hayat M (2017) Overview of autophagy. In: Hayat M (ed) Autophagy: Cancer, Other Pathologies, Inflammation, Immunity, Infection, and Aging, vol 12. Academic Press, pp 3–123
He C, Klionsky DJ (2009) Regulation mechanisms and signaling pathways of autophagy. Annu Rev Genet 43:67–93. https://doi.org/10.1146/ANNUREV-GENET-102808-114910
Herber S, Schwartz IVD, Nalin T et al (2014) Maple syrup urine disease in Brazil: a panorama of the last two decades. J Pediatr (Rio J) 91:292–298. https://doi.org/10.1016/j.jpedp.2015.03.006
Itakura E, Mizushima N (2010) Characterization of autophagosome formation site by a hierarchical analysis of mammalian Atg proteins. Autophagy 6:764–776. https://doi.org/10.4161/AUTO.6.6.12709
Kabeya Y, Mizushima N, Ueno T et al (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 19:5720. https://doi.org/10.1093/EMBOJ/19.21.5720
Levine B, Kroemer G (2019) Biological Functions of Autophagy Genes: A Disease Perspective. Cell 176:11–42. https://doi.org/10.1016/J.CELL.2018.09.048
Levonen AL, Hill BG, Kansanen E et al (2014) Redox regulation of antioxidants, autophagy, and the response to stress: implications for electrophile therapeutics. Free Radic Biol Med 71:196–207. https://doi.org/10.1016/J.FREERADBIOMED.2014.03.025
Liang JH, Jia JP (2014) Dysfunctional autophagy in Alzheimer’s disease: Pathogenic roles and therapeutic implications. Neurosci Bull 30:308–316. https://doi.org/10.1007/s12264-013-1418-8
Lowry OH, Rosebrogh NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275. https://doi.org/10.1016/s0021-9258(19)52451-6
Luo F, Sandhu AF, Rungratanawanich W et al (2020) Melatonin and Autophagy in Aging-Related Neurodegenerative Diseases. Int J Mol Sci 21:1–31. https://doi.org/10.3390/IJMS21197174
Mizushima N, Kuma A, Kobayashi Y et al (2003) Mouse Apg16L, a novel WD-repeat protein, targets to the autophagic isolation membrane with the Apg12-Apg5 conjugate. J Cell Sci 116:1679–1688. https://doi.org/10.1242/JCS.00381
Murray R, Granner D, Mayes P, Rodwell V (2006) Harper: bioquímica, 27a. Atheneu, São Paulo
Nemoto T, Tanida I, Tanida-Miyake E et al (2003) The mouse APG10 homologue, an E2-like enzyme for Apg12p conjugation, facilitates MAP-LC3 modification. J Biol Chem 278:39517–39526. https://doi.org/10.1074/JBC.M300550200
Scherz-Shouval R, Elazar Z (2007) ROS, mitochondria and the regulation of autophagy. Trends Cell Biol 17:422–427. https://doi.org/10.1016/J.TCB.2007.07.009
Serra JD, Sánchez FA, Visus FSV (2010) Enfermidades de Orina de Jarabe Arce. In: Sanjurjo P, Baldellou A. Diagnóstico y Tratamiento de las Enfermidades Hereditárias. Madri: Edicciones Ergon, 3th ed
Serra JD, Sánchez F, Visus FS-V, Miñana IV (2014) Enfermedad de orina de jarabe de arce. In: Sanjurgo P, Baldellou A, Couce ML, et al. (eds) Diagnóstico y tratamiento de las enfermedades metabólicas hereditárias, 4a. Ergon, Majadahonda, pp 531–542
Sperringer JE, Addington A, Hutson SM (2017) Branched-Chain Amino Acids and Brain Metabolism. Neurochem Res 42:1697–1709. https://doi.org/10.1007/S11064-017-2261-5
Strauss KA, Morton DH (2003) Branched-chain Ketoacyl Dehydrogenase Deficiency: Maple Syrup Disease. Curr Treat Options Neurol 5:329–341. https://doi.org/10.1007/S11940-003-0039-3
Strauss KA, Wardley B, Robinson D et al (2010) Classical maple syrup urine disease and brain development: principles of management and formula design. Mol Genet Metab 99:333–345. https://doi.org/10.1016/j.ymgme.2009.12.007
Strauss KA, Puffenberger EG, Carson VJ (2020) Maple Syrup Urine Disease - GeneReviews® - NCBI Bookshelf. In: Maple Syrup Urin. Dis. https://www.ncbi.nlm.nih.gov/books/NBK1319/. Accessed 2 Aug 2022
Wajner M, Vargas C (2002) Aminoacidopatias e Acidemias orgânicas. In: Fonseca L, Piaretti G, Xavier C (eds) Compêndio de Neurologia Infantil, 2a. Medsi, Rio de Janeiro, p 984
Xie Z, Klionsky DJ (2007) Autophagosome formation: core machinery and adaptations. Nat Cell Biol 9:1102–1109. https://doi.org/10.1038/NCB1007-1102
Acknowledgements
This research was supported by the Coordination for the Improvement of Higher Education Personnel (CAPES), the Brazilian National Council for Scientific and Technological Development (CNPq), Santa Catarina State Research and Innovation Support Foundation (Fapesc), and the University of Southern Santa Catarina (UNESC).
Funding
This research was supported by the (CAPES), the Brazilian National Council for Scientific and Technological Development (CNPq), Santa Catarina State Research and Innovation Support Foundation (Fapesc) and University of Southern Santa Catarina (UNESC).
Author information
Authors and Affiliations
Contributions
Conception and design of the research: Hemelin Resende Farias, Emílio Luiz Streck. Acquisition of data: Karoline Teixeira Fermo, Isabela da Silva Lemos, Hemelin Resende Farias, Marina Peyrot Rosso. Analysis and interpretation of the data: Isabela da Silva Lemos, Hemelin Resende Farias. Statistical analysis: Hemelin Resende Farias. Obtaining financing: Emílio Luiz Streck. Writing of the manuscript: Karoline Teixeira Fermo, Hemelin Resende Farias, Pauline Souza Effting, Emílio Luiz Streck. Critical revision of the manuscript for intellectual content: Pauline Souza Effting, Guilhian Leipnitz, Emílio Luiz Streck.
Corresponding author
Ethics declarations
Ethics approval
All procedures were performed according to the Brazilian guidelines for the use of animals in research and approved by the local Ethics Committee (CEUA protocol #77/2018/1).
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fermo, K.T., da Silva Lemos, I., Farias, H.R. et al. Branched-chain amino acids (BCAA) administration increases autophagy and the autophagic pathway in brain tissue of rats submitted to a Maple Syrup Urine Disease (MSUD) protocol. Metab Brain Dis 38, 287–293 (2023). https://doi.org/10.1007/s11011-022-01109-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-022-01109-y