Abstract
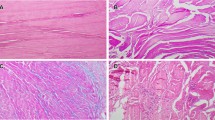
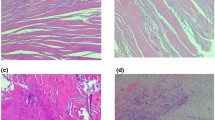
Tendonosis is characterized by an increase in collagen type III relative to collagen type I, but the precise ratio of collagen type I to collagen type III in pathologic shoulder tendons has not been reported. A tendon continuously bathed in an OA synovial environment may exhibit histomorphologic differences as compared to those from shoulders with a preserved articular surface. The purpose of this study was to evaluate the biochemical and histological differences in the proximal portion of the long head of the biceps tendon (LHBT) from a non-arthritic versus an arthritic glenohumeral joint. Sixteen patients undergoing shoulder surgery were prospectively enrolled in the study. Group-1 consisted of patients with glenohumeral OA and Group-2 consisted of patients without OA. The LHBT was tenodesed and excised tendon was fixed, embedded, sectioned (5 µm), deparaffinized, and used for histology (H&E and Masson trichrome staining) and immunofluorescence analysis to determine levels of collagen III, MMP-1, MMP-9, TIMP-1, and TIMP-2. Compared to the patients without arthritis, group-1 exhibited significant inflammation with extracellular matrix disorganization and significantly higher (P < 0.05) collagen III, MMP-2, and MMP-9 levels. No considerable difference in collagen I was observed between the two groups. TIMP-1 and TIMP-2 were completely absent in both the groups. The level of collagen subtypes varied markedly between LHBT tendons from patients with and without OA. Increased collagen III in the LHBT is associated with glenohumeral arthritis. Measuring collagen type in the LHBT may serve as a useful metric in the diagnosis and treatment of musculoskeletal pathology.





Similar content being viewed by others
Abbreviations
- LHBT:
-
Long head of the biceps tendon
- RCI:
-
Rotator cuff injury
- OA:
-
Osteoarthritis
- ECM:
-
Extracellular matrix
- HIER:
-
Heat-induced epitope retrieval
- MMP:
-
Matrix metalloproteases
- TIMP:
-
Tissue inhibitors of matrix metalloproteases
References
Ahrens PM, Boileau P (2007) The long head of biceps and associated tendinopathy. Br Vol 89–B:1001–1009. https://doi.org/10.1302/0301-620X.89B8.19278
Berenson MC, Blevins FT, Plaas AHK, Vogel KG (1996) Proteoglycans of human rotator cuff tendons. J Orthop Res 14:518–525. https://doi.org/10.1002/jor.1100140404
Lakemeier S, Schwuchow SA, Peterlein CD et al (2010) Expression of matrix metalloproteinases 1, 3, and 9 in degenerated long head biceps tendon in the presence of rotator cuff tears: an immunohistological study. BMC Musculoskelet Disord. https://doi.org/10.1186/1471-2474-11-271
Joseph M, Maresh CM, McCarthy MB et al (2009) Histological and molecular analysis of the biceps tendon long head post-tenotomy. J Orthop Res 27:1379–1385. https://doi.org/10.1002/jor.20868
Mazzocca AD, McCarthy MBR, Ledgard FA et al (2013) Histomorphologic changes of the long head of the biceps tendon in common shoulder pathologies. Arthrosc J Arthrosc Relat Surg 29:972–981. https://doi.org/10.1016/j.arthro.2013.02.002
Thankam FG, Dilisio MF, Agrawal DK (2016) Immunobiological factors aggravating the fatty infiltration on tendons and muscles in rotator cuff lesions. Mol Cell Biochem 417:17–33. https://doi.org/10.1007/s11010-016-2710-5
Sharma P, Maffulli N (2005) Tendon injury and tendinopathy: healing and repair. J Bone Joint Surg Am 87:187–202. https://doi.org/10.2106/JBJS.D.01850
Kannus P, Józsa L (1991) Histopathological changes preceding spontaneous rupture of a tendon. A controlled study of 891 patients. J Bone Joint Surg Am 73:1507–1525
Shishani Y, Streit J, Rodgers M, Gobezie R (2015) Tendinopathy of the long head of the biceps tendon: histopathologic analysis of the extra-articular biceps tendon and tenosynovium. Open Access J Sports Med 6:63. https://doi.org/10.2147/OAJSM.S76325
Carr RM, Shishani Y, Gobezie R (2016) How accurate are we in detecting biceps tendinopathy? Clin Sports Med 35:47–55. https://doi.org/10.1016/j.csm.2015.08.002
Thankam FG, Boosani CS, Dilisio MF et al (2016) MicroRNAs associated with shoulder tendon matrisome disorganization in glenohumeral arthritis. PLoS ONE 11:e0168077. https://doi.org/10.1371/journal.pone.0168077
Thankam FG, Boosani CS, Dilisio MF, Agrawal DK (2017) MicroRNAs associated with inflammation in shoulder tendinopathy and glenohumeral arthritis. Mol Cell Biochem. https://doi.org/10.1007/s11010-017-3097-7
Gupta GK, Agrawal T, DelCore MG et al (2012) Vitamin D deficiency induces cardiac hypertrophy and inflammation in epicardial adipose tissue in hypercholesterolemic swine. Exp Mol Pathol 93:82–90. https://doi.org/10.1016/j.yexmp.2012.04.006
Tang JB (2008) Adeno-associated virus-2-mediated bFGF gene transfer to digital flexor tendons significantly increases healing strength: an in vivo study. JBJS 90:1078. https://doi.org/10.2106/JBJS.F.01188
Thankam FG, Dilisio MF, Dietz NE, Agrawal DK (2016) TREM-1, HMGB1 and RAGE in the shoulder tendon: dual mechanisms for inflammation based on the coincidence of glenohumeral arthritis. PLoS ONE 11:e0165492. https://doi.org/10.1371/journal.pone.0165492
Rao VH, Kansal V, Stoupa S, Agrawal DK (2014) MMP-1 and MMP-9 regulate epidermal growth factor-dependent collagen loss in human carotid plaque smooth muscle cells. Physiol Rep. https://doi.org/10.1002/phy2.224
Loiselle AE, Bragdon GA, Jacobson JA et al (2009) Remodeling of murine intrasynovial tendon adhesions following injury: MMP and neotendon gene expression. J Orthop Res 27:833–840. https://doi.org/10.1002/jor.20769
Bigg HF, Rowan AD, Barker MD, Cawston TE (2007) Activity of matrix metalloproteinase-9 against native collagen types I and III: MMP-9 activity against collagens I and III. FEBS J 274:1246–1255. https://doi.org/10.1111/j.1742-4658.2007.05669.x
Woessner JF (2002) MMPs and TIMPs-an historical perspective. Mol Biotechnol 22:033–050. https://doi.org/10.1385/MB:22:1:033
Ireland D, Harrall R, Curry V et al (2001) Multiple changes in gene expression in chronic human Achilles tendinopathy. Matrix Biol 20:159–169. https://doi.org/10.1016/S0945-053X(01)00128-7
Fu SC, Chan BP, Wang W et al (2002) Increased expression of matrix metalloproteinase 1 (MMP1) in 11 patients with patellar tendinosis. Acta Orthop Scand 73:658–662. https://doi.org/10.1080/000164702321039624
Alfredson H, Lorentzon M, Bäckman S et al (2003) cDNA-arrays and real-time quantitative PCR techniques in the investigation of chronic achilles tendinosis. J Orthop Res 21:970–975. https://doi.org/10.1016/S0736-0266(03)00107-4
Thankam FG, Dilisio MF, Dougherty KA et al (2016) Triggering receptor expressed on myeloid cells and 5′adenosine monophosphate-activated protein kinase in the inflammatory response: a potential therapeutic target. Expert Rev Clin Immunol 12: 1239–1249. https://doi.org/10.1080/1744666X.2016.1196138
Karousou E, Ronga M, Vigetti D et al (2008) Collagens, proteoglycans, MMP-2, MMP-9 and TIMPs in human achilles tendon rupture. Clin Orthop 466:1577–1582. https://doi.org/10.1007/s11999-008-0255-y
Casagrande D, Stains JP, Murthi AM (2015) Identification of shoulder osteoarthritis biomarkers: comparison between shoulders with and without osteoarthritis. J Shoulder Elbow Surg 24:382–390. https://doi.org/10.1016/j.jse.2014.11.039
Kurdziel MD, Moravek JE, Wiater BP et al (2015) The impact of rotator cuff deficiency on structure, mechanical properties, and gene expression profiles of the long head of the biceps tendon (LHBT): Implications for management of the LHBT during primary shoulder arthroplasty: THE IMPACT OF ROTATOR CUFF DEFICIENCY. J Orthop Res 33:1158–1164. https://doi.org/10.1002/jor.22895
Haseeb A, Haqqi TM (2013) Immunopathogenesis of osteoarthritis. Clin Immunol 146:185–196. https://doi.org/10.1016/j.clim.2012.12.011
Acknowledgements
This work was supported by Creighton University LB692 Grant to MF Dilisio from the State of Nebraska. The content of this research article is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have read the journal’s policy on disclosure of potential conflicts of interest and the authors have no other relevant affiliations or financial involvement with any organization or entity with financial interest or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed. All authors declare that there is no conflict. No writing assistance was utilized in the production of this manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Thankam, F.G., Evan, D.K., Agrawal, D.K. et al. Collagen type III content of the long head of the biceps tendon as an indicator of glenohumeral arthritis. Mol Cell Biochem 454, 25–31 (2019). https://doi.org/10.1007/s11010-018-3449-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-018-3449-y