Abstract
Anthropogenic disturbances, including land use change and exotic species, can alter the diversity and dynamics of ant communities. To examine foraging behavior in an urbanized habitat in northern California, we surveyed the presence of 9 ant species on 876 trees across 4 seasons during both day and night in a 9.5-hectare urbanized oak-exotic woodland. Ants were more likely to be observed on native, evergreen trees, suggesting that native evergreen species may help maintain ant diversity. Species showed clear patterns of temporal partitioning of foraging activity. Ant species varied in their use of native evergreen Quercus agrifolia trees across season and day/night axes. Of the 3 ant species most frequently observed, Camponotus semitestaceus was most active during spring and summer nights, Formica moki was most active during spring and summer days, and Prenolepis imparis was most active during both day and night during fall and winter. Liometopum occidentale was the second most active species during summer day and night, and winter day. Our findings demonstrate that an oak-exotic urban woodland in Northern California was able to maintain a native ant community, and strong temporal partitioning within that community.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Anthropogenic disturbances, such as pollution, climate change, habitat modification, and invasive species, can decrease ant species diversity and alter community dynamics (Folgarait 1998; Holway 1998; Floren et al. 2008) and create novel ecosystems (Lach et al. 2010; Majer et al. 2013; Stahlschmidt and Johnson 2018). For example, unique assemblages of native and exotic tree species are often associated with urbanization (Kunick 1987; Jim 1993; Sjöman et al. 2012; Aronson et al. 2015; Avolio et al. 2015). Native ant biodiversity in northern California is strongly linked to urbanization; disturbed, urbanized habitats have lower native ant species richness but support higher levels of invasive ant species (Vonshak and Gordon 2015).
Urban habitats can alter resource availability and affect ants’ nutritional preferences (Stahlschmidt and Johnson 2018). Here we extend previous studies of the effects of urbanization on California ant communities (Vonshak and Gordon 2015; Stahlschmidt and Johnson 2018), to ask whether California native ant species preferentially occupy native or exotic trees, and deciduous or evergreen trees. Urban ecosystems often contain exotic plant species (Avolio et al. 2015) and assemblages of native and exotic plants vary heterogeneously across urban ecosystems (Aronson et al. 2015). Many ant species have strong associations with native host plant species, suggesting that ants may prefer to use native plants due to existing coevolutionary relationships (Beattie 1985; Hölldobler and Wilson 1990; Rosumek et al. 2009). Conversely, ants may preferentially use exotic plants over native plant species in cases where the exotic plants harbor a higher abundance of honeydew-producing hemipterans (Lescano and Farji-Brener 2011). We studied ants’ occupancy patterns on native and exotic trees in an oak-exotic urban woodland to determine how these trends may extend to an urbanized ecosystem.
Ants often rely on stable populations of sap-sucking hemipterans for honeydew (Bluthgen et al. 2000; Davidson et al. 2003; Styrsky and Eubanks 2007). In seasonal forests, evergreen trees can play a key role in supporting populations of sap-sucking insects (Silva et al. 2017). Evergreen and deciduous plants exhibit different leaf traits (Pringle et al. 2011; Piper and Fajardo 2014), and herbivorous insects may prefer evergreen trees (Silva et al. 2017). We examined ants’ occupancy patterns on evergreen vs. deciduous trees, as well as native vs. exotic trees, to determine which type of trees are most important to conserving ant diversity. We also address a knowledge gap on ants’ occupancy patterns on trees in modified, urban ecosystems.
Ants may partition shared tree resources across niche axes. Observational studies can provide information about how species differ in resource use, and can suggest which niche axes may be important in preventing direct competition for resources (Hutchinson 1957; MacArthur 1958). In modified, urban ecosystems, the axes used for partitioning in a natural ecosystem may or may not be present. In a California urban ant community, Stahlschmidt and Johnson (2018) found that seasonality and species-level differences influenced nutritional, thermal, and spatial niche axes. Relatively few previous studies have examined how ant communities partition resource use in modified ecosystems (Bestelmeyer 2000; Carval et al. 2016).
To examine partitioning in an ant community in a modified ecosystem, we investigated whether ant species in an oak-exotic urban woodland showed temporal partitioning in foraging activity. In native ant communities in natural ecosystems, ant species partition foraging according to season (Fellers 19871989; Stuble et al. 2013), time of day (Houadria et al. 2015; Narendra et al. 2016), temperature (Cerda et al. 1997; Bestelmeyer 2000), and space (Albrecht and Gotelli 2001; Wittman et al. 2010). Some of these partitioning mechanisms may persist even in heavily modified agroecosystems (Bestelmeyer 2000; Carval et al. 2016). The question remains whether temporal partitioning will persist in urbanized ecosystems, which share some types of disturbance with agroecosystems (e.g. higher temperatures, Oke 1973; Parida et al. 2008)), but may have some distinct types of disturbance (e.g. high light levels at night in urban areas, (Hopkins et al. 2018)).
Within our study site, an oak-exotic urban woodland, the most common tree is Quercus agrifolia, the coast live oak, a native evergreen species (Kershner et al. 2008). Critically, Quercus agrifolia is vulnerable to sudden oak death (Phytophthora ramorum), a pathogen of concern in northern California with the potential to spread onto our study site (Rizzo and Garbelotto 2003). Oaks with symptoms of P. ramorum infection harbored a lower abundance of phytophagous insects than asymptomatic oaks, suggesting that oak health may affect insect community dynamics (Apigian et al. 2005). Since native oaks help maintain biodiversity and influence ecosystem structure (Rizzo and Garbelotto 2003; Mahall et al. 2009; Mclaughlin and Zavaleta 2012), we sought to investigate whether ants preferentially occupy native oaks in urbanized ecosystems, which could lend further support for oak conservation.
In our study system, five common native ant species (Camponotus semitestaceus, Formica moki, Liometopum occidentale, Prenolepis imparis, and Tapinoma sessile) co-occur in an oak-exotic urban woodland. All five ant species forage on trees and most apparently forage on aphid honeydew (Lynch et al. 1980; Gano and Rogers 1983; Holway 1999). Despite the relatively modified plant community at this site, the ant community is similar to those of nearby natural areas (Human and Gordon 1997). The combination of tree and ant communities present at our site allowed us to investigate how a relatively natural, native ant community selected among native/exotic and deciduous/evergreen trees, and how this community partitioned the most heavily occupied trees.
We conducted seasonal surveys of ants in an oak-exotic urban woodland in Northern California to examine whether ants preferred native or exotic trees, and whether ants preferentially used evergreen or deciduous trees. We compared the presence of different ant species to investigate how ant species varied in their daily and seasonal activity. We asked the following questions: (1) Do ants prefer foraging on native evergreen trees, exotic evergreen trees, or native deciduous trees? (2) Within the preferred tree category, do ants differ in foraging activity across season and time of day? We predicted that ants would prefer native evergreen trees and that temporal partitioning would persist in an urban ecosystem.
Materials and Methods
Study Site
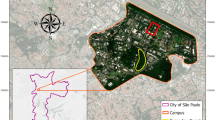
Our study site was located in a 9.53 ha region of an urbanized mixed oak-exotic woodland on the Stanford University campus (37.43, -122.16), (California, USA; Fig. 1a) (Cain and Nilan 2003). The inclusion of this area as part of the Stanford Arboretum led to the formation of an oak-exotic urban woodland inhabited by different types of trees (native evergreen, native deciduous, and exotic evergreen; Fig. 1b). Urbanized features of our study site included roads, and paved and gravel walking paths. The woodland lacked brushy understory.
We surveyed ant activity on tree trunks at our study site. Trees were identified using maps from the Stanford Maps and Records office (340 Bonair Siding Rd., Stanford, CA), which included GPS coordinates, tree species, tree diameter at breast height (DBH), and an assessment of tree health through vigor ratings for each tree. The site has a mixture of native and exotic tree species planted over the last 200 years; previously the site was a mixture of grassland and oak savanna (Cain and Nilan 2003). Of the trees surveyed at our study site, 51.6% were native evergreen, 5.1% were native deciduous, 43.2% were exotic evergreen, and 0.2% were exotic deciduous. We observed ant activity on nine species of native, evergreen trees, although a single species (Quercus agrifolia) represented 93% of observations on native, evergreen trees. We observed ants on two species of native, deciduous trees: Quercus lobata (61% of observations), and Quercus douglasii (39%). We observed ants on 23 species of exotic, evergreen trees; typical exotic species included Eucalyptus spp. (31% of observations), Olea europaea (22%), and Schinus molle (15%).
Data on tree health were obtained from the Stanford Maps and Records Office. Tree vigor ratings were previously assigned by Stanford University arborists using industry standards to rank each tree’s health based on the quality of the tree’s canopy and wood. A tree with fresh leaves in its canopy, expanding foliage, and healthy wood was considered to have a “good” vigor rating. A tree with an abundance of dying canopy foliage and peeling bark was considered to have a “poor” vigor rating. A tree with an appearance intermediate between good and poor was considered to have a “fair” vigor rating. Vigor ratings were collected between July 11th, 2014 and July 8th, 2015 and were 1–3 years old at the time of ant observations. We removed all trees with a “dead” vigor rating from our dataset prior to analysis.
Common ant species, inhabiting > 10 trees in any one season, included Camponotus semitestaceus (CS), Formica moki (FM), Liometopum occidentale (LO), Prenolepis imparis (PI), and Tapinoma sessile (TS). Rarer ant species included Crematogaster coarctata (CC) (1 observation), Camponotus laevigatus (2 observations), Camponotus vicinus (CV) (1 observation), and Pheidole californica (PC) (1 observation). We observed Linepithema humile (LH) 18 times total during spring, summer, and fall surveys in < 10 trees on the northwestern edge of the survey region, which adjoined a small patch of irrigated lawn. The rest of our field site did not appear to be irrigated.
Field Surveys
Four seasonal surveys, each lasting up to 2 weeks, were conducted in 2016–2017 during each season: Summer (July 29th, 2016 – August 8th, 2016), Fall (November 12th, 2016 –November 13th), Winter (February 22nd, 2017 – February 26th, 2017, and Spring (May 12th, 2017 – May 19th, 2017). We surveyed each of 876 trees twice during each season, once during the day and once at night, for a total of 7008 individual tree observations. We did not survey on rainy days as ants were not active, and we did not survey in the hour before or after sunrise and sunset to avoid surveying at intermediate light levels. Daytime observations were made at all times of day, while the majority of nighttime observations were made in the first few hours after darkness fell. We recorded day/night as a binary factor and did not consider any more granular distinctions within night or day. We eliminated a small number of trees from the sample set that died between surveys for unknown reasons. For about 20 trees, growth around the base was too dense to allow observation, and those trees were not included in our final analysis.
During each survey, we observed the bottom 2 m of the tree trunk for approximately 30 s—1 min. Observations were made by the authors and six research assistants. Ants were identified to species in the field or after observations using specimens collected at the time of observation. In some cases, we were able to observe ants (Liometopum occidentale, Prenolepis imparis) directly tending aphids and harvesting honeydew. On other occasions, when ants were foraging inaccessibly in the tree canopy, we sometimes observed ants returning down the trunk of trees with abdomens noticeably swollen with clear liquid (assumed to be honeydew).
Data Analysis
All statistical tests were performed in RStudio Version 1.2.5033. For each individual tree, we defined seasonal ant presence/absence as the presence or absence of at least one ant on that tree in a given season (combining daytime and nighttime observations). To examine whether ants tended to occupy trees of a certain type overall, we constructed a generalized linear mixed model with a binomial distribution: Seasonal Ant Presence/Absence ~ DBH + Tree_Health + Tree_Native + Tree_Evergreen + Tree_Native:Tree_Evergreen + (1|Tree_Genus). Our study area included 25 distinct tree genera, all of which contained only native or only exotic species. We found only one exotic deciduous tree present at the study site, so we excluded this category from analysis.
Because we found that ants tended to occupy native evergreen trees, we then examined whether ant species differed in their use of Quercus agrifolia, the most abundant native evergreen tree, across season and day/night. We constructed a generalized linear mixed model with a binomial distribution: Ant Presence/Absence ~ Season + Day/Night + Ant Species + Season:Ant Species + Day/Night:Ant Species + (1|Individual Tree). For this temporal model, we used the unique identifier number of each individual tree as a random effect to control for observations made of the same tree across time. For the temporal model, we included the five most abundant species: Camponotus semitestaceus (CS), Formica moki (FM), Liometopum occidentale (LO), Prenolepis imparis (PI), and Tapinoma sessile (TS). We excluded any ant species from the temporal model that we observed on fewer than 25 trees across all surveys on the assumption that we would have been unlikely to detect temporal activity patterns without a minimum number of observations.
We generated both the tree trait and the temporal partitioning models using the ‘glmmTMB’ package (Brooks et al. 2017). We used the Akaike Information Criterion (AIC) test to evaluate whether our final temporal model differed significantly from models with the season-species interaction dropped and the day/night-species interaction dropped respectively. We further compared models using the Maximum Likelihood Ratio Test. We ran Dunn post-hoc tests with a Bonferroni correction on both the tree trait and temporal models using the package “dunn.test” (Dinno 2017). We used chi-squared tests to determine if occupancy of deciduous and evergreen trees differed within each season and used a Benjamini–Hochberg correction to account for multiple tests.
Results
Question 1: Do Ants Prefer to Occupy Native Evergreen Trees, Exotic Evergreen Trees, or Native Deciduous Trees?
Ants of all species tended to occupy a higher mean proportion of native evergreen trees (81.7% seasonally occupied), than native deciduous (23.3%) and exotic evergreen trees (57.7%; Fig. 2a). Ants occupied native evergreen trees significantly more often than native deciduous trees (Dunn test; p = 0.004), while ant occupation of exotic evergreen trees did not significantly differ from either category of native tree (Fig. 2a). The generalized linear mixed binomial model yielded a significant interaction of evergreen and native (GLMM; p = 0.015).
Ants were more likely to occupy healthier trees (Dunn Test; p = 0.09; Fig. 2b), although this trend was not statistically significant. Ants occupied 73.7% of trees with a good vigor rating, 66.5% of trees with a fair vigor rating, and 50.0% of trees with a poor vigor rating. Ants did not significantly differ in their seasonal occupancy of trees of differing sizes as measured by diameter at breast height (Dunn Test; p = 0.2; Fig. 2c).
Ants occupied a significantly higher proportion of evergreen trees than deciduous trees in every season (chi-squared tests, p < 0.001 for all seasons, Fig. 3). The proportional difference between deciduous and evergreen tree occupancy was greatest during our winter survey. During the winter, when deciduous trees did not have leaves, evergreen trees were 8.8x more likely to be occupied by ants compared to deciduous trees. During fall, when deciduous trees were in the process of losing leaves, evergreen trees were 2.6x more likely to be occupied by ants than deciduous trees. During spring and summer, when deciduous trees had leaves, evergreen trees were 2.1 × and 3.1 × more likely to be occupied by ants than deciduous trees respectively.
Proportion of evergreen and deciduous trees occupied by ants during summer, fall, winter, and spring. Deciduous trees mostly had leaves during the spring and summer survey, no leaves during the winter survey, and were in the process of losing leaves during the fall survey. *** represents significant differences, chi squared test, p < 0.001 level
Question 2: In Native Evergreen Trees, Do Ants Differ in Foraging Activity Across Season and Time of Day?
Ant occupation of native evergreen trees differed across seasons and time of day (Fig. 4). Adding an ant species x season interaction and an ant species x day/night interaction both improved the final binomial generalized linear model (∆AIC of -306 and -154 respectively; complete model AIC = 1441). Different ant species were observed at different rates in different seasons (season:species interaction, p < 0.001) as well as different times of day (day/night:species interaction, p < 0.001).
Proportion of trees in which each species was observed for day and night, for: (a) Summer, (b) Fall, (c) Winter, (d) Spring. Data are shown for Camponotus semitestaceus (CS), Formica moki (FM), Liometopum occidentale (LO), Prenolepis imparis (PI), and Tapinoma sessile (TS). Gray bars represent observations for day, and black bars represent observations for night. Different letters represent significant differences from Dunn tests at the p < 0.05 level
During summer days, Formica moki was the species most often observed, in 19.8% of trees (Fig. 4a). During summer nights, however, Camponotus semitestaceus was most often observed, in 54.9% of trees (Fig. 4a). Liometopum occidentale exhibited weaker temporal tree occupancy patterns, but occupied slightly more trees on summer days and nights than during other seasons (Fig. 4a-d). Prenolepis imparis was more active during the night than during the day in winter and spring and was very active during both night and day in fall (Fig. 4b-d). During the fall, P. imparis was the most commonly observed species, both during the day (63.1% of trees) and during the night (65% of trees; Fig. 4b). Similarly, during the winter, P. imparis was the most commonly observed species, both during the day (33.1% of trees) and during the night (45%; Fig. 4c). During spring days, P. imparis was observed in 22.7% of trees and F. moki was observed in 22.7% of trees (Fig. 4d). During spring nights C. semitestaceus was the most commonly observed species (48.9% of trees), followed by P. imparis (28.4% of trees; Fig. 4d). Activity in Tapinoma sessile did not exhibit significant seasonal or daily variation. All post hoc Dunn test results are presented in Fig. 4.
Discussion
Native ant species in an urbanized oak-exotic woodland preferentially occupy native evergreen trees, and temporally partition foraging activity among seasons and from day to night. These results indicate that temporal resource partitioning persists in an urbanized ecosystem, building off of previous studies that found that ant communities maintained temporal partitioning in human-modified agroecosystems (Bestelmeyer 2000; Carval et al. 2016).
Ants preferred foraging on native evergreen trees, suggesting that the distribution of honeydew resources may differ between evergreen and deciduous, as well as native and exotic trees. We saw ants of Liometopum occidentale and Prenolepis imparis directly tending aphids and harvesting honeydew, and saw ants of P. imparis returning down the trunks of trees with abdomens noticeably swollen with clear liquid, presumably honeydew. Four of the species we observed, C. semitestaceus, L. occidentale, P. imparis, and T. sessile have been demonstrated to forage on scale insects for honeydew (Wheeler 1930; Gano and Rogers 1983; Hoey-Chamberlain et al. 2013). Our finding that ants preferred to occupy native trees is consistent with previous work that found that exotic trees may contain a lower diversity of honeydew producing hemipterans (Southwood et al. 1982).
At our field site, we observed many ant colonies to remain in the same place throughout the year. Throughout each season, ants consistently occupied a greater proportion of evergreen trees than deciduous trees, suggesting that foraging on evergreen trees could provide more consistent access to food resources. Further work is needed to examine how hemipteran richness and abundance, as well as the nutritional quality of honeydew, vary among native evergreen, native deciduous, and evergreen exotic trees as well as among seasons. Moreover, future research should investigate the dietary niches of these ant species to determine whether they are partitioning the same food resource. Some ant species may prefer foraging on evergreen native trees due to established ant-plant coevolutionary relationships; predator, pathogen and parasite avoidance; and microclimate. Future work should investigate whether dense evergreen trees have a cooler and more humid microclimate that may be more favorable to certain species of ants.
While urbanized ecosystems often have lower native ant species richness and more invasive ant species (Vonshak and Gordon 2015), we observed only a few instances of the invasive Argentine ant Linepithema humile in our surveys, perhaps due to a lack of consistent water availability throughout the year at our study site (Heller et al. 2008). The presence of an intact native evergreen tree resource may help maintain native ant diversity throughout the season. We observed six of eight epigeic ant species that were identified in a survey at a nearby natural preserve, including four of the five most abundant species at our study site (Human and Gordon 1997).
The most common tree species in the urbanized woodland was Quercus agrifolia, a native, evergreen oak species, suggesting that this species may play an important role in maintaining ant biodiversity. Q. agrifolia is susceptible to sudden oak death (Rizzo and Garbelotto 2003), and the sudden oak death pathogen can affect insect assemblages by changing the distribution of honeydew-producing scale insects and ants (Apigian et al. 2005). All ant species occupied healthier trees more often. Although we did not directly test the trees for the sudden oak death pathogen, trees assigned a poor vigor rating had peeling bark and dying foliage similar to the symptoms of sudden oak death. Future research could investigate how ant community dynamics vary between healthy woodlands and woodlands experiencing Q. agrifolia mortality due to sudden oak death. For example, if certain ant species prefer using Q. agrifolia, their populations may decrease in response to decreasing Q. agrifolia abundance. Since Q. agrifolia appears to play a key role in supporting ant diversity in this urbanized oak-exotic woodland, sudden oak death may pose a serious threat to the diversity of native ants in this ecosystem.
Differences among species in thermal tolerance may account for the seasonal and day/night patterns we observed (Fig. 5). The most common ant species occupied trees during different seasons and times of day, suggesting that ant species may be partitioning their use of native evergreen trees across temporal scales. Prenolepis imparis was active all day during fall, winter, and spring, which is consistent with other work showing that P. imparis is more active in cooler temperatures. In other regions of North America, P. imparis is active nocturnally in warm months and diurnally in cold months (Talbot 1943; Fellers 1989). Formica moki is active only during the day in warmer months, which could indicate a preference for warmer temperatures. In contrast, Camponotus semitestaceus was most active during spring and summer nights, consistent with a previous study showing nocturnal activity in this species (Gano and Rogers 1983). These results indicate F. moki and C. semitestaceus are partitioning activity across day/night during the summer. As in previous studies (Smith 1927; Fellers 1989; Barbani 2003), we did not observe strong day/night or seasonal activity patterns in Liometopum occidentale and Tapinoma sessile. Cogni and Freitas (2002) suggest that thermal fluctuations on daily scales influence daily turnover patterns in forest ants. Thus, differences in thermal tolerances between ant species may contribute to the observed patterns of temporal partitioning between ant species.
Other research has suggested that ant communities may be structured by a dominance-discovery tradeoff between species’ ability to discovery food resources and their ability to defend resources from other species through behavioral dominance (Parr and Gibb 2012). Several of the ant species we observed, including F. moki, L. occidentale, and P. imparis, have been described as dominant based on bait assays (Fellers 1987, 1989; Hoey-Chamberlain et al. 2013; Lynch et al. 1980; Wittman et al. 2010). Though we did not offer baits, we rarely observed aggressive behavior such as biting or chasing when more than one species was present on the same tree. Thus, we found no evidence for a dominance-discovery tradeoff (Feener et al. 2008). The ants we observed appeared to be foraging on trees for aphid honeydew, a spatially stable food source for which repeated discovery may not be needed. Parr and Gibb (2012) suggest that ant communities that rely on honeydew do not demonstrate dominance-discovery tradeoffs. However, further research is needed to assess to extent to which aggressive interactions occur between the ant species in our study system.
Our study shows that ants preferentially use native trees in an urbanized oak-exotic woodland community and vary in their foraging behavior for shared tree resources across season and day/night axes. Throughout all seasons, ants consistently occupied a greater proportion of evergreen trees than deciduous trees, and this difference was greatest in the winter when deciduous trees lacked leaves. Preferences for native evergreen trees may be driven by differences in aphid species composition and honeydew availability. Understanding the degree of dietary overlap could elucidate how the quality and availability of honeydew resources may influence ant community structure and temporal activity patterns, as in other ant communities (Anjos et al. 2017). Moreover, few studies have examined ant community patterns in urbanized ecosystems (Vonshak and Gordon 2015; Stahlschmidt and Johnson 2018), underscoring the need for further research on how urbanized ecosystems affect ant partitioning and ant-plant coevolutionary relationships. Our findings demonstrate that the native ant community was maintained within an urbanized oak-exotic woodland, suggesting that modified ecosystems can help conserve native ant diversity.
Data Availability
All data related to this project are available from the Stanford Library Digital Repository at: https://purl.stanford.edu/qz365hg2497.
Code Availability
Code in R associated with this project is available from the Stanford Library Digital Repository at: https://purl.stanford.edu/qz365hg2497.
References
Albrecht M, Gotelli NJ (2001) Spatial and temporal niche partitioning in grassland ants. Oecologia 126:134–141
Anjos DV, Caserio B, Bexende FT et al (2017) Extrafloral-nectaries and interspecific aggressiveness regulate day/night turnover of ant species foraging for nectar on Bionia coriacea. Austral Ecol 42:317–328
Apigian KO, Allen-Diaz B, Stephens S, Sousa W (2005) Forest Disturbance Effects on Insect and Bird Communities: Insectivorous Birds in Coast Live Oak Woodlands and Leaf Litter Arthropods in the Sierra Nevada. Dissertation, University of California Berkeley
Aronson MFJ, Handel SN, La Puma IP, Clemants SE (2015) Urbanization promotes non-native woody species and diverse plant assemblages in the New York metropolitan region. Urban Ecosyst 18:31–45
Avolio ML, Pataki DE, Gillespie TW et al (2015) Tree diversity in southern California’s urban forest: the interacting roles of social and environmental variables. Front Ecol Evol 3:73
Barbani LE (2003) Foraging activity and food preferences of the odorous house ant (Tapinoma sessile Say) (Hymenoptera: Formicidae). Thesis, Virginia Polytechnic Institute
Beattie AJ (1985) The Evolutionary Ecology of Ant-Plant Mutualisms. Cambridge University Press, Cambridge
Bestelmeyer BT (2000) The trade-off between thermal tolerance and behavioural dominance in a subtropical South American ant community. J Anim Ecol 69:998–1009
Bluthgen N, Verhaagh M, Goitia W et al (2000) How plants shape the ant community in the Amazonian rainforest canopy: the key role of extrafloral nectaries and homopteran honeydew. Oecologia 125:229–240
Brooks ME, Kristensen K, van Benthem KJ et al (2017) glmmTMB balances speed and flexibility among packages for zero-inflated generalized linear mixed modeling. The R Journal 9:378–400
Cain J, Nilan R (2003) Every tree, shrub, and vine: Leland Stanford’s arboretum, 1879–1905. Sandstone and Tile 27:15–30
Carval D, Cotté V, Resmond R et al (2016) Dominance in a ground-dwelling ant community of banana agroecosystem. Ecol Evol 6:8617–8631
Cerda X, Retana J, Cros S et al (1997) Thermal disruption of transitive hierarchies in Mediterranean ant communities. J Anim Ecol 66:363–374
Cogni R, Freitas AVL (2002) The ant assemblage visiting extrafloral nectaries of Hibiscus pernambucensis (Malvaceae) in a mangrove forest in Southeast Brazil (Hymenoptera: Formicidae). Sociobiology 40:373–383
Davidson D, Cook S, Snelling R, Chua T (2003) Explaining the abundance of ants in lowland tropical rainforest canopies. Science 300:969–972
Dinno A (2017) dunn.test: Dunn's Test of Multiple Comparisons Using Rank Sums. R package version 1.3.5. https://CRAN.R-project.org/package=dunn.test
Esri “World Light Grey Base [basemap]. Scale Not Given. September 6, 2011. https://www.arcgis.com/home/item.html?id=ed712cb1db3e4bae9e85329040fb9a49. (August 18, 2020)
Feener DH, Matthew OR, Wackford K et al (2008) Geographic variation in resource dominance-discovery in Brazilian ant communities. Ecology 89:1824–1836
Fellers JH (1987) Interference and exploitation in a guild of woodland ants. Ecology 68:1466–1478
Fellers JH (1989) Daily and seasonal activity in woodland ants. Oecologia 78:69–76
Floren A, Freking A, Biehl M, Linsenmair KE (2008) Anthropogenic disturbance changes the structure of arboreal tropical ant communities. Ecography 24:547–554
Folgarait PJ (1998) Ant biodiversity and its relationship to ecosystem functioning: A review. Biodivers Conserv 7:1221–1244
Gano KA, Rogers LE (1983) Colony density and activity times of the ant Camponotus semitestaceus (Hymenoptera: Formicidae) in a shrub steppe community. Ann Entomol Soc Am 76:958–963
Heller NE, Sanders NJ, Shors JW, Gordon DM (2008) Rainfall facilitates the spread, and time alters the impact, of the invasive Argentine ant. Oecologia 155:385–395
Hoey-Chamberlain R, Rust MK, Klotz JH (2013) A Review of the Biology, Ecology, and Behavior of Velvety Tree Ants of North America. Sociobiology 60:1–10
Hölldobler B, Wilson EO (1990) The Ants. Harvard University Press, Cambridge
Holway DA (1998) The effect of Argentine ant invasions on ground-dwelling arthropods in northern California riparian woodlands. Oecologia 116:252–258
Holway DA (1999) Competitive mechanisms underlying the displacement of native ants by the invasive Argentine ant. Ecology 80:238–251
Hopkins GR, Gaston KJ, Visser ME et al (2018) Artificial light at night as a driver of evolution across urban-rural landscapes. Front Ecol Environ 16:1–8
Houadria M, Salas-Lopez A, Orivel J et al (2015) Dietary and temporal niche differentiation in tropical ants - can they explain local ant coexistence? Biotropica 47:208–217
Human KG, Gordon DM (1997) Effects of Argentine ants on invertebrate biodiversity in Northern California. Conserv Biol 11:1242–1248
Hutchinson GE (1957) Concluding remarks. Cold Spring Harb Symp Quant Biol 22:415–427
Jim CY (1993) Trees and landscape of a suburban residential neighbourhood in Hong Kong. Landsc Urban Plan 23:119–143
Kershner B, Tufts C, Nelson G (2008) National Wildlife Federation field guide to trees of North America. Sterling Publishing Company, New York
Kunick W (1987) Woody vegetation in settlements. Landsc Urban Plan 14:57–78
Lach L, Tillberg CV, Suarez AV (2010) Contrasting effects of an invasive ant on a native and an invasive plant. Biol Invasions 12:3123–3133
Lescano MN, Farji-Brener AG (2011) Exotic thistles increase native ant abundance through the maintenance of enhanced aphid populations. Ecol Res 26:827–834
Lynch JF, Balinsky EC, Vail SG (1980) Foraging patterns in three sympatric forest ant species, Prenolepis imparis, Paratrechina melanderi and Aphaenogaster rudis (Hymenoptera: Formicidae). Ecol Entomol 5:353–371
MacArthur RH (1958) Population ecology of some warblers of Northeastern Coniferous forests. Ecology 39:599–619
Mahall BE, Tyler CM, Cole ES, Mata C (2009) A comparative study of oak (Quercus, Fagaceae) seedling physiology during summer drought in southern California. Am J Bot 96:751–761
Majer JD, Heterick B, Gohr T et al (2013) Is thirty-seven years sufficient for full return of the ant biota following restoration? Ecol Process 2:1–12
Mclaughlin BC, Zavaleta ES (2012) Predicting species responses to climate change: demography and climate microrefugia in California valley oak (Quercus lobata). Glob Chang Biol 18:2301–2312
Narendra A, Greiner B, Ribi WA, Zeil J (2016) Light and dark adaptation mechanisms in the compound eyes of Myrmecia ants that occupy discrete temporal niches. J Exp Biol 219:2435–2442
Oke TR (1973) City Size and the Urban Heat Island. Atmos Environ 7:769–779
Parida BR, Oinam B, Patel NR et al (2008) Land surface temperature variation in relation to vegetation type using MODIS satellite data in Gujarat state of India. Int J Remote Sens 29:4219–4235
Parr CL, Gibb H (2012) The discovery-dominance trade-off is the exception, rather than the rule. J Anim Ecol 81:233–241
Piper FI, Fajardo A (2014) Foliar habit, tolerance to defoliation and their link to carbon and nitrogen storage. J Ecol 102:1101–1111
Pringle EG, Adams RI, Broadbent E et al (2011) Distinct leaf-trait syndromes of evergreen and deciduous trees in a seasonally dry tropical forest. Biotropica 43:299–308
Rizzo DM, Garbelotto M (2003) Sudden oak death : endangering California and Oregon forest ecosystems. Front Ecol Env 1:197–204
Rosumek F, Silveira F, de S. Neves F, et al (2009) Ants on plants: a meta-analysis of the role of ants as plant biotic defenses. Oecologia 160:537–549
Silva JO, Leal CRO, Espírito-Santo MM, Morais HC (2017) Seasonal and diel variations in the activity of canopy insect herbivores differ between deciduous and evergreen plant species in a tropical dry forest. J Insect Conserv 21:667–676
Sjöman H, Östberg J, Bühler O (2012) Diversity and distribution of the urban tree population in ten major Nordic cities. Urban For Urban Green 11:31–39
Smith MR (1927) The biology of Tapinoma sessile Say, an important house-infesting ant. Ann Entomol Soc Am 21:307–330
Southwood TRE, Moran VC, Kennedy CEJ (1982) The Richness, Abundance and Biomass of the Arthropod Communities on Trees. J Anim Ecol 51:635–649
Stahlschmidt ZR, Johnson D (2018) Moving targets : determinants of nutritional preferences and habitat use in an urban ant community. Urban Ecosyst 21:1151–1158
Stuble KL, Rodriguez-Cabal MA, McCormick GL et al (2013) Tradeoffs, competition, and coexistence in eastern deciduous forest ant communities. Oecologia 171:981–992
Styrsky JD, Eubanks MD (2007) Ecological consequences of interactions between ants and honeydew-producing insects. Proc R Soc B Biol Sci 274:151–164
Talbot M (1943) Population studies of the ant, Prenolepis imparis say. Ecology 24:31–44
Vonshak M, Gordon DM (2015) Intermediate disturbance promotes invasive ant abundance. Biol Conserv 186:359–367
Wheeler WM (1930) The ant Prenolepis imparis Say. Ann Entomol Soc Am 23:1–26
Wittman SE, Sanders NJ, Ellison AM et al (2010) Species interactions and thermal constraints on ant community structure. Oikos 119:551–559
Acknowledgements
We gratefully acknowledge Angela Gu, Duncan Coleman, Eleanor Glockner, Julie Fukunaga, Katie Lan, and Miranda Vogt for many hours of field work assistance. We thank Rodolfo Dirzo, Daniel Friedman, Maria Wojakowski, Gianalberto Losapio, Marina LaForgia, and Louie Yang for providing advice and assistance with statistical analysis. Marina LaForgia, Leander D.L. Anderegg, Talia Borofsky, Kaleda Denton, Tyler McFadden, Louie Yang, Elizabeth Postema, and Tracie Hayes provided helpful comments on the manuscript. The Stanford Maps and Records Office (340 Bonair Siding, Stanford, CA 94305) provided detailed maps of trees on the Stanford grounds that were invaluable for this project. We would especially like to thank arborist Michael Gladden from the Stanford Maps and Records Office who provided us with information about the tree maps. We thank past anonymous peer reviewers for their helpful feedback on earlier versions of this paper. This project was supported by a Small Grant from the Stanford Office for Undergraduate Advising and Research.
Funding
This project was supported by a Small Grant from the Stanford Office for Undergraduate Advising and Research.
Author information
Authors and Affiliations
Contributions
Study design, data collection, and data analysis were carried out by DM, RN, GL, and DG. Authors DM, RN and DG contributed equally to the writing of this manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
MacArthur-Waltz, D.J., Nelson, R.A., Lee, G. et al. Tree Preference and Temporal Activity Patterns for a Native Ant Community in an Urbanized California Woodland. J Insect Behav 34, 211–222 (2021). https://doi.org/10.1007/s10905-021-09778-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10905-021-09778-w