Abstract
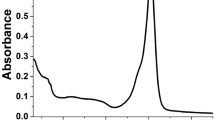
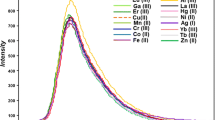
A fluorescent turn-on chemosensor (BA) was constructed by attaching bis(pyridin-2-ylmethyl)-amine (DPA) unit to the BODIPY scaffold. It can give a prominent green/yellow fluorescent response selectivity with each of Zn2+/Hg2+/Cd2+/Ca2+/Mn2+/Pb2+/Al3+. The 1:1 stoichiometry of BA and metal ions was drawn from the analysis of Job’s plot. The limit detection of BA in recognition of Zn2+/Hg2+/Cd2+/Ca2+/Mn2+/Pb2+/Al3+ is ranged in 50.8-146.6 nM. There exists a linear relationship between the fluorescence intensity and concentration of metal ions (Zn2+: 4-15 µM). The mechanism of fluorescence signal “turn-on” is based on the photo induced transfer (PET) in the excited state of BA. The coordinated metal ions significantly weakened the electron-donating ability nitrogen atom in DPA, thus recovering the emission character of BODIPY. The substituted group at the phenyl ring in meso-position of BODIPY scaffold determines the recognizable list of metal ions.











Similar content being viewed by others
Data Availability
No datasets were generated or analysed during the current study.
References
Baroncini M, Silvi S, Credi A (2020) Photo- and redox-driven artificial molecular motors. Chem Rev 120:200–268. https://doi.org/10.1021/acs.chemrev.9b00291
Langton MJ, Beer PD (2014) Rotaxane and catenane host structures for sensing charged guest species. Acc Chem Res 47:1935–1949. https://doi.org/10.1021/ar500012a
Li X, Liu X, Li F (2024) Configuration of super-fast Cu2+-responsive chemosensor by attaching diaminomaleonitrile to BODIPY scaffold for high-contrast fluorescence imaging of living cells. Spectrochim Acta Part A 304:123377. https://doi.org/10.1016/j.saa.2023.123377
Luo S, Zheng B, Dong S (2023) Natural light-regulated switchable self-adhesive supramolecular films for smart windows. Chem Eur J 29:e202301277. https://doi.org/10.1002/chem.202301277
Qiu S, Cui S, Shi F, Pu S (2019) Novel diarylethene-based fluorescent switching for the detection of Al3+ and construction of logic circuit. ACS Omega 4:14841–14848. https://doi.org/10.1021/acsomega.9b01432
Karmakar S, Mardanya S, Maity D, Baitalik S (2016) Polypyridyl-imidazole based Os(II) complex as optical chemosensor for anions and cations and multi-readout molecular logic gates and memory device: experimental and DFT/TDDFT study. Sens Actuators B Chem 226:388–402. https://doi.org/10.1016/j.snb.2015.11.104
Zheng X, Liu X, Liu L, Li X, Jiang S, Niu C, Xie P, Liu G, Cao Z, Ren Y, Qin Y, Wang J (2022) Multi-stimuli-induced mechanical bending and reversible fluorescence switching in a single organic crystal. Angew Chem Int Ed 61:e202113073. https://doi.org/10.1002/anie.202113073
Yuan L, Su Y, Cong H, Yu B, Shen Y (2022) Application of multifunctional small molecule fluorescent probe BODIPY in life science. Dye Pigm 208:110851. https://doi.org/10.1016/j.dyepig.2022.110851
Loudet A, Burgess K (2007) BODIPY dyes and their derivatives: synthesis and spectroscopic properties. Chem Rev 107:4891–4932. https://doi.org/10.1021/cr078381n
Li X, Cai Q, Zhang J, Kim H, Son YA (2020) An electron lock toward the photochromic activity of phenylacetylene appended bisthienylethene. Mol Cryst Liq Cryst 706:141–149. https://doi.org/10.1080/15421406.2020.1743450
Li X, Qian Q, Jiang W (2023) Photo-induced fluorochromism of a star-shaped photochromic dye with 2,4-dimethylthiazole attaching to triangle terthiophene. J Fluoresc 33:1907–1915. https://doi.org/10.1007/s10895-023-03196-1
Takeuchi M, Gnanasekaran K, Friedrich H, Imai H, Sommerdijk NAJM, Oaki Y (2018) Tunable stimuli-responsive color-change properties of layered organic composites. Adv Funct Mater 28:1804906. https://doi.org/10.1002/adfm.201804906
Ren M, Cai W, Xu J (2020) Tailorable dynamics in nonlinear optical metasurfaces. Adv Mater 32:180637. https://doi.org/10.1002/adma.201806317
Rashid U, Chatir E, Sandonas LM, Sreelakshmi PA, Dianat A, Gutierrez R, Cuniberti G, Cobo S, Kaliginedi V (2023) Dithienylethene-based single molecular photothermal linear actuator. Angew Chem Int Ed 62:e202218767. https://doi.org/10.1002/anie.202218767
Ji G, Hou Q, Jiang W, Li X (2024) Investigating the properties of triangle terthiophene and triphenylamine configured propeller-like photochromic dye with ethyne bridge. J Fluoresc (in press, https://doi.org/10.1007/s10895-023-03557-w)
Li X, Zou Y, Heo G, Son YA (2020) Emission shift of an imidazole bridged diethylaminocoumarin and diphenyl. Mol Cryst Liq Cryst 704:48–56. https://doi.org/10.1080/15421406.2020.1741801
Xie P, Zhou Y, Li X, Liu X, Liu L, Cao Z, Wang J, Zheng X (2023) Strong dual-state emission of unsymmetrical and symmetrical thiazolothiazole-bridged imidazolium salts. Chin Chem Lett 34:107582. https://doi.org/10.1016/j.cclet.2022.06.005
Ji G, Hou Q, Zhang J, Li X (2023) Investigation of triangle terthiophene and hydroxyphenylbenzothiazole configured fluorescent dye with a triple bond bridge. J Fluoresc 33:153–159. https://doi.org/10.1007/s10895-022-03049-3
Fang J, Li X, Gao C, Tao J, Li W, Seidu MA (2023) Oxidative cleavage of alkene: a new strategy to construct a mitochondria-targeted fluorescent probe for hydrogen peroxide imaging in vitro and in vivo. Sens Actuators B Chem 395:134504
Chopra T, Sasan S, Devi L, Parkesh R, Kapoor KK (2022) A comprehensive review on recent advances in copper sensors. Coord Chem Rev 470:214704. https://doi.org/10.1016/j.ccr.2022.214704
Li X, Han Y, Sun S, Shan D, Ma X, He G, Mergu N, Park JS, Kim CH, Son YA (2020) A diaminomaleonitrile-appended BODIPY chemosensor for the selective detection of Cu2+ via oxidative cyclization and imaging in SiHa cells and zebrafish. Spectrochim Acta Part A 233:118179. https://doi.org/10.1016/j.saa.2020.118179
Li Y, Liu K, Zhang W, Wang Y, Wang B, Wang Y, Li X (2023) Two 3D ln(III)-MOFs based on phosphineoxide ligand: synthesis, structure luminescent and photocatalytic properties. J Fluoresc 33:2119–2129. https://doi.org/10.1007/s10895-023-03218-y
Liu L, Wang J, Wang X, Wang H, Li M, Wu T, Gao G, Zheng X, Liu G, Fan L, Shen W, Ru G, Zhao Z, Tang B (2023) High-resolution imaging of latent fingerprints through near-infrared organoboron AIEgens. Chin J Chem 41:1465–1470. https://doi.org/10.1002/cjoc.202200677
Ji G, Hou Q, Jiang W, Li X (2023) Investigating the properties of double triangle terthiophene configured dumbbell-like photochromic dye with ethyne and 1,3-butadiene bridge. J Fluoresc 33:1495–1503. https://doi.org/10.1007/s10895-023-03171-w
Guo Z, Par S, Yoon J, Shin I (2014) Recent progress in the development of near-infrared fluorescent probes for bioimaging applications. Chem Soc Rev 43:16–29. https://doi.org/10.1039/C3CS60271K
Li ZQ, Zhou X, Chen DX, Jin J, Lv YY (2024) Facile and highly selective near-infrared emitting fluorescent indicator for hypochlorous acid signaling and bioimaging. Sens Actuators B Chem 398:134752. https://doi.org/10.1016/j.snb.2023.134752
Cao Z, Yang F, Wu D, Wu L, Liu L, Liu G, Li X, Zheng X, Zheng X, Qu D (2023) Supramolecular aggregates constructed by pillar[5]arene-based host–guest interaction with aggregation-induced emission. Polym Chem 14:1318–1322. https://doi.org/10.1039/d3py00026e
Li X, Wang Y, Jia C, Kim H, Son YA (2019) Photochromic reactivity induced by electron distribution: active or inactive. Mol Cryst Liq Cryst 689:83–91. https://doi.org/10.1080/15421406.2019.1597556
Gaggelli E, Kozlowski H, Vaniela D, Valensin G (2006) Copper homeostasis and neurodegenerative disorders (alzheimer’s, prion, and Parkinson’s diseases and amyotrophic lateral sclerosis). Chem Rev 106:1995–2044. https://doi.org/10.1021/cr040410w
Sivaraman G, Iniya M, Anand T, Kotla NG, Sunnapu O, Singaravadivel S, Gulyani A, Chellappa D (2018) Chemically diverse small molecule fluorescent chemosensors for copper ion. Coord Chem Rev 357:50–104. https://doi.org/10.1016/j.ccr.2017.11.020
Zhang J, Zhao X, Liu X, Dong C (2020) Enhanced chemical sensing for Cu2+ based on composites of ZIF-8 with small molecules. RSC Adv 10:13998–14006. https://doi.org/10.1039/C9RA10695B
Li X, Guo X, Chen Y, Cui T, Xing L (2021) Double 3-ethyl-2,4-diemthylpyrrole configured fluorescent dye with fluorine-boron as the bridge. J Fluoresc 31:1797–1803. https://doi.org/10.1007/s10895-021-02819-9
Li X, Yao C, Jiang W (2023) Emission and energy transfer investigation of non-conjugated total carbon configuration between BODIPY and naphthalimide. J Chem Sci 135:65. https://doi.org/10.1007/s12039-023-02181-2
He W, Li X, Kim H, Son YA (2020) Shifting the emission of proton transfer fluorescence with fluorine-boron as the rotation lock. Mol Cryst Liq Cryst 704:41–47. https://doi.org/10.1080/15421406.2020.1741800
Li X, Liao M, Sun J, Heo G, Son YA (2019) Thiophene modulated BODIPY dye as a light harvester. Mol Cryst Liq Cryst 679:127–136. https://doi.org/10.1080/15421406.2019.1597557
Li S, Chang X, Kong X, Wang Q, Zhao F, Han J, Liu Y (2024) A visible BODIPY-based sensor for ‘naked-eye’ recognition of Ag+ and its application on test paper strips. Spectrochim Acta Part A 304:123446. https://doi.org/10.1016/j.saa.2023.123446
Hernández-Contreras J, Madrigal P, Arroyo P, Liu-González M, Gil S, Parra M, Sáez J, Gaviña P (2024) Optical detection of scopolamine and ketamine with a BODIPY-Phen conjugate and Cu(II). Dyes Pigm 221:111806. https://doi.org/10.1016/j.dyepig.2023.111806
Li X, Tian G, Shao D, Xu Y, Wang Y, Ji G, Ryu J, Son YA (2020) A BODIPY based emission signal turn-on probe toward multiple heavy metals. Mol Cryst Liq Cryst 706:38–46. https://doi.org/10.1080/15421406.2020.1743436
Xue X, Fang H, Chen H, Zhang C, Zhu C, Bai Y, He W, Guo Z (2016) In vivo fluorescence imaging for Cu2+ in live mice by a new NIR fluorescent sensor. Dyes Pigm 130:116–121. https://doi.org/10.1016/j.dyepig.2016.03.017
Li X, Li F, Ji G (2023) A fluorescent turn-on sensor toward multiple heavy metal ions based on meso-anisole modified BODIPY scaffold. J Fluoresc 33:631–637. https://doi.org/10.1007/s10895-022-03110-1
Raveendran AV, Sankeerthana P, Jayaraj A, Swamy PCA (2022) Recent developments on BODIPY base chemosensors for the detection of group IIB metal ions. Results Chem 4:100297. https://doi.org/10.1016/j.rechem.2022.100297
Bastug E, Kursunlu AN, Guler E (2020) A fluorescent clever macrocycle: deca-bodipy bearing a pillar[5]arene and its selective binding of asparagine in half-aqueous medium. J Lumin 225:117343. https://doi.org/10.1016/j.jlumin.2020.117343
Kursunlu AN, Ozmen M, Guler E (2019) A novel fluorescent chemosensor for Cu (II) ion: click synthesis of dual-bodipy including the triazole groups and bioimaging of yeast cells. J Fluoresc 29:1321–1329. https://doi.org/10.1007/s10895-019-02456-3
Lin JR, Chu CJ, Venkatesan P, Wu SP (2015) Zinc(II) and pyrophosphate selective fluorescence probe and its application to living cell imaging. Sens Actuators B Chem 207:563–570. https://doi.org/10.1016/j.snb.2014.10.109
Atilgan S, Ozdemir T, Akkaya EU (2008) A sensitive and selective ratiometric near IR fluorescent probe for zinc ions based on the distyryl-bodipy fluorophore. Org Lett 10:4065–4067. https://doi.org/10.1021/ol801554t
Kang G, Son H, Lim JM, Kweon HS, Lee IS, Kang D, Jong H (2012) Functionalized Fe O nanoparticles for detecting zinc ions in living cells and their cytotoxicity. Chem Eur J 18:5843–5847. https://doi.org/10.1002/chem.201200294
Zhao C, Zhang Y, Feng P, Cao J (2012) Development of a borondipyrromethene-based Zn2+ fluorescent probe: solvent effects on modulation sensing ability. Dalton T 41:831–838. https://doi.org/10.1039/C1DT10797F
Wu Y, Peng X, Guo B, Fan J, Zhang Z, Wang J, Cui A, Gao Y (2005) Boron dipyrromethene fluorophore based fluorescence sensor for the selective imaging of zn(ii) in living cells. Org Biomol Chem 3:1387–1392. https://doi.org/10.1039/B501795E
Raveendran AV, Sankeerthana PA, Jayaraj A, Swamy CA (2022) Recent developments on BODIPY based chemosensors for the detection of group IIB metal ions. Results Chem 4:100297. https://doi.org/10.1016/j.rechem.2022.100297
Qin W, Dou W, Leen V, Dehaen W, Auweraer MV, Boens N (2016) A ratiometric, fluorescent BODIPY-based probe for transition and heavy metal ions. RSC Adv 6:7806–7816. https://doi.org/10.1039/C5RA23751C
Song Y, Tao J, Wang Y, Cai Z, Fang X, Wang S, Xu H (2021) A novel dual-responsive fluorescent probe for the detection of copper(II) and nickel(II) based on BODIPY derivatives. Inorg Chim Acta 516:120099. https://doi.org/10.1016/j.ica.2020.120099
Zhu S, Zhang J, Janjanam J, Vegesna G, Luo FT, Tiwari A, Liu H (2013) Highly water-soluble BODIPY-based fluorescent probes for sensitive fluorescent sensing of zinc(ii). J Mater Chem B 1:1722–1728. https://doi.org/10.1039/C3TB00249G
Jia X, Yu X, Zhang G, Liu W, Qin W (2013) Preparation of a fluorescent sensor based on BODIPY-functionalized hydroxyapatite nanoparticles and spectroscopic study of the Cd2+ and Zn2+ complex formation. J Coord Chem 66:662–670. https://doi.org/10.1080/00958972.2013.767896
Jiao L, Yu C, Li J, Wang Z, Wu M, Hao E (2009) ) β-Formyl-BODIPYs from the vilsmeier-haack reaction. J Org Chem 74:7525–7528. https://doi.org/10.1021/jo901407h
Li X, Ji G, Jung JM, Son YA (2016) A BODIPY based highly selective fluorescence turn-on sensor toward VIB and IIB metal ions. Mol Cryst Liq Cryst 636:159–167. https://doi.org/10.1080/15421406.2016.1201406
Wang KP, Lei Y, Zhang SJ, Zheng WJ, Chen JP, Chen S, Zhang Q, Zhang YB, Hu ZQ (2017) Fluorescent probe for Fe(III) with high selectivity and its application in living cells. Sens Actuators B Chem 252:1140–1145. https://doi.org/10.1016/j.snb.2017.07.184
Nie K, Dong B, Shi H, Chao L, Huang Z, Long M, Xu H, Liu Z, Liang B (2019) Pyridyl DPP based soluble nanoaggregates for ratiometric/fluorescent detection of Cu2+/Hg2+ in water. J Lumin 208:408–414. https://doi.org/10.1016/j.jlumin.2018.12.046
Benesi HA, Hildebrand JH (1949) A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J Am Chem Soc 71:2703–2707. https://doi.org/10.1021/ja01176a030
Funding
This work was supported by the National Natural Science Foundation of China (grant no. 21772034) We also thank the financial support from Henan Key Laboratory of Organic Functional Molecules and Drug Innovation.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by X. Liu. The draft of the manuscript was written by X. Li. X. Liu. prepared figures and Schemes. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, X., Liu, X. A Sensitive Probe of Meso-Cyanophenyl Substituted BODIPY Derivative as Fluorescent Chemosensor for the Detection of Multiple Heavy Metal Ions. J Fluoresc (2024). https://doi.org/10.1007/s10895-024-03581-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10895-024-03581-4