Abstract
Echocardiography is a versatile, accurate and noninvasive tool suited to examination of shocked patients. Since the 1980s, intensive care practitioners have used ultrasound widely for hemodynamic evaluation and for cardiac anatomy visualization. This article will describe transthoracic and transesophageal echocardiography, their scope, and the classic windows needed to interpret the examination properly. We will also report the main indications of echocardiography and the corresponding parameters. Finally, we will indicate educational programs and define minimum training that enable self-sufficiency.
Similar content being viewed by others
1 Introduction
Echocardiography has become a true bedside clinical device for full hemodynamic evaluation, monitoring and diagnosis in patients with circulatory failure. It allows real-time anatomical and functional cardiac assessment in very different situations, as in sepsis or acute respiratory distress syndrome (ARDS). Edler et al. [1] first described echocardiography in 1954, as an “ultrasonic reflectoscope for the continuous recording of the movements of heart walls”. Many advances followed, including time motion mode in 1963 [2], real time 2D in 1973 [3], spectral Doppler in 1979 [4] and tissue Doppler in 1989 [5], each time enriching the information available for physicians.
Some pioneers started to use echocardiography in the ICU in the 1980s, especially in sepsis where it was able to detect profound cardiac failure [6–8]. After many years of development and clinical studies in critically ill patients, scientific societies defined critical care echocardiography (CCE) as echocardiography performed by intensivists themselves at the bedside for diagnosis of cardiac failure and hemodynamic monitoring, with 2 different skill levels: basic and advanced [9].
2 Transthoracic (TTE) or transesophageal echocardiography (TEE)?
Transthoracic echocardiography is often used as a first-line approach as it is well tolerated by patients, safe, and widely available [10–12]. It is especially suitable for basic CCE, where intensivists have to perform a quick and focused examination to diagnose or to exclude “gross” cardiac abnormalities, as acute cor pulmonale (ACP), cardiac tamponade or major left ventricular systolic dysfunction. However, TEE is especially accurate in mechanically ventilated patients with ARDS or septic shock where advanced CCE is required. Intensivists have to repeat a full hemodynamic evaluation at various times in different situations with different ventilatory settings [10]. In these situations, TEE has a better diagnostic capability and is more reproducible than TTE. Whereas TTE is totally non-invasive, TEE is considered as semi-invasive because of esophageal intubation, even though most complications have been described in spontaneously breathing patients [13].
3 Main echocardiographic views for hemodynamic evaluation
3.1 TTE
Even though many views are available in cardiology for diagnosis and evaluation of cardiac diseases, few views are necessary in the ICU, as a first step for evaluation and hemodynamic monitoring. The apical four-chamber view allows intensivists to detect right ventricular (RV) dilatation (Fig. 1a). Using the pulsed Doppler, it also allows recording of left ventricular (LV) inflow for evaluation of LV filling pressure (Fig. 1b). From this view, by tilting the probe up, the LV outflow track may be visualized and LV stroke volume calculated (Fig. 1c). The parasternal short-axis view is perfectly suitable for evaluation of LV contraction and for calculating LV fractional area contraction, a surrogate of LV ejection fraction. It also allows detection of paradoxical septal motion (Fig. 2), leading to the diagnosis of ACP when associated with RV dilatation. Finally, a subcostal view visualizes the inferior vena cava (IVC). Whereas a small or virtual IVC (Fig. 3a) is highly suggestive of hypovolemia and the need for fluids in a context of shock, a dilated or congestive IVC is often related to cardiac failure (Fig. 3b).
Apical four-chamber view by a transthoracic approach. a RV dilatation in a patient with acute cor pulmonale. b Pulsed-Doppler at the mitral valve with the E and the A waves, demonstrating a restrictive pattern of flow, reflecting a high LV filling pressure. c Pulsed-Doppler in the LV outflow tract, allowing calculation of the LV stroke volume. RV right ventricle, LV left ventricle
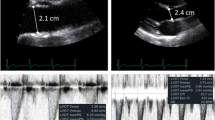
Subcostal view of the inferior vena cava (IVC), coupled with time-motion study. a Very small IVC with significant respiratory variations of its diameter during tidal ventilation, reflecting hypovolemia in this patient in shock. b Dilated IVC without any respiratory variations reflecting the absence of preload responsiveness and occurrence of cardiac failure
3.2 TEE
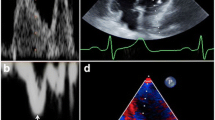
Even though a lot of views may be obtained by TEE using a multiplane probe, from 0 to 180° in each position, 3 views are especially required to monitor hemodynamics in the mechanically ventilated patient: the mid-esophageal 4-chamber view (Fig. 4), the transgastric short-axis view (Fig. 5) and the longitudinal view of the superior vena cava (SVC) (Fig. 6). Whereas the first view gives information similar to that provided by the apical 4-chamber view, the second view is similar to the parasternal short-axis view. The longitudinal view of the SVC is more specific and interesting since SVC respiratory variations have been demonstrated to predict the response to fluids accurately [14]. An inspiratory collapse in a ventilated patient perfectly adapted to the ventilator is highly suggestive of the need for fluids (Fig. 6).
Long-axis view of the superior vena cava by a transesophageal approach, coupled with the time-motion study. a Before volume expansion, note the collapse of the vessel during each tidal ventilation. b After volume expansion, blood pressure was normalized and the collapse disappeared. SVC superior vena cava
4 Functional hemodynamic monitoring: the concept
Over the last few years, a new approach to hemodynamic evaluation called functional hemodynamic monitoring [15] has been developed. It is less based on numbers and invasive tools, and more on qualitative and “functional” assessment, and on prediction of treatment effect. Echocardiography is perfectly adapted to this new approach. Vieillard-Baron et al. [16] have validated such a functional approach in patients with septic shock using TEE. The 3 previously described views were fully able to detect LV systolic dysfunction, preload responsiveness and RV failure [16]. More recently, Bouferrache et al. [17] reported how this functional approach is weakly related to the Surviving Sepsis Campaign guidelines for fluids and inotropic drug management.
5 Some usual indications of echocardiography in the ICU
5.1 Assessment of fluid responsiveness
Hypovolemia is a crucial problem for intensivists. It leads to organ ischemia and potentially organ failure. However, fluid overload also has marked deleterious effects, as pulmonary edema and finally increased mortality [18, 19]. This explains why intensivists progressively move from a “fluid challenge” approach to a prediction of fluid responsiveness. In this field, new echocardiographic parameters have been validated, based on heart–lung interactions, i.e. the IVC distensibility index (Fig. 3a) [20, 21], the SVC collapsibility index [14] (Fig. 6) and respiratory variations of the velocity–time integral of aortic flow [22]. The latter requires that patients be in sinus rhythm.
Static parameters, as cardiac filling pressure evaluation by pulsed Doppler at the mitral valve, are not accurate in detecting the need for fluids, but may be used to evaluate tolerance of volume expansion and to exclude (or diagnose) a cardiogenic factor in a patient with acute respiratory failure (Fig. 1b).
5.2 Adaptation of respiratory settings to RV function
Acute cor pulmonale occurs when the RV is overloaded [23, 24]. Common causes in the ICU are ARDS, “aggressive” mechanical ventilation and massive pulmonary embolism. Because ACP has been reported to have a significant impact on prognosis in ARDS patients [24], its demonstration may lead to adaptation of the ventilatory settings to RV function, by decreasing the plateau pressure, and by limiting the PEEP and hypercapnia [25, 26]. In this setting in ARDS patients, it is now recommended to perform at least one echocardiographic examination per day during the first 3 days to evaluate RV function and to adapt the ventilator.
5.3 Hemodynamic management in septic shock
Septic shock is a complex process where different mechanisms, alone or associated, may be responsible for circulatory failure throughout disease progression: hypovolemia, vasoplegia and septic cardiomyopathy. As discussed above, echocardiography is perfectly adapted to optimize volemia. But echocardiography has also proven valuable in accurately detecting LV systolic dysfunction in patients with septic shock [27]. In our usual practice, we use serial echocardiographic examinations, in association with invasive blood pressure monitoring and lactate assay, to adapt and to optimize hemodynamic resuscitation.
6 Acquisition of the skills required for CCE
Echocardiography is as an indispensable tool in the ICU for diagnosis and also for hemodynamic monitoring, and the question then is how can intensivists acquire the necessary skills? Recently, a round table of experts representing most of the international societies of intensive care medicine in the world proposed guidelines [28]. Briefly, basic CCE requires intensivists to participate in a 10-hour training course and to perform at least 30 fully supervised TTE examinations. Competence-based training in basic CCE should be included in the curriculum of all intensivists. For advanced CCE, intensivists must complete 40 h of course work and perform 150 fully supervised TTE and 25–50 TEE examinations. A process of certification, accreditation, or delivery of a diploma validating the acquisition of the required skills is essential.
In France, two types of certifications have been developed for intensivists. The first was initially intended for cardiologists, but was enlarged to include intensivists through a collaboration between the societies of cardiology and of intensive care. It is a 2-year training program that teaches the skills needed by future trainers. The second certification is specifically intended for intensivists, and is awarded after a one-year course.
7 Conclusion
Echocardiography should be widely used first line in the ICU. It safely provides hemodynamic information about acutely ill patients, as well as functional information about the condition of the heart. It can also be used as a diagnostic tool for circulatory or respiratory failure. Every intensivist should receive training at least to the basic level, and acquire an appropriate diploma approved by an intensive care society.
References
Edler I, Hertz CH. The use of ultrasonic reflectoscope for the continuous recording of the movements of heart walls. 1954. Clin Physiol Funct Imaging. 2004;24(3):118–36.
Joyner CR Jr, Reid JM. Applications of ultrasound in cardiology and cardiovascular physiology. Prog Cardiovasc Dis. 1963;5:482–97.
Bom N, Lancée CT, van Zwieten G, Kloster FE, Roelandt J. Multiscan echocardiography I. Technical description. Circulation. 1973;48(5):1066–74.
Hatle L, Angelsen B, Tromsdal A. Noninvasive assessment of atrioventricular pressure half-time by Doppler ultrasound. Circulation. 1979;60(5):1096–104.
Isaaz K, Thompson A, Ethevenot G, Cloez JL, Brembilla B, Pernot C. Doppler echocardiographic measurement of low velocity motion of the left ventricular posterior wall. Am J Cardiol. 1989;64(1):66–75.
Ozier Y, Guéret P, Jardin F, Farcot JC, Bourdarias JP, Margairaz A. Two-dimensional echocardiographic demonstration of acute myocardial depression in septic shock. Crit Care Med. 1984;12(7):596–9.
Jardin F, Gueret P, Dubourg O, Farcot JC, Margairaz A, Bourdarias JP. Two-dimensional echocardiographic evaluation of right ventricular size and contractility in acute respiratory failure. Crit Care Med. 1985;13(11):952–6.
Jardin F, Brun-Ney D, Auvert B, Beauchet A, Bourdarias JP. Sepsis-related cardiogenic shock. Crit Care Med. 1990;18(10):1055–60.
Mayo PH, Beaulieu Y, Doelken P, et al. American college of chest physicians/La Société de Réanimation de Langue Française statement on competence in critical care ultrasonography. Chest. 2009;135(4):1050–60.
Price S, Nicol E, Gibson DG, Evans TW. Echocardiography in the critically ill: current and potential roles. Intensive Care Med. 2006;32(1):48–59.
Douglas PS, Khandheria B, Stainback RF, et al. ACCF/ASE/ACEP/ASNC/SCAI/SCCT/SCMR 2007 appropriateness criteria for transthoracic and transesophageal echocardiography: a report of the American College of Cardiology Foundation Quality Strategic Directions Committee Appropriateness Criteria Working Group, American Society of Echocardiography, American College of Emergency Physicians, American Society of Nuclear Cardiology, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Computed Tomography, and the Society for Cardiovascular Magnetic Resonance. Endorsed by the American College of Chest Physicians and the Society of Critical Care Medicine. J Am Soc Echocardiogr. 2007;20(7):787–805.
Douglas PS, Garcia MJ, Haines DE, et al. ACCF/ASE/AHA/ASNC/HFSA/HRS/SCAI/SCCM/SCCT/SCMR 2011 Appropriate Use Criteria for Echocardiography. A Report of the American College of Cardiology Foundation Appropriate Use Criteria Task Force, American Society of Echocardiography, American Heart Association, American Society of Nuclear Cardiology, Heart Failure Society of America, Heart Rhythm Society, Society for Cardiovascular Angiography and Interventions, Society of Critical Care Medicine, Society of Cardiovascular Computed Tomography, Society for Cardiovascular Magnetic Resonance American College of Chest Physicians. J Am Soc Echocardiogr. 2011;24(3):229–67.
Hüttemann E, Schelenz C, Kara F, Chatzinikolaou K, Reinhart K. The use and safety of transoesophageal echocardiography in the general ICU: a minireview. Acta Anaesthesiol Scand. 2004;48(7):827–36.
Vieillard-Baron A, Chergui K, Rabiller A, Peyrouset O, Page B, Beauchet A, Jardin F. Superior vena caval collapsibility as a gauge of volume status in ventilated septic patients. Intensive Care Med. 2004;30(9):1734–9.
Pinsky MR, Payen D. Functional hemodynamic monitoring. Crit Care. 2005;9(6):566–72.
Vieillard-Baron A, Charron C, Chergui K, Peyrouset O, Jardin F. Bedside echocardiographic evaluation of hemodynamics in sepsis: is a qualitative evaluation sufficient? Intensive Care Med. 2006;32(10):1547–52.
Bouferrache K, Amiel J-B, Chimot L, Caille V, Charron C, Vignon P, Vieillard-Baron A. Initial resuscitation guided by the Surviving Sepsis Campaign recommendations and early echocardiographic assesment of hemodynamics in ICU septic patients: a pilot study. Crit Care Med (in press).
Vincent J-L, Weil MH. Fluid challenge revisited. Crit Care Med. 2006;34(5):1333–7.
Wiedemann HP, Wheeler AP, Bernard GR, Thompson BT, Hayden D, deBoisblanc B, Connors AF Jr, Hite RD, Harabin AL. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med. 2006;354(24):2564–75.
Barbier C, Loubières Y, Schmit C, Hayon J, Ricôme J-L, Jardin F, Vieillard-Baron A. Respiratory changes in inferior vena cava diameter are helpful in predicting fluid responsiveness in ventilated septic patients. Intensive Care Med. 2004;30(9):1740–6.
Feissel M, Michard F, Faller J-P, Teboul J-L. The respiratory variation in inferior vena cava diameter as a guide to fluid therapy. Intensive Care Med. 2004;30(9):1834–7.
Feissel M, Michard F, Mangin I, Ruyer O, Faller JP, Teboul JL. Respiratory changes in aortic blood velocity as an indicator of fluid responsiveness in ventilated patients with septic shock. Chest. 2001;119(3):867–73.
Jardin F, Dubourg O, Bourdarias JP. Echocardiographic pattern of acute cor pulmonale. Chest. 1997;111(1):209–17.
Jardin F, Vieillard-Baron A. Acute cor pulmonale. Curr Opin Crit Care. 2009;15(1):67–70.
Jardin F, Vieillard-Baron A. Is there a safe plateau pressure in ARDS? The right heart only knows. Intensive Care Med. 2007;33(3):444–7.
Mekontso Dessap A, Charron C, Devaquet J, Aboab J, Jardin F, Brochard L, Vieillard-Baron A. Impact of acute hypercapnia and augmented positive end-expiratory pressure on right ventricle function in severe acute respiratory distress syndrome. Intensive Care Med. 2009;35(11):1850–8.
Vieillard-Baron A, Caille V, Charron C, Belliard G, Page B, Jardin F. Actual incidence of global left ventricular hypokinesia in adult septic shock. Crit Care Med. 2008;36(6):1701–6.
International expert statement on training standards for critical care ultrasonography. Intensive Care Med. 2011;37(7):1077–1083.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Au, SM., Vieillard-Baron, A. Bedside echocardiography in critically ill patients: a true hemodynamic monitoring tool. J Clin Monit Comput 26, 355–360 (2012). https://doi.org/10.1007/s10877-012-9385-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-012-9385-6