Abstract
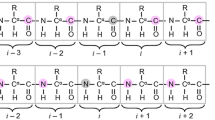
An experiment that provides a simple procedure to assign backbone nuclei in proteins is presented. The method relies on time-shared evolution of the coherences present in the (HN)NCAHA and (HA)CANNH experiments. By exploiting the fact that some of the coherences are common to both experiments the alpha and amide protons that are simultaneously detected are correlated with each other and with nitrogen and carbon nuclei. Thus, simultaneous assignment of Hα, HN, Cα and N signals is achieved in a single 3D spectrum. The experiment was tested on the streptococcal protein G B1 domain (GB1) which was easily assigned using a “stairway” procedure and on an 11 kDa domain of the yeast transcriptional co-activator Gal11.
Similar content being viewed by others
References
B. Bersch E. Rossy J. Coves B. Brutscher (2003) J. Biomol. NMR 27 57–67 Occurrence Handle10.1023/A:1024746306675 Occurrence Handle1:CAS:528:DC%2BD3sXlsVeitrc%3D
G. Bodenhausen R. Ernst (1981) J. Magn. Reson. 45 319
G. Bodenhausen R. Ernst (1982) J. Am. Chem. Soc. 104 1304–1309 Occurrence Handle10.1021/ja00369a027 Occurrence Handle1:CAS:528:DyaL38Xpt1ChsQ%3D%3D
W. Boucher E. Laue (1992) J. Am. Chem. Soc. 114 2262–2264 Occurrence Handle10.1021/ja00032a053 Occurrence Handle1:CAS:528:DyaK38XhsVOhtbs%3D
J. Cavanagh W.J. Fairbrother A.G. Palmer N.J. Skelton (1996) Protein NMR Spectroscopy Principles and Practice, Academic Press San Diego
E. Chiarparin P. Pelupessy R. Ghose G. Bodenhausen (1999) J. Am. Chem. Soc. 121 6876–6883 Occurrence Handle10.1021/ja984390p Occurrence Handle1:CAS:528:DyaK1MXkt1artrk%3D
R. Clubb V. Thanabal G. Wagner (1992a) J. Magn. Reson. 97 213 Occurrence Handle1:CAS:528:DyaK38XitFSmt7k%3D
R. Clubb V. Thanabal G. Wagner (1992b) J. Biomol. NMR 2 203–210 Occurrence Handle1:CAS:528:DyaK38XkvVKisbw%3D
L. Emsley G. Bodenhausen (1992) J. Magn. Reson. 97 135–148 Occurrence Handle1:CAS:528:DyaK38XhvVKgsLc%3D
B. Farmer SuffixII (1991) J. Magn. Reson. 93 635–641 Occurrence Handle1:CAS:528:DyaK3MXltVCnur8%3D
A.E. Ferentz G. Wagner (2000) Q. Rev. Biophys. 33 29–65 Occurrence Handle1:CAS:528:DC%2BD3cXnvVeqsLY%3D
H. Geen R. Freeman (1991) J. Magn. Reson. 93 93–141
T.D. Goddard D.G. Kneller (2004) SPARKY 3 University of California San Fransisco
S. Grzesiek A. Bax (1993a) J. Biomol. NMR 3 185–204 Occurrence Handle1:CAS:528:DyaK3sXks1Kqur4%3D
S. Grzesiek A. Bax (1993b) J. Am. Chem. Soc. 115 12593–12594 Occurrence Handle1:CAS:528:DyaK2cXis1OltA%3D%3D
B.A. Johnson R.A. Blevins (1994) J. Biomol. NMR 4 603–614 Occurrence Handle10.1007/BF00404272 Occurrence Handle1:CAS:528:DyaK2cXmt1Gkurw%3D
L. Kay M. Ikura A. Bax (1991) J. Magn. Reson. 91 84–92 Occurrence Handle1:CAS:528:DyaK3MXnvVSluw%3D%3D
L.E. Kay (1998) Nat. Struct. Biol. 5 Suppl 513–7 Occurrence Handle1:STN:280:DyaK1czivVWiug%3D%3D
L.E. Kay M. Ikura R. Tschudin A. Bax (1990) J. Magn. Reson. 89 496–514 Occurrence Handle1:CAS:528:DyaK3MXivFWg
L. Kay M. Wittekind M. McCoy M. Friedrichs L. Mueller (1992a) J. Magn. Reson. 98 443–450 Occurrence Handle1:CAS:528:DyaK38XltVOmu7s%3D
L.E. Kay P. Keifer T. Saarinen (1992b) J. Am. Chem. Soc. 114 10663–10665 Occurrence Handle10.1021/ja00052a088 Occurrence Handle1:CAS:528:DyaK3sXjt1eg
Keller R. (2004) CANTINA verlag, Goldau.
E. Kupce R. Freeman (2003a) J. Am. Chem. Soc. 125 13958–13959 Occurrence Handle10.1021/ja038297z Occurrence Handle1:CAS:528:DC%2BD3sXot12msL4%3D
E. Kupce R. Freeman (2003b) J. Magn. Reson. 162 300–10 Occurrence Handle1:CAS:528:DC%2BD3sXks1WgsLw%3D
T.M. Logan E.T. Olejniczak R.X. Xu S.W. Fesik (1993) J. Biomol. NMR 3 225–231 Occurrence Handle10.1007/BF00178264 Occurrence Handle1:CAS:528:DyaK3sXks1Kqur0%3D
D. Marion M. Ikura R. Tschudin A. Bax (1989) J. Magn. Reson. 85 393–399 Occurrence Handle1:CAS:528:DyaK3cXntlWrsg%3D%3D
V.Y. Orekhov I.V. Ibraghimov M. Billeter (2001) J. Biomol. NMR 20 49–60 Occurrence Handle10.1023/A:1011234126930 Occurrence Handle1:CAS:528:DC%2BD3MXksFCitL8%3D
A.G. Palmer J. Cavanagh P.E. Wright M. Rance (1991) J. Magn. Reson. 93 151–170 Occurrence Handle1:CAS:528:DyaK3MXkt1Sksbs%3D
S. Pascal D. Muhandiram T. Yamazaki J. Forman-Kay L. Kay (1994) J. Magn. Reson. Ser. B 103 197–201 Occurrence Handle10.1006/jmrb.1994.1031 Occurrence Handle1:CAS:528:DyaK2cXitFWrs7s%3D
M. Piotto V. Saudek V. Sklenar (1992) J. Biomol. NMR 2 661–665 Occurrence Handle10.1007/BF02192855 Occurrence Handle1:CAS:528:DyaK3sXitVyktrY%3D
D. Rovnyak D.P. Frueh M. Sastry Z.Y. Sun A.S. Stern J.C. Hoch G. Wagner (2004) J. Magn. Reson. 170 15–21 Occurrence Handle10.1016/j.jmr.2004.05.016 Occurrence Handle1:CAS:528:DC%2BD2cXmvVOmsbw%3D
M. Sattler M. Maurer J. Schleucher C. Griesinger (1995) J. Biomol. NMR 5 97–102 Occurrence Handle1:CAS:528:DyaK2MXjtlyju7Y%3D
M. Sattler J. Schleucher C. Griesinger (1999) Progr. NMR Spectrosc. 34 93–158 Occurrence Handle1:CAS:528:DyaK1MXisF2jsbk%3D
P. Schmieder A.S. Stern G. Wagner J.C. Hoch (1993) J. Biomol. NMR 3 569–56 Occurrence Handle10.1007/BF00174610 Occurrence Handle1:CAS:528:DyaK2cXhvVKnu7w%3D
A.J. Shaka P. Barker R. Freeman (1983) J. Magn. Reson. 51 547
A.J. Shaka C.J. Lee A. Pines (1988) J. Magn. Reson. 77 274–293
T. Spitzer G. Martin R. Crouch J. Shockor B. Farmer SuffixII (1992) J. Magn. Reson. 99 433–438 Occurrence Handle1:CAS:528:DyaK38XmtVektLk%3D
T. Szyperski G. Wider J.H. Bushweller K. Wuthrich (1993) J. Biomol. NMR 3 127–132 Occurrence Handle1:CAS:528:DyaK3sXkt1eqs7o%3D
A. Wang S. Grzesiek S. Tschudin R. Lodi A. Bax (1995) J. Biomol. NMR 5 376–382 Occurrence Handle1:CAS:528:DyaK2MXnt1ajtrY%3D
E.R. Zuiderweg (2002) Biochem. 41 1–7 Occurrence Handle1:CAS:528:DC%2BD3MXoslygsr8%3D
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Frueh, D.P., Arthanari, H. & Wagner, G. Unambiguous Assignment of NMR Protein Backbone Signals with a Time-shared Triple-resonance Experiment. J Biomol NMR 33, 187–196 (2005). https://doi.org/10.1007/s10858-005-3204-z
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s10858-005-3204-z