Abstract
The demand for natural food colorants is growing as consumers question the use of artificial colorants more and more. The phycobiliprotein C-phycocyanin of Arthospira platensis is used as a natural blue colorant in certain food products. The thermoacidophilic red microalga Cyanidioschyzon merolae might provide an alternative source of phycocyanin. Cyanidioschyzon merolae belongs to the order Cyanidiophyceae of the phylum Rhodophyta. Its natural habitat are sulfuric hot springs and geysers found near volcanic areas in, e.g., Yellowstone National Park in the USA and in Java, Indonesia. It grows optimally at a pH between 0.5 and 3.0 and at temperatures up to 56 °C. The low pH at which C. merolae grows minimizes the risk of microbial contamination and could limit production loss. As C. merolae lacks a cell wall, phycocyanin with a high purity number of 9.9 could be extracted by an osmotic shock using a simple ultrapure water extraction followed by centrifugation. The denaturation midpoint at pH 5 was 83 °C, being considerably higher than the A. platensis phycocyanin (65 °C). The C. merolae phycocyanin was relatively stable at pH 4 and 5 up to 80 °C. The high thermostability at slightly acidic pH makes the C. merolae phycocyanin an interesting alternative to A. platensis phycocyanin as a natural blue food colorant.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Synthetic dyes are used to provide color to all kinds of food products, confectionary, and beverages (Antello et al. 2008). Consumers have become suspicious about the use of these synthetic colorants as they are linked to having negative effect on children’s behavior (McCann et al. 2007; Arnold et al. 2012). Several large retailers are following major food producers by banning food products containing artificial colorants from their stores. This leads to a growing demand for natural colorants derived from plants and algae. For most of the artificial colorants, natural alternatives are relatively easily available (Wrolstad and Culter 2012). More challenging is to find natural alternatives to the artificial blue colorants such as Patent Blue V (E131) or Brilliant Blue FCF (E133). Blue colors are widespread in nature but it turns out that it is difficult to replicate the blue color. At neutral pH, natural blue colorants are stable but, especially at pH values below 5, they are much less stable and fade quickly (Newsome et al. 2014). Recently the FDA and EFSA have given approval for use of a Spirulina (Arthrospira) platensis extract containing high levels of phycocyanin as natural blue food colorant for coloring candy and chewing gum (Code of Federal Regulation 2016). Besides the potential use as food colorant, phycocyanin has also been described as having interesting pharmaceutical and nutraceutical properties (Eriksen 2008).
Phycocyanin is a pigment-protein complex that is part of the phycobilisomes found in cyanobacteria and microalga. Phycobilisomes are large, water-soluble protein complexes attached to the cytoplasmic surface of the thylakoid membrane (Biggins and Bruce 1989) and serve as the major antenna complex harvesting light. Phycobilisomes can make up as much as 20% of the cellular protein content (Glazer 1989). Phycocyanin is an oligomeric protein composed of α and β subunit to which several open chain tetrapyroles are attached (Stec et al. 1999; Padyana et al. 2001; Coyler et al. 2005). The tetrapyrole structures give the typical blue color to phycocyanin while the protein part confers the stability with respect to pH and temperature.
Arthrospira platensis phycocyanin has a limited thermostability as it denatures at temperatures above 60 °C (Jespersen et al. 2005; Martelli et al. 2014). In the search for a more thermostable and/or acid stable phycocyanin, the thermoacidophilic red microalga Cyanidioschyzon merolae was explored as a source of a blue colorant. Cyanidioschyzon merolae is a unicellular microalga belonging to the order Cyanidiophyceae of the phylum Rhodophyta. This species inhabits hot sulfuric springs and geysers in volcanic areas; it grows best at temperatures between 40 and 56 °C and acidic condition of pH 0.5 to 3 (Ciniglia et al. 2004). The phycocyanin of another red microalga, Galdieria sulphuraria, has been investigated (Sloth et al. 2006; Sørensen et al. 2012) as this species not only grows at relatively high temperatures and low pH, but also because it can grow heterotrophically in the dark on sugar as well as autotrophically in the light (Gross and Schnarrenberger 1995). Arthrospira platensis is grown outdoor in open ponds or raceway systems (Lee 1997; Belay 2013), suffering from productivity loss due to infection as it is difficult to maintain strict hygienic conditions (Richmond and Grobbelaar 1986). The extreme conditions applied to grow red microalga could be advantageous as infections are unlikely at the low pH levels applied. Sloth et al. (2006) and Sørensen et al. (2012) investigated the heterotrophic growth of G. sulphuraria on glucose in closed fermenters and found a reasonable phycocyanin productivity but concluded that the polysaccharide rich cell wall of G. sulphuraria makes it difficult to disrupt the cells and extract the phycocyanin. In addition, Sørensen et al. (2012) found that the phycocyanin extracted from G. sulphuraria cultures was not pure, as a considerable amount of protein and chlorophyll was also extracted. Cyanidioschyzon merolae could be a more effective source of phycocyanin as it lacks a cell wall (Lee 2004) making the extraction of the phycocyanin probably more efficient. This paper reports on the production and extraction of phycocyanin from C. merolae by a simple ultrapure water treatment.
Materials and methods
Growth of C. merolae and phycocyanin extraction
The red unicellular microalga Cyanidioschyzon merolae was obtained from National Institute for Environmental Studies (NIES, Japan), catalog no 1332. A stock culture was maintained in Allen medium pH 2 under constant light (100 μmol photons m−2 s−1) on a shaker at 150 rpm and 40 °C. Allen medium (Allen 1959) consisted of 1.32 g L−1 (NH4)2SO4, 0.27 g L−1 KH2PO4, 0.25 g L−1 MgSO4.7H2O, 0.073 g L−1 CaCl2.2H2O, 11 mg L−1 FeCl3, 2.8 mg L−1 H3BO3, 1.8 mg L−1 MnCl2, 0.218 mg L−1 ZnSO4.7H2O, 0.05 mg L−1 CuSO4, 0.023 mg L−1 NH4VO3, and 0.024 mg L−1 Na2MoO4.2H2O. The pH of the medium was adjusted to 2.0 with 4 M H2SO4 and autoclaving at 120 °C for 20 min. A 1-L photo-bioreactor (approximately 100 μmol photons m−2 s−1 at 40 °C) was used to grow C. merolae for phycocyanin production. Cultures were harvested by centrifugation at 10,000×g for 10 min at 4 °C. The pellet was resuspended in ultrapure water, mixed well for 5 min, and kept for several hours at room temperature. Cell debris was removed by centrifugation at 15,000×g at 4 °C for 15 min; the blue colored supernatant was transferred to new tube for further analysis.
Purification of phycocyanin
To investigate the influence of the time of exposure to ultrapure water on the yield of phycocyanin, 200 mg wet biomass were mixed well with 2-mL ultrapure water (Milli Q purification system) and left at room temperature for up to 300 min. Blue colored supernatant was collected by centrifugation at 15,000×g and transferred into new tube. Besides mixing, two other extraction methods were used, bead beating by shaking the suspended cells with a small metal ball at high speed and high-pressure homogenization by means of implosion of the cells.
The crude phycocyanin extract obtained after mixing was further purified by ammonium sulfate precipitation in three steps (20, 40, and 60% saturation). The precipitate was recovered by centrifugation at 10,000×g for 30 min, the colorless supernatant was discarded, and the precipitate was dissolved in 50 mM citrate buffer pH 5 at room temperature.
Determination of concentration
The phycocyanin concentration was estimated by using a spectrophotometer, DR 3900 (Hach-Lange, The Netherlands). Measurement was conducted at 624 and 652, at which phycocyanin and allophycocyanin respectively show maximum absorption (Bennett and Bogorad 1973). The purity of phycocyanin was assessed by calculating the ratio of A 624 to A 280, where A 280 is the absorbance of total protein. The concentration phycocyanin was calculated using the following equation (Silveira et al. 2007):
Thermostability of phycocyanin
To determine the denaturation midpoint, 1 mL of phycocyanin solution (initial OD A 620 = 0.8) was incubated at pH 5 and different temperatures in a waterbath for 30 min (intervals of 10 °C from 30 to 100 °C). The thermostability of the phycocyanin was measured by incubating samples at pH 5 and 80 °C followed by measuring the absorbance at 624 nm at regular intervals (0–150 min). The remaining concentration of phycocyanin (C R, %) relative to the initial concentration was calculated using the following equation: C R (%) = C/C 0 × 100. To determine the pH stability of phycocyanin, the samples were incubated at 80 °C at different pH values from 2 to 5, and the absorbance at 624 nm was measured at regular intervals (0 to 60 min).
Results and discussion
Growth of C. merolae and phycocyanin extraction
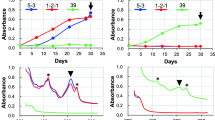
The extremophilic red microalga C. merolae was grown autotrophically in mineral medium at pH 2 and 40 °C with constant illumination (Fig. 1a). After about 3 days, it started to grow with a specific growth rate of 0.15 ± 0.01 day−1. At day 21, the cells had reached an OD800 of about 1.8, which corresponds to a dry weight of 1.051 ± 0.14 g L−1. An in vivo absorption spectrum (300 to 800 nm) was made every day, showing that there were three main absorption maxima, at 430, 620, and 680 nm (Fig. 2). The 430 and 680 nm maxima are typical for chlorophyll (Giltelson et al. 1999), while the 620 nm maximum is typical for phycocyanin (Patel et al. 2005). The amount of phycocyanin per cell did not seem to vary as a constant value of about 17.5 to 18 mg phycocyanin per unit of absorption at 800 nm was found for almost all time points (Fig. 1b).
To extract phycocyanin from the cells, several cell disruption methods were tested. Bead beating and high-pressure homogenization, both disruptive techniques, were compared with exposure of the cells to an osmotic shock by mixing them with ultrapure water (Fig. 3). The latter method worked best as most phycocyanin (0.55 mg mL−1) was found in solution. Cyanidioschyzon merolae is known to lack a cell wall (Albertano et al. 2000), making it susceptible to osmotic shocks. When exposed to ultrapure water, the cells take up water and finally lyse, releasing the cellular contents into the water. The water phase mainly contained phycocyanin (A 624, Table 1) with a purity index of 9.92, calculated as the ratio of A 624 to A 280 (protein). Small amounts of chlorophyll were also detected in the water phase (A 562 and A 652, Table 1).
C. merolae phycocyanin extracted by osmotic shock has a high purity number
The duration of the osmotic shock had a clear effect on the amount of phycocyanin extracted (Fig. 4). Incubating C. merolae cells in ultrapure water for up to 100 min gave relatively low amounts of phycocyanin in the water phase (up to 0.28 mg mL−1). However, a steep increase in the amount of phycocyanin was observed between 100 and 150 min incubation; after 150 min, almost 0.81 ± 0.09 mg mL−1 phycocyanin was released. Longer incubation did not result in a significant increase in the amount of phycocyanin released. The purity of the phycocyanin extract obtained with ultrapure water treatment is considerably higher than that of extracts obtained from Spirulina, with a purity index ranging from 0.46 to 2.78 (Silveira et al. 2007; Liao et al. 2011), from Synechococcus with a purity index of 2.2 (Gupta and Sainis 2010), and from the closely related red microalga G. sulphuraria of only 1 (Sørensen et al. 2012). The reason for the much higher purity index found for the phycocyanin extracted from C. merolae compared to other phototrophs is that the latter have a (thick) cell wall requiring a mechanical treatment to disrupt the cells and release the phycocyanin. As a result of the mechanical treatment of G. sulphuraria, also chlorophyll a and carotenoids are released into solution (Sloth et al. 2006), resulting in much lower purity number for phycocyanin. Applying different purification techniques such as aqueous two phase extraction with polyethylene glycol and water (Rito-Palomares et al. 2004), ammonium sulfate precipitation of expended bed absorption in combination with chromatography resulted in higher purity numbers (Zhang and Chen 1999; Niu et al. 2007; Yan et al. 2011), but these are still considerably lower than the 9.9 found for C. merolae. Phycocyanin solution with a purity index of at least 0.7 are considered to be a food grade, while a purity index of at least 4 is considered to be analytical grade (Cisneros and Rito-Palomares 2004). The phycocyanin extracted from C. merolae grown cells has a high purity index any further purification is not required to use it as analytical grade material.
The ultrapure water extract containing phycocyanin was further purified by ammonium sulfate precipitation at 20–40% saturation resulting in a concentrated phycocyanin solution (54% yield) with a purity index of 18.07 (Table 1). The pellet fraction 20–40% ammonium sulfate was dissolved in citrate buffer (pH 5) and was used to further characterize the C. merolae phycocyanin. The absorption spectrum of this fraction showed a clear λ max at 624 nm (Fig. 5), being the phycocyanin, and shoulder at 562 nm, indicative for phycoerythrin (Cisneros and Rito-Palomares 2004). The A. platensis phycocyanin has a λ max of 616 nm at pH 5 and 620 at pH 7 (Jespersen et al. 2005). The thermophile Synechococcus lividus phycocyanin has λ max of 609 nm at pH 6 (Edwards et al. 1997), while Phormidium luridum phycocyanin has a λ max at pH 6 of 622 nm (Edwards et al. 1996). Glazer and Fang (1996) even reported a λ max of about 650 nm at pH 3 for the phycocyanin of Synechococcus sp.
C. merolae phycocyanin is thermostable
The alpha and beta subunit of the phycocyanin having a molecular weight of 17–18 kDa were clearly visible on an SDS-polyacrylamide gel (data not shown). This is in agreement with the molecular weight of other phycocyanin reported so far (Glazer and Fang 1996; Chaiklahan et al. 2011). As C. merolae can grow at relatively high temperatures up to 55 °C, it is to be expected that the phycocyanin is relatively thermostable. The purified phycocyanin was incubated for 30 min in citrate buffer (pH 5) at temperatures varying from 20 to 100 °C (Fig. 6a). Up to 75 °C, the phycocyanin was soluble and remained clearly blue; at temperatures above 75 °C, it started to precipitate and at 90 °C, it had completely precipitated. The denaturation midpoint, which can be defined as that temperature (T m) at which 50% of the phycocyanin is still in solution (C R = 50%), of C. merolae phycocyanin is at 83 °C (Fig. 6a). At room temperature and pH 5, the 40% ammonium sulfate purified phycocyanin fraction was stable; more than 85% stayed in solution for 180 min (Fig. 6b). At pH 4, this fraction was already less stable; about 50% of the phycocyanin was lost after 180 min of incubation (Fig. 6b). At 80 °C and pH of 4 or 5, the color faded much more rapidly (Fig. 6c). At pH of 2 and 3, the blue color disappeared within several minutes both at room temperature and at 80 °C (Fig. 6b, c). The half-life of the 40% ammonium sulfate purified phycocyanin at room temperature was several hundred minutes at pH 4 and 5 while at pH 2 and 3, the phycocyanin faded to colorless in less than 5 min (Table 2). At high temperature of 80 °C, the phycocyanin had a half-life of 29 to 40 min at pH 4 and 5, respectively. At pH 2 and 3 and 80 °C, the phycocyanin lost its color within several minutes (Table 2).
Phycocyanin predominantly exists as a hexamer at pH 5, and it is believed that the hexameric form gives some protection against denaturation (Edwards et al. 1996). At pH 7, it is predominantly in a monomeric or trimeric form, resulting in lower thermostability (Edwards et al. 1997; Jespersen et al. 2005). Very likely, the C. merolae phycocyanin is also present in monomeric or trimeric form at low pH, as it looses its thermostability rapidly at these low pH values. The stability of the C. merolae phycocyanin at higher temperatures and a pH of 4 or 5 is much better than that of the S. platensis phycocyanin. The T m of the A. platensis phycocyanin is between 55 and 62 °C (Jespersen et al. 2005; Martelli et al. 2014). The C. merolae phycocyanin protein sequence contains four cysteine residues, whereas the A. platensis phycocyanin sequence, being 75% identical to the C. merolae sequence, has two cysteine residues (UniProtKB-P72509). Cysteine can form covalent disulfide bonds that contribute to the thermostability of a protein (Fass 2012). Adding high amounts of sugars (40 to 55%) such as fructose or glucose improved the thermostability of A. platensis phycocyanin, indicating that it could be used in high sugar food products such as confectionary and pastries (Martelli et al. 2014). As C. merolae phycocyanin already has a higher thermostability of its own, it could be used in low sugar products that are exposed to higher temperatures during production.
Conclusions
Phycocyanin can easily be extracted from autotrophically grown C. merolae cells by an osmotic shock procedure with ultrapure water. The phycocyanin obtained in this way has a high purity number (9.9) and is thermostable up to 83 °C at neutral and slight acidic pH. These properties make the C. merolae phycocyanin an interesting alternative to A. platensis phycocyanin as a natural blue food colorant.
References
Albertano P, Ciniglia C, Pinto G, Pollio A (2000) The taxonomic position of Cyanidium, Cyanidioschyzon and Galdieria: an update. Hydrobiologia 433:137–143
Allen MB (1959) Studies with Cyanidium caldarium, an anomalously pigmented chlorophyte. Arch Mikrobiol 32:270–277
Antello FS, Costa JAV, Kal SJ (2008) Thermal degradation kinetics of the phycocyanin from Spirulina platensis. Biochem Eng 41:43–47
Arnold LE, Lofthouse N, Hurt E (2012) Artificial food colors and attention-deficit/hyperactivity symptoms: conclusion to dye for. Neurotherapeutics 9:599–609
Belay A (2013) Biology and industrial production of Arthrospira (Spirulina). In: Richmond A, Hu Q (eds) Handbook of microalgal culture: applied phycology and biotechnology. Blackwell, Oxford, pp. 339–358
Bennett A, Bogorad L (1973) Complementary chromatic adaptation in a filamentous blue-green alga. J Cell Biol 58:419–435
Biggins J, Bruce D (1989) Regulation of excitation energy transfer in organism containing phycobilins. Photosynth Res 20:1–34
Chaiklahan R, Chirasuwan N, Loha V, Tia S, Bunnag B (2011) Separation and purification of phycocyanin from Spirulina sp. using a membrane process. Bioresource Technol 102:7159–7164
Ciniglia C, Yoon HS, Pollio A, Pinto G, Bhayyacharya D (2004) Hidden biodiversity of the extremophilic Cyanidiales red algae. Mol Ecol 13:1827–1838
Cisneros M, Rito-Palomares M (2004) A simplified strategy for the release and pimary recovery of C-phycocyanin produced by Spirulina platensis. Chem Biochem Eng Q 18:385–390
Code of Federal Regulation (2016) Spirulina extract. https://www.govregs.com/regulations/title21_chapterI_part73_subpartA_section73.530, accessed 13 July 2016
Coyler CL, Kinkade CS, Viskari PJ, Landers JP (2005) Analysis of cyanobacterial pigment and proteins by electrophoretic and chromatographic method. Anal Biochem Chem 382:559–569
Edwards MR, MacColl R, Eisele LE (1996) Some physical properties of an usual C-phycocyanin isolated from a photosynthethic thermopile. Biochem Biophys Acta 1276:64–70
Edwards MR, Houer C, Stack RF, Eisele LE, MacColl R (1997) Thermophilic C-phycocyanin: effect of temperature, monomer stability, and structure. Biochem Biophys Acta 1321:157–164
Eriksen NT (2008) Production of phycocyanin—a pigment with application in biology, biotechnology, foods, and medicine. Appl Microbial Biotechnol 80:1–14
Jespersen L, Strømdahl LD, Olsen K, Skibsted LH (2005) Heat and light stability of natural blue colorants for use in confectionery and beverages. Eur Food Res Technol 220:261–266
Fass D (2012) Disulfide bonding in protein biophysics. Annu Rev Biophys 41:63–79
Giltelson AA, Buschmann C, Lichtenthaler HK (1999) The chlorophyll fluorescen ratio F735/F700 as an accurate measure of the chlorophyll content in plants. Remote Sens Environ 69:296–302
Glazer AN (1989) The light guide: directional energy transfer in a photosynthetic antenna. J. Biol Chem 264:1–4
Glazer AN, Fang S (1996) Chromophore content of blue-green algal phycobiliproteins. J Biol Chem 248:659–662
Gross W, Schnarrenberger C (1995) Heterotrophic growth of two strains of the acidophilic red algae Galdieria sulphuraria. Plant Cell Physiol 36:633–638
Gupta A, Sainis JK (2010) Isolation of C-phycocyanin from Synechococcus sp. (Anacystis nidulans BD1). J Appl Phycol 22:231–233
Lee YK (1997) Commercial production of microalga in Asia-Pacific rim. J Appl Phycol 9:403–411
Lee RE (2004) Phycology, 4th edn. Cambridge University Press, Cambridge, pp. 108–109
Liao X, Zhang B, Wang X, Yan H, Zhang X (2011) Purification of C-phycocyanin from Spirulina platensis by single-step ion-exchange chromatography. Chromatographia 73:291–296
Martelli G, Folli C, Vista L, Daglia M, Ferrari D (2014) Thermal stability improvement of blue colorant C-phycocyanin from Spirulina platensis for food industry application. Process Biochem 49:154–159
McCann D, Barret A, Cooper A, Crumpler D, Dalne L, Grimshaw K, Kitchin E, Lok K, Porteous L, Prince E, Sanuga-Barke E, Warnet JO, Stevenson J (2007) Food additives and hyperactive in 3-year-old and 8/9-year-old children in the community: a randomized, double-blinded, placebo-controlled trial. Lancet 370:1560–1567
Newsome AG, Culter CA, van Breemen RB (2014) Nature’s palette: the search for natural blue colorants. J Agric Food Chem 62:6498–6511
Niu JF, Wang GC, Lin X, Zhou BC (2007) Large-scale recovery of phycocyanin from Spirulina platensis using expanded bed adsorption chromatography. J Chromatogr B 850:267–276
Padyana AK, Bhat VB, Madyastha KM, Rajashankar KB, Ramakumar S (2001) Crystal structure of light-harvesting protein C-phycocyanin from Spirulina platensis. Biochem Biophys Res Commun 282:893–898
Patel A, Mishra S, Pawar R, Ghosh PK (2005) Purification and characterization of phycocyanin from cyanobacterial species of marine and freshwater habitat. Prot Expr Pur 40:248–255
Richmond A, Grobbelaar JU (1986) Factor affecting the output rate of Spirulina platensis with reference to mass cultivation. Biomass 10:253–264
Rito-Palomares M, Nunez L, Amadr D (2004) Practical application of aqueous two-phase system for development of a prototype process for c-phycocyanin recovery from Spirulina maxima. J Chem Technol Biotechnol 76:1273–1280
Silveira S, Burkert JFM, Costa JAV, Burkert CAV, Kalil SJ (2007) Optimization of phycocyanin extraction from Spirulina platensis using factorial design. Bioresource Technol 98:1629–1634
Sloth JK, Wiebe MK, Eriksen NT (2006) Accumulation of phycocyanin in heterotrophic and mixotrophic cultures of the acidophilic red algae Galdieria sulphuraria. Enz Microb Technol 38:168–175
Sørensen L, Hantke A, Eriksen NT (2012) Purification of the photosynthetic pigment C-phycocyanin from heterotrophic Galdieria sulphuraria. J Sci Food Agric 93:2933–2938
Stec B, Troxler RF, Teeter MM (1999) Crystal structure of C-phycocyanin from Cyanidium caldarium provides a new perspective on phycobilisomes assembly. Biophys J 76:2912–2921
Wrolstad RE, Culter CA (2012) Alternatives to those artificial FD and C food colorant. Annu Rev Food Sci Technol 3:59–77
Yan S, Zhu L, Su H, Zhang X, Chen X, Zhou B, Zhang Y (2011) Single step chromatography for simultaneous purification of C-phycocyanin and allophycocyanin with high purity and recovery from Spirulina (Arthrospira) platensis. J Appl Phycol 3:1–6
Zhang Y, Chen F (1999) A simple method for efficient separation and purification of C-phycocyanin and allophycocyanin from Spirulina platensis. Biotechnol Technol 13:601–603
Acknowledgements
DYR was supported by the Ubbo Emmius PhD scholarship program of the University of Groningen.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Rahman, D.Y., Sarian, F.D., van Wijk, A. et al. Thermostable phycocyanin from the red microalga Cyanidioschyzon merolae, a new natural blue food colorant. J Appl Phycol 29, 1233–1239 (2017). https://doi.org/10.1007/s10811-016-1007-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-016-1007-0