Abstract
Despite the number of available methods to predict prognosis in patients with heart failure, prognosis remains poor, likely because of marked patient heterogeneity and varied heart failure etiologies. Thus, identification of novel prognostic indicators to stratify risk in patients with heart failure is of paramount importance. The spleen is emerging as a potential novel prognostic indicator for heart failure. In this article, we provide an overview of the current prognostic tools used for heart failure. We then introduce the spleen as a potential novel prognostic indicator, before outlining the structure and function of the spleen and introducing the concept of the cardiosplenic axis. This is followed by a focused discussion on the function of the spleen in the immune response and in hemodynamics, as well as a review of what is known about the usefulness of the spleen as an indicator of heart failure. Expert insight into the most effective spleen-related measurement indices for the prognostication of patients with heart failure is provided, and suggestions on how these could be measured in clinical practice are considered. In future, studies in humans will be required to draw definitive links between specific splenic measurements and different heart failure manifestations, as well as to determine whether splenic prognostic measurements differ between heart failure classes and etiologies. These contributions will provide a step forward in our understanding of the usefulness of the spleen as a prognostic predictor in heart failure.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Cardiovascular diseases contribute to an estimated 17.9 million deaths each year [1]. Despite advances in heart failure management and treatment, its prevalence was 64.63 million cases worldwide in 2020, and this figure continues to increase owing to population aging [2]. An estimated 1–3% of adults are living with heart failure [3], and prognosis is poor, with 5- and 10-year mortality rates of 50% and 90%, respectively [4].
There is substantial heterogeneity in the clinical progression of patients with heart failure because of marked patient heterogeneity [5,6,7,8] and varied heart failure etiologies, and a deeper understanding of this heterogeneity is needed to improve patient care. This limited understanding impedes the development of effective strategies to address heart failure as a public health concern [8]. The complexity of heart failure dictates that personalized medicine is fundamental for treatment and management. Precision medicine is also key, and improving the accuracy of risk prediction requires the incorporation of several clinical variables that are routinely assessed in clinical practice [9]. As such, prognostic models for heart failure that incorporate several clinical variables tend to be more useful than assessment of individual clinical parameters [9].
Heart failure prognostic prediction
There are a number of traditional and established predictors of prognosis in heart failure; however, many have their shortcomings. Raphael et al. [10] emphasize high inter-operator variability in the methods used to assess New York Heart Association (NYHA) functional class in clinical practice. Another study has shown comparable mortality rates between patients with NYHA functional class I or II and those with NYHA functional class III or IV [11]. In terms of prior heart failure hospitalization, Blumer et al. [12] showed that prior heart failure hospitalization is not associated with 180-day mortality and suggest that clinical variables may be a more direct means of predicting prognosis in patients with heart failure.
Other factors have also been investigated as prognostic determinants in patients with heart failure. A lower resting cardiac power output has been demonstrated as a prognostic factor in patients with advanced disease after adjusting for certain variables, including age, sex, left ventricular ejection fraction (LVEF), and mean arterial pressure [13]. Moreover, a low cardiac power index has been shown to predict a high cardiac event rate in patients with NYHA functional class II/III heart failure [14]. Right heart catheterization (RHC) is also used to assess hemodynamics in patients with heart failure; however, owing to its invasive nature, RHC is associated with an increased risk of complications [15]. Moreover, in patients with heart failure undergoing implantable cardioverter-defibrillator implantation, an increase in heart rate is strongly associated with poor outcomes [16, 17]. Recently, the value of the plasma amino acid profile as a novel prognostic marker for heart failure has been investigated, including the leucine/phenylalanine ratio, which was an independent predictor of future cardiac events in patients with heart failure [18, 19]. Despite the number of prognostic prediction methods to stratify risk and the number of treatment options in patients with heart failure, prognosis remains poor, which may be due to the substantial inter-patient heterogeneity, variation in heart failure etiologies, and the focus on symptom control with existing therapies. Thus, both treatment and prognostic prediction require improvement [20]. Novel prognostic indicators that can be incorporated into effective prognostic models are of paramount importance to effectively stratify risk and improve outcomes for patients.
Although the spleen has received limited research attention thus far, it is emerging as a potential prognostic indicator in patients with heart failure based on new evidence that highlights crosstalk between the heart and spleen, termed the “cardiosplenic axis” [18, 21,22,23]. Although the cardiosplenic axis has been widely demonstrated in animals, only a few studies have demonstrated its presence in humans [21,22,23]. Therefore, little is known about the potential of the cardiosplenic axis for clinical application [24].
This review will describe the structure and function of the spleen and discuss the concept of the cardiosplenic axis. This will be followed by a focused discussion on the function of the spleen in the immune response and in hemodynamics, as well as a review of what is known about the usefulness of the spleen as an indicator of cardiovascular disease and heart failure. Expert insight into the most effective spleen-related measurement indices for the prognostication of patients with heart failure will be provided, and suggestions on how these could be measured in clinical practice will be considered. In this review, we aim to collate current knowledge to provide insight into how this area of research could be carried forward for potential application in the clinic.
Biology of the spleen
The structure and function of the spleen
The spleen performs a number of functions that are facilitated by its structural components. The spleen varies in size between individuals, but it is typically ~ 10 × 6 × 3 cm3, weighs ~ 120 g, and is highly vascularized, storing 20–30% of total blood volume [21]. Larger-than normal spleen sizes have been reported in “Sea Nomads” (or Bajau people) who engage in breath-hold diving and in elite endurance athletes [25, 26]. Although splenic contraction is a part of the human diving response, it is thought that those who engage in breath-hold diving have physiologically and genetically adapted to the hypoxic environment, which has led to the development of a larger-than normal sized spleen. The larger splenic volume in elite endurance athletes is possibly functional and helps to cope with oxygen demands and deficits. Structurally, the spleen can be divided into two main regions, which are distinguished as white pulp and red pulp. The latter comprises the majority of splenic tissue. In the red pulp, pathogens, cellular debris, and old erythrocytes are efficiently removed from the circulation, and iron is recycled by an abundance of macrophages. The white pulp is the primary immunological region of the spleen in rodents and humans. It mostly consists of lymphoid cells in a highly organized compartment in which adaptive immune responses are initiated.
Functionally, the red pulp plays a role in immunity that is distinct from that of the white pulp [27]. Leukocytes residing in the red pulp, including neutrophils, monocytes, dendritic cells, and macrophages [28, 29], exit the spleen mostly through splenic veins in the red pulp [30]. The white pulp functions as a secondary lymphoid organ of the circulatory system and contains T and B cell zones in which antigen-specific immune responses are generated. The spleen regulates innate and adaptive immunity, contributing to immune responses that can either protect the host or contribute to disease, and can effectively clear pathogens from the blood [31].
The cardiosplenic axis
The relationship between the spleen and heart was first identified by Rein et al. in 1949 [32], who showed that electrical stimulation of splenic nerves restored ventricular function in dogs. Rein and colleagues also showed that splenectomy worsened myocardial contractile failure [33]. On the basis of these observations, the authors speculated a role for the spleen in protecting and maintaining cardiac function. Attention to this topic subsequently declined until 2009 when Swirski et al. investigated the role of the spleen as a reservoir for inflammatory macrophages and monocytes [34]. The authors showed that following myocardial infarction, splenic activation by increased angiotensin II mobilized splenic monocytes in mice, which infiltrated the myocardium.
Since these initial observations, the spleen has been further shown to influence the function of the heart, providing further evidence of the cardiosplenic axis. A key observation is that splenic contraction and volume reduction counteract tissue hypoperfusion. Splenic contraction is mediated by sympathetic control and is intended to compensate for systemic oxygen demand. This is associated with various states, such as hypovolemic shock [35, 36], sepsis [37, 38], hypoxia, trauma [39], and exercise [40, 41], supporting a hemodynamic link [21] between the spleen and the heart.
Acute myocardial infarction is one example of a cardiovascular event that increases sympathetic nervous system activity, which mobilizes progenitor cells from the bone marrow, in turn boosting monocyte production in the spleen [42]. Beta-adrenoreceptors are involved in leukocyte mobilization from the spleen [42]. Moreover, beta-adrenoreceptor activation increases interleukin-10 (IL-10) mRNA expression in the spleen and reduces infarct size in mice, which is abrogated by splenectomy; thus, IL-10 produced in the spleen might exert a cardioprotective effect [43, 44].
Further links between the spleen and the heart have also been noted. For example, positive inotropy has been reported in isolated cardiac preparations when exposed to splenic extracts [45]. Moreover, Emami et al. [23] have shown that the metabolic activity of the spleen markedly increases after acute coronary syndrome in humans and is associated with an increased risk of subsequent cardiovascular events. Additionally, in a mouse model of myocardial infarction with pre-existing chronic inflammation, angiotensin-converting enzyme inhibition has been shown to prevent monocyte release from the splenic reservoir [46]. All of the above evidence suggests a close link between the spleen and the heart.
The cardiosplenic axis in heart failure
The function of the spleen in the immune response
The spleen is an important reservoir for the deployment of leukocytes to tissue sites of injury and inflammation [34, 47]. In a study by Prabhu [24], mice with ischemic heart failure demonstrated expansion of proinflammatory monocytes and macrophages, as well as splenic remodeling. However, splenic macrophages are proinflammatory and promote tissue injury [24]. For instance, activation of splenic immune cells underlies the chronic inflammatory response in heart failure, and these cells induce tissue injury, leading to cardiac remodeling (Fig. 1). This process is facilitated by heightened antigen processing in the spleen [48]. Another study by Fujinami et al. [49] showed that the number of peripheral blood monocytes, which is positively correlated with maximum spleen diameter and spleen index, is related to the response to cardiac resynchronization therapy in chronic heart failure. Cardiac tissue damage, stress signals, and inflammation can activate the innate immune system, triggering the release of splenic immune cells, which can further contribute to chronic inflammation and cardiac remodeling in heart failure; thus, immune cell-mediated inflammation can play both a causative and consequent role in cardiac remodeling and heart failure [50]. Therefore, the interactions between the spleen and heart in terms of immune-mediated inflammation appear to be complex and bidirectional.
Proposed mechanisms of the cardiosplenic axis in heart failure. The cardiosplenic axis can be described as a cycle involving hemodynamic and immune processes. Heart failure is caused by reversible or irreversible damage to cardiac muscle. This tissue damage leads to immune cell recruitment from the circulation, triggering inflammation through cross-talk with the spleen. In terms of hemodynamic changes, the spleen holds a significant amount of the increased intravascular blood volume and regulates fluid distribution. As a result, splenomegaly and increased spleen stiffness are often observed in heart failure. This structural remodeling of the spleen is accompanied by functional remodeling, with changes in the composition of spleen-derived immune cells. Splenic immune cells, such as splenic macrophages, are released into the blood and migrate to the heart. This can lead to cardiac remodeling at the macroscopic and microscopic levels, which may be maladaptive or adaptive. The former can lead to cardiac fibrosis, while the latter is associated with suppression of inflammation in myocardial tissue via cardiac macrophage activity, which supports normalization of cardiac rhythm and suppression of arrhythmia development. These seemingly contradictory mechanisms have antagonistic effects, and their balance may affect cardiac function and prognosis. Owing to these wide-ranging changes, the spleen may be a clinical prognostic indicator in heart failure. For example, changes at the macroscopic level (spleen size and stiffness), as well as at the microscopic level (specific subsets of circulating spleen-derived immune cells), could be utilized as biomarkers to predict the prognosis of patients with heart failure
In mice, the cancer therapeutic doxorubicin disrupted immunometabolic pathways, triggered by persistent inflammation in the spleen and heart [51]. Specifically, doxorubicin dysregulated cyclooxygenase in the spleen and left ventricle. As a result, cyclooxygenase-mediated prostaglandin species were decreased in the left ventricle [51]. Doxorubicin causes cardiac toxicity and heart failure [52]; thus, an understanding of the immunometabolism pathways within the spleen and heart may be important for improving protection from heart failure, and this may be a key component of the cardiosplenic axis.
Like the spleen, the myocardium has its own resident macrophages; however, unlike spleen-derived macrophages, cardiac macrophages are minimally inflammatory and promote angiogenesis and tissue repair [24]. During the course of heart failure, macrophages gradually expand within the remote myocardium [53]. These immune cells are thought to phagocytose material from cardiomyocytes, such as remnants of dysfunctional mitochondria, through a process driven by cardiomyocyte autophagy during cardiac stress [54]. Cardiac macrophages also facilitate electrical conduction through the atrioventricular node. Cardiac macrophages, which reside between cardiomyocytes and in the atrioventricular node, form gap junctions with cardiomyocytes in the steady state and regulate conduction between cardiomyocytes [55]. Specifically, they have been shown to accelerate cardiomyocyte repolarization [55]. Cardiac macrophages are also involved in the normalization of cardiac rhythm and the suppression of arrhythmia development [55]. In a mouse model of heart failure, macrophages have been shown to act not only on cardiomyocytes, but also on fibroblasts via secretion of oncostatin-m [56], which directly inhibits tumor growth factor-β1-mediated activation of cardiac fibroblasts to suppress cardiac fibrosis.
It is important to note that the role of macrophages in fibrosis is not well understood owing to conflicting evidence. The conflicting observations may be due to differences in gene expression between different macrophage populations. For example, Haider et al. [57] showed that macrophages can transition to fibroblast-like cells in the heart following myocardial infarction with respect to their expression of fibroblastic markers. The triggers for such changes in gene expression are likely complex and multifactorial. For example, Nacu et al. [58] showed that macrophage phagocytosis of dead cells and debris leads to the release of pro-fibrotic mediators. Thus, it is possible that pro-fibrotic mediators may be expressed at a higher level in more advanced heart failure where tissue damage can be more severe, leading to fibrosis and maladaptive cardiac remodeling. Another study showed that sphingosine-1-phosphate (S1P) and S1P receptor expression were increased in the heart and spleen of mice. Selective activation of the S1P receptor in macrophages suppressed the inflammatory markers tumor necrosis factor-alpha and monocyte chemoattractant protein-1 (CCL2), expedited the reparative marker arginase-1, and was indicative of cardiac repair in the acute phase of heart failure. The study suggested that alterations in the S1P pathway in macrophages may contribute to pathological remodeling in heart failure [59]. There are a multitude of anti- and pro-inflammatory mediators that contribute to remodeling in the different stages (inflammatory, reparative, proliferative, and maturation), and some cell populations, such as macrophages, have two or more subsets with opposite or complementary actions, which have been discussed in detail previously [60, 61]. Hence, the balance between different macrophage populations may determine the status of remodeling and inform heart failure prognosis.
The function of the spleen in hemodynamics
In addition to its function in immunity, the spleen also plays a role in hemodynamics. A previous study has shown that splenic size increases in patients with advanced heart failure after left ventricular assist device (LVAD) implantation [21]. Another study in patients with advanced heart failure found a significant negative correlation between splenic volume and systemic vascular resistance prior to LVAD implantation, which was absent after LVAD implantation. These observations suggest that the spleen serves as a blood volume reservoir for systemic volume regulation (Fig. 1). Ibrahim et al. [62] demonstrated splenic enlargement in congestive heart failure. As such, spleen size may change in patients with heart failure undergoing LVAD support as a response to a change in cardiac preload and systemic volume status. Whether spleen size could be useful to monitor preload and systemic volume status as indicators of heart failure prognosis remains to be clarified.
Fujinami et al. [49] observed that chronic heart failure induces splenomegaly and that spleen size could predict the response to cardiac resynchronization therapy. A previous study has shown that splenectomy in mice with heart failure results in left ventricular reverse remodeling, with a reduction in left ventricular chamber size and an improvement in LVEF [24]. This research group proposed that a pathological cardiosplenic axis develops during heart failure, which is essential for disease progression. Furthermore, Erdem et al. [63] reported on the clinical course of five children with asplenia and concomitant cardiac abnormalities, and showed that congenital asplenia was associated with ventricular and atrial septal defects. Conversely, another study has shown that splenectomy reduces inflammatory infiltration and improves cardiac geometry and function [48]. Hence, there are conflicting reports as to whether splenectomy has a positive or negative effect on cardiac function. Moreover, it is not clear what effect splenectomy has on prognosis, which may be worthy of further investigation.
A recent study has shown that splenic volume is correlated with right arterial pressure and pulmonary capillary wedge pressure [64]. Moreover, heart rate and splenic volume are independent determinants of pulsatility index, reflecting cardiac preload in patients with heart failure [64]. This study concluded that spleen measurements may help to estimate systemic volume status and to understand hemodynamics in patients with LVADs for heart failure. Another recent study has shown an association between spleen volume index and outcomes in patients with heart failure [65], supporting the spleen as a potentially useful prognostic indicator in heart failure.
Other functions of the spleen in the cardiovascular system
The most widely known function of the spleen is to provide macrophages that remove old or damaged erythrocytes and platelets from the circulation for subsequent degradation. Erythrocyte-derived hemoglobin is degraded into heme and globin, and iron molecules within the heme are subsequently recycled for the synthesis of de novo hemoglobin molecules. The spleen also stores erythrocytes. Hypoxia and exercise separately trigger spleen contraction, resulting in the release of stored erythrocytes [41]. A study in rats has shown that intermittent hypoxia results in a reversible increase in blood hemoglobin concentration during hypoxia via splenic contraction [66]. In a study of spleen volume and hemoglobin concentration in healthy individuals, the spleen contracted and mobilized stored erythrocytes at increased altitudes during rested conditions. Additionally, the spleen contracted further during exercise, increasing oxygen delivery to tissues during acute hypoxia [41]. From these observations, it was concluded that the attenuated hemoglobin response to exercise at high altitude was likely due to greater recruitment of the spleen reserve during rest and that maximal spleen contraction was reached during exercise [41]. Therefore, the spleen appears to play a role in regulating the circulating hemoglobin concentration in response to varying degrees of hypoxia in humans. In the context of chronic heart failure, patients with reduced ejection fraction experience dyspnea from fluid overload that may result in hypoxemia [67], which may in turn trigger the spleen to contract, mobilizing stored erythrocytes to regulate circulating hemoglobin concentration. Moreover, a recent study on the relationship between the spleen and exercise tolerance in patients with heart failure has shown that the link between splenic size and peak VO2 may be hemoglobin concentration [68]. Anemia and hemoglobin concentration [69,70,71] have previously been reported as prognostic factors in patients with heart failure. Thus, it is possible that spleen-mediated regulation of hemoglobin concentration is associated with heart failure prognosis.
Clinical utility of the spleen as a prognostic biomarker in heart failure
In this paper, we have reviewed the available evidence on the cardiosplenic axis and the splenic manifestations that have been observed in heart failure to date. However, it is important that a consensus be reached on how these parameters can be measured clinically and how they may be useful to examine heart failure prognosis.
Of note, it is necessary to identify novel biomarkers to predict prognosis in patients with heart failure, for whom predicting risk is often challenging, and to provide a personalized strategy with precision medicine to improve outcomes.
The cardiosplenic axis represents a reservoir of immunological and hemodynamic processes that play a central pathophysiological role in heart failure. Therefore, it is our opinion that the spleen may be a useful tool to predict prognosis in patients with heart failure. At the macroscopic level, the size, volume, stiffness, and function of the spleen should be considered. At the microscopic level, there is a need to explore and develop molecular and cellular markers that are specific to the spleen, particularly those that can be evaluated by simple practical analyses, such as blood tests. Some of these factors are outlined in more detail below.
There is no established method to clinically assess spleen function in patients with heart failure at present; however, we speculate that certain immune cell types, such as proinflammatory C–C chemokine receptor 2 (CCR2)-positive macrophages, CD4 + T helper (Th; Th1, Th2, and Th17) cells, regulatory T cells, and CD8 + T cells, might be useful to predict heart failure prognosis. As explained by Prabhu [24], cardiac injury stimulates the death and substitution of cardiac-resident cells by infiltrating blood monocyte-derived macrophages, which express CCR2. Thus, CCR2-positive macrophages may be a useful marker of cardiac damage in heart failure. Sager et al. [53] have also shown that in chronic ischemic heart failure, the proportion of monocyte-derived macrophages increases. Expansion of CD4 + T helper cells and CD8 + T cells has also been observed in the spleen in heart failure [48, 72]. It is important to note that circulating immune cells are not necessarily specific to the heart and spleen; thus, suitable markers of the cardiosplenic axis that can be used to examine heart failure prognosis need to be identified.
Furthermore, when considering which factors might be most useful for prognostic prediction in patients with heart failure, Halade et al. [73] showed that leukocyte trafficking can be used to distinguish the status of inflammation in mice. Activated splenic immune cells modulate local tissue injury, cell death, and fibrosis in the heart, which influences cardiac remodeling, a topic previously reviewed by Prabhu [24]. Therefore, these cell types may be potentially useful for the assessment of inflammation and cardiac remodeling, and thus heart failure prognosis. It is also important to note that there are similarities and differences between different types of heart failure in terms of the correlative biomarkers. For example, Tromp et al. [74] showed that heart failure with preserved ejection fraction (HFpEF) or heart failure with mid-range ejection fraction is more closely related to inflammation than heart failure with reduced ejection fraction (HFrEF). In their study, pentraxin-3 (which regulates the inflammatory activity of macrophages [75]) was more associated with HFpEF. Another study [76] also showed that HFpEF is more associated with inflammation, while HFrEF is more associated with cardiac stretch. Given that HFpEF appears to be more closely related to inflammation, the spleen may play a greater role in this type of heart failure. However, further studies are needed to define the role of the spleen in HFpEF compared with HFrEF.
As described earlier, in patients with advanced heart failure, splenic size increases after LVAD implantation, and a significant negative correlation has been observed between splenic volume and systemic vascular resistance [21], which is often high in patients with heart failure. Thus, in terms of the clinical manifestations of heart failure that could be measured using the cardiosplenic axis, it is possible that splenic size and volume could inform on the functioning of the heart, such as its pumping efficiency or hemodynamics, and thus predict prognosis. However, when assessing the spleen in the context of heart failure, it would also be important to consider the association of the spleen with other organs and in different diseases. For example, the spleen has shown associations with the liver [77], obesity [78], and resting energy expenditure [78]. Thus, the spleen likely has a much wider range of functions than previously understood. When using the spleen as a prognostic tool in heart failure, such considerations would be particularly important in patients with co-existing diseases in which the spleen has demonstrated involvement. Further research is also needed to identify other functions of the spleen.
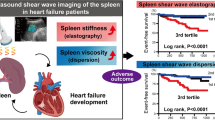
In terms of imaging approaches to assess spleen size and volume, computed tomography of the spleen could be used to monitor at-risk patients, although this method is relatively expensive and uses ionizing radiation. Ultrasound can also be used to measure spleen size, is more widely available than computed tomography, and does not use radiation, thus avoiding any potential damage from repeated measurements [79]. Doppler ultrasound may also be useful in measuring spleen function by evaluating spleen blood flow [80]. However, it is important to note that there is marked inter-user variability with ultrasound imaging. Moreover, shear-wave elastography to measure spleen stiffness could be used to quantify the severity of splenic congestion and is a viable method for predicting adverse events in patients with acute decompensated heart failure [81]. The use of ultrasound and shear-wave elastography to assess spleen size/volume/function and stiffness, respectively, as potential indicators of pumping efficiency and hemodynamics could provide a non-invasive alternative to invasive RHC, which would reduce the risk of complications.
Future perspectives
A summary of the future perspectives and directions that should be taken to address the current knowledge gaps is shown in Table 1.
First, it has been hypothesized that all organs possess endocrine function and may therefore contribute to whole-body homeostasis [82]. For example, the heart produces atrial natriuretic peptide and BNP; adipose tissues produce and secrete adipokines, such as adiponectin; and skeletal muscle produces and secretes myokines, such as myonectin, all of which may induce cardioprotective effects [83,84,85,86]. As such, further research is required to determine the role of the spleen within this hypothesized multi-organ endocrine system. In heart failure, the spleen may be instrumental in maintaining the functions of multiple organs, not just the heart. Currently, the hormones and endocrine substances that are specific to the spleen are unknown. It may be that the spleen secretes other factors in addition to macrophages and cytokines, such as hormones and chemical messengers, that may play a protective role in heart failure, which is an area worthy of further study.
Second, much of the existing evidence demonstrating the role of the spleen in heart failure is from animal studies; thus, further studies should be conducted in humans to clarify the relevance of the cardiosplenic axis and to identify potentially useful spleen-related prognostic predictors for heart failure.
Third, the physiological mechanisms of the role of the spleen in heart failure remain unclear, and the relationships between physiological indices and cellular and molecular markers specific to the spleen have not yet been clarified. Further knowledge in this area would be useful for the potential application of the spleen as a tool for prognostic prediction in heart failure.
Fourth, it has been emphasized that heart failure varies in etiology and pathology and can be highly variable; hence, splenic involvement may depend on the cause of heart failure. Therefore, its assessment may only be relevant for specific subpopulations of patients. If the cardiosplenic axis is demonstrated to play a definitive role in heart failure in humans, research to identify subpopulations of patients in whom the spleen may be a useful prognostic tool will be a valuable step toward precision medicine.
Finally, it has been established that prognostic models for heart failure are more effective than individual clinical variables alone [9]. Thus, if the spleen is confirmed as a useful tool for prognostic prediction in heart failure, future studies should investigate the most effective way to incorporate spleen assessment into existing prognostic models.
Conclusions
In this study, we reviewed evidence of the interplay between the heart and spleen, termed the cardiosplenic axis, in cardiovascular disease and heart failure. The spleen forms a major part of the immune system, and evidence suggests a role for the spleen in immune cell mobilization as a mediator of inflammation and cardiac remodeling. Other evidence suggests a relationship between splenic size and systemic blood volume regulation and hemodynamics. Most studies have been conducted in animals; thus, future studies in humans should aid our understanding of the importance of the cardiosplenic axis and drawing of definitive links between specific splenic measurements and different heart failure manifestations, including hemodynamic changes, inflammation, fibrosis, and remodeling. It should also be determined whether splenic measurements have variable importance between different heart failure classes and etiologies. These contributions will represent a step forward in understanding the usefulness of the spleen as a prognostic predictor in heart failure.
References
World Health Organization (2021) Cardiovascular disease. https://www.who.int/health-topics/cardiovascular-diseases/#tab=tab_1. Accessed 23 July 2021
Lippi G, Sanchis-Gomar F (2020) Global epidemiology and future trends of heart failure. AME Med J 5. https://doi.org/10.21037/amj.2020.03.03
Dunlay SM, Roger VL, Redfield MM (2017) Epidemiology of heart failure with preserved ejection fraction. Nat Rev Cardiol 14:591–602. https://doi.org/10.1038/nrcardio.2017.65
Bowen RES, Graetz TJ, Emmert DA, Avidan MS (2020) Statistics of heart failure and mechanical circulatory support in 2020. Ann Transl Med 8:827. https://doi.org/10.21037/atm-20-1127
Iorio A, Pozzi A, Senni M (2017) Addressing the heterogeneity of heart failure in future randomized trials. Curr Heart Fail Rep 14:197–202. https://doi.org/10.1007/s11897-017-0332-1
Tromp J, Shen L, Jhund PS, Anand IS, Carson PE, Desai AS, Granger CB, Komajda M, McKelvie RS, Pfeffer MA, Solomon SD, Køber L, Swedberg K, Zile MR, Pitt B, Lam CSP, McMurray JJV (2019) Age-related characteristics and outcomes of patients with heart failure with preserved ejection fraction. J Am Coll Cardiol 74:601–612. https://doi.org/10.1016/j.jacc.2019.05.052
Spencer FA, Meyer TE, Gore JM, Goldberg RJ (2002) Heterogeneity in the management and outcomes of patients with acute myocardial infarction complicated by heart failure. Circulation 105:2605–2610. https://doi.org/10.1161/01.CIR.0000017861.00991.2F
Shah AM, Solomon SD (2012) Phenotypic and pathophysiological heterogeneity in heart failure with preserved ejection fraction. Eur Heart J 33:1716–1717. https://doi.org/10.1093/eurheartj/ehs124
Aaronson KD, Cowger J (2012) Heart failure prognostic models: why bother?. Circ Heart Fail 5:6–9. https://doi.org/10.1161/CIRCHEARTFAILURE.111.965848
Raphael C, Briscoe C, Davies J, Whinnett ZI, Manisty C, Sutton R, Mayet J, Francis DP (2007) Limitations of the New York Heart Association functional classification system and self-reported walking distances in chronic heart failure. Heart 93:476–482. https://doi.org/10.1136/hrt.2006.089656
Ahmed A (2007) A propensity matched study of New York Heart Association class and natural history end points in heart failure. Am J Cardiol 99:549–553. https://doi.org/10.1016/j.amjcard.2006.08.065
Blumer V, Mentz RJ, Sun JL, Butler J, Metra M, Voors AA, Hernandez AF, O’Connor CM, Greene SJ (2021) Prognostic role of prior heart failure hospitalization among patients hospitalized for worsening chronic heart failure. Circ Heart Fail 14:e007871
Yildiz O, Aslan G, Demirozu ZT, Yenigun CD, Yazicioglu N (2017) Evaluation of resting cardiac power output as a prognostic factor in patients with advanced heart failure. Am J Cardiol 120:973–979. https://doi.org/10.1016/j.amjcard.2017.06.028
Morimoto R, Mizutani T, Araki T, Oishi H, Kimura Y, Kazama S, Shibata N, Kuwayama T, Hiraiwa H, Kondo T, Furusawa K, Okumura T, Murohara T (2021) Prognostic value of resting cardiac power index depends on mean arterial pressure in dilated cardiomyopathy. ESC Heart Fail 8:3206–3213. https://doi.org/10.1002/ehf2.13446
Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE Jr, Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi JL, Johnson MR, Kasper EK, Levy WC, Masoudi FA, McBride PE, McMurray JJV, Mitchell JE, Peterson PN, Riegel B, Sam F, Stevenson LW, Tang WHW, Tsai EJ, Wilkoff BL, College A, of Cardiology Foundation; American Heart Association Task Force on Practice Guidelines (2013) ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol 62:e147-239. https://doi.org/10.1016/j.jacc.2013.05.019
Ahmadi-Kashani M, Kessler DJ, Day J, Bunch TJ, Stolen KQ, Brown S, Sbaity S, Olshansky B, INTRINSIC RV Study Investigators (2009) Heart rate predicts outcomes in an implantable cardioverter-defibrillator population. Circulation 120:2040–2045. https://doi.org/10.1161/CIRCULATIONAHA.108.847608
Vukadinović AN, Vukadinović D, Borer J, Cowie M, Komajda M, Lainscak M, Swedberg K, Böhm M (2017) Heart rate and its reduction in chronic heart failure and beyond. Eur J Heart Fail 19:1230–1241. https://doi.org/10.1002/ejhf.902
Hiraiwa H, Okumura T, Kondo T, Kato T, Kazama S, Kimura Y, Ishihara T, Iwata E, Shimojo M, Kondo S, Aoki S, Kanzaki Y, Tanimura D, Sano H, Awaji Y, Yamada S, Murohara T (2021) Prognostic value of leucine/phenylalanine ratio as an amino acid profile of heart failure. Heart Vessels 36:965–977. https://doi.org/10.1007/s00380-020-01765-z
Hiraiwa H, Okumura T, Kondo T, Kato T, Kazama S, Ishihara T, Iwata E, Shimojo M, Kondo S, Aoki S, Kanzaki Y, Tanimura D, Sano H, Awaji Y, Yamada S, Murohara T (2020) Usefulness of the plasma branched-chain amino acid/aromatic amino acid ratio for predicting future cardiac events in patients with heart failure. J Cardiol 75:689–696. https://doi.org/10.1016/j.jjcc.2019.12.016
Taylor CJ, Ordóñez-Mena JM, Roalfe AK, Lay-Flurrie S, Jones NR, Marshall T, Hobbs FDR (2019) Trends in survival after a diagnosis of heart failure in the United Kingdom 2000–2017: population based cohort study. BMJ 364:I223. https://doi.org/10.1136/bmj.I223
Hiraiwa H, Okumura T, Sawamura A, Kondo T, Kazama S, Kimura Y, Shibata N, Arao Y, Oishi H, Kato H, Kuwayama T, Yamaguchi S, Furusawa K, Morimoto R, Murohara T (2020) Spleen size improvement in advanced heart failure patients using a left ventricular assist device. Artif Organs 44:700–708
Maeda D, Sakane K, Kanzaki Y, Horai R, Akamatsu K, Tsuda K, Ito T, Sohmiya K, Hoshiga M (2021) Splenic volume index determined using computed tomography upon admission is associated with readmission for heart failure among patients with acute decompensated heart failure. Int Heart J 62:584–591. https://doi.org/10.1536/ihj.20-564
Emami H, Singh P, MacNabb M, Vucic E, Lavender Z, Rudd JHF, Fayad ZA, Lehrer-Graiwer J, Korsgren M, Figueroa AL, Fredrickson J, Rubin B, Hoffmann U, Truong QA, Min JK, Baruch A, Nasir K, Nahrendorf M, Tawakol A (2015) Splenic metabolic activity predicts risk of future cardiovascular events: demonstration of a cardiosplenic axis in humans. JACC Cardiovasc Imaging 8:121–130. https://doi.org/10.1016/j.jcmg.2014.10.009
Prabhu SD (2018) The cardiosplenic axis is essential for the pathogenesis of ischemic heart failure. Trans Am Clin Climatol Assoc 129:202–214. PMCID: PMC6116633
Ilardo MA, Moltke I, Korneliussen TS, Cheng J, Stern AJ, Racimo F, de Barros DP, Sikora M, Seguin-Orlando A, Rasmussen S, van den Munckhof IC, ter Horst R, Joosten LAB, Netea MG, Salingkat S, Nielsen R, Willerslev E (2018) Physiological and genetic adaptations to diving in sea nomads. Cell 173:569-580.e15. https://doi.org/10.1016/j.cell.2018.03.054
Holmström PK, Karlsson Ö, Lindblom H, McGawley K, Schagatay EK (2021) Enhanced splenic volume and contraction in elite endurance athletes. J Appl Physiol 131:474–486. https://doi.org/10.1152/japplphysiol.01066.2020
Lewis SM, Williams A, Eisenbarth SC (2019) Structure and function of the immune system in the spleen. Sci Immunol 4:eaau6085. https://doi.org/10.1126/sciimmunol.aau6085
Nolte MA, Hoen EN, van Stijn A, Kraal G, Mebius RE (2000) Isolation of the intact white pulp. Quantitative and qualitative analysis of the cellular composition of the splenic compartments. Eur J Immunol 30:626–634. https://doi.org/10.1002/1521-4141(2000002)30:2%3c626::AID-IMMU626%3e3.0.CO;2-H
Mebius RE, Kraal G (2005) Structure and function of the spleen. Nat Rev Immunol 5:606–616. https://doi.org/10.1038/nri1669
Pellas TC, Weiss L (1990) Deep splenic lymphatic vessels in the mouse: a route of splenic exit for recirculating lymphocytes. Am J Anat 187:347–354. https://doi.org/10.1002/aja.1001870404
Bronte V, Pittet MJ (2013) The spleen in local and systemic regulation of immunity. Immunity 39:806–818. https://doi.org/10.1016/j.immuni.2013.10.010
Rein H (1949) Via a regulation system “spleen-liver” for the oxidative metabolism of the body tissues and especially the heart. Nat Sci 36:260–268 (in German). https://doi.org/10.1007/BF00584977
Rein H, Dohrn A (1951) The role of the spleen and liver in coronary or hypoxic myocardial insufficiency. Pflugers Arch Gesamte Physiol Menschen Tiere 253:435–458 (in German). https://doi.org/10.1007/BF00370032
Swirski FK, Nahrendorf M, Etzrodt M, Wildgruber M, Cortez-Retamozo V, Panizzi P, Figueiredo J-L, Kohler RH, Chudnovskiy A, Waterman P, Aikawa E, Mempel TR, Libby P, Weissleder R, Pittet MJ (2009) Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science 325:612–616. https://doi.org/10.1126/science.1175202
Enslow MS, Preece SR, Wildman-Tobriner B, Enslow RA, Mazurowski M, Nelson RC (2018) Splenic contraction: a new member of the hypovolemic shock complex. Abdom Radiol (New York) 43:2375–2783. https://doi.org/10.1007/s00261-018-1478-3
Kiguchi T, Higuchi T, Takahashi N, Shimokoshi T, Yamazaki M, Yoshimura N, Aoyama H (2015) CT measurement of splenic volume changes as a result of hypovolemic shock. Jpn J Radiol 33:645–649. https://doi.org/10.1007/s11604-015-0470-x
Koga Y, Fujita M, Nakahara T, Yagi T, Miyauchi T, Kaneda K, Kawamura Y, Oda Y, Tsuruta R (2016) Splenic volume in severe sepsis is associated with disease severity and pneumococcal infection. Acute Med Surg 3:339–344. https://doi.org/10.1002/ams2.204
Zha A, Vahidy F, Randhawa J, Parsha K, Bui T, Aronowski J, Savitz SI (2018) Association between splenic contraction and the systemic inflammatory response after acute ischemic stroke varies with age and race. Transl Stroke Res 9:484–492. https://doi.org/10.1007/s12975-017-0596-5
Cruz-Romero C, Agarwal S, Abujudeh HH, Thrall J, Hahn PF (2016) Spleen volume on CT and the effect of abdominal trauma. Emerg Radiol 23:315–323. https://doi.org/10.1007/s10140-016-1402-5
Schagatay E, Hubinette A, Lodin-Sundström A, Engan H, Stenfors N (2015) Exercise induced hemoconcentration following spleen contraction in subjects with COPD. COPD Res Pract 1:13. https://doi.org/10.1186/s40749-015-0015-9
Schagatay E, Lunde A, Nilsson S, Palm O, Lodin-Sundström A (2020) Spleen contraction elevates hemoglobin concentration at high altitude during rest and exercise. Eur J Appl Physiol 120:2693–2704. https://doi.org/10.1007/s00421-020-04471-w
Dutta P, Courties G, Wei Y, Leuschner F, Gorbatov R, Robbins CS, Iwamoto Y, Thompson B, Carlson AL, Heidt T, Majmudar MD, Lasitschka F, Etzrodt M, Waterman P, Waring MT, Chicoine AT, van der Laan AM, Niessen HWM, Piek JJ, Rubin BB, Butany J, Stone JR, Katus HA, Murphy SA, Morrow DA, Sabatine MS, Vinegoni C, Moskowitz MA, Pittet MJ, Libby P, Lin CP, Swirski FK, Weissleder R, Nahrendorf M (2012) Myocardial infarction accelerates atherosclerosis. Nature 487:325–329. https://doi.org/10.1038/nature11260
Tian Y, Miao B, Charles EJ, Wu D, Kron IL, French BA, Yang Z (2018) Stimulation of the beta2 adrenergic receptor at reperfusion limits myocardial reperfusion injury via an interleukin-10-dependent anti-inflammatory pathway in the spleen. Circ J 82:2829–2836. https://doi.org/10.1253/circj.CJ-18-0061
Markowski P, Boehm O, Goelz L, Haesner AL, Ehrentraut H, Bauerfeld K, Tran N, Zacharowski K, Weisheit C, Langhoff P, Schwederski M, Hilbert T, Klaschik S, Hoeft A, Baumgarten G, Meyer R, Knuefermann P (2013) Pre-conditioning with synthetic CpG-oligonucleotides attenuates myocardial ischemia/reperfusion injury via IL-10 up-regulation. Basic Res Cardiol 108:376. https://doi.org/10.1007/s00395-013-0376-7
Heusch G (2019) The spleen in myocardial infarction. Circ Res 124:26–28. https://doi.org/10.1161/CIRCRESAHA.118.314331
Leuschner F, Panizzi P, Chico-Calero I, Lee WW, Ueno T, Cortez-Retamozo V, Waterman P, Gorbatov R, Marinelli B, Iwamoto Y, Chudnovskiy A, Figueiredo J-L, Sosnovik DE, Pittet MJ, Swirski FK, Weissleder R, Nahrendorf M (2010) Angiotensin-converting enzyme inhibition prevents the release of monocytes from their splenic reservoir in mice with myocardial infarction. Circ Res 107:1364–1373. https://doi.org/10.1161/CIRCRESAHA.110.227454
Seifert HA, Hall AA, Chapman CB, Collier LA, Willing AE, Pennypacker KR (2012) A transient decrease in spleen size following stroke corresponds to splenocyte release into systemic circulation. J Neuroimmune Pharmacol 7:1017–1024. https://doi.org/10.1007/s11481-012-9406-8
Ismahil MA, Hamid T, Bansal SS, Patel B, Kingery JR, Prabhu SD (2014) Remodeling of the mononuclear phagocyte network underlies chronic inflammation and disease progression in heart failure: critical importance of the cardiosplenic axis. Circ Res 114:266–282. https://doi.org/10.1161/CIRCRESAHA.113.301720
Fujinami M, Kondo H, Yufu K, Shinohara T, Ishii Y, Teshima Y, Nakagawa M, Takahashi N (2018) Association between the baseline peripheral blood monocyte counts, the size of spleen, and the response to cardiac resynchronization therapy. J Cardiol 71:299–304. https://doi.org/10.1016/j.jjcc.2017.09.004
Van Linthout S, Tschöpe C (2017) Inflammation — cause of consequence of heart failure or both?. Curr Heart Fail Rep 14:251–265. https://doi.org/10.1007/s11897-017-0337-9
Jadapalli JK, Wright GW, Kain V, Asif Sherwani MA, Sonkar R, Yusuf N, Halade GV (2018) Doxorubicin triggers splenic contraction and irreversible dysregulation of COX and LOX that alters the inflammation-resolution program in the myocardium. Am J Physiol Heart Circ 315:H1091–H1100. https://doi.org/10.1152/ajpheart.00290.2018
Von Hoff DD, Layard MW, Basa P (1979) Risk factors for doxorubicin-induced congestive heart failure. Ann Intern Med 91:710–717. https://doi.org/10.7326/0003-4819-91-5-710
Sager HB, Hulsmans M, Lavine KJ, Moreira MB, Heidt T, Courties G, Sun Y, Iwamoto Y, Tricot B, Khan OF, Dahlman JE, Borodovsky A, Fitzgerald K, Anderson DG, Weissleder R, Libby P, Swirski FK, Nahrendorf M (2016) Proliferation and recruitment contribute to myocardial macrophage expansion in chronic heart failure. Circ Res 119:853–864. https://doi.org/10.1161/CIRCRESAHA.116.309001
Nicolás-Ávila JA, Lechuga-Vieco AV, Esteban-Martínez L, Sánchez-Díaz M, Díaz-García E, Santiago DJ, Rubio-Ponce A, Li JLY, Balachander A, Quintana JA, Martínez-de-Mena R, Castejón-Vega B, Pun-García A, Través PG, Bonzón-Kulichenko E, García-Marqués F, Cussó L, A-González N, González-Guerra A, Roche-Molina M, Martin-Salamanca S, Crainiciuc G, Guzmán G, Larrazabal J, Herrero-Galán E, Alegre-Cebollada J, Lemke G, Rothlin CV, Jimenez-Borreguero LJ, Reyes G, Castrillo A, Desco M, Muñoz-Cánoves P, Ibáñez B, Torres M, Ng LG, Priori SG, Bueno H, Vázquez J, Cordero MD, Bernal JA, Enríquez JA, Hidalgo A, (2020) A network of macrophages supports mitochondrial homeostasis in the heart. Cell 183:94–109. https://doi.org/10.1016/j.cell.2020.08.031
Hulsmans M, Clauss S, Xiao L, Aguirre AD, King KR, Hanley A, Hucker WJ, Wülfers EM, Seemann G, Courties G, Iwamoto Y, Sun Y, Savol AJ, Sager HB, Lavine KJ, Fishbein GA, Capen DE, Da Silva N, Miquerol L, Wakimoto H, Seidman CE, Seidman JG, Sadreyev RI, Naxerova K, Mitchell RN, Brown D, Libby P, Weissleder R, Swirski FK, Kohl P, Vinegoni C, Milan DJ, Ellinor PT, Nahrendorf M (2017) Macrophages facilitate electrical conduction in the heart. Cell 169:510–522. https://doi.org/10.1016/j.cell.2017.03.050
Abe H, Takeda N, Isagawa T, Semba H, Nishimura S, Morioka MS, Nakagama Y, Sato T, Soma K, Koyama K, Wake M, Katoh M, Asagiri M, Neugent ML, Kim J-W, Stockmann C, Yonezawa T, Inuzuka R, Hirota Y, Maemura K, Yamashita T, Otsu K, Manabe I, Nagai R, Komuro I (2019) Macrophage hypoxia signaling regulates cardiac fibrosis via oncostatin M. Nat Commun 10:2824. https://doi.org/10.1038/s41467-019-10859-w
Haider N, Boscá L, Zandbergen R, Kovacic JC, Narula N, González-Ramos S, Fernandez-Velasco M, Agrawal S, Paz-García M, Gupta S, DeLeon-Pennell K, Fuster V, Ibañez B, Narula J (2019) Transition of macrophages to fibroblast-like cells in healing myocardial infarction. J Am Coll Cardiol 74:2134–3135. https://doi.org/10.1016/j.jacc.2019.10.036
Nacu N, Luzina IG, Highsmith K, Lockatell V, Pochetuhen K, Cooper ZA, Gillmeister MP, Todd NW, Atamas SP (2008) Macrophages produce TGF-β-induced (β-ig-h3) following ingestion of apoptotic cells and regulate MMP14 levels and collagen turnover in fibroblasts. J Immunol 180:5036–5044. https://doi.org/10.4049/jimmunol.180.7.5036
Gowda SGB, Gowda D, Kain V, Chiba H, Hui S-P, Chalfant CE, Parcha V, Arora P, Halade GV (2021) Sphingosine-1-phosphate interactions in the spleen and heart reflect extent of cardiac repair in mice and failing human hearts. Am J Physiol Heart Circ Physiol 321:H599–H611. https://doi.org/10.1152/ajpheart.00314.2021
Okyere AD, Tilley DG (2020) Leukocyte-dependent regulation of cardiac fibrosis. Front Physiol 11:301. https://doi.org/10.3389/fphys.2020.00301
Kologrivova I, Shtatolkina M, Suslova T, Ryabov V (2021) Cells of the immune system in cardiac remodeling: main players in resolution of inflammation and repair after myocardial infarction. Front Immunol 12:664457. https://doi.org/10.3389/fimmu.2021.664457
Ibrahim M, Sorour A, Elsherif A (1951) Splenic enlargement in congestive heart failure and active rheumatic infection. Br Heart J 13:212–214. https://doi.org/10.1136/hrt.13.2.212
Erdem SB, Genel F, Erdur B, Ozbek E, Gulez N, Mese T (2015) Asplenia in children with congenital heart disease as a cause of poor outcome. Cent Eur J Immunol 40:266–269. https://doi.org/10.5114/ceji.2015.52841
Hiraiwa H, Okumura T, Sawamura A, Kondo T, Kazama S, Kimura Y, Shibata N, Arao Y, Oishi H, Kato H, Kuwayama T, Yamaguchi S, Furusawa K, Morimoto R, Fujimoto K, Mutsuga M, Usui A, Murohara T (2021) Association between splenic volume and pulsatility index in patients with left ventricular assist devices. Int J Artif Organs 44:282–287. https://doi.org/10.1177/0391398820957019
Hiraiwa H, Okumura T, Sawamura A, Araki T, Mizutani T, Kazama S, Kimura Y, Shibata N, Oishi H, Kuwayama T, Kondo T, Furusawa K, Morimoto R, Murohara T (2022) Splenic size as an indicator of hemodynamics and prognosis in patients with heart failure. Heart Vessels. https://doi.org/10.1007/s00380-022-02030-1
Kuwahira I, Kamiya U, Iwamoto T, Moue Y, Urano T, Ohta Y, Gonzalez NC (1999) Splenic contraction-induced reversible increase in hemoglobin concentration in intermittent hypoxia. J Appl Physiol 86:181–187. https://doi.org/10.1152/jappl.1999.86.1.181
Jung M, Riley P, Drozdzewski A, Pressler SJ (2017) Mild to moderate hypoxemia among stable heart failure patients with reduced ejection fraction: 24-hour oxygen monitoring. J Card Fail 23:S81. https://doi.org/10.1016/j.cardfail.2017.07.233
Hiraiwa H, Okumura T, Sawamura A, Araki T, Mizutani T, Kazama S, Kimura Y, Shibata N, Oishi H, Kuwayama T, Kondo T, Furusawa K, Morimoto R, Adachi T, Yamada S, Mutsuga M, Usui A, Murohara T (2022) Relationship between spleen size and exercise tolerance in advanced heart failure patients with a left ventricular assist device. BMC Res Notes 15:40. https://doi.org/10.1186/s13104-022-05939-y
Abebe TB, Gebreyohannes EA, Bhagavathula AS, Tefera YG, Abegaz TM (2017) Anemia in severe heart failure patients: does it predict prognosis?. BMC Cardiovasc Dis 17:248. https://doi.org/10.1186/s12872-017-0680-5
Sharma R, Francis DP, Pitt B, Poole-Wilson PA, Coats AJS, Anker SD (2004) Haemoglobin predicts survival in patients with chronic heart failure: a substudy of the ELITE II trial. Eur Heart J 25:1021–1028. https://doi.org/10.1016/j.ehj.2004.04.023
Varadarajan P, Gandhi S, Sharma S, Umakanthan B, Pai RG (2006) Prognostic significance of hemoglobin level in patients with congestive heart failure and normal ejection fraction. Clin Cardiol 29:444–449. https://doi.org/10.1002/clc.4960291006
Bansal SS, Ismahil MA, Goel M, Patel B, Hamid T, Rokosh G, Prabhu SD (2017) Activated T lymphocytes are essential drivers of pathological remodeling in ischemic heart failure. Circ Heart Fail 10:e003688. https://doi.org/10.1161/CIRCHEARTFAILURE.116.003688
Halade GV, Norris PC, Kain V, Serhan CN, Ingle KA (2018) Splenic leukocytes define the resolution of inflammation in heart failure. Sci Signal 11:eaao1818. https://doi.org/10.1126/scisignal.aao1818
Tromp J, Khan MAF, Mentz RJ, O’Connor CM, Metra M, Dittrich HC, Ponikowski P, Teerlink JR, Cotter G, Davison B, Cleland JGF, Givertz MM, Bloomfield DM, van Veldhuisen DJ, Hillege HL, Voors AA, van der Meer P (2017) Biomarker profiles of acute heart failure patients with a mid-range ejection fraction. JACC: Heart Fail 5:507–517. https://doi.org/10.1016/j.jchf.2017.04.007
Shiraki A, Kotooka N, Komoda H, Hirase T, Oyama JI, Node K (2016) Pentraxin-3 regulates the inflammatory activity of macrophages. Biochem Biophys Rep 5:290–295. https://doi.org/10.1016/j.bbrep.2016.01.009
Sanders-van Wijk S, van Empel V, Davarzani N, Maeder MT, Handschin R, Pfisterer ME, Brunner-La Rocca HP, Investigators TIME-CHF (2015) Circulating biomarkers of distinct pathophysiological pathways in heart failure with preserved vs. reduced left ventricular ejection fraction. Eur J Heart Fail 17:1006–1014. https://doi.org/10.1002/ejhf.414
Tarantino G, Scalera A, Finelli C (2013) Liver-spleen axis: intersection between immunity, infections and metabolism. World J Gastroenterol 19:3534–3542. https://doi.org/10.3748/wjg.v19.i23.3534
Tarantino G, Citro V, Conforti P, Balsano C, Capone D (2019) Is there a link between basal metabolic rate, spleen volume and hepatic growth factor levels in patients with obesity-related NAFLD?. J Clin Med 8:1510. https://doi.org/10.3390/jcm8101510
Yetter EM, Acosta KB, Olson MC, Blundell K (2003) Estimating splenic volume: sonographic measurements correlated with helical CT determination. Gastrointest Radiol 181:1615–1620. https://doi.org/10.2214/ajr.181.6.1811615
Manoharan A, Gill RW, Griffiths KA (1987) Splenic blood flow measurements by Doppler ultrasound: a preliminary report. Cardiovasc Res 21:779–782. https://doi.org/10.1093/cvr/21.10.779
Saito Y, Matsumoto N, Aizawa Y, Fukamachi D, Kitano D, Kazuto T, Tamaki T, Fujito H, Sezai A, Okumura Y (2020) Clinical significance of spleen stiffness in patients with acute decompensated heart failure. ESC Heart Fail 7:4005–4014. https://doi.org/10.1002/ehf2.13001
Chrousos GP (2008) Organization and integration of the endocrine system. Sleep Med Clin 2:125–145. https://doi.org/10.1016/j.jsmc.2007.04.004
Shibata R, Ouchi N, Ohashi K, Murohara T (2017) The role of adipokines in cardiovascular disease. J Cardiol 70:329–334. https://doi.org/10.1016/j.jjcc.2017.02.006
Lau WB, Ohashi K, Wang Y, Ogawa H, Murohara T, Ma XL, Ouchi N (2017) Role of adipokines in cardiovascular disease. Circ J 81:920–928. https://doi.org/10.1253/circj.CJ-17-0458
Otaka N, Shibata R, Ohashi K, Uemura Y (2018) Myonectin is an exercise-induced myokine that protects the heart from ischemia-reperfusion injury. Circ Res 123:1326–1338. https://doi.org/10.1161/CIRCRESAHA.118.313777
Ouchi N, Ohashi K, Shibata R, Murohara T (2016) Protective roles of adipocytokines and myokines in cardiovascular disease. Circ J 80:2073–2080. https://doi.org/10.1253/circj.CJ-16-0663
Acknowledgements
The authors would like to thank Ryota Morimoto, MD, PhD; Akinori Sawamura, MD, PhD; Toru Kondo, MD, PhD; Tasuku Kuwayama, MD, PhD; Hideo Oishi, MD, PhD; Takashi Araki, MD; Takashi Mizutani, MD; Shingo Kazama, MD; Yuki Kimura, MD; and Ryota Ito, MD, for their useful discussion and/or helpful comments on the manuscript. The authors would also like to thank Nila Bhana, MSc, and Emily Woodhouse, PhD, of Edanz (https://jp.edanz.com/ac) for providing medical writing support.
Funding
This work was supported in part by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (JSPS KAKENHI) (grant number JP21K16085 to H. H.).
Author information
Authors and Affiliations
Contributions
H. H. contributed to the conceptualization, investigation, and visualization of this review. T. O. and T. M. contributed to the investigation and visualization. All authors contributed to the writing and review of, and read and approved, the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
Takahiro Okumura received research grants from Ono Pharmaceutical, Bayer Pharmaceutical, Daiichi-Sankyo Pharma, and Amgen Astellas BioPharma, as well as honoraria from Ono Pharmaceutical, Otsuka Pharmaceutical, Novartis Pharma, and Medtronic Japan. Toyoaki Murohara received lecture fees from Bayer Pharmaceutical, Daiichi-Sankyo, Dainippon Sumitomo Pharma, Kowa, MSD, Mitsubishi Tanabe Pharma, Nippon Boehringer Ingelheim, Novartis Pharma, Pfizer Japan, Sanofi-Aventis, and Takeda Pharmaceutical. Toyoaki Murohara also received an unrestricted research grant from the Department of Cardiology of Nagoya University Graduate School of Medicine, as well as honoraria from Astellas Pharma, Daiichi-Sankyo, Dainippon Sumitomo Pharma, Kowa, MSD, Mitsubishi Tanabe Pharma, Nippon Boehringer Ingelheim, Novartis Pharma, Otsuka Pharma, Pfizer Japan, Sanofi-Aventis, Takeda Pharmaceutical, and Teijin Pharma. All other authors declare that they have no relationships with industry relevant to the contents of this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hiraiwa, H., Okumura, T. & Murohara, T. The cardiosplenic axis: the prognostic role of the spleen in heart failure. Heart Fail Rev 27, 2005–2015 (2022). https://doi.org/10.1007/s10741-022-10248-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10741-022-10248-4