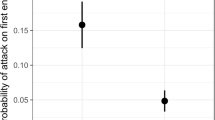
Abstract
Some phytophagous insects gain defense from natural enemies by associating with otherwise potentially harmful top predators. Many lycaenid butterfly caterpillars are involved in such interactions with ants: larvae provide carbohydrate rewards from the dorsal nectary organ (DNO) to associated ants in return for protection from natural enemies. The stability of these interactions involves signals that identify the lycaenid caterpillar as a mutualist. However, larvae of some lycaenid species, such as Lycaena xanthoides, are found in close association with ants but do not possess the reward producing DNO. Evaluating the relationship in a phylogenetic framework, we show that the association between L. xanthoides and ants likely evolved from a non-ant-associated ancestor. Behavioral trials also show that L. xanthoides larvae are capable of influencing ant behavior to increase ant tending when faced with a simulated predator attack, without providing DNO-derived rewards to ant associates. These results demonstrate that the DNO is not necessary to maintain associations between lycaenid larvae and ants. Third-party interactions may affect the evolution of mutualisms and consideration of underlying evolutionary history is necessary to understand contemporary species associations.


Similar content being viewed by others
References
Agarwal AA, Fordyce JA (2000) Induced indirect defense in a lycaenid-ant association: the regulation of a resource in a mutualism. Proc R Soc Lond B 267:1857–1861
Akino T, Knapp JJ, Thomas JA, Elmes GW (1999) Chemical mimicry and host specificity in the butterfly Maculinea rebeli, a social parasite of Myrmica ant colonies. Proc R Soc Lond B 266:1419–1426
Allen TJ, Brock JP, Glassberg J (2005) Caterpillars in the field and garden: a field guide to the butterfly caterpillars of North America. Oxford University Press, Oxford
Atsatt PR (1981) Lycaenid butterflies and ants: selection for enemy-free space. Am Nat 118:638–654
Ballmer GR, Pratt GF (1988) A survey of the last instar larvae of the Lycaenidae (Lepidoptera) of California. J Res Lepid 27:1–81
Ballmer GR, Pratt GF (1991) Quantification of ant attendance (myrmecophily) of lycaenid larvae. J Res Lepid 30:95–112
Bozano GC, Weidenhoffer Z (2001) Lycaenidae part I: subfamily Lycaeninae. In: Bozano GC (ed) Guide to the butterflies of the Palearctic region. Omnes Artes, Milan, pp 1–62
Bronstein JL (2001) The exploitation of mutualisms. Ecol Lett 4:277–287
Caterino MS, Sperling FAH (1999) Papilio phylogeny based on mitochondrial cytochrome oxidase I and II genes. Mol Phylogenet Evol 11:122–137
Cushman JH, Rashbrook VK, Beattie AJ (1994) Assessing benefits to both participants in a lycaenid-ant association. Ecology 75:1031–1041
Daniels, H. 2004. Facultative butterfly-ant interactions—the role of variation in composition of nectar secretions. Ph.D. dissertation, University of Bayreuth, Bayreuth
Dettner K, Liepert C (1994) Chemical mimicry and camouflage. Annu Rev Entomol 39:129–154
DeVries PJ (1984) Of crazy-ants and Curetinae: are Curetis butterflies tended by ants? Zool J Linnean Soc 80:59–66
Eliot JN (1973) The higher classification of the Lycaenidae (Lepidoptera): a tentative arrangement. Bull Brit Mus (Nat Hist), Entomol 28:1–505
Fiedler K, Maschwitz U (1989) Functional analysis of the myrmecophilous relationships between ants (Hymenoptera: Formicidae) and Lycaenids (Lepidoptera: Lycaenidae). Ethology 80:71–80
Fiedler K, Saam C (1995) Ants benefit from attending facultatively myrmecophilous Lycaenidae caterpillars: evidence from a survival study. Oecologia 104:316–322
Fiedler K, Seufert P, Pierce NE, Pearson JG, Baumgarten H-T (1992) Exploitation of lycaenid-ant mutualisms by braconid parasitoids. J Res Lepid 31:153–168
Fiedler K, Hölldobler B, Seufert P (1996) Butterflies and ants: the communicative domain. Experientia 52:14–24
Francoeur A (1973) Revision taxonomique des especes nearctiques du group fusca, genre Formica (Hymenoptera: Formicidae). Mem Soc Entomol Québec 3:1–316
Fraser AM, Axén AH, Pierce NE (2001) Assessing the quality of different ant species as partners of a myrmecophilous butterfly. Oecologia 129:452–460
Green P (1999) Phrap. http://phrap.org
Green P, Ewing B (2002) Phred. http://phrap.org
Gross P (1993) Insect behavioral and morphological defenses against parasitoids. Ann Rev Entomol 38:251–273
Henning FF (1983) Chemical communication between lycaenid larvae (Lepidoptera: Lycaenidae) and ants (Hymenoptera: Formicidae). J Entomol Soc S Afr 46:341–366
Hinton HE (1951) Myrmecophilous Lycaenidae and other Lepidoptera—a summary. Proc Trans S London Entomol Nat Hist Soc 1949–50:111–175
Hodges RW, Dominick T, Davis DR, Ferguson DC, Franclemont JG, Munroe EG, Powell JA (eds) (1983) Check list of the lepidoptera of America North of Mexico (Including Greenland). E. W. Classey Ltd. and The Wedge Entomological Research Foundation, London
Kitching RL, Luke B (1985) The myrmecophilous organs of the larvae of some British Lycaenidae (Lepidoptera): a comparative study. J Nat Hist 19:259–276
Leimar O, Axén AH (1993) Strategic behaviour in an interspecific mutualism: interactions between lycaenid larvae and ants. Anim Behav 46:1177–1182
Maddison WP (1997) Gene trees in species trees. Syst Biol 46:523–536
Maddison WP, Knowles LL (2006) Inferring phylogeny despite incomplete lineage sorting. Syst Biol 55:21–30
Maddison DR, Maddison WP (2007a) Chromaseq: a Mesquite module for analyzing sequence chromatograms. Version 0.91. http://mesquiteproject.org/packages/chromaseq
Maddison WP, Maddison DR (2007b) Mesquite: a modular system for evolutionary analysis. Version 2.0bi44. http://mesquiteproject.org
Maschwitz U, Wüst M, Schurian K (1975) Bläulingsraupen als Zuckerlieferanten für Ameisen. Oecologia 18:17–21
Megens H-J, de Jong R, Fiedler K (2005) Phylogenetic patterns in larval host plant and ant association of Indo-Australian Arhopalini butterflies (Lycaenidae: Theclinae). Biol J Linn Soc 84:225–241
Monteiro A, Pierce NE (2001) Phylogeny of Bicyclus (Lepidoptera: Nymphalidae) inferred from COI, COII and EF-1alpha gene sequences. Mol Phylogenet Evol 18:264–281
Nault LR, Montgomery ME, Bowers WS (1976) Ant-aphid association: role of aphid alarm pheromone. Science 192:1349–1351
Newcomer EJ (1912) Some observations on the relation of ants and lycaenid caterpillars, and a description of the relational organs of the latter. J NY Entomol Soc 20:31–36
Oliver JC (2008) AUGIST: Inferring species trees while accommodating gene tree uncertainty. Bioinformatics 24:2932–2933
Oliver JC, Shapiro AM (2007) Genetic isolation and cryptic variation within the Lycaena xanthoides species group (Lepidoptera: Lycaenidae). Mol Ecol 16:4308–4320
Oliver JC, Prudic KL, Pauly GB (2007) Parasitism rates in larval Lycaena xanthoides (Godart) (Lepidoptera: Lycaenidae) and a new host record for Cotesia theclae (Riley) (Hymenoptera: Braconidae). Pan-Pac Entomol 83:262–264
Osborn F, Jaffé K (1997) Cooperation vs. exploitation: interactions between Lycaenid (Lepidoptera: Lycaenidae) larvae and ants. J Res Lepid 34:69–82
Pasteels JM, Grégoire J-C, Rowell-Rahier M (1983) The chemical ecology of defense in arthropods. Ann Rev Entomol 28:263–289
Pierce NE (1983) Ecology and evolution of symbioses between lycaenid butterflies and ants. Ph.D. thesis, Harvard University, Cambridge, Massachusetts
Pierce NE, Mead PS (1981) Parasitoids as selective agents in the symbiosis between lycaenid butterfly larvae and ants. Science 211:1185–1187
Pierce NE, Braby MF, Heath A, Lohman DJ, Mathew J, Rand DB, Travassos MA (2002) The ecology and evolution of ant association in the Lycaenidae (Lepidoptera). Ann Rev Entomol 47:733–771
Pratt GF, Wright DM (2002) Allozyme phylogeny of North American coppers (Lycaeninae: Lycaenidae). Pan-Pac Entomol 78:219–229
R Development Core Team (2007) R: a language and environment for statistical computing (R Foundation for Statistical Computing, Vienna, Austria) ISBN 3-900051-07-0, URL http://www.R-project.org
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Ruxton GD, Sherratt TN, Speed MP (2004) Avoiding attack: the evolutionary ecology of crypsis, Warning Signals and Mimicry. Oxford University Press, Oxford
Saarinen EV (2006) Differences in worker caste behaviour of Oecophylla smaragdina (Hymenoptera: Formicidae) in response to larvae of Anthene emolus (Lepidoptera: Lycaenidae). Biol J Linnean Soc 88:391–395
Scott JA (1986) The Butterflies of North America: a natural history and field guide. Stanford University Press, Stanford
Stadler B, Dixon AFG (2005) Ecology and evolution of aphid-ant interactions. Ann Rev Ecol Evol Syst 36:345–372
Stadler B, Dixon AFG (2008) Mutualism: ants and their insect partners. Cambridge University Press, Cambridge
Stamatakis A (2006) RAxML-VI-HPC: maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 22:2688–2690
Stanton ML (2003) Interacting guilds: moving beyond the pairwise perspective on mutualisms. Am Nat 162:S10–S23
Travassos MA, Pierce NE (2000) Acoustics, context and function of vibrational signalling in a lycaenid butterfly–ant mutualism. Anim Behav 60:13–26
van Dorp K (2004) Molecular systematics of Lycaena F., 1807 (Lepidoptera: Lycaenidae)—Some preliminary results. Proc Neth Entomol Soc 15:65–70
Vane-Wright RI (1976) A unified classification of mimetic resemblances. Biol J Linnean Soc 8:25–56
Wahlberg N, Braby MF, Brower AVZ, de Jong R, Lee M-M, Nylin S, Pierce NE, Sperling FAH, Vila R, Warren AD, Zakharov E (2005) Synergistic effects of combining morphological and molecular data in resolving the phylogeny of butterflies and skippers. Proc R Soc B 272:1577–1586
Weeks JA (2003) Parasitism and ant protection alter the survival of the lycaenid Hemiargus isola. Ecol Entomol 28:228–232
Wickler W (1968) Mimicry in plants and animals. McGraw-Hill Book Company, New York
Acknowledgments
We thank K.L. Prudic, E.C. Snell-Rood, J.M. Davis, and especially D.R. Papaj for discussions concerning behavioral analyses and species interactions. D.R. Maddison and M.J. Sanderson provided insight on analyses of character evolution. We also thank G. Anweiler, G.C. Bozano, A.M. Shapiro, and E. Weingartner for providing specimens. G.F. Pratt and G.R. Ballmer provided invaluable information regarding field sites and larval rearing conditions. This work was funded by the Center for Insect Science and an NSF DDIG to J.C.O.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Oliver, J.C., Stein, L.R. Evolution of influence: signaling in a lycaenid-ant interaction. Evol Ecol 25, 1205–1216 (2011). https://doi.org/10.1007/s10682-011-9478-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10682-011-9478-6