Abstract
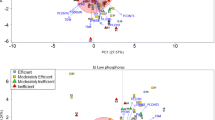
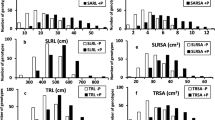
Aegilops tauschii, as one of wheat wild relatives, possesses a wide range of resistance to biotic/abiotic factors. To better utilize and provide the genetic basis of breeding for improving phosphorus deficiency tolerance, large-scale screening for tolerant A. tauschii to phosphorus deficiency was conducted using hydroponic experiment. A total of 380 A. tauschii genotypes were used to evaluate their phosphorus deficiency tolerance using root system at seedling stage. The results indicate that compared to applied phosphorus condition, genetic variation of the tested traits among 380 A. tauschii genotypes was significant under non-applied phosphorus condition. Heritability estimates showed that root dry weight, shoot dry weight, and total dry weight is highly inheritable. Principal component (PC) analysis showed that first four PCs explained 81.387 % of total variation. S value and phosphorus deficiency tolerance index (PDTI) showed that all A. tauschii genotypes can be divided into three groups, high (17 %), moderate (58 %) and low (25 %) tolerance groups. Compared with other cultivated wheat using S value and PDTI, the top five tolerant A. tauschii genotypes have stronger ability of phosphorus deficiency tolerance than other cultivated wheat. In conclusion, we systematically evaluated large-scale A. tauschii genotypes, and 63 tolerant lines and some root traits used as selection criteria were selected. The top five tolerant A. tauschii genotypes have relative higher tolerance than other wheat, and can be further used for wheat genetic improvement and molecular breeding for abiotic stress tolerance.




Similar content being viewed by others
References
Annamária S, Istvá M, Márta ML (2008) Utilisation of Aegilops (goatgrass) species to widen the genetic diversity of cultivated wheat. Euphytica 163:1–19
Bai DY, Feng GH, Zhang HY, Chen RZ, Wang XJ (2007) Screening of wheat genotypes with high phosphorus efficiency under low phosphorus stress. J Triticeae Crops 27:407–410
Bouma TJ, Nielsen KL, Koutstaal B (2000) Sample preparation and scanning protocol for computerized analysis of root length and diameter. Plant Soil 218:185–196
CAY (1991) China Agriculture Yearbook. Chinese Agricultural Press, Beijing
CAY (2006) China Agriculture Yearbook. Chinese Agricultural Press, Beijing
Colmer TD, Flowers TJ, Munns R (2006) Use of wild relatives to improve salt tolerance in wheat. J Exp Bot 57:1059–1078
Cox TS, Hatchett JH (1994) Hessian fly resistance gene H26 transferred from Triticum tauschii to common wheat. Crop Sci 34:958–960
Cox TS, Raupp WJ, Wilson DL, Gill BS, Leath S, Bockus WW et al (1992) Resistance to foliar diseases in a collection of Triticum tauschii germplasm. Plant Dis 76:1061–1064
Cui DQ, Hao X, Li Y, Liu YY (2004) Progress of genetic study on phosphorus efficiency of wheat. J Triticeae Crops 24:110–113
Damania AB, Pecetti L (1990) Varialility in a collection of Aegilops species and evaluation for yellow rust resistance at two location in northern Syria. J Genet Breed 44:97–102
Dimov A, Zaharieva M, Mihova S (1993) Rust and powdery mildew resistance in Aegilops accessions from Bulgaria. In: Damania AB (ed) Biodiversity and wheat improvement. Wiley, New York, pp 165–169
Draper NR, Smith H (1966) Applied regression analysis. Wiley, New York, p 7407
Friebe B, Jiang J, Raupp WJ, McIntosh RA, Gill BS (1996) Characterization of wheat alien translocations conferring resistance to diseases and pests: current status. Euphytica 71:59–83
George TS, Richardson AE (2008) Potential and limitations to improving crops for enhanced phosphorus utilization. In: White PJ, Hammond JP (eds) Ecophysiology of plant-phosphorus interactions. Springer, Dordrecht, pp 247–270
Gill BS, Browder LE, Hatchett JH, Harvey TL, Martin TJ, Raupp WJ, Sharma HC, Waines JG (1983) Disease and insect resistance in wild wheats. In: Sakamoto S (ed) Proc 6th Int Wheat Genet Symp, Faculty of Agriculture, Kyoto University, Japan, pp 783–792
Gill BS, Sharma HC, Raupp WJ, Browder LE, Hatchett JH, Harvey TL (1985) Evaluation of Aegilops species for resistance to wheat powdery mildew, wheat leaf rust, Hessian fly and greenbuy. Plant Breed 69:314–316
Gill BS, Hatchett JH, Raupp WJ (1987) Chromosomal mapping of Hessian fly resistance gene H13 in the D genome of wheat. J Hered 78:97–100
Grant CA, Flaten DN, Tomasiewicz DJ, Sheppard SC (2001) The importance of early season phosphorus nutrition. Can J Plant Sci 81:211–224
Guo Y, Kong FM, Xu YF, Zhao Y, Liang X, Wang YY et al (2012) QTL mapping for seedling traits in wheat grown under varying concentrations of N, P and K nutrients. Theor Appl Genet 124:851–865
Hammond JP, White PJ (2008) Sucrose transport in the phloem: integrating root responses to phosphorus starvation. J Exp Bot 59:93–109
Hammond JP, Broadley MR, White PJ, King GJ, Bowen HC, Hayden R, Meacham MC, Mead A, Overs T, Spracklen WP, Greenwood DJ (2009) Shoot yield drives phosphorus use efficiency in Brassica oleracea and correlates with root architecture traits. J Exp Bot 60:1953–1968
Hermans C, Hammond JP, White PJ, Verbruggen N (2006) How do plants respond to nutrient shortage by biomass alocaion? Trends Plant Sci 11:610–617
Hoagland DR, Arnon IR (1950) The water culture method for growing plants without soils. Circ Calif Agric Exp Stn 347:4–32
Horst WJ, Abdou M, Wiesler F (1993) Genotypic differences in phophorus efficiency of wheat. Plant Soil 155:293–296
Iyamuremye F, Dick RP, Baham J (1996) Organic amendment and phosphorus dynamics: II. Distribution of soil phosphorus fractions. Soil Sci 161:436–443
Jain A, Vasconcelos MJ, Raghothama KG, Sahi SV (2007) Molecular mechanisms of plant adaptation to phosphate deficiency. In: Janick J (ed) Plant breeding reviews. Wiley, NJ, pp 359–419
Jolliffe IT (2002) Principal component analysis, 2nd edn. Springer, New York, pp 1–10
Kong ZX, Yang LL, Zhang ZZ, Ma ZQ (2010) Screening of wheat germplasms tolerant to low phosphorus. J Triticeae Crops 30:591–595
Levent O, Selim E, Bulent T, Ismail C (2005) Variation in phosphorus efficiency among 73 bread and durum wheat genotypes grown in a phosphorus-deficient calcareous soil. Plant Soil 269:69–80
Li YF, Luo AC, Wang WM, Cai BX, Hu XY, Yang X (2005) Genotypic variation of rice in yield, phosphorus uptake and utilization at different phosphorus supply. Chin J Soil Sci 36:365–368
Li GQ, Li ZF, Yang WY, Zhang Y, He ZH, Xu SC, Singh RP, Qu YY, Xia XC (2006) Molecular mapping of stripe rust resistance gene YrCH42 in Chinese wheat cultivar Chuanmai 42 and its allelism with Yr 24 and Yr26. Theor Appl Genet 112:1434–1440
Li CY, Zhang H, Ma L, Li C (2012) Comprehensive evaluation on physiological indices of nitrogen absorbion and utilization in winter wheat at the seedling stage. Plant Nutr Fertil Sci 18:523–530
Liu Y, Li ZC, Mi GH, Zhang HL, Mu P, Wang XK (2005) Screening and identification for tolerance to low-phosphorus stress of rice germplasm (Oryza sativa L.). Acta Agron Sin 31:238–242
Lu DL, Yan FB, Lu WP (2010) Genotypic difference of phosphorus uptake and utilization in sweet maize varieties. J Yangzhou Univ 31:60–64
Lynch JP (2007) Roots of the second green revolution. Aust J Bot 55:493–512
Ma WQ, Zhang FS, Zhang WF (2005) Fertilizer production and consumption and the resources, environment, food security and sustainable development in China. Resour Sci 27:33–40
Magalhães PC, De Souza TC, Cantão FRO (2011) Early evaluation of root morphology of maize genotypes under phosphorus deficiency. Plant Soil Environ 57:135–138
Marschner H (1995) Functions of mineral nutrients: macronutrients. Miner Nutr High Plants 2:379–396
Molnár I, Gáspár L, Sárvári É, Dulai S, Hoffmann B, Molnár-Láng M, Galiba G (2004) Physiological and morphological responses to water stress in Aegilops biuncialis and Triticum aestivum genotypes with differing tolerance to drought. Funct Plant Biol 31:1149–1159
Raghothama KG, Karthikeyan AS (2005) Phosphate acquisition. Plant Soil 274:37–49
Raupp WJ, Amri A, Hatchett JH, Gill BS, Wilson DL, Cox TS (1993) Chromosomal location of Hessian fly-resistance genes H22, H23 and H24 derived from Triticum tauschii in the D genome of wheat. J Hered 84:142–145
Raupp WJ, Gill BS, Friebe B, Wilson DL, Cox TS, Sears RG (1995) The Wheat Genetic Resource Center: Germplasm conservation, evaluation and utilization. Proc 8th Int Wheat Genet Symp, China Agricultural Scientech Press, Beijing, China pp 469–475
Raupp WJ, Friebe B, Wilson DL, Cox TS, Gill BS (1997) Kansas State’s Wheat Genetics Resource Center provides unique oasis for germplasm research. Diversity 13:21–23
Ren YZ (2012) Effects of low phosphorus stress on different wheat varieties at seedling stage. Chin Agric Sci Bull 28:40–44
Smith SE, Kuehl RO, Ray IM, Hui R, Soleri D (1998) Evaluation of simple methods for estimating broad-sense heritability in stands of randonly planted genotypes. Crop Sci 38:1125–1129
Su JY, Xiao YM, Li M, Liu QY, Li B, Tong YP, Jia JZ, Li ZS (2006) Mapping QTLs for phosphorus-deficiency tolerance at wheat seedling stage. Plant Soil 281:25–36
Sun HG, Zhang FS (2002) Morphology of wheat roots under low-phosphorus stress. Chin J Appl Ecol 13:295–299
Tahir S, Alam SM, Ahmad Z (2011) Wheat yield and phosphorus fertilizer efficiency as influenced by pre-incubated use of single superphosphate and poultry litter and its time of application. J Plant Nutr 34:1034–1040
Teng W, Deng Y, Chen XP, Xu XF, Chen RY, Lv Y et al (2013) Characterization of root response to phosphorus supply from morphology to gene analysis in field-grown wheat. Exp Bot 64:1403–1411
Turner BL, Paphazy MJ, Haygarth PM, Mckelvie ID (2002) Inositol phosphates in the environment. Philos Trans R Soc Lond B Biol Sci 357:449–469
Valkoun J, Dostal J, Kucerová D (1990) Triticum × Aegilops hybrids through embryo culture. Biotechnol Agric For 13:152–166
Van Slageren MW (1994) Wild wheats: a monograph of Aegilops L. and Amblyopyrum (Jaub & Spach) Eig (Poaceae). Wageningen Agricultural Univerisity Papers, Wageningen, The Netherlands, pp 7–94
Vance CP (2001) Plant nutrition in a world of declining renewable resources. Plant Physiol 127:390–397
Vance CP, Uhde-Stone C, Allan DL (2003) Phosphorus acquisition and use: critical adaptations by plants for securing a nonrenewable resource. New Phytol 157:423–447
Wang XR, Shen JB, Liao H (2010) Acquisition or utilization, which is more critical for enhancing phosphorus efficiency in modern crops. Plant Sci 179:302–305
Wu ZS (1990) Wheat breeding. Chinese Agricultural Press, Beijing
Xiong YS, Yuan JF, Hao FX, Zhao SJ, Peng CL, Xu XY (2008) Advance on wheat genotype diversity with high nutrient efficiency. Hubei Agric Sci 47:1084–1087
Yang M, Ding GD, Shi L, Feng J, Xu FS, Meng JL (2010) Quantitative trait loci for root morphology in response to low phosphorus stress in Brassica napus. Theor Appl Genet 121:181–193
Yang XB, Zhang XZ, Li TX, Wu DY (2012) Differences in phosphorus utilization efficiency among wheat cultivars. Chin J Appl Ecol 23:60–66
Yen J, Yang JL (1984) Aegilops tauschii in Yili Xinjiang region. Crops J 3:1–8
Zaharieva M, Monneveux P, Henry M, Rivoal R, Valkoun J, Nachit MM (2001) Evaluation of a collection of wild wheat relative Aegilops geniculata Roth and identification of potential sources for useful traits. Euphytica 119:33–38
Zhang LM, He LY, Li JS, Xu SZ (2004a) Investigation of Maize Inbred Lines on Tolerance to Low-Phosphorus Stress at Seedling Stage. Sci Agric Sin 37:1955–1959
Zhang Y, Yang WY, Hu XR, Yu Y, Zou YC, Li QM (2004b) Anaylsis of agronomic characters of new wheat variety Chuanmai 42 derived from synthetics (Triticum duru Aegilops tauschii). Southwest China J Agric Sci 17:141–145
Zhang JH, Gao SB, Yang KC, Zhang ZM, Lin HJ, Huang N et al (2008) Screening and identification for tolerance to low phosphorus stress of Maize germplasm resources. J Plant Genet Resour 9:335–339
Zhao HT, Wang RF, Xu YF, An DG (2011) Screening and evaluating low nitrogen tolerant wheat genotype at seedling stage. Chin J Eco-Agric 19:1199–1204
Zhong GC, Mu SM, Zhang ZB (2003) Distant hybridization of triticeae. Science Press, Beijing
Acknowledgments
We thank Assad Siham (ICARDA, Syria), Jon W. Raupp (Kansas State University, USA), Shuhei Nasuda (Komugi, Japan) and Harold Bockelman (USDA, USA) for plant materials. This work was supported by the National Natural Science Foundation of China (31301317) and the National Basic Research Program of China (2014CB147200).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, L., Liu, K., Mao, S. et al. Large-scale screening for Aegilops tauschii tolerant genotypes to phosphorus deficiency at seedling stage. Euphytica 204, 571–586 (2015). https://doi.org/10.1007/s10681-014-1327-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-014-1327-6