Abstract
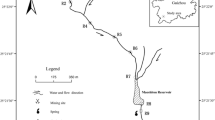
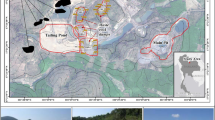
Acid mine drainage (AMD) has resulted in significant risks to both human health and the environment of the Han River watershed. In this study, water and sediment samples from typical mine adits were selected to investigate the hydrogeochemical characteristics and assess the environmental impacts of AMD. The interactions between coexisting chemical factors, geochemical processes in the mine adit, and the causes of AMD formation are discussed based on statistical analysis, mineralogical analysis, and geochemical modeling. The results showed that the hydrochemical types of AMD consisted of SO4–Ca–Mg, SO4–Ca, and SO4–Mg, with low pH and extremely high concentrations of Fe and SO42–. The release behaviors of most heavy metals are controlled by the oxidation of sulfide minerals (mainly pyrite) and the dissolution/precipitation of secondary minerals. Along the AMD pathway in the adit, the species of Fe-hydroxy secondary minerals tend to initially increase and later decrease. The inverse model results indicated that (1) oxidative dissolution of sulfide minerals, (2) interconversion of Fe-hydroxy secondary minerals, (3) precipitation of gypsum, and (4) neutralization by calcite are the main geochemical reactions in the adit, and chlorite might be the major neutralizing mineral of AMD with calcite. Furthermore, there were two sources of AMD in abandoned mine adits: oxidation of pyrite within the adits and infiltration of AMD from the overlying waste rock dumps. The findings can provide deeper insight into hydrogeochemical processes and the formation of AMD contamination produced in abandoned mine adits under similar mining and hydrogeological conditions.











Similar content being viewed by others
Data availability
The data used during the current research are available from the corresponding author on reasonable request.
References
Acero, P., Ayora, C., Torrentó, C., & Nieto, J.-M. (2006). The behavior of trace elements during schwertmannite precipitation and subsequent transformation into goethite and jarosite. Geochimica Et Cosmochimica Acta, 70, 4130–4139. https://doi.org/10.1016/j.gca.2006.06.1367
Acharya, B. S., & Kharel, G. (2020). Acid mine drainage from coal mining in the United States: An overview. Journal of Hydrology, 588, 125061. https://doi.org/10.1016/j.jhydrol.2020.125061
Akcil, A., & Koldas, S. (2006). Acid mine drainage (AMD): Causes, treatment and case studies. Journal of Cleaner Production, 14, 1139–1145. https://doi.org/10.1016/j.jclepro.2004.09.006
Alumaa, P., Kirso, U., Petersell, V., & Steinnes, E. (2002). Sorption of toxic heavy metals to soil1 1Following a presentation given as part of the International Congress on Environmental Health and the 4th Annual Meeting of the International Society of Environmental Medicine (ISEM), 1–4 October, 2000, Hannover Germany. International Journal of Hygiene and Environmental Health, 204, 375–376. https://doi.org/10.1078/1438-4639-00114
Asta, M. P., Calleja, M. L., Pérez-López, R., & Auqué, L. F. (2015). Major hydrogeochemical processes in an acid mine drainage affected estuary. Marine Pollution Bulletin, 91, 295–305. https://doi.org/10.1016/j.marpolbul.2014.11.023
Baleeiro, A., Fiol, S., Otero-Fariña, A., & Antelo, J. (2018). Surface chemistry of iron oxides formed by neutralization of acidic mine waters: Removal of trace metals. Applied Geochemistry, 89, 129–137. https://doi.org/10.1016/j.apgeochem.2017.12.003
Ball J. W., Nordstrom D. K. (1991). User's manual for WATEQ4F, with revised thermodynamic data base and text cases for calculating speciation of major, trace, and redox elements in natural waters. 91–183, Survey USG. http://pubs.er.usgs.gov/publication/ofr91183
Bigham, J. M., Schwertmann, U., & Pfab, G. (1996a). Influence of pH on mineral speciation in a bioreactor simulating acid mine drainage. Applied Geochemistry, 11, 845–849. https://doi.org/10.1016/S0883-2927(96)00052-2
Bigham, J. M., Schwertmann, U., Traina, S. J., Winland, R. L., & Wolf, M. (1996b). Schwertmannite and the chemical modeling of iron in acid sulfate waters. Geochimica Et Cosmochimica Acta, 60, 2111–2121. https://doi.org/10.1016/0016-7037(96)00091-9
Bradl, H. B. (2004). Adsorption of heavy metal ions on soils and soils constituents. Journal of Colloid and Interface Science, 277, 1–18. https://doi.org/10.1016/j.jcis.2004.04.005
Brandt, F., Bosbach, D., Krawczyk-Bärsch, E., Arnold, T., & Bernhard, G. (2003). Chlorite dissolution in the acid ph-range: A combined microscopic and macroscopic approach. Geochimica Et Cosmochimica Acta, 67, 1451–1461. https://doi.org/10.1016/S0016-7037(02)01293-0
Burton, E. D., Bush, R. T., Sullivan, L. A., & Mitchell, D. R. G. (2007). Reductive transformation of iron and sulfur in schwertmannite-rich accumulations associated with acidified coastal lowlands. Geochimica Et Cosmochimica Acta, 71, 4456–4473. https://doi.org/10.1016/j.gca.2007.07.007
Cánovas, C. R., Macías, F., & Pérez-López, R. (2016). Metal and acidity fluxes controlled by precipitation/dissolution cycles of sulfate salts in an anthropogenic mine aquifer. Journal of Contaminant Hydrology, 188, 29–43. https://doi.org/10.1016/j.jconhyd.2016.02.005
Caraballo, M. A., Macías, F., Rötting, T. S., Nieto, J. M., & Ayora, C. (2011). Long term remediation of highly polluted acid mine drainage: A sustainable approach to restore the environmental quality of the Odiel river basin. Environmental Pollution, 159, 3613–3619. https://doi.org/10.1016/j.envpol.2011.08.003
Chen, M., Lu, G., Wu, J., Sun, J., Yang, C., Xie, Y., Wang, K., Deng, F., Yi, X., & Dang, Z. (2020). Acidity and metallic elements release from AMD-affected river sediments: Effect of AMD standstill and dilution. Environmental Research, 186, 109490. https://doi.org/10.1016/j.envres.2020.109490
Chidambaram, S., Prasanna, M. V., Singaraja, C., Thilagavathi, R., Pethaperumal, S., & Tirumalesh, K. (2012). Study on the saturation index of the carbonates in the groundwater using WATEQ4F, in layered coastal aquifers of Pondicherry. Journal of the Geological Society of India, 80, 813–824. https://doi.org/10.1007/s12594-012-0210-0
Davila, J. M., Sarmiento, A. M., Aroba, J., Fortes, J. C., Grande, J. A., Santisteban, M., Cordoba, F., Leiva, M., & Luís, A. T. (2021). Application of a fuzzy logic based methodology to validate the hydrochemical characterization and determining seasonal influence of a watershed affected by acid mine drainage. International Journal of Environmental Research and Public Health, 18, 4693. https://doi.org/10.3390/ijerph18094693
Dong, Y., Lu, H., & Lin, H. (2023). Release characteristics of heavy metals in high-sulfur coal gangue: Influencing factors and kinetic behavior. Environmental Research, 217, 114871. https://doi.org/10.1016/j.envres.2022.114871
Fan, R., Qian, G., Li, Y., Short, M. D., Schumann, R. C., Chen, M., Smart, R. S. C., & Gerson, A. R. (2022). Evolution of pyrite oxidation from a 10-year kinetic leach study: Implications for secondary mineralisation in acid mine drainage control. Chemical Geology, 588, 120653. https://doi.org/10.1016/j.chemgeo.2021.120653
Feng, B., Zhong, Y., He, J., Sha, X., Fang, L., Xu, Z., & Qi, Y. (2023). Nitrogen sources and conversion processes in shallow groundwater around a plain lake (Northwest China): Evidenced by multiple isotopes and water chemistry. Chemosphere, 337, 139322. https://doi.org/10.1016/j.chemosphere.2023.139322
Ficklin W., Plumlee G., Smith K. & McHugh J. (1992). Geochemical classification of mine drainages and natural drainages in mineralized areas, International symposium on water-rock interaction (pp. 381–384).
Galhardi, J. A., & Bonotto, D. M. (2016). Hydrogeochemical features of surface water and groundwater contaminated with acid mine drainage (AMD) in coal mining areas: A case study in southern Brazil. Environmental Science and Pollution Research, 23, 18911–18927. https://doi.org/10.1007/s11356-016-7077-3
Grande, J. A., de la Torre, M. L., Cerón, J. C., Beltrán, R., & Gómez, T. (2010). Overall hydrochemical characterization of the Iberian Pyrite Belt. Main acid mine drainage-generating sources (Huelva, SW Spain). Journal of Hydrology, 390, 123–130. https://doi.org/10.1016/j.jhydrol.2010.06.001
Grande, J. A., De La Torre, M. L., Santisteban, M., Valente, T., Fernandez, J. P., & Pérez-Ostalé, E. (2016). Spatial evolution of an AMD stream in the Iberian Pyrite Belt: Process characterization and control factors on the hydrochemistry. Hydrological Sciences Journal, 61, 1503–1511. https://doi.org/10.1080/02626667.2014.983515
Gray, N. F. (1996). The Use of an Objective Index for the Assessment of the Contamination of Surface Water and Groundwater by Acid Mine Drainage. Water Environment Journal, 10, 332–340. https://doi.org/10.1111/j.1747-6593.1996.tb00061.x
Han, Y.-S., Youm, S.-J., Oh, C., Cho, Y.-C., & Ahn, J. S. (2017). Geochemical and eco-toxicological characteristics of stream water and its sediments affected by acid mine drainage. CATENA, 148, 52–59. https://doi.org/10.1016/j.catena.2015.11.015
Hermassi, M., Granados, M., Valderrama, C., Ayora, C., & Cortina, J. L. (2022). Recovery of rare earth elements from acidic mine waters: An unknown secondary resource. Science of the Total Environment, 810, 152258. https://doi.org/10.1016/j.scitotenv.2021.152258
Holmström, H., Salmon, U. J., Carlsson, E., Petrov, P., & Öhlander, B. (2001). Geochemical investigations of sulfide-bearing tailings at Kristineberg, northern Sweden, a few years after remediation. Science of the Total Environment, 273, 111–133. https://doi.org/10.1016/S0048-9697(00)00850-0
Hu, J., Zhu, C., Long, Y., Yang, Q., Zhou, S., Wu, P., Jiang, J., Zhou, W., & Hu, X. (2021). Interaction analysis of hydrochemical factors and dissolved heavy metals in the karst Caohai Wetland based on PHREEQC, cooccurrence network and redundancy analyses. Science of the Total Environment, 770, 145361. https://doi.org/10.1016/j.scitotenv.2021.145361
Kim, D.-M., Kwon, H.-L., & Im, D.-G. (2023). Determination of contamination sources and geochemical behaviors of metals in soil of a mine area using Cu, Pb, Zn, and S isotopes and positive matrix factorization. Journal of Hazardous Materials, 447, 130827. https://doi.org/10.1016/j.jhazmat.2023.130827
Kim, K.-H., Yun, S.-T., Mayer, B., Lee, J.-H., Kim, T.-S., & Kim, H.-K. (2015). Quantification of nitrate sources in groundwater using hydrochemical and dual isotopic data combined with a Bayesian mixing model. Agriculture, Ecosystems & Environment, 199, 369–381. https://doi.org/10.1016/j.agee.2014.10.014
Kotalik, C. J., Cadmus, P., & Clements, W. H. (2021). Before-After Control-Impact field surveys and novel experimental approaches provide valuable insights for characterizing stream recovery from acid mine drainage. Science of the Total Environment, 771, 145419. https://doi.org/10.1016/j.scitotenv.2021.145419
Li, J., Kawashima, N., Fan, R., Schumann, R. C., Gerson, A. R., & Smart, R. S. (2014). Method for distinctive estimation of stored acidity forms in acid mine wastes. Environmental Science and Technology, 48, 11445–11452. https://doi.org/10.1021/es502482m
Li, S., Xu, Z., Wang, H., Wang, J., & Zhang, Q. (2009). Geochemistry of the upper Han River basin, China: 3: Anthropogenic inputs and chemical weathering to the dissolved load. Chemical Geology, 264, 89–95. https://doi.org/10.1016/j.chemgeo.2009.02.021
Li, S., & Zhang, Q. (2010). Risk assessment and seasonal variations of dissolved trace elements and heavy metals in the Upper Han River China. Journal of Hazardous Materials, 181, 1051–1058. https://doi.org/10.1016/j.jhazmat.2010.05.120
Liu, X., Zuo, R., Wang, J., He, Z., & Li, Q. (2021). Advances in researches on ammonia, nitrite and nitrate on migration and transformation in the groundwater level fluctuation zone. Hydrogeology & Engineering Geology, 48, 27–36. https://doi.org/10.16030/j.cnki.issn.1000-3665.202007033
Liu, Q., Chen, B., Haderlein, S., Gopalakrishnan, G., & Zhou, Y. (2018). Characteristics and environmental response of secondary minerals in AMD from Dabaoshan Mine, South China. Ecotoxicology and Environmental Safety, 155, 50–58. https://doi.org/10.1016/j.ecoenv.2018.02.017
Liu, Y., Wei, L., Wu, Q., Luo, D., Xiao, T., Wu, Q., Huang, X., Liu, J., Wang, J., & Zhang, P. (2023). Impact of acid mine drainage on groundwater hydrogeochemistry at a pyrite mine (South China): A study using stable isotopes and multivariate statistical analyses. Environmental Geochemistry and Health, 45, 771–785. https://doi.org/10.1007/s10653-022-01242-8
López, J., Reig, M., Gibert, O., & Cortina, J. L. (2019). Recovery of sulphuric acid and added value metals (Zn, Cu and rare earths) from acidic mine waters using nanofiltration membranes. Separation and Purification Technology, 212, 180–190. https://doi.org/10.1016/j.seppur.2018.11.022
Mayo, A. L., Petersen, E. C., & Kravits, C. (2000). Chemical evolution of coal mine drainage in a non-acid producing environment, Wasatch Plateau, Utah USA. Journal of Hydrology, 236, 1–16. https://doi.org/10.1016/S0022-1694(00)00277-8
McGregor, R. G., Blowes, D. W., Jambor, J. L., & Robertson, W. D. (1998). Mobilization and attenuation of heavy metals within a nickel mine tailings impoundment near Sudbury, Ontario. Canada. Environmental Geology, 36, 305–319. https://doi.org/10.1007/s002540050346
Moreno-González, R., Macías, F., Olías, M., & Ruiz, C. C. (2022). Temporal evolution of acid mine drainage (AMD) leachates from the abandoned tharsis mine (Iberian Pyrite Belt, Spain). Environmental Pollution, 295, 118697. https://doi.org/10.1016/j.envpol.2021.118697
Naidu, G., Ryu, S., Thiruvenkatachari, R., Choi, Y., Jeong, S., & Vigneswaran, S. (2019). A critical review on remediation, reuse, and resource recovery from acid mine drainage. Environmental Pollution, 247, 1110–1124. https://doi.org/10.1016/j.envpol.2019.01.085
Newman, C. P., Walton-Day, K., Runkel, R. L., & Wilkin, R. T. (2023). Mechanisms of water-rock interaction and implications for remediating flooded mine workings elucidated from environmental tracers, stable isotopes, and rare earth elements. Applied Geochemistry, 157, 105769. https://doi.org/10.1016/j.apgeochem.2023.105769
Nordstrom D., Alpers C. (1999). Geochemistry of acid mine waters, The Environmental Geochemistry of Mineral Deposits, pp. 133–160
Parkhurst, D. L., & Appelo, C. (1999). User’s guide to PHREEQC (Version 2): A computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations. Water-Resources Investigations Report, 99, 312.
Qian, G., Fan, R., Short, M. D., Schumann, R. C., Li, J., Li, Y., Smart, R. S. C., & Gerson, A. R. (2019). Evaluation of the rate of dissolution of secondary sulfate minerals for effective acid and metalliferous drainage mitigation. Chemical Geology, 504, 14–27. https://doi.org/10.1016/j.chemgeo.2018.12.003
Sáinz, A., Grande, J. A., & de la Torre, M. L. (2003). Analysis of the impact of local corrective measures on the input of contaminants from the Odiel River to the Ría of Huelva (Spain). Water, Air, and Soil Pollution, 144, 375–389. https://doi.org/10.1023/A:1022905502320
Sánchez, E. J., López, P. E., Santofimia, E., Aduvire, O., Reyes, J., & Barettino, D. (2005). Acid mine drainage in the Iberian Pyrite Belt (Odiel river watershed, Huelva, SW Spain): Geochemistry, mineralogy and environmental implications. Applied Geochemistry, 20, 1320–1356. https://doi.org/10.1016/j.apgeochem.2005.01.011
Santisteban, M., Luís, A., Grande, J., Aroba, J., Dávila, J., Sarmiento, A., Garrido, J. C., Cordoba, F., & Rodriguez, A. (2022). Hydrochemical characterization of an acid mine effluent from Concepcion mine using classical statistic and fuzzy logic techniques. Minerals, 12, 464. https://doi.org/10.3390/min12040464
Sarmiento, A. M., Nieto, J. M., Olías, M., & Cánovas, C. R. (2009). Hydrochemical characteristics and seasonal influence on the pollution by acid mine drainage in the Odiel river Basin (SW Spain). Applied Geochemistry, 24, 697–714. https://doi.org/10.1016/j.apgeochem.2008.12.025
Schmidt, M. J., Pankavich, S. D., Navarre-Sitchler, A., Engdahl, N. B., Bolster, D., & Benson, D. A. (2020). Reactive particle-tracking solutions to a benchmark problem on heavy metal cycling in lake sediments. Journal of Contaminant Hydrology, 234, 103642. https://doi.org/10.1016/j.jconhyd.2020.103642
Schoepfer, V. A., & Burton, E. D. (2021). Schwertmannite: A review of its occurrence, formation, structure, stability and interactions with oxyanions. Earth-Science Reviews. https://doi.org/10.1016/j.earscirev.2021.103811
Servida, D., Grieco, G., & De Capitani, L. (2009). Geochemical hazard evaluation of sulphide-rich iron mines: The Rio Marina district (Elba Island, Italy). Journal of Geochemical Exploration, 100, 75–89. https://doi.org/10.1016/j.gexplo.2008.03.005
Sharif, M. U., Davis, R. K., Steele, K. F., Kim, B., Kresse, T. M., & Fazio, J. A. (2008). Inverse geochemical modeling of groundwater evolution with emphasis on arsenic in the Mississippi River Valley alluvial aquifer, Arkansas (USA). Journal of Hydrology, 350, 41–55. https://doi.org/10.1016/j.jhydrol.2007.11.027
Shum, M., & Lavkulich, L. (1999). Speciation and solubility relationships of Al, Cu and Fe in solutions associated with sulfuric acid leached mine waste rock. Environmental Geology, 38, 59–68. https://doi.org/10.1007/s002540050401
Simate, G. S., & Ndlovu, S. (2014). Acid mine drainage: Challenges and opportunities. Journal of Environmental Chemical Engineering, 2, 1785–1803. https://doi.org/10.1016/j.jece.2014.07.021
Soltani, N., Moore, F., Keshavarzi, B., & Sharifi, R. (2014). Geochemistry of trace metals and rare earth elements in stream water, stream sediments and acid mine drainage from Darrehzar Copper Mine, Kerman. Iran. Water Qual. Exposure Health, 6, 97–114. https://doi.org/10.1007/s12403-014-0114-x
Tomiyama, S., Igarashi, T., Tabelin, C. B., Tangviroon, P., & Ii, H. (2020). Modeling of the groundwater flow system in excavated areas of an abandoned mine. Journal of Contaminant Hydrology, 230, 103617. https://doi.org/10.1016/j.jconhyd.2020.103617
Torres-Martínez, J. A., Mora, A., Mahlknecht, J., Daesslé, L. W., Cervantes-Avilés, P. A., & Ledesma-Ruiz, R. (2021). Estimation of nitrate pollution sources and transformations in groundwater of an intensive livestock-agricultural area (Comarca Lagunera), combining major ions, stable isotopes and MixSIAR model. Environmental Pollution, 269, 115445. https://doi.org/10.1016/j.envpol.2020.115445
Tu, Z., Wu, Q., He, H., Zhou, S., Liu, J., He, H., Liu, C., Dang, Z., & Reinfelder, J. R. (2022). Reduction of acid mine drainage by passivation of pyrite surfaces: A review. Science of the Total Environmen. https://doi.org/10.1016/j.scitotenv.2022.155116
Wu, Y., Shen, Z., & Zhong, Z. (2006). Chemical origin of acid mine drainage and its computer simulation with Phreeqc—A case study of Zibo coal mining district. Chinese Journal of Geochemistry, 25, 26–26. https://doi.org/10.1007/BF02839783
Xiying, G., Binghui, C., Lina, C., Wen, L., & Yao, Z. (2020). The relationship among iron ion, secondary mineral assemblage and heavy metals distribution in the acid mine drainage (AMD) at Dabaoshan mine, Guangdong province. Acta Scientiarum Naturalium Universitatis Sunyatseni, 59, 12. https://doi.org/10.13471/j.cnki.acta.snus.2020.03.002
Yang, C., Lu, G., Xie, Y., Guo, L., Chen, M., Ge, L., & Dang, Z. (2021). Sulfate migration and transformation characteristics in paddy soil profile affected by acid mine drainage. Environmental Research, 200, 111732. https://doi.org/10.1016/j.envres.2021.111732
Yue, M., Zhao, F. H., & Ren, D. Y. (2006). Simulating experiments to the soluble metals migration of Co, Ni, Zn, Cd, Al, Cr, As and Pb in acid coal-mine drainage. Acta Scientiae Circumstantiae, 26, 949–953. https://doi.org/10.13671/j.hjkxxb.2006.06.013
Zhang, Y., Zhang, R., Wu, P., Chen, S., Yang, Y., An, L., Fu, Y., & Zhang, Y. (2022). Adsorption mechanisms and effect factors of As(V) by AMD sludge composite material. Environmental Science. https://doi.org/10.13227/j.hjkx.202108230
Zhao, H., Xia, B., Qin, J., & Zhang, J. (2012). Hydrogeochemical and mineralogical characteristics related to heavy metal attenuation in a stream polluted by acid mine drainage: A case study in Dabaoshan Mine. Chinese Journal of Environmental Science, 24, 979–989. https://doi.org/10.1016/S1001-0742(11)60868-1
Acknowledgements
This research was mainly supported by the Geological and Mineral Engineering Survey Institute, Shaanxi. All the authors sincerely thank the researchers and workers for their help. We also thank the anonymous referees for their thorough reviews and constructive comments. They have helped improve the quality of the original manuscript.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
WC took part in investigation; formal analysis; methodology; writing—original draft; writing—review and editing. XK involved in investigation; methodology; writing—review and editing. WW took part in resources; writing—reviewing and editing. PL took part in investigation; formal analysis. All authors contributed to the article and approved the submitted version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
This article does not involve studies with animals or human participation.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chang, W., Ke, X., Wang, W. et al. Identifying sources of acid mine drainage and major hydrogeochemical processes in abandoned mine adits (Southeast Shaanxi, China). Environ Geochem Health 46, 60 (2024). https://doi.org/10.1007/s10653-024-01858-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10653-024-01858-y