Abstract
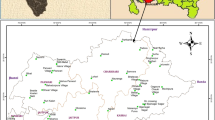
A study was conducted to bring out the relationship and behavior of different Saturation Index (SI) of carbonate minerals in layered coastal aquifers. Carbonates present in groundwater aids in different nature of the water like hardness, partial pressure of carbon-di-oxide (pCO2), pH and saturation index of different carbonate minerals at various temperatures. The SI of the carbonates helps us to define the thermodynamic stability of water and to find out the geochemical behavior of water. 98 groundwater samples were collected from specific aquifers (alluvium, upper Cuddalore, lower Cuddalore and Cretaceous) during Pre-Monsoon (May 2007) and Post-Monsoon (January 2008) seasons. The physicochemical parameters such as pH, EC, Ca, Mg, Na, K, Cl, HCO3, SO4 and PO4 were analyzed. Geochemical model, WATEQ4F was used to calculate the SI of different minerals. The SI was studied in relation to mHCO3 concentration, pCO2 and correlation between SI of different minerals.
Similar content being viewed by others
References
APHA (American Public Health Association) 1998 Standard methods for the examination of water and wastewater. 19th edition, APHA, Washington DC, USASS.
Barack, P. and Chen, Y.N. (1992) Equivalent radii of humic macromolecules from acid base titration. Soil Sci., v.154, pp.184–195.
Bartschat, B.M., Cabaniss, S.E. and Morel, F.M.M. (1992) Oligoeletrolyte model for cation binding by humic substances. Environ. Sci. Tech., v.26, pp.284–294.
Benedetti, M.F., Van Riemsdijk, W.H. and Koopal, L.K. (1996) Humic substances considered as a heterogeneous Donnan gel phase. Envion. Sci. Tech., v.30, pp.1805–1813.
CGWB (Centre for Groundwater Board). (1993) Groundwater Resources and Development Prospects in Pondicherry Region, Union Territory of Pondicherry, 63p.
Charlet, L., Wersion, P. and Srumm, W. (1990) Surface charge of MnCO3 and FeCO3. Geochim. Acta, v.54, pp.2329–2336.
Chidambaram, S., Karmegam, U., Sasidhar, P., Prasanna, M.V., Manivannan, R., Arunachalam, S., Manikandan, S. and Anandhan, P. (2011a) Significance of saturation index of certain clay minerals in shallow coastal groundwater, in and around Kalpakkam, Tamil Nadu, India. Jour. Earth Sys. Sci., v.120, no. 5, pp.897–909.
Chidambaram, S., Prasanna, M.V., Karmegam, U., Singaraja, C., Pethaperumal, S., Manivannan, R., Anandhan, P. and Tirumalesh, K. (2011b) Significance of pCO2 values in determining carbonate chemistry in groundwater of Pondicheery region, India. Front. Earth Sci. v.5, no.2, pp.197–206.
Chidambaram, S., Vijayakumar, V., Srinivasamoorthy, K., Anandhan, P., Prasanna, M.V. and Vasudevan, S. (2007) A study on variation in ionic composition of aqueous system in different lithounits around Perambalur Region, Tamil Nadu. Jour. Geol. Soc. India, v.70, pp.1061–1069.
Clarkson, Jr., Price, Tj. and Adams, Cj. (1992) Role of Metastable phases in the Spontaneous precipitation of calcium carbonate. Jour. Chem. Soc. Faraday Trans., v.88, pp.243–249.
De Wit, J.C.M., Van Reimsdijk, W.H., Koopal, L. and Proton, K. (1993) Binding to humic substances. I. Electrostatic effects. Environ. Sci. Tech., v.27, pp.2005–2014.
De Wit, J.C.M., Van Riemsdijk., W.H., Nederlof, L.K., Kinniburgh, D.G. and Koopal, L.K. (1990) Analysis of ionbinding on humic substances and the determination of intrinsic affinity distributions. Anal. Chim Acta, pp.198–207.
Domenico, P.A. and Schwartz, F.W. (1998) Physical and Chemical Hydrogeology, 2nd edn. (New York: Wiley) 506p.
Engebreston, R,R., Amos, T. and Von Wandruszka, R. (1996) Quantitative approach to humic acid associations. Environ. Sci. Tech., v.30, pp.990–997.
Gache, N. and Gal, Jy. (1998) Formation mechanisms of scale I. On the existence of the uncharged complex CaCO3 (Mecanisms de formation du tatre. I ZSur I’existence du complexnon charge CaCO3) Trib Eau, v.51(9).
Gal, Jy., Bollinger, Jc., Tolosa, H. and Gache, N. (1996) Calcium carbonate solubility: a reappraisal of scale formation and inhibition. Talanta, v.43, pp.1497–509.
Gal, Jy., Yannick Fovet. and Nathalie Gache. (2002) Mechanisms of scale formation and carbon dioxide partial pressure influence. Part I. Elaboration of experimental method and a scaling model. Water Res., v.36, pp.755–763.
Helgeson, H. (1967) Thermodynamics of complex dissociation in aqueous solution at elevated temperature. Jour. Phys. Chem., v.7, no.10, 3121p.
Helgeson, H. (1969) Thermodynamics of hydrothermal system at elevated temperature and pressure. Amer. Jour. Sci., v.267, 729p.
Helgeson, H. (1971) Kinetics of mass transfer among silicates in aqueous solution. Geochim. Comochim. Acta, v.35, 729p.
Marinsky, J. and Ephrain J. (1986) A unified physicochemical description of the protonation and metal ion complexation equilibria of natural organic acids (humic and fulvic acids).1analysis of the influence of polyelectrolyte properties on protonation equilibria in ionic media: fundamental concepts. Environ. Sci. Tech., v.20, pp.349–354.
Milne, C.J., Kinniburgh, D.G., De Witj, C.M., Van Reimsdijk, W.H. and Koopal, L.K. (1995) Analysis of proton binding by proton binding by a peat humic acid using a simple electrostatic model. Geochim. Cosmochim. Acta, v.59, pp.1101–1112.
Murphy, E.M. and Zachara, J.M. (1995) The role of Sorbed humic substances on the distribution of organic and inorganic contaminants in groundwater. Geoderma, v.67, pp.103–124.
Nair, K.M. and Rao, V.P. (1971) Result of shallow drilling in the area, north of Pondicherry, unpublished ONGC field season report of 1969–1970.
Nordstrom, D.K. and Ball. (1989) Ground water chemistry and water-rock intraction at stripa. Geochim. Cosmochim. Acta, v.53, pp.1727–1740.
Ogino, T., Suzuki, T. and Sawada, K. (1987) The formation and transformation mechanism of calcium carbonate in water. Geochim Cosmochim Acta, v.51, pp.2757–67.
Pethaperumal, S., Chidambaram, S., Prasanna, M.V., Verma, V.N., Balaji, K., Ramesh, R., Karmegam, U. and Paramaguru, P. (2008) A study on groundwater quality in the Pondicherry Region. Eco-Chronicle, v.3, no. 2, pp.85–90.
Pethaperumal, S. (2010) Study on groundwater chemistry in the Pondicherry region. Ph.D. thesis, Annamalai University, India.
Plummer, L.M. and Busenberg, E. (1982) The solubilities of calcite, aragonite and vertirite in CO2-H2O solutions between 0 and 90° C, and an evolution of the aquous model for the system CaCO2-CO2-H2O. Geochim. Cosmochim. Acta, v.46, pp.1011–40.
Prasanna, M.V., Chidambaram, S., Shahul Hameed, A. and Srinivasamoorthy, K. (2010) Study of evaluation of groundwater in Gadilam basin using hydrogeochemical and isotope data. Environ. Monit. Assess., v.168, pp.63–90.
Ryzhenko, B.N., Melnikova, G.L. and Shvarov. Yu, V. (1977) Main features in forming chemical composition of aqueous solution in the Earth’ crust. Geochimiya, v.8, 819p.
Ryzhenko, B.N., Melnikova, G.L. and Shvarov. Yu, V. (1981) Simulating formation of chemical composition in natural solutions with interactions in water-rock system. Geochimiya, v.4, pp.481–495.
Tipping, E. and Hurtly, M.A. (1992) A unifying model of cation binding by humic substances. Geochim. Cosmochim. Acta, v.56, pp.3627–3641.
Vasanthavigar, M., Srinivasamoorthy, K., Vijaragavan, K., Rajiv Ganthi, R., Chidambaram, S., Sarama, V.S., Anandhan, P., Manivannan, R. and Vasudevan, S. (2009) Hydrogeochemistry of Thirumanimuttar Basin: An indication of weathering and anthropogenic impact. Internat. Jour. Environ. Res., v.3, no.4, pp.617–628.
Vendorbosch, H.J. and Overmen, L.J. (1986) Critical pH as major parameters in the formation of calcium carbonate. Kema Tech Report, pp.119–124.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chidambaram, S., Prasanna, M.V., Singaraja, C. et al. Study on the saturation index of the carbonates in the groundwater using WATEQ4F, in layered coastal aquifers of Pondicherry. J Geol Soc India 80, 813–824 (2012). https://doi.org/10.1007/s12594-012-0210-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12594-012-0210-0