Abstract
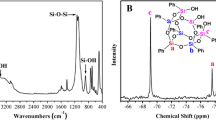
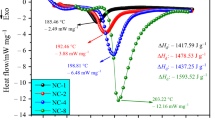
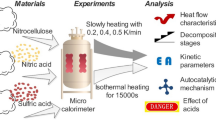
The thermal behavior of nitrocellulose (NC) with different nitrogen contents has been widely investigated in previous works. However, most of the experiments were carried out under high heating rates, a situation in which it is difficult to capture minor changes during the thermal decomposition process. In the present study, the thermal characteristics of NC with different nitrogen contents were theoretically and experimentally investigated using a CALVET heat flux calorimeter C80 at heating rates from 0.2 to 0.8 °C/min. Before thermal analysis, the results of high-precision scanning electron microscopy showed that more cracks and coarser surfaces were detected on NC fibers with higher nitrogen contents. It was found that a “turning point” was detected on the heat flow curves of NC with higher nitrogen contents. Furthermore, the heat flow curves of NC transformed from “bell shape” to “right triangles” with increases in the nitrogen content, which suggested that NC with high nitrogen contents exhibited autocatalysis characteristics under low heating rates. This characteristic was also confirmed by the isothermal experiment results that showed the autocatalytic properties became increasingly obvious by increasing the isothermal temperature. Moreover, the relevant chemical kinetic and thermodynamic parameters were obtained. The heat of the reaction (ΔH) increased and the activation energy values in the initial stages of NC decomposition decreased as the nitrogen content was increased. Furthermore, the critical explosion temperature (Tb), which is as an important parameter to evaluate the thermal hazard of NC, decreased by elevating the nitrogen content.












Similar content being viewed by others
References
Abd-Elghany M, Klapötke TM, Elbeih A (2017) Thermal behavior and decomposition kinetics of Bis (2, 2, 2-trinitroethyl)-oxalate as a high energy dense oxidizer and its mixture with nitrocellulose. Propellant Explos Pyrotech 42:1373–1381. https://doi.org/10.1002/prep.201700179
Batten JJ (1985) The agent of the autocatalytic thermal decomposition of aliphatic nitrate ester explosives. Int J Chem Kinet 17:1085–1090. https://doi.org/10.1002/kin.550171005
Bessières D, Lafitte T, Daridon JL, Randzio SL (2005) High pressure thermal expansion of gases: measurements and calibration. Thermochim Acta 428:25–30. https://doi.org/10.1016/j.tca.2004.09.020
Cuissinat C, Navard P, Heinze T (2008) Swelling and dissolution of cellulose, Part V: cellulose derivatives fibres in aqueous systems and ionic liquids. Cellulose 15:75–80. https://doi.org/10.1007/s10570-007-9159-3
Guo S, Wang QS, Sun JH, Liao X, Wang ZS (2009) Study on the influence of moisture content on thermal stability of propellant. J Hazard Mater 168:536–541. https://doi.org/10.1016/j.jhazmat.2009.02.073
He Y, He YP, Liu JH, Li P, Chen MY, Wei RC, Wang J (2017) Experimental study on the thermal decomposition and combustion characteristics of nitrocellulose with different alcohol humectants. J Hazard Mater 340:202–212. https://doi.org/10.1016/j.jhazmat.2017.06.029
Hurley MJ, Gottuk DT, Hall JR Jr, Harada K, Kuligowski ED, Puchovsky M, Torero JL, Watts JM Jr, Wieczorek CJ (2015) SFPE handbook of fire protection engineering. Springer, Berlin
Janković B (2010) The kinetic analysis of isothermal curing reaction of an unsaturated polyester resin: estimation of the density distribution function of the apparent activation energy. Chem Eng J 162:331–340. https://doi.org/10.1016/j.cej.2010.05.010
Jenkins PJ, Donald AM (1998) Gelatinisation of starch: a combined SAXS/WAXS/DSC and SANS study. Carbohydr Res 308:133–147. https://doi.org/10.1016/S0008-6215(98)00079-2
Jiang L, Xiao HH, He JJ, Sun Q, Gong L, Sun JH (2015) Application of genetic algorithm to pyrolysis of typical polymers. Fuel Process Technol 138:48–55. https://doi.org/10.1016/j.fuproc.2015.05.001
Jiang L, He JJ, Sun JH (2018) Sample width and thickness effects on upward flame spread over PMMA surface. J Hazard Mater 342:114–120. https://doi.org/10.1016/j.jhazmat.2017.08.022
Katoh K, Ito S, Kawaguchi S, Higashi E, Nakano K, Ogata Y, Wada Y (2010) Effect of heating rate on the thermal behavior of nitrocellulose. J Therm Anal Calorim 100:303–308. https://doi.org/10.1007/s10973-009-0478-5
Krause H (2013) Conversion concepts for commercial applications and disposal technologies of energetic systems. Springer, Berlin
Li KY, Huang X, Fleischmann C, Rein G, Ji J (2014) Pyrolysis of medium-density fiberboard: optimized search for kinetics scheme and parameters via a genetic algorithm driven by Kissinger’s method. Energy Fuels 28:6130–6139. https://doi.org/10.1021/ef501380c
Lin CP, Li JS, Tseng JM, Mannan MS (2016) Thermal runaway reaction for highly exothermic material in safe storage temperature. J Loss Prev Proc 40:259–265. https://doi.org/10.1016/j.jlp.2016.01.006
Lv JY, Chen LP, Chen WH, Gao HS, Peng MJ (2013) Kinetic analysis and self-accelerating decomposition temperature (SADT) of dicumyl peroxide. Process Saf Environ 571:60–63. https://doi.org/10.1016/j.tca.2013.08.029
Miyake A, Sumino M, Oka Y, Ogawa T, Iizuka Y (2000) Prediction and evaluation of the reactivity of self-reactive substances using microcalorimetries. Thermochim Acta 352:181–188. https://doi.org/10.1016/S0040-6031(99)00465-7
Mundhwa M, Henni A (2007) Molar excess enthalpy for various alkanolamine(1) + water(2) systems at T = (298.15, 313.15, and 323.15) K. J Chem Thermodyn 39:1439–1451. https://doi.org/10.1016/j.jct.2007.03.010
Muravyev NV, Kiselev VG (2017) Cheaper, faster, or better: are simple estimations of safety parameters of hazardous materials reliable? Comments on “thermal behaviors, nonisothermal decomposition reaction kinetics, thermal safety and burning rates of BTATz-CMDB propellant” by Zhao et al. (2010). J Hazard Mater 334:267–270. https://doi.org/10.1016/j.jhazmat.2017.03.063
Nguyen GT, Tran KT, Pham TV, Nguyen HT (2017) Kinetic study of synthesis reaction of lignosulfonate using isothermal differential scanning calorimetry method. Vietnam J Sci Technol 55:493. https://doi.org/10.15625/2525-2518/55/4/9029
Ning BK, Hu RZ, Zhang H, Xia ZM, Guo PJ, Liu R, Lu G, Jiang JY (2004) Estimation of the critical rate of temperature rise for thermal explosion of autocatalytic decomposing reaction of nitrocellulose using non-isothermal DSC. Thermochim Acta 416:47–50. https://doi.org/10.1016/j.tca.2003.11.029
Onésippe C, Passe-Coutrin N, Toro F, Delvasto S, Bilba K, Arsène MA (2010) Sugar cane bagasse fibres reinforced cement composites: thermal considerations. Compos Part A-Appl S 41:549–556. https://doi.org/10.1016/j.compositesa.2010.01.002
Ping P, Wang QS, Huang PF, Sun JH, Chen CH (2014) Thermal behaviour analysis of lithium-ion battery at elevated temperature using deconvolution method. Appl Energy 129:261–273. https://doi.org/10.1016/j.apenergy.2014.04.092
Poel GV, Mathot VBF (2006) High-speed/high performance differential scanning calorimetry (HPer DSC): temperature calibration in the heating and cooling mode and minimization of thermal lag. Thermochim Acta 446:41–54. https://doi.org/10.1016/j.tca.2006.02.022
Pourmortazavi SM, Hosseini SG, Rahimi-Nasrabadi M, Hajimirsadeghi SS, Momenian H (2009) Effect of nitrate content on thermal decomposition of nitrocellulose. J Hazard Mater 162:1141–1144. https://doi.org/10.1016/j.jhazmat.2008.05.161
Press WH, Flannery BP, Teukolsky SA, Veterling WT (1989) Numerical recipes in pascal. Cambridge University Press, Cambridge
Shao ZQ, Wang WJ (2011) Structure and properties of nitrocellulose. National Defense Industry Press, Beijing (in Chinese)
Sovizi MR, Hajimirsadeghi SS, Naderizadeh B (2009) Effect of particle size on thermal decomposition of nitrocellulose. J Hazard Mater 168:1134–1139. https://doi.org/10.1016/j.jhazmat.2009.02.146
Sun JH, Ding H (2007) Evaluation of the thermal risk of chemical substances, 2nd edn. Science Press, Beijing (in Chinese)
Sun JH, Sun ZH, Wang QS, Ding H, Wang T, Jiang CS (2005) Catalytic effects of inorganic acids on the decomposition of ammonium nitrate. J Hazard Mater 127:204–210. https://doi.org/10.1016/j.jhazmat.2005.07.028
Sun ZD, Fu XL, Yu HJ, Fan XZ, Ju XH (2017) Theoretical study on stabilization mechanisms of nitrate esters using aromatic amines as stabilizers. J Hazard Mater 339:401–408. https://doi.org/10.1016/j.jhazmat.2017.06.025
Tang Z, Ren Y, Yang L, Zhang TL, Qiao XJ, Zhang JG, Zhou ZN (2011) A new way to estimate the thermal decomposition mechanism function and thermal safety of RDX. Chin J Explos Propellants 34:19–25
Trache D (2016) Comments on “thermal degradation behavior of hypochlorite-oxidized starch nanocrystals under different oxidized levels”. Carbohyd Polym 151:535–537. https://doi.org/10.1016/j.carbpol.2016.05.106
Trache D, Khimeche K (2013) Study on the influence of ageing on thermal decomposition of double-base propellants and prediction of their in-use time. Fire Mater 37:328–336. https://doi.org/10.1002/fam.2138
Trache D, Tarchoun AF (2018) Stabilizers for nitrate ester-based energetic materials and their mechanism of action: a state-of-the-art review. J Mater Sci 53:100–123. https://doi.org/10.1007/s10853-017-1474-y
Trache D, Khimeche K, Mezroua A, Benziane M (2016) Physicochemical properties of microcrystalline nitrocellulose from Alfa grass fibres and its thermal stability. J Therm Anal Calorim 124:1485–1496. https://doi.org/10.1007/s10973-016-5293-1
Trache D, Abdelaziz A, Siouani B (2017) A simple and linear isoconversional method to determine the pre-exponential factors and the mathematical reaction mechanism functions. J Therm Anal Calorim 128:335–348. https://doi.org/10.1007/s10973-016-5962-0
Trache D, Maggi F, Palmucci I, DeLuca LT (2018) Thermal behavior and decomposition kinetics of composite solid propellants in the presence of amide burning rate suppressants. J Therm Anal Calorim 132:1601–1615. https://doi.org/10.1007/s10973-018-7160-8
Vincent L, Barale S, Sandeau A, Castillo RV, Genet N, Sbirrazzuoli N (2017) Monitoring grease production by reaction calorimetry and thermoanalytical methods as an alternative to dropping point determination. Energy Fuels 31:11489–11494. https://doi.org/10.1021/acs.energyfuels.7b01920
Vyazovkin S (2001) Modification of the integral isoconversional method to account for variation in the activation energy. J Comput Chem 22:178–183. https://doi.org/10.1002/1096-987X(20010130)22:2%3c178:AID-JCC5%3e3.0.CO;2-%23
Vyazovkin S, Burnham AK, Criado JM, Pérez-Maqueda LA, Popescu C, Sbirrazzuoli N (2011) ICTAC kinetics committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta 520:1–19. https://doi.org/10.1016/j.tca.2011.03.034
Wang QS, Sun JH, Yao XL, Chen CH (2006) C80 calorimeter studies of the thermal behavior of LiPF6 solutions. J Solut Chem 35:179–189. https://doi.org/10.1007/s10953-006-9377-6
Wang QS, Ping P, Zhao XJ, Chu GQ, Sun JH, Chen CH (2012) Thermal runaway caused fire and explosion of lithium ion battery. J Power Sources 208:210–224. https://doi.org/10.1016/j.jpowsour.2012.02.038
Wang XY, Fleischmann C, Spearpoint M, Li KY (2017) A simple hand calculation method to estimate the pyrolysis kinetics of plastic and wood materials. Fire science and technology 2015. Springer, Singapore, pp 455–462. https://doi.org/10.1007/978-981-10-0376-9_46
Wei RC, He YP, Liu JH, He Y, Mi WZ, Yuen R, Wang J (2017) Experimental study on the fire properties of nitrocellulose with different structures. Materials 10:316. https://doi.org/10.3390/ma10030316
Weir J (1946) Nitroglycerine and guncotton: a double centenary. Nature 158:83. https://doi.org/10.1038/158083a0
Wu TQ, Chen HD, Wang QS, Sun JH (2018) Comparison analysis on the thermal runaway of lithium-ion battery under two heating modes. J Hazard Mater. https://doi.org/10.1016/j.jhazmat.2017.11.022
Xinhua News Agency (2016) Tianjin Port “8.12” Ruihai dangerous goods warehouse special fire and explosion investigation report. http://www.chinasafety.gov.cn/newpage/newfiles/201600812baogao.pdf. (in Chinese)
Xu KZ, Song JR, Zhao FQ, Ma HX, Gao HX, Chang CR, Ren YH, Hu RZ (2008) Thermal behavior, specific heat capacity and adiabatic time-to-explosion of G (FOX-7). J Hazard Mater 158:333–339. https://doi.org/10.1016/j.jhazmat.2008.01.077
Xue L, ZhaoFQ HuRZ, Gao HX (2010) A simple method to estimate the critical temperature of thermal explosion for energetic materials using nonisothermal DSC. J Energ Mater 28:17–34. https://doi.org/10.1080/07370650903124518
Zhang X, Weeks BL (2014) Preparation of sub-micron nitrocellulose particles for improved combustion behavior. J Hazard Mater 268:224–228. https://doi.org/10.1016/j.jhazmat.2014.01.019
Zhang TL, Hu RZ, Xie Y, Li FP (1994) The estimation of critical temperatures of thermal explosion for energetic materials using non-isothermal DSC. Thermochim Acta 244:171–176. https://doi.org/10.1016/0040-6031(94)80216-5
Zhang YH, Shao ZQ, Gao KZ, Li J, Wu X, Wang WJ, Wang FJ, Liu YH (2014) Rheological characteristics of nitrate glycerol ether cellulose gel based on phase separation in ternary system. Cellulose 21:4135–4143. https://doi.org/10.1007/s10570-014-0355-7
Zhao FQ, Hu RZ, Gao HX (2009) A simple method based on Harcourt-Esson’s equation to estimate the critical temperature of thermal explosion for energetic materials using non-isothermal DSC. Chin J Chem 27:1067–1072. https://doi.org/10.1002/cjoc.200990178
Zhou JH, Zheng YP, Liu JJ, Bing X, Hua JJ, Zhang HY (2016) A paper-based detection method of cancer cells using the photo-thermal effect of nanocomposite. J Pharm Biomed 117:333–337. https://doi.org/10.1016/j.jpba.2015.09.017
Acknowledgments
This work is supported by the National Key Research and Development Plan (Projects Nos. 2016YFC0801502 and 2016YFC0801505). These supports are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chai, H., Duan, Q., Jiang, L. et al. Theoretical and experimental study on the effect of nitrogen content on the thermal characteristics of nitrocellulose under low heating rates. Cellulose 26, 763–776 (2019). https://doi.org/10.1007/s10570-018-2100-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-018-2100-0